
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Domenico De Berardis | + 2094 word(s) | 2094 | 2021-12-10 03:07:52 | | | |
| 2 | Dean Liu | Meta information modification | 2094 | 2021-12-14 01:53:45 | | | | |
| 3 | Dean Liu | Meta information modification | 2094 | 2021-12-14 01:54:20 | | | | |
| 4 | Lindsay Dong | Meta information modification | 2094 | 2022-03-28 04:10:36 | | |
Video Upload Options
C-reactive protein (CRP) is an acute-phase protein that is produced by hepatocytes and other cell types, including immune cells, endothelial cells, and smooth muscle cells, after stimulation of the gene encoding for CRP by Interleukin-6 (IL-6).
1. Role of CRP in Development of Schizophrenia
C-reactive protein (CRP) is an acute-phase protein that is produced by hepatocytes and other cell types, including immune cells, endothelial cells, and smooth muscle cells, after stimulation of the gene encoding for CRP by Interleukin-6 (IL-6) [38]. The circulating levels of CRP in serum rise and fall according to the inflammatory status of the body and therefore it is the most commonly used biomarker of systemic inflammation worldwide [38,39]. CRP is a standard laboratory exam and can be measured in the peripheral blood and analyzed in any clinical laboratory around the globe. Therefore, CRP has a very promising potential in being a clinical biomarker for psychiatric disorders [40]. The high-sensitivity CRP (hs-CRP) assay has a lower limit of detection of 0.1 mg/L. However, these detection limits may vary from manufacturer to manufacturer. The measurement of CRP is useful in the diagnosis and monitoring of many acute and chronic inflammatory conditions of both infectious and non-infectious etiology [41].
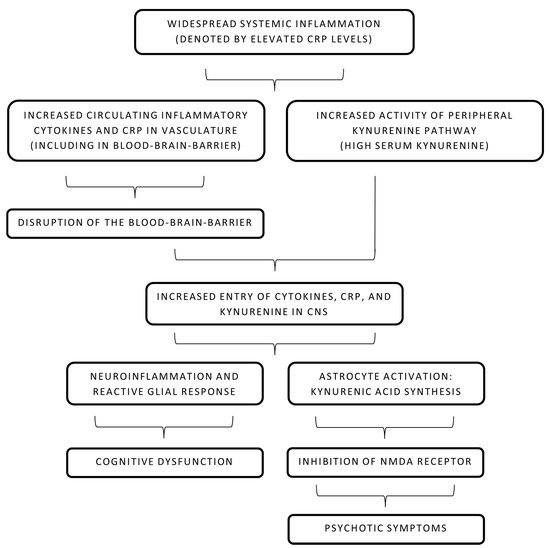
The role of CRP in the causality of schizophrenia has been a subject of interest for many years. It is known that CRP and other acute-phase reactants cause disruption of the BBB and alter its permeability for inflammatory mediators and antibodies [1][2]. As described above, this compromise in the integrity of the BBB in the backdrop of widespread systemic inflammation has been linked to developing psychotic symptoms [3][4]. Increased CRP has also been recently linked with a significant decline in multiple cognitive domains, including working memory and learning ability, in individuals of all ages suffering from schizophrenia [5]. Further reviews were consistent in highlighting a marked cognitive impairment in schizophrenic patients with even low-grade or subclinical inflammation [6][7]. Interestingly, a study in Finland elucidated the role of elevated maternal CRP levels in increasing the risk of psychosis in the offspring [8]. In summary, an inverse association of CRP levels and cognitive ability is noted in acute psychosis. However, a clinical trial published in 2019 provided valuable insight into the prognostic value of CRP in six months following the resolution of acute psychosis [9]. Cognitive abilities improved with time and a noticeable drop in CRP levels was noted in the later periods, continuing the inverse relationship [9]. The pathophysiology behind increased CRP levels and subsequent cognitive dysfunction is not fully known but some studies have implicated CRP in increasing BBB permeability during acute inflammatory phases and causing neuroinflammation [10]. Mouse models revealed that CRP has no virtual effect on BBB permeability in smaller amounts but impairs its function and increases paracellular permeability in higher amounts that may be achieved during high systemic inflammation or pro-inflammatory states like psychosis [10]. Multiple complex molecular pathways have been described to explain CRP-led increased permeability of the BBB and endothelial dysfunction [10]. Notably, it is widely believed that the disruption of tight junctions is involved in the increased permeation of CRP [2]. CRP in blood activates the surface Fc-gamma receptors (CD16/32) on endothelial cells and leads to the formation of reaction oxygen species (via p38-mitogen-activated protein kinase mechanism) that causes an alteration in the myosin light chain kinase activity [2]. This implies that impairment of tight junctions by CRP is most likely achieved via modification of the cytoskeletal structure and induction of abnormal contractility [2][10]. After entering the CNS, CRP causes a reactive microglial reaction, astrogliosis, and neuroinflammation [11]. Neuroinflammation in psychiatric conditions is invariably associated with short-term and lasting cognitive deficits [12]. Therefore, this mechanism may explain how elevated CRP levels (reflecting systemic inflammation) in psychosis may lead to decreased cognitive abilities.
Apart from CRP’s direct role in the disruption of the BBB, its role in the increment of the kynurenine pathway is still unclear. High kynurenic acid levels, as described earlier in this text, act as an antagonist at the NMDA receptor and possibly contribute to psychotic symptoms [13]. Both CNS and peripheral kynurenine pathways are tightly regulated by the immune status of the body [14]. With increased BBB permeability in the setting of elevated CRP levels, infiltration of CRP and other cytokines in CNS leads to neuroinflammation and increased activation of astrocytes and microglia [15]. Tryptophan degeneration and kynurenic acid production are increased and the antagonistic effect on NMDA is more pronounced, leading to psychotic symptoms [14]. It is also important to consider that some studies have concluded that CRP levels are not associated with the kynurenine pathway or levels of kynurenic acid in CNS [16][17]. It is important to note that CRP levels have been largely associated with cognitive symptoms in psychosis, while kynurenic acid levels are associated with psychiatric symptoms [18]. Therefore, it is unclear whether CRP levels and kynurenic acid levels act as two independent markers occurring coincidentally during schizophrenia or have a complex interaction that leads to a possible cause–effect relationship.
2. Associations of CRP with Psychotic Symptoms and Role in Clinical Evaluation
3. CRP and Treatment of Schizophrenia
References
- Nehring, S.M.; Goyal, A.; Bansal, P.; Patel, B.C. C Reactive Protein, In StatPearls, Treasure Island (FL), StatPearls Publishing. 2021. Available online: https://www.ncbi.nlm.nih.gov/books/NBK441843 (accessed on 10 May 2021).
- Fond, G.; Lançon, C.; Auquier, P.; Boyer, L. C-Reactive Protein as a Peripheral Biomarker in Schizophrenia. An Updated Systematic Review. Front. Psychiatry 2018, 9, 392.
- Fond, G.; D’Albis, M.-A.; Jamain, S.; Tamouza, R.; Arango, C.; Fleischhacker, W.W.; Glenthøj, B.; Leweke, M.; Lewis, S.; McGuire, P.; et al. The Promise of Biological Markers for Treatment Response in First-Episode Psychosis: A Systematic Review. Schizophr. Bull 2015, 41, 559–573.
- Windgassen, E.B.; Funtowicz, L.; Lunsford, T.N.; Harris, L.A.; Mulvagh, S.L. C-Reactive Protein and High-Sensitivity C-Reactive Protein: An Update for Clinicians. Postgrad. Med. 2011, 123, 114–119.
- Wium-Andersen, M.K.; Ørsted, D.D.; Nordestgaard, B.G. Elevated C-reactive protein associated with late- and very-late-onset schizophrenia in the general population: A prospective study. Schizophr. Bull. 2014, 40, 1117–1127.
- Kuhlmann, C.R.; Librizzi, L.; Closhen, D.; Pflanzner, T.; Lessmann, V.; Pietrzik, C.U.; de Curtis, M.; Luhmann, H.J. Mechanisms of C-reactive protein-induced blood-brain barrier disruption. Stroke 2009, 40, 1458–1466.
- Benros, M.E.; Nielsen, P.R.; Nordentoft, M.; Eaton, W.W.; Dalton, S.O.; Mortensen, P.B. Autoimmune diseases and severe infections as risk factors for schizophrenia: A 30-year population-based register study. Am. J. Psychiatry 2011, 168, 1303–1310.
- Steiner, J.; Frodl, T.; Schiltz, K.; Dobrowolny, H.; Jacobs, R.; Fernandes, B.S.; Guest, P.C.; Meyer-Lotz, G.; Borucki, K. Innate Immune Cells and C-Reactive Protein in Acute First-Episode Psychosis and Schizophrenia: Relationship to Psychopathology and Trehatment. Schizophr. Bull 2020, 46, 363–373.
- Park, S.; Miller, B.J. Meta-analysis of cytokine and C-reactive protein levels in high-risk psychosis. Schizophr. Res. 2020, 226, 5–12.
- Misiak, B.; Stanczykiewicz, B.; Kotowicz, K.; Rybakowski, J.K.; Samochowiec, J.; Frydecka, D. Cytokines and C-reactive protein alterations with respect to cognitive impairment in schizophrenia and bipolar disorder: A systematic review. Schizophr. Res. 2018, 192, 16–29.
- Bora, E. Peripheral inflammatory and neurotrophic biomarkers of cognitive impairment in schizophrenia: A meta-analysis. Psychol. Med. 2019, 49, 1971–1979.
- Canetta, S.; Sourander, A.; Surcel, H.-M.; Hinkka-Yli-Salomäki, S.; Leiviskä, J.; Kellendonk, C.; McKeague, I.W.; Brown, A.S. Elevated maternal C-reactive protein and increased risk of schizophrenia in a national birth cohort. Am. J. Psychiatry. 2014, 171, 960–968.
- Fathian, F.; Løberg, E.-M.; Gjestad, R.; Steen, V.M.; Kroken, R.A.; Jørgensen, H.A.; Johnsen, E. Associations between C-reactive protein levels and cognition during the first 6 months after acute psychosis. Acta Neuropsychiatr. 2019, 31, 36–45.
- Hsuchou, H.; Kastin, A.J.; Mishra, P.K.; Pan, W. C-reactive protein increases BBB permeability: Implications for obesity and neuroinflammation. Cell Physiol. Biochem. 2012, 30, 1109–1119.
- Hsuchou, H.; Kastin, A.J.; Pan, W. Blood-borne metabolic factors in obesity exacerbate injury-induced gliosis. J. Mol. Neurosci. 2012, 47, 267–277.
- Fourrier, C.; Singhal, G.; Baune, B.T. Neuroinflammation and cognition across psychiatric conditions. CNS Spectr. 2019, 24, 4–15.
- Pedraz-Petrozzi, B.; Elyamany, O.; Rummel, C.; Mulert, C. Effects of inflammation on the kynurenine pathway in schizophrenia —A systematic review. J. Neuroinflamm. 2020, 17, 56.
- Schwarcz, R.; Bruno, J.P.; Muchowski, P.J.; Wu, H.Q. Kynurenines in the mammalian brain: When physiology meets pathology. Nat. Rev. Neurosci. 2012, 13, 465–477.
- Owe-Young, R.; Webster, N.L.; Mukhtar, M.; Pomerantz, R.J.; Smythe, G.; Walker, D.; Armati, P.J.; Crowe, S.M.; Brew, B.J. Kynurenine pathway metabolism in human blood-brain-barrier cells: Implications for immune tolerance and neurotoxicity. J. Neurochem. 2008, 105, 1346–1357.
- Khandaker, G.M.; Pearson, R.M.; Zammit, S.; Lewis, G.; Jones, P.B. Association of serum interleukin 6 and C-reactive protein in childhood with depression and psychosis in young adult life: A population-based longitudinal study. JAMA Psychiatry 2014, 71, 1121–1128.
- Wurfel, B.E.; Drevets, W.C.; Bliss, S.A.; McMillin, J.R.; Suzuki, H.; Ford, B.N.; Morris, H.M.; Teague, T.K.; Dantzer, B.; Savitz, J.B. Serum kynurenic acid is reduced in affective psychosis. Transl. Psychiatry 2017, 7, e1115.
- Dickerson, F.; Stallings, C.; Origoni, A.; Boronow, J.; Yolken, R. C-reactive protein is associated with the severity of cognitive impairment but not of psychiatric symptoms in individuals with schizophrenia. Schizophr. Res. 2007, 93, 261–265.
- Ligthart, S. Commentary: CRP and schizophrenia: Cause, consequence or confounding? Int. J. Epidemiol. 2019, 48, 1514–1515.
- Boozalis, T.; Teixeira, A.L.; Cho, R.Y.; Okusaga, O. C-Reactive Protein Correlates with Negative Symptoms in Patients with Schizophrenia. Front. Public Health 2018, 5, 360.
- Micoulaud-Franchi, J.A.; Faugere, M.; Boyer, L.; Fond, G.; Richieri, R.; Faget, C.; Cermolacce, M.; Philip, P.; Vion-Dury, J.; Lancon, C. Elevated C-reactive protein is associated with sensory gating deficit in schizophrenia. Schizophr. Res. 2015, 165, 94–96.
- Wang, Z.; Li, P.; Chi, D.; Wu, T.; Mei, Z.; Cui, G. Association between C-reactive protein and risk of schizophrenia: An updated meta-analysis. Oncotarget 2017, 8, 75445–75454.
- Fernandes, B.S.; Steiner, J.; Bernstein, H.-G.; Dodd, S.; Pasco, J.A.; Dean, O.M.; Nardin, P.; Gonçalves, C.A.; Berk, M. C-reactive protein is increased in schizophrenia but is not altered by antipsychotics: Meta-analysis and implications. Mol. Psychiatry 2016, 21, 554–564.
- Akanji, A.O.; Ohaeri, J.U.; Al-Shammri, S.; Fatania, H.R. Association of blood levels of C-reactive protein with clinical phenotypes in Arab schizophrenic patients. Psychiatry Res. 2009, 169, 56–61.
- Suvisaari, J.; Loo, B.M.; Saarni, S.E.; Haukka, J.; Perälä, J.; Saarni, S.I.; Viertiö, S.; Partti, K.; Lönnqvist, J.; Jula, A. Inflammation in psychotic disorders: A population-based study. Psychiatry Res. 2011, 189, 305–311.
- Fawzi, M.H.; Fawzi, M.M.; Fawzi, M.M.; Said, N.S. C-reactive protein serum level in drug-free male Egyptian patients with schizophrenia. Psychiatry Res. 2011, 190, 91–97.
- Miola, A.; Dal Porto, V.; Tadmor, T.; Croatto, G.; Scocco, P.; Manchia, M.; Carvalho, A.F.; Maes, M.; Vieta, E.; Sambataro, F. Increased C-reactive protein concentration and suicidal behavior in people with psychiatric disorders: A systematic review and meta-analysis. Acta Psychiatr. Scand. 2021, 144, 537–552.
- Horsdal, H.T.; Köhler-Forsberg, O.; Benros, M.E.; Gasse, C. C-reactive protein and white blood cell levels in schizophrenia, bipolar disorders and depression—Associations with mortality and psychiatric outcomes: A population-based study. Eur. Psychiatry 2017, 44, 164–172.
- Misiak, B.; Bartoli, F.; Carrà, G.; Stańczykiewicz, B.; Gładka, A.; Frydecka, D.; Samochowiec, J.; Jarosz, K.; Hadryś, T.; Miller, B.J. Immune-inflammatory markers and psychosis risk: A systematic review and meta-analysis. Psychoneuroendocrinology 2021, 127, 105200.
- Osimo, E.F.; Baxter, L.; Stochl, J.; Perry, B.I.; Metcalf, S.A.; Kunutsor, S.K.; Laukkanen, J.A.; Wium-Andersen, M.K.; Jones, P.B.; Khandaker, G.M. Longitudinal association between CRP levels and risk of psychosis: A meta-analysis of population-based cohort studies. NPJ Schizophr. 2021, 7, 31.
- Fond, G.; Lançon, C.; Korchia, T.; Auquier, P.; Boyer, L. The Role of Inflammation in the Treatment of Schizophrenia. Front. Psychiatry. 2020, 11, 160.
- Na, K.-S.; Jung, H.-Y.; Kim, Y.-K. The role of pro-inflammatory cytokines in the neuroinflammation and neurogenesis of schizophrenia. Prog. Neuropsychopharmacol. Biol. Psychiatry 2014, 48, 277–286.
- Muench, J.; Hamer, A.M. Adverse effects of antipsychotic medications. Am. Fam. Physician 2010, 81, 617–622.
- Bulzacka, E.; Boyer, L.; Schürhoff, F.; Godin, O.; Berna, F.; Brunel, L.; Andrianarisoa, M.; Aouizerate, B.; Capdevielle, D.; Chéreau-Boudet, I.; et al. Chronic Peripheral Inflammation is Associated with Cognitive Impairment in Schizophrenia: Results from the Multicentric FACE-SZ Dataset. Schizophr. Bull. 2016, 42, 1290–1302.
- Schwieler, L.; Erhardt, S.; Erhardt, C.; Engberg, G. Prostaglandin-mediated control of rat brain kynurenic acid synthesis--opposite actions by COX-1 and COX-2 isoforms. J. Neural. Transm. 2005, 112, 863–872.
- Müller, N. Inflammation in Schizophrenia: Pathogenetic Aspects and Therapeutic Considerations. Schizophr. Bull. 2018, 44, 973–982.
- Sommer, I.E.; van Westrhenen, R.; Begemann, M.J.; de Witte, L.D.; Leucht, S.; Kahn, R.S. Efficacy of anti-inflammatory agents to improve symptoms in patients with schizophrenia: An update. Schizophr. Bull. 2014, 40, 181–191.
- Çakici, N.; van Beveren, N.J.M.; Judge-Hundal, G.; Koola, M.M.; Sommer, I.E.C. An update on the efficacy of anti-inflammatory agents for patients with schizophrenia: A meta-analysis. Psychol. Med. 2019, 49, 2307–2319.
- Gillespie, A.L.; Samanaite, R.; Mill, J.; Egerton, A.; MacCabe, J.H. Is treatment-resistant schizophrenia categorically distinct from treatment-responsive schizophrenia? a systematic review. BMC Psychiatry 2017, 17, 12.
- Nucifora, F.C.; Woznica, E.; Lee, B.J.; Cascella, N.; Sawa, A. Treatment resistant schizophrenia: Clinical, biological, and therapeutic perspectives. Neurobiol. Dis. 2019, 131, 104257.
- Fond, G.; Godin, O.; Boyer, L.; Berna, F.; Andrianarisoa, M.; Coulon, N.; Brunel, L.; Bulzacka, E.; Aouizerate, B.; Capdevielle, D.; et al. Chronic low-grade peripheral inflammation is associated with ultra resistant schizophrenia. Results from the FACE-SZ cohort. Eur. Arch. Psychiatry Clin. Neurosci. 2019, 269, 985–992.





