
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Jaume Garcia-Amorós | + 4185 word(s) | 4185 | 2021-12-08 07:38:36 | | | |
| 2 | Vivi Li | Meta information modification | 4185 | 2021-12-20 03:28:55 | | |
Video Upload Options
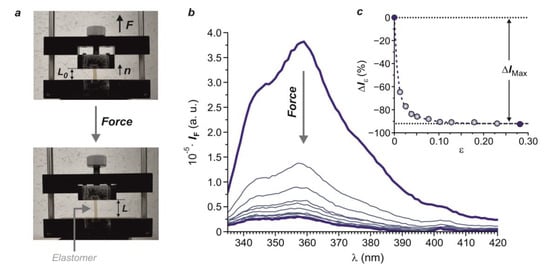
Liquid single crystal elastomers (LSCEs) containing carbazole fluorogenic components alter their luminescence when they are stretched along the director direction. The differential luminescent behavior arises from the distinct interaction between the carbazole fluorophores and their local environment before and after the application of the mechanical input. Indeed, the uniaxial deformation of the material, along its anisotropic direction, forces a closer mesogen–fluorophore interaction, which leads to the quenching of the carbazole luminescence. Importantly, this intermolecular interaction is intimately related to the intrinsic order present in the LSCE. As a result, the amount of light emitted by the material in the form of fluorescence diminishes upon deformation. Thus, the application of mechanical stimuli to liquid-crystalline elastomers furnishes to two interconvertible states for the system with distinct optical properties (with either different emission color or fluorescence intensity). The initial state of the material is completely restored once the applied force is removed. In this way, this kind of macromolecular system can transduce mechanical events into detectable and processable optical signals, thus, having great potential as optical force sensors. In this context, the realization of the distinct structural factors that govern the interactions established between the mesogenic and fluorogenic units at the supramolecular level upon deformation is essential for the development of efficient LSCE-based force sensors. In fact, not only the density of carbazole units and their connection to the main polymer backbone, but also the presence of long range molecular order in the system and the type of mesophase exhibited by the LSCE are key factors for the conception of efficient force sensors based on these self-organized polymer networks.
1. Introduction
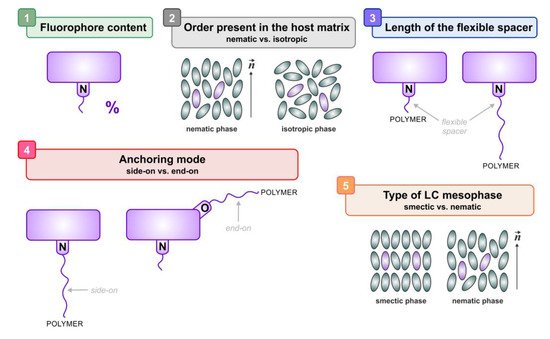
-
Fluorophore content.
-
Presence of long range orientational order in the host matrix.
-
Length of the flexible spacer connecting the carbazole fluorophores to the polymer backbone.
-
Anchoring mode (side-on or end-on) of the fluorogenic moieties.
-
Type of mesophase exhibited by the elastomeric material.
2. Discussion
2.1. Design, Synthesis and Characterization of the Mechanofluorescent Elastomers
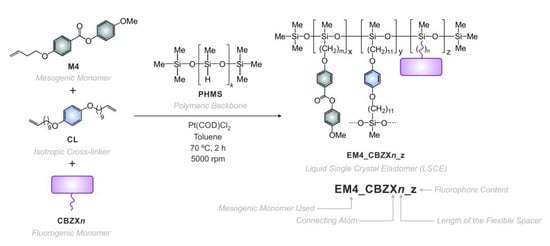
2.2. Structural Factors That Impact the Mechanofluorescent Behavior
2.2.1. Fluorophore Content in the Elastomeric Network
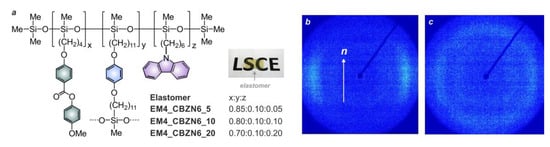

2.2.2. Presence of Long Range Orientational Order in the Host Matrix
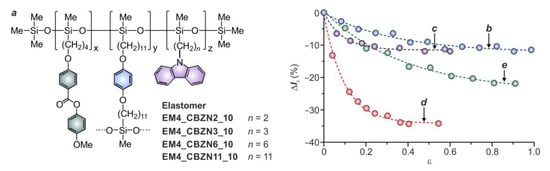
2.2.3. Length of the Fluorophore Flexible Spacer
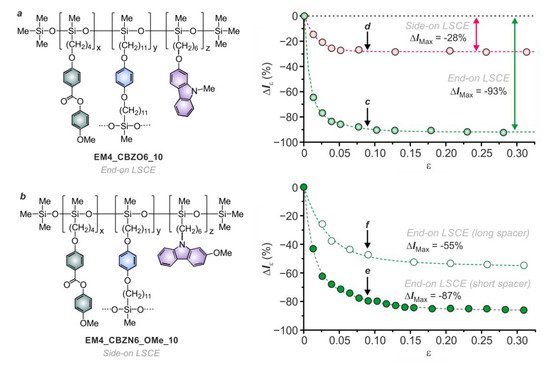
2.2.4. Anchoring Mode (End-on Versus Side-on)
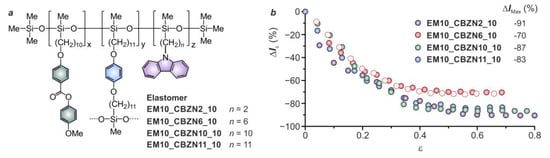
2.2.5. Type of Mesophase Exhibited by the LSCE
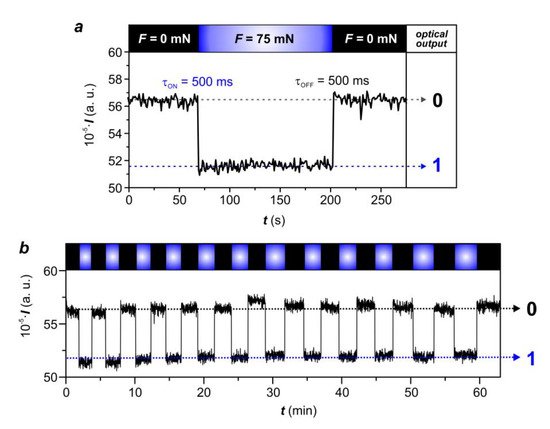
2.2.6. Performance of the LSCE-Based Optical Force Sensors
References
- Warner, M.; Terentjev, E.M. Liquid Crystal Elastomers; Clarendon Press: Oxford, UK, 2007.
- Demus, D.; Goodby, J.W.; Gray, G.W.; Spiess, H.W.; Vill, V. Handbook of Liquid Crystals; Wiley-VCH: Weinheim, Germany, 1998.
- Fleischmann, E.K.; Zentel, R. Liquid-Crystalline Ordering as a Concept in Materials Science: From Semiconductors to Stimuli-Responsive Devices. Angew. Chem. Int. Ed. 2013, 52, 8810–8827.
- Finkelmann, H.; Nishikawa, E.; Pereira, G.G.; Warner, M. A New Opto-Mechanical Effect in Solids. Phys. Rev. Lett. 2001, 87, 015501-1–015501-4.
- Hogan, P.M.; Tajbakhsh, A.R.; Terentjev, E.M. UV Manipulation of Order and Macroscopic Shape in Nematic Elastomers. Phys. Rev. E 2002, 65, 0411720.
- Broer, D.J.; Crawford, G.P.; Zumer, S. The Liquid Crystals Book Series. Cross-Linked Liquid Crystalline Systems: From Rigid Polymer Networks to Elastomers; CRC Press: Boca Raton, FL, USA, 2011.
- Camacho-López, M.; Finkelmann, H.; Palffy-Muhoray, P.; Shelley, M. Fast Liquid-Crystal Elastomer Swims into the Dark. Nat. Mater. 2004, 3, 307–310.
- Garcia-Amorós, J.; Finkelmann, H.; Velasco, D. Influence of the Photo-Active Azo Cross-Linker Spacer on the Opto-Mechanics of Polysiloxane Elastomer Actuators. J. Mater. Chem. 2011, 21, 1094–1101.
- Garcia-Amorós, J.; Piñol, A.; Finkelmann, H.; Velasco, D. Azophenol-Based Liquid-Crystalline Elastomers for Light-Driven Actuators. Org. Lett. 2011, 13, 2282–2285.
- Garcia-Amorós, J.; Martínez, M.; Finkelmann, H.; Velasco, D. Photoactuation and Thermal Isomerisation Mechanism of Cyanoazobenzene-Based Liquid Crystal Elastomers. Phys. Chem. Chem. Phys. 2014, 16, 8448–8454.
- Chi, Z.; Zhang, X.; Xu, B.; Zhou, X.; Ma, C.; Zhang, Y.; Liu, S.; Xu, J. Recent Advances in Organic Mechanofluorochromic Materials. Chem. Soc. Rev. 2012, 41, 3878–3896.
- Ma, Z.; Wang, Z.; Teng, M.; Xu, Z.; Jia, X. Mechanically Induced Multicolor Change of Luminescent Materials. ChemPhysChem 2015, 16, 1811–1828.
- Xu, J.; Chi, Z. (Eds.) Mechanochromic Fluorescent Materials: Phenomena, Materials and Applications; RSC Smart Materials Series, No. 8; Royal Society of Chemistry: Cambridge, UK, 2014.
- Dong, Y.Q.; Lam, J.W.Y.; Tang, B.Z. Mechanochromic Luminescence of Aggregation-Induced Emission Luminogens. J. Phys. Chem. Lett. 2015, 6, 3429–3436.
- Chen, J.; Tang, B.Z. Aggregation-induced Emission: Fundamentals and Applications; Qin, A., Tang, B.Z., Eds.; Wiley-VCH: Chichester, UK, 2014; Volume 1, pp. 307–322.
- Huang, X.; Qian, L.; Zhou, Y.; Liu, M.; Cheng, Y.; Wu, H. Effective Structural Modification of Traditional Fluorophores to Obtain Organic Mechanofluorochromic Molecules. J. Mater. Chem. C 2018, 6, 5075–5096.
- Di, B.-H.; Chen, Y.-L. Recent Progress in Organic Mechanoluminescent Materials. Chin. Chem. Lett. 2018, 29, 245–251.
- Jin, M.; Seki, T.; Ito, H. Mechano-Responsive Luminescence via Crystal-to-Crystal Phase Transitions between Chiral and Non-Chiral Space Groups. J. Am. Chem. Soc. 2017, 139, 7452–7455.
- Garcia-Amorós, J.; Velasco, D. Optical mechanotransduction with carbazole-based liquid single-crystal elastomers. Macromol. Rapid Commun. 2015, 36, 755–761.
- Wei, R.B.; He, Y.N.; Wang, X.G.; Keller, P. Photoluminescent Nematic Liquid Crystalline Elastomer with a Thermomechanical Emission Variation Function. Macromol. Rapid Commun. 2014, 35, 1571–1577.
- Wei, R.B.; Zhang, H.X.; He, Y.N.; Wang, X.G.; Keller, P. Photoluminescent Nematic Liquid Crystalline Elastomer Actuators. Liq. Cryst. 2014, 41, 1821–1830.
- Davis, D.A.; Hamilton, A.; Yang, J.; Cremar, L.D.; Van Gough, D.; Potisek, S.L.; Ong, M.T.; Braun, P.V.; Martínez, T.J.; White, S.R.; et al. Force-Induced Activation of Covalent Bonds in Mechanoresponsive Polymeric Materials. Nature 2009, 459, 68–72.
- Kingsbury, C.M.; May, P.A.; Davis, D.A.; White, S.R.; Moore, J.S.; Sottos, N.R. Shear Activation of Mechanophore-Crosslinked Polymers. J. Mater. Chem. 2011, 21, 8381–8388.
- Kim, T.A.; Robb, M.J.; Moore, J.S.; White, S.R.; Sottos, N.R. Mechanical Reactivity of Two Different Spiropyran Mechanophores in Polydimethylsiloxane. Macromolecules 2018, 51, 9177–9183.
- Imato, K.; Kanehara, T.; Ohishi, T.; Nishihara, M.; Yajima, H.; Ito, M.; Takahara, A.; Otsuka, H. Mechanochromic Dynamic Covalent Elastomers: Quantitative Stress Evaluation and Autonomous Recovery. ACS Macro Lett. 2015, 4, 1307–1311.
- Ducrot, E.; Chen, Y.; Bulters, M.; Sijbesma, R.P.; Creton, C. Toughening Elastomers with Sacrificial Bonds and Watching them Break. Science 2014, 344, 186–189.
- Crenshaw, B.R.; Weder, C.J. Deformation Induced Color Changes in Melt-Processed Photoluminescent Polymer Blends. Chem. Mater. 2003, 15, 4717–4724.
- Weder, C.J. Mechanoresponsive Materials. Mater. Chem. 2011, 21, 8235–8236.
- Calvino, C.; Neumann, L.; Weder, C.; Schrettl, S. Approaches to Polymeric Mechanochromic Materials. J. Polym. Sci. Pol. Chem. 2017, 55, 640–652.
- Muramatsu, T.; Sagara, Y.; Traeger, H.; Tamaoki, N.; Weder, C. Mechanoresponsive Behavior of a Polymer-Embedded Red-Light Emitting Rotaxane Mechanophore. ACS Appl. Mater. Interfaces 2019, 11, 24571–24576.
- Sagara, Y.; Karman, M.; Seki, A.; Pannipara, M.; Tamaoki, N.; Weder, C. Rotaxane-Based Mechanophores Enable Polymers with Mechanically Switchable White Photoluminescence. ACS Cent. Sci. 2019, 5, 874–881.
- Sulkanen, A.R.; Sung, J.; Robb, M.J.; Moore, J.S.; Sottos, N.R.; Liu, G.Y. Spatially Selective and Density-Controlled Activation of Interfacial Mechanophores. J. Am. Chem. Soc. 2019, 141, 4080–4085.
- Zhao, D.; Li, G.; Wu, D.; Qin, X.; Neuhaus, P.; Cheng, Y.; Yang, S.; Lu, Z.; Pu, X.; Long, C.; et al. Regiospecific N Heteroarylation of Amidines for Full Color Tunable Boron Difluoride Dyes with Mechanochromic Luminescence. Angew. Chem. Int. Ed. 2013, 52, 13676–13680.
- Li, G.; Song, F.; Wu, D.; Lan, J.; Liu, X.; Wu, J.; Yang, S.; Xiao, D.; You, J. Cation-Anion Interaction Directed Molecular Design Strategy for Mechanochromic Luminescence. Adv. Funct. Mater. 2014, 24, 747–753.
- Zhang, Z.; Xue, P.; Gong, P.; Zhang, G.; Peng, J.; Lu, R. Mechanofluorochromic Behaviors of Biminoenolate Boron Complexes Functionalized with Carbazole. J. Mater. Chem. C 2014, 2, 9543–9551.
- Tanioka, M.; Kamino, S.; Muranaka, A.; Ooyama, Y.; Ota, H.; Shirasaki, Y.; Horigome, J.; Ueda, M.; Uchiyama, M.; Sawada, D.; et al. Reversible Near-Infrared/Blue Mechanofluorochromism of Aminobenzopyranoxanthene. J. Am. Chem. Soc. 2015, 137, 6436–6439.
- Matsunaga, Y.; Yang, J.-S. Multicolor Fluorescence Writing Based on Host-Guest Interactions and Force Induced Fluorescence Color Memory. Angew. Chem. Int. Ed. 2015, 54, 7985–7989.
- Jiang, Y.; Gindre, D.; Allain, M.; Liu, P.; Cabanetos, C.; Roncali, J. A Mechanofluorochromic Push-Pull Small Molecule with Aggregation Controlled Linear and Nonlinear Optical Properties. Adv. Mater. 2015, 27, 4285–4289.
- Yoshii, R.; Suenaga, K.; Tanaka, K.; Chujo, Y. Mechanofluorochromic Materials Based on Aggregation Induced Emission Active Boron Ketoiminates: Regulation of the Direction of the Emission Color Changes. Chem. Eur. J. 2015, 21, 7231–7237.
- Nishida, T.; Ohta, S.; Xu, F.; Shinohara, K.; Kamada, T.; Akashi, H.; Takezaki, M.; Wakamatsu, K.; Orita, A. Dual Emission and Mechanofluorochromism of a V-Shaped π-System Composed of Disulfonyl-Substituted Dibenzocyclooctatetraenes. Org. Lett. 2016, 18, 3988–3991.
- Xue, P.; Yao, B.; Liu, X.; Sun, J.; Gong, P.; Zhang, Z.; Qian, C.; Zhang, Y.; Lu, R. Reversible Mechanochromic Luminescence of Phenothiazine-based 10,10′-Bianthracene Derivatives with Different Lengths of Alkyl Chains. J. Mater. Chem. C 2015, 3, 1018–1025.
- Zhang, Z.; Wu, Z.; Sun, J.; Yao, B.; Xue, P.; Lu, R. β-Iminoenolate Boron Complex with Terminal Triphenylamine Exhibiting Polymorphism and Mechanofluorochromism. J. Mater. Chem. C 2016, 4, 2854–2861.
- Yang, H.; Ye, K.; Sun, J.; Gong, P.; Lu, R. Mechanofluorochromic Behaviors of Salicylaldimine Difluoroboron Complexes. Asian J. Org. Chem. 2017, 6, 199–206.
- Zhao, J.; Chi, Z.; Yang, Z.; Mao, Z.; Zhang, Y.; Ubba, E.; Chi, Z. Recent Progress in the Mechanofluorochromism of Distyrylanthracene Derivatives with Aggregation-Induced Emission. Mater. Chem. Front. 2018, 2, 1595–1608.
- Zhao, W.; He, Z.; Peng, Q.; Lam, J.W.Y.; Ma, H.; Qiu, Z.; Chen, Y.; Zhao, Z.; Shuai, Z.; Dong, Y.; et al. Highly Sensitive Switching of Solid-State Luminescence by Controlling Intersystem Crossing. Nat. Commun. 2018, 9, 3044.
- Sharber, S.A.; Shih, K.-C.; Mann, A.; Frausto, F.; Haas, T.E.; Nieh, M.-P.; Thomas, S.W. Reversible Mechanofluorochromism of Aniline-Terminated Phenylene Ethynylenes. Chem. Sci. 2018, 9, 5415–5426.
- Sagara, Y.; Karman, M.; Verde-Sesto, E.; Matsuo, K.; Kim, Y.; Tamaoki, N.; Weder, C. Rotaxanes as Mechanochromic Fluorescent Force Transducers in Polymers. J. Am. Chem. Soc. 2018, 140, 1584–1587.
- Calvino, C.; Sagara, Y.; Buclin, V.; Haehnel, A.P.; del Prado, A.; Aeby, C.; Simon, Y.C.; Schrettl, S.; Weder, C. Mechanoresponsive, Luminescent Polymer Blends Based on an Excimer Forming Telechelic Macromolecule. Macromol. Rapid Commun. 2019, 40, 1800705.
- Louis, M.; Sethy, R.; Kumar, J.; Katao, S.; Guillot, R.; Nakashima, T.; Allain, C.; Kawai, T.; Métivier, R. Mechano-Responsive Circularly Polarized Luminescence of Organic Solid-State Chiral Emitters. Chem. Sci. 2019, 10, 843–847.
- Hirai, Y.; Wrona-Piotrowicz, A.; Zakrzewski, J.; Brosseau, A.; Guillot, R.; Métivier, R.; Allain, C. Mechanofluorochromism of pyrene-derived amidophosphonates. Photochem. Photobiol. Sci. 2020.
- Li, Y.-X.; Chen, Y.-G.; Yu, Z.-F.; Yang, X.-F.; Nie, Y.; Cui, Y.; Sun, G.-X. Polymorph-Dependent Multicolor-Switchable Mechanofluorochromism of 1-(9-Anthryl)vinyl-3-(3-pyridyl)vinylbenzene: Molecular Conformation versus Intermolecular Interaction. J. Phys. Chem. C 2020, 124, 3784–3792.
- Küpfer, J.; Finkelmann, H. Nematic Liquid Single Crystal Elastomers. Makromol. Chem. Rapid Commun. 1991, 12, 717–726.
- Kim, H.; Zhu, B.; Adetiba, O.; Agrawal, A.; Ajayan, P.; Jacot, J.G.; Verduzco, R. Preparation of Monodomain Liquid Crystal Elastomers and Liquid Crystal Elastomers Composites. J. Vis. Exp. 2016, 108, 53688.
- Lovell, R.; Mitchell, G.R. Molecular Orientation Distribution Derived from an Arbitrary Reflection. Acta Cryst. 1981, A37, 135–137.
- Mitchell, G.R.; Windle, A. Orientation in Liquid Crystal Polymers. In Developments in Crystal Polymers—2; Basset, D.C., Ed.; Science: London, UK; New York, NY, USA, 1988; pp. 115–175.
- Schatzle, J.; Kaufhold, W.; Finkelmann, H. Nematic Elastomers: The Influence of External Mechanical Stress on the Liquid-Crystalline Phase Behavior. Makromol. Chem. 1989, 190, 3269–3284.
- Garcia-Amorós, J.; Bassaganyas, S.; Velasco, D. Exploring Optical Mechanotransduction in Fluorescent Liquid Crystal Elastomers. Phys. Chem. Chem. Phys. 2016, 18, 5108–5111.
- Heras, D.; Reig, M.; Llorca-Isern, N.; Garcia-Amorós, J.; Velasco, D. Highly Efficient Elastomeric Fluorescence Sensors for Force Detection. ACS Appl. Polym. Mater. 2019, 1, 535–541.
- Escalera-López, D.; Garcia-Amorós, J.; Velasco, D. Smectic-B Liquid Single Crystal Elastomers as Efficient Optical Mechanotransducers. Macromol. Chem. Phys. 2018, 219, 1700550.





