
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Felix Broecker | + 3957 word(s) | 3957 | 2021-12-13 04:34:16 | | | |
| 2 | Bruce Ren | Meta information modification | 3957 | 2021-12-14 02:30:26 | | |
Video Upload Options
Viral infections as well as changes in the composition of the intestinal microbiota and virome have been linked to cancer. Moreover, the success of cancer immunotherapy with checkpoint inhibitors has been correlated with the intestinal microbial composition of patients. The transfer of feces—which contain mainly bacteria and their viruses (phages)—from immunotherapy responders to non-responders, known as fecal microbiota transplantation (FMT), has been shown to be able to convert some non-responders to responders. Since phages may also increase the response to immunotherapy, for example by inducing T cells cross-reacting with cancer antigens, modulating phage populations may provide a new avenue to improve immunotherapy responsiveness.
1. Introduction—The Human Virome
2. The Roles of the Eukaryotic Virome in Cancer
| Virus Family | Virus | Cancer Type | Observations |
|---|---|---|---|
| Papillomaviridae | HPV-16 | Anal | HPV, especially HPV-16, is a possible risk factor for anal and rectal cancer [8][9] and a significant prognostic marker, especially for locally advanced disease [10] |
| HPV | Bladder | HPV (different serotypes) may be linked to bladder cancer in a small number of cases [11] | |
| HPV-16, -18 | Cervical | Association between infection with high-risk HPV serotype (mainly HPV-16 and-18) and development of cervical cancer [11][12][13] | |
| HPV-18 | Colorectal | HPV, especially HPV-18, is a possible risk factor for colorectal cancer [14][15], however, another study found no association [16] | |
| HPV-16, -18, -26, -57 | Esophageal | HPV-16 is a risk factor for esophageal carcinoma [17][18]; HPV infection (mainly HPV-16, -18, -26 and-57) is common in esophageal carcinoma [19] | |
| HPV-16 | Head and neck (SCC) | HPV infection, especially HPV-16, is associated with head and neck cancer [11][20][21] and better long-term outcome [22] | |
| HPV-6 | Oral | Association of HPV-6 with oral cancer [23] | |
| HPV-16 | Prostate | Association of HPV-16 with prostate cancer [24] | |
| HPV-16, -18, -58 | Renal | Association of HPV-16, -18 and-58 with renal cell carcinoma [25] | |
| HPV-5, -8 | Skin and mucosal | Papillomavirus DNA frequently detected in skin-and mucosa-associated cancers [26]; HPV-5 and-8 are associated with epidermodysplasia verruciformis associated with a high risk of skin cancer [27][28] | |
| HPV-16 | Vulvar | Association between HPV, especially HPV-16, and vulvar squamous cell carcinoma [29] | |
| Herpesviridae | CMV (HHV5) | Colorectal | CMV DNA is more abundant cancer tissues compared to healthy tissues [30]; CMV-positive tumors in non-elderly patients are associated with increased disease-free survival rate [31]; specific genetic polymorphisms of CMV are linked to different clinical outcomes [32] |
| EBV (HHV4) | Colorectal | Possible association of EBV with colorectal carcinoma [33], however, no association found in another study [16] | |
| EBV (HHV4) | Esophageal | EBV is associated with esophageal cancer [34] | |
| EBV (HHV4) | Gastric | Possible involvement of EBV in gastric cancer and precursor lesions [35]; patients with EBV-positive gastric cancer had a better response to chemotherapy and better survival [36] | |
| EBV (HHV4) | Hepatic | EBV infections detected in HCC tissues [37] | |
| EBV (HHV4) | Lymphoma (Burkitt) | EBV infections contribute to Burkitt lymphoma [38] | |
| EBV (HHV4) | Lymphoma (DLBCL) | EBV RNA detected in B-cell lymphoma samples [39] | |
| EBV (HHV4) | Lymphoma (PTCL) | EBV expression associated with some subtypes of peripheral T-cell lymphomas [40] | |
| EBV (HHV4) | Oral | Higher proportion of EBV-positive oral squamous cell carcinoma in industrialized countries [41] | |
| EBV (HHV4) | Skin and mucosal | EBV DNA frequently detected in skin and mucosal cancers [26] | |
| HHV6 | Lymphoma (DLBCL) | HHV6 RNA detected in B-cell lymphoma samples [39] | |
| HHV6 | Malignant melanoma | HHV6 DNA frequently detected in malignant melanoma [26] | |
| HHV7 | Bladder | HHV7 DNA frequently detected in bladder cancer [26] | |
| HHV7 | Lymphoma (CTCL) | HHV7 DNA frequently detected in cutaneous T-cell lymphoma (Mycosis fungoides) [26] | |
| HHV7 | Oral | HHV7 DNA frequently detected in oral cavity cancer [26] | |
| HSV (HHV1/2) | Oral | Higher proportion of HSV-positive oral squamous cell carcinoma in industrialized countries [41] | |
| KSHV (HHV8) | Kaposi sarcoma | In HIV-infected individuals, KSHV infection is associated with Kaposi sarcoma [42] | |
| Polyomaviridae | BKV | Bladder | Possible association of BKV with bladder cancer [26] |
| BKV | Colorectal | Possible association of BKV with colorectal cancer [43][44], however, other studies found no association [16][45] | |
| JCV | Colorectal | JCV is associated with colorectal cancer [45][46] and may be involved in carcinogenesis [47], specifically in chromosomal instability [48]; JCV T-antigen is expressed in early-stage colorectal cancer [49], however, another study found no association [16] | |
| MCV | Merkel cell carcinoma | MCV is the major causative factor for Merkel cell carcinoma [50][51] | |
| Retroviridae | HIV | Anal | HIV-positive people have increased risk for anal cancer [52][53] and worse overall colostomy-free survival rates [54] |
| HIV | Cervical | Cervical cancer is more prevalent in HIV-positive individuals, likely because of increased susceptibility to HPV infection [55][56] | |
| HIV | Kaposi sarcoma | Kaposi sarcoma is more prevalent in HIV-positive individuals, likely because of increased susceptibility to KSHV infection [55][56] | |
| HIV | Lymphoma (NHL) | Aggressive B cell non-Hodgkin lymphoma is more prevalent in HIV-positive individuals, likely because of increased susceptibility to EBV infection [55][56] | |
| HTLV-1 | Lymphoma (ATLL) | HTLV-1 induces adult T-cell leukemia/lymphoma in 5% of infected individuals [57] through random integration into the host genome [58] | |
| Others | HBV | Bile duct | HBV is a risk factor for bile duct cancer [59] |
| HBV | Colorectal | Chronic HBV infection is a risk factor for colorectal cancer [60] | |
| HBV | Hepatic | Liver cancer is associated with HBV [11] | |
| HBV | Pancreatic | Chronic HBV infection [61] or past exposure [62] are risk factors for pancreatic cancer | |
| HCV | Bile duct | HCV is a risk factor for bile duct cancer [59] | |
| HCV | Hepatic | Liver cancer is associated with HCV [11] | |
| TTV | Hepatic | TTV is a risk factor for hepatocellular carcinoma [63] | |
| HBoV | Colorectal | Some colorectal cancers are associated with HBoV [64] | |
| HBoV | Lung | Some lung cancers are associated with HBoV [64] | |
| HBoV | Tonsillar | Association of HBoV with tonsil squamous cell carcinoma [65] | |
| Orthobunyaviruses | Colorectal | High abundance of orthobunyaviruses in colorectal cancer [66] | |
| Parvoviruses | Skin | Parvovirus DNA frequently detected in skin-associated cancers [26] | |
| Anelloviruses | Mucosal | Anellovirus DNA frequently detected in mucosal cancers [26] | |
| Anelloviruses | Leukemias | Anellovirus DNA frequently detected in leukemias [26] |
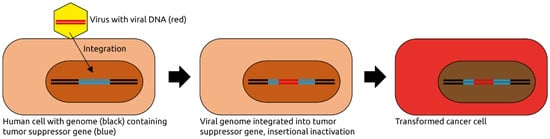
2.1. Papillomaviridae
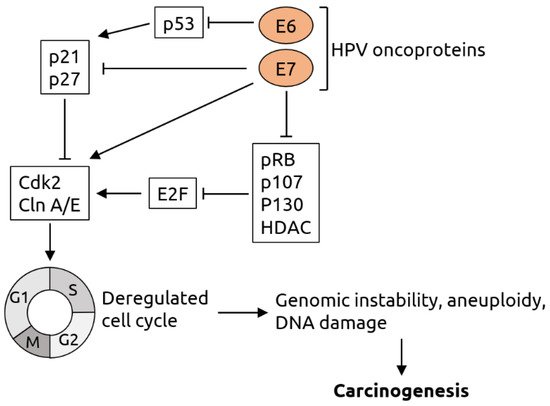
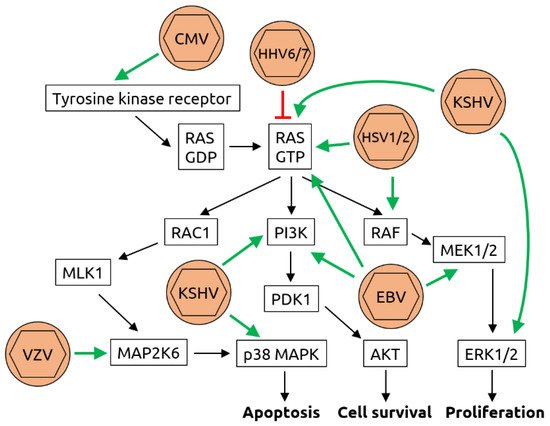
2.2. Herpesviridae
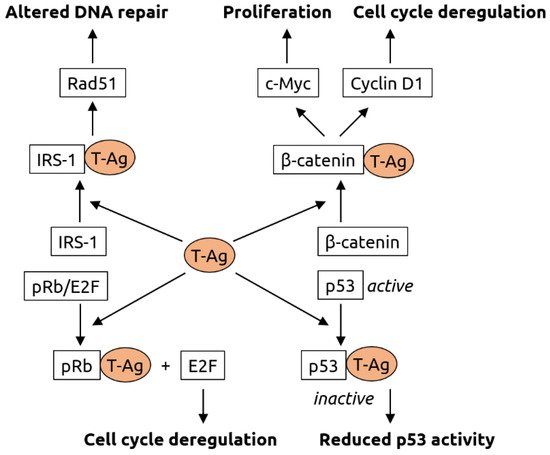
2.3. Polyomaviridae
2.4. Retroviridae
2.5. Other Viruses
3. The Human Intestinal Virome and Its Links to Cancer
References
- Shkoporov, A.N.; Hill, C. Bacteriophages of the Human Gut: The “Known Unknown“ of the Microbiome. Cell Host Microbe 2019, 25, 195–209.
- Liang, G.; Bushman, F.D. The human virome: Assembly, composition and host interactions. Nat. Rev. Microbiol. 2021, 19, 514–527.
- Sender, R.; Fuchs, S.; Milo, R. Revised Estimates for the Number of Human and Bacteria Cells in the Body. PLoS Biol. 2016, 14, e1002533.
- Plummer, M.; de Martel, C.; Vignat, J.; Ferlay, J.; Bray, F.; Franceschi, S. Global burden of cancers attributable to infections in 2012: A synthetic analysis. Lancet Glob. Health 2016, 4, e609–e616.
- Mesri, E.A.; Feitelson, M.A.; Munger, K. Human viral oncogenesis: A cancer hallmarks analysis. Cell Host Microbe 2014, 15, 266–282.
- Zhang, L.L.; Wie, J.Y.; Wang, L.; Huang, S.L.; Chen, J.L. Human T-cell lymphotropic virus type 1 and its oncogenesis. Acta Pharmacol. Sin. 2017, 38, 1093–1103.
- Marônek, M.; Link, R.; Monteleone, G.; Gardlík, R.; Stolfi, C. Viruses in Cancers of the Digestive System: Active Contributors or Idle Bystanders? Int. J. Mol. Sci. 2020, 21, 8133.
- Bodaghi, S.; Yamanegi, K.; Xiao, S.Y.; Da Costa, M.; Palefsky, J.M.; Zheng, Z.M. Colorectal papillomavirus infection in patients with colorectal cancer. Clin. Cancer Res. 2005, 11, 2862–2867.
- Muresu, N.; Sotgiu, G.; Saderi, L.; Sechi, I.; Cossu, A.; Marras, V.; Meloni, M.; Martinelli, M.; Cocuzza, C.; Tanda, F.; et al. Distribution of HPV Genotypes in Patients with a Diagnosis of Anal Cancer in an Italian Region. Int. J. Environ. Res. Public Health 2020, 17, 4516.
- Kabarriti, R.; Brodin, N.P.; Ohri, N.; Narang, R.; Huang, R.; Chuy, J.W.; Rajdev, L.N.; Kalnicki, S.; Guha, C.; Garg, M.K. Human papillomavirus, radiation dose and survival of patients with anal cancer. Acta Oncol. 2019, 58, 1745–1751.
- Cantalupo, P.G.; Katz, J.P.; Pipas, J.M. Viral sequences in human cancer. Virology 2018, 513, 208–216.
- de Sanjose, S.; Quint, W.G.; Alemany, L.; Geraets, D.T.; Klaustermeier, J.E.; Lloveras, B.; Tous, S.; Felix, A.; Bravo, L.E.; Shin, H.R.; et al. Human papillomavirus genotype attribution in invasive cervical cancer: A retrospective cross-sectional worldwide study. Lancet Oncol. 2010, 11, 1048–1056.
- Clifford, G.; Franceschi, S.; Diaz, M.; Muñoz, N.; Villa, L.L. Chapter 3: HPV type-distribution in women with and without cervical neoplastic diseases. Vaccine 2006, 24 (Suppl. S3), 26–34.
- Kirgan, D.; Manalo, P.; Hall, M.; McGregor, B. Association of human papillomavirus and colon neoplasms. Arch. Surg. 1990, 125, 862–865.
- Lee, Y.M.; Leu, S.Y.; Chiang, H.; Fung, C.P.; Liu, W.T. Human papillomavirus type 18 in colorectal cancer. J. Microbiol. Immunol. Infect. 2001, 34, 87–91.
- Fiorina, L.; Ricotti, M.; Vanoli, A.; Luinetti, O.; Dallera, E.; Riboni, R.; Paolucci, S.; Brugnatelli, S.; Paulli, M.; Pedrazzoli, P.; et al. Systematic analysis of human oncogenic viruses in colon cancer revealed EBV latency in lymphoid infiltrates. Infect. Agent. Cancer 2014, 9, 18.
- Bjørge, T.; Hakulinen, T.; Engeland, A.; Jellum, E.; Koskela, P.; Lehtinen, M.; Luostarinen, T.; Paavonen, J.; Sapp, M.; Schiller, J.; et al. A prospective, seroepidemiological study of the role of human papillomavirus in esophageal cancer in Norway. Cancer Res. 1997, 57, 3989–3992.
- Zhang, S.K.; Guo, L.W.; Chen, Q.; Zhang, M.; Liu, S.Z.; Quan, P.L.; Lu, J.B.; Sun, X.B. The association between human papillomavirus 16 and esophageal cancer in Chinese population: A meta-analysis. BMC Cancer 2015, 15, 1096.
- Wang, X.; Tian, X.; Liu, F.; Zhao, Y.; Sun, M.; Chen, D.; Lu, C.; Wang, Z.; Shi, X.; Zhang, Q.; et al. Detection of HPV DNA in esophageal cancer specimens from different regions and ethnic groups: A descriptive study. BMC Cancer 2010, 10, 19.
- Khoury, J.D.; Tannir, N.M.; Williams, M.D.; Chen, Y.; Yao, H.; Zhang, J.; Thompson, E.J.; TCGA Network; Meric-Bernstam, F.; Medeiros, L.J.; et al. Landscape of DNA virus associations across human malignant cancers: Analysis of 3775 cases using RNA-Seq. J. Virol. 2013, 87, 8916–8926.
- Parfenov, M.; Pedamallu, C.S.; Gehlenborg, N.; Freeman, S.S.; Danilova, L.; Bristow, C.A.; Lee, S.; Hadjipanayis, A.G.; Ivanova, E.V.; Wilkerson, M.D.; et al. Characterization of HPV and host genome interactions in primary head and neck cancers. Proc. Natl. Acad. Sci. USA 2014, 111, 15544–15549.
- Curado, M.P.; Hashibe, M. Recent changes in the epidemiology of head and neck cancer. Curr. Opin. Oncol. 2009, 21, 194–200.
- Maden, C.; Beckmann, A.M.; Thomas, D.B.; McKnight, B.; Sherman, K.J.; Ashley, R.L.; Corey, L.; Daling, J.R. Human papillomaviruses, herpes simplex viruses, and the risk of oral cancer in men. Am. J. Epidemiol. 1992, 135, 1093–1102.
- Bae, J.M. Human papillomavirus 16 infection as a potential risk factor for prostate cancer: An adaptive meta-analysis. Epidemiol. Health 2015, 37, e2015005.
- Farhadi, A.; Behzad-Behbahani, A.; Geramizadeh, B.; Sekawi, Z.; Rahsaz, M.; Sharifzadeh, S. High-risk human papillomavirus infection in different histological subtypes of renal cell carcinoma. J. Med. Virol. 2014, 86, 1134–1144.
- Mollerup, S.; Asplund, M.; Friis-Nielsen, J.; Kjartansdóttir, K.R.; Fridholm, H.; Hansen, T.A.; Herrera, J.A.R.; Barnes, C.J.; Jensen, R.H.; Richter, S.R.; et al. High-Throughput Sequencing-Based Investigation of Viruses in Human Cancers by Multienrichment Approach. J. Infect. Dis. 2019, 220, 1312–1324.
- Gewirtzman, A.; Bartlett, B.; Tyring, S. Epidermodysplasia verruciformis and human papilloma virus. Curr. Opin. Infect. Dis. 2008, 21, 141–146.
- Ramoz, N.; Rueda, L.A.; Bouadjar, B.; Montoya, L.S.; Orth, G.; Favre, M. Mutations in two adjacent novel genes are associated with epidermodysplasia verruciformis. Nat. Genet. 2002, 32, 579–581.
- Rakislova, N.; Saco, A.; Sierra, A.; Del Pino, M.; Ordi, J. Role of Human Papillomavirus in Vulvar Cancer. Adv. Anat. Pathol. 2017, 24, 201–214.
- Dimberg, J.; Hong, T.T.; Skarstedt, M.; Löfgren, S.; Zar, N.; Matussek, A. Detection of cytomegalovirus DNA in colorectal tissue from Swedish and Vietnamese patients with colorectal cancer. Anticancer Res. 2013, 33, 4947–4950.
- Chen, H.P.; Jiang, J.K.; Chen, C.Y.; Yang, C.Y.; Chen, Y.C.; Lin, C.H.; Chou, T.Y.; Cho, W.L.; Chan, Y.J. Identification of human cytomegalovirus in tumour tissues of colorectal cancer and its association with the outcome of non-elderly patients. J. Gen. Virol. 2016, 97, 2411–2420.
- Chen, H.P.; Jiang, J.K.; Chan, C.H.; Teo, W.H.; Yang, C.Y.; Chen, Y.C.; Chou, T.Y.; Lin, C.H.; Chan, Y.J. Genetic polymorphisms of the human cytomegalovirus UL144 gene in colorectal cancer and its association with clinical outcome. J. Gen. Virol. 2015, 96, 3613–3623.
- Song, L.B.; Zhang, X.; Zhang, C.Q.; Zhang, Y.; Pan, Z.Z.; Liao, W.T.; Li, M.Z.; Zeng, M.S. Infection of Epstein-Barr virus in colorectal cancer in Chinese. Ai Zheng 2006, 25, 1356–1360.
- Awerkiew, S.; Bollschweiler, E.; Metzger, R.; Schneider, P.M.; Hölscher, A.H.; Pfister, H. Esophageal cancer in Germany is associated with Epstein-Barr-virus but not with papillomaviruses. Med. Microbiol. Immunol. 2003, 192, 137–140.
- Martínez-López, J.L.; Torres, J.; Camorlinga-Ponce, M.; Mantilla, A.; Leal, Y.A.; Fuentes-Pananá, E.M. Evidence of Epstein-Barr virus association with gastric cancer and non-atrophic gastritis. Viruses 2014, 6, 301–318.
- Corallo, S.; Fucà, G.; Morano, F.; Salati, M.; Spallanzani, A.; Gloghini, A.; Volpi, C.C.; Trupia, D.V.; Lobefaro, R.; Guarini, V.; et al. Clinical Behavior and Treatment Response of Epstein-Barr Virus-Positive Metastatic Gastric Cancer: Implications for the Development of Future Trials. Oncologist 2020, 25, 780–786.
- Li, W.; Wu, B.A.; Zeng, Y.M.; Chen, G.C.; Li, X.X.; Chen, J.T.; Guo, Y.W.; Li, M.H.; Zeng, Y. Epstein-Barr virus in hepatocellular carcinogenesis. World J. Gastroenterol. 2004, 10, 3409–3413.
- Brady, G.; MacArthur, G.J.; Farrell, P.J. Epstein-Barr virus and Burkitt lymphoma. J. Clin. Pathol. 2007, 60, 1397–1402.
- Strong, M.J.; O’Grady, T.; Lin, Z.; Xu, G.; Baddoo, M.; Parsons, C.; Zhang, K.; Taylor, C.M.; Flemington, E.K. Epstein-Barr virus and human herpesvirus 6 detection in a non-Hodgkin’s diffuse large B-cell lymphoma cohort by using RNA sequencing. J. Virol. 2013, 87, 13059–13062.
- Nakhoul, H.; Lin, Z.; Wang, X.; Roberts, C.; Dong, Y.; Flemington, E. High-Throughput Sequence Analysis of Peripheral T-Cell Lymphomas Indicates Subtype-Specific Viral Gene Expression Patterns and Immune Cell Microenvironments. mSphere 2019, 4, e00248-19.
- Jalouli, J.; Jalouli, M.M.; Sapkota, D.; Ibrahim, S.O.; Larsson, P.A.; Sand, L. Human papilloma virus, herpes simplex virus and epstein barr virus in oral squamous cell carcinoma from eight different countries. Anticancer Res. 2012, 32, 571–580.
- Goncalves, P.H.; Montezuma-Rusca, J.M.; Yarchoan, R.; Uldrick, T.S. Cancer prevention in HIV-infected populations. Semin. Oncol. 2016, 43, 173–188.
- Narayanan, M.; Szymanski, J.; Slavcheva, E.; Rao, A.; Kelly, A.; Jones, K.; Jaffers, G. BK virus associated renal cell carcinoma: Case presentation with optimized PCR and other diagnostic tests. Am. J. Transplant. 2007, 7, 1666–1671.
- Abend, J.R.; Jiang, M.; Imperiale, M.J. BK virus and human cancer: Innocent until proven guilty. Semin. Cancer Biol. 2009, 19, 252–260.
- Shoraka, H.R.; Abobakri, O.; Naghibzade Tahami, A.; Mollaei, H.R.; Bagherinajad, Z.; Malekpour Afshar, R.; Shahesmaeili, A. Prevalence of JC and BK viruses in Patients with Colorectal Cancer: A Systematic Review and Meta- Analysis. Asian Pac. J. Cancer Prev. 2020, 21, 1499–1509.
- Coelho, T.R.; Gaspar, R.; Figueiredo, P.; Mendonça, C.; Lazo, P.A.; Almeida, L. Human JC polyomavirus in normal colorectal mucosa, hyperplastic polyps, sporadic adenomas, and adenocarcinomas in Portugal. J. Med. Virol. 2013, 85, 2119–2127.
- Hori, R.; Murai, Y.; Tsuneyama, K.; Abdel-Aziz, H.O.; Nomoto, K.; Takahashi, H.; Cheng, C.M.; Kuchina, T.; Harman, B.V.; Takano, Y. Detection of JC virus DNA sequences in colorectal cancers in Japan. Virchows Arch. 2005, 447, 723–730.
- Laghi, L.; Randolph, A.E.; Chauhan, D.P.; Marra, G.; Major, E.O.; Neel, J.V.; Boland, C.R. JC virus DNA is present in the mucosa of the human colon and in colorectal cancers. Proc. Natl. Acad. Sci. USA 1999, 96, 7484–7489.
- Jung, W.T.; Li, M.S.; Goel, A.; Boland, C.R. JC virus T-antigen expression in sporadic adenomatous polyps of the colon. Cancer 2008, 112, 1028–1036.
- Hashida, Y.; Nakajima, K.; Nakajima, H.; Shiga, T.; Tanaka, M.; Murakami, M.; Matsuzaki, S.; Naganuma, S.; Kuroda, N.; Seki, Y.; et al. High load of Merkel cell polyomavirus DNA detected in the normal skin of Japanese patients with Merkel cell carcinoma. J. Clin. Virol. 2016, 82, 101–107.
- Moore, P.S.; Chang, Y. Why do viruses cause cancer? Highlights of the first century of human tumour virology. Nat. Rev. Cancer 2010, 10, 878–889.
- Jin, F.; Vajdic, C.M.; Law, M.; Amin, J.; van Leeuwen, M.; McGregor, S.; Poynten, I.M.; Templeton, D.J.; Grulich, A.E. Incidence and time trends of anal cancer among people living with HIV in Australia. AIDS 2019, 33, 1361–1368.
- Colón-López, V.; Shiels, M.S.; Machin, M.; Ortiz, A.P.; Strickler, H.; Castle, P.E.; Pfeiffer, R.M.; Engels, E.A. Anal Cancer Risk Among People with HIV Infection in the United States. J. Clin. Oncol. 2018, 36, 68–75.
- Grew, D.; Bitterman, D.; Leichman, C.G.; Leichman, L.; Sanfilippo, N.; Moore, H.G.; Du, K. HIV Infection Is Associated With Poor Outcomes for Patients With Anal Cancer in the Highly Active Antiretroviral Therapy Era. Dis. Colon Rectum 2015, 58, 1130–1136.
- Grulich, A.E.; van Leeuwen, M.T.; Falster, M.O.; Vajdic, C.M. Incidence of cancers in people with HIV/AIDS compared with immunosuppressed transplant recipients: A meta-analysis. Lancet 2007, 370, 59–67.
- Hernández-Ramírez, R.U.; Shiels, M.S.; Dubrow, R.; Engels, E.A. Cancer risk in HIV-infected people in the USA from 1996 to 2012: A population-based, registry-linkage study. Lancet HIV 2017, 4, e495–e504.
- Panfil, A.R.; Martinez, M.P.; Ratner, L.; Green, P.L. Human T-cell leukemia virus-associated malignancy. Curr. Opin. Virol. 2016, 20, 40–46.
- Giam, C.Z.; Semmes, O.J. HTLV-1 Infection and Adult T-Cell Leukemia/Lymphoma—A Tale of Two Proteins: Tax and HBZ. Viruses 2016, 8, 161.
- Zhou, Y.; Zhao, Y.; Li, B.; Huang, J.; Wu, L.; Xu, D.; Yang, J.; He, J. Hepatitis viruses infection and risk of intrahepatic cholangiocarcinoma: Evidence from a meta-analysis. BMC Cancer 2012, 12, 289.
- Su, F.H.; Le, T.N.; Muo, C.H.; Te, S.A.; Sung, F.C.; Yeh, C.C. Chronic Hepatitis B Virus Infection Associated with Increased Colorectal Cancer Risk in Taiwanese Population. Viruses 2020, 12, 97.
- Iloeje, U.H.; Yang, H.I.; Jen, C.L.; Su, J.; Wang, L.Y.; You, S.L.; Lu, S.N.; Chen, C.J. Risk of pancreatic cancer in chronic hepatitis B virus infection: Data from the REVEAL-HBV cohort study. Liver Int. 2010, 30, 423–429.
- Hassan, M.M.; Li, D.; El-Deeb, A.S.; Wolff, R.A.; Bondy, M.L.; Davila, M.; Abbruzzese, J.L. Association between hepatitis B virus and pancreatic cancer. J. Clin. Oncol. 2008, 26, 4557–4562.
- Tokita, H.; Murai, S.; Kamitsukasa, H.; Yagura, M.; Harada, H.; Takahashi, M.; Okamoto, H. High TT virus load as an independent factor associated with the occurrence of hepatocellular carcinoma among patients with hepatitis C virus-related chronic liver disease. J. Med. Virol. 2002, 67, 501–509.
- Schildgen, V.; Malecki, M.; Tillmann, R.L.; Brockmann, M.; Schildgen, O. The Human Bocavirus Is Associated with Some Lung and Colorectal Cancers and Persists in Solid Tumors. PLoS ONE 2013, 8, e68020.
- Höpken, M.; Förster, I.; Maune, S.; Brockmann, M.; Schildgen, O.; Schildgen, V. Association of the Human Bocavirus With Tonsil Squamous Cell Carcinomas. Front. Microbiol. 2018, 9, 2450.
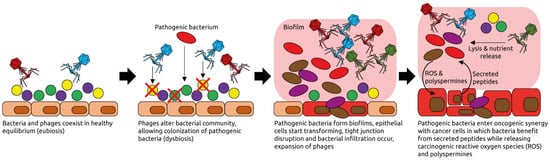
- Nakatsu, G.; Zhou, H.; Wu, W.K.K.; Wong, S.H.; Coker, O.O.; Dai, Z.; Li, X.; Szeto, C.H.; Sugimura, N.; Lam, T.Y.; et al. Alterations in Enteric Virome Are Associated with Colorectal Cancer and Survival Outcomes. Gastroenterology 2018, 155, 529–541.
- De Villiers, E.M.; Fauquet, C.; Broker, T.R.; Bernard, H.U.; zur Hausen, H. Classification of papillomaviruses. Virology 2004, 324, 17–27.
- Doorbar, J.; Egawa, N.; Griffin, H.; Kranjec, C.; Murakami, I. Human papillomavirus molecular biology and disease association. Rev. Med. Virol. 2015, 25 (Suppl. S1), 2–23.
- Buitrago-Pérez, A.; Garaulet, G.; Vázquez-Carballo, A.; Paramio, J.M.; García-Escudero, R. Molecular Signature of HPV-Induced Carcinogenesis: pRb, p53 and Gene Expression Profiling. Curr. Genomics. 2009, 10, 26–34.
- Lehoux, M.; D’Abramo, C.M.; Archambault, J. Molecular Mechanisms of Human Papillomavirus-Induced Carcinogenesis. Public Health Genom. 2009, 12, 268–280.
- Emlet, C.; Ruffin, M.; Lamendella, R. Enteric Virome and Carcinogenesis in the Gut. Dig. Dis. Sci. 2020, 65, 852–864.
- Braaten, K.P.; Laufer, M.R. Human Papillomavirus (HPV), HPV-Related Disease, and the HPV Vaccine. Rev. Obstet. Gynecol. 2008, 1, 2–10.
- Davison, A.J.; Eberle, R.; Ehlers, B.; Hayward, G.S.; McGeoch, D.J.; Minson, A.C.; Pellett, P.E.; Roizman, B.; Studdert, M.J.; Thiry, E. The order Herpesvirales. Arch. Virol. 2009, 154, 171–177.
- Van Zyl, D.G.; Mautner, J.; Delecluse, H.J. Progress in EBV Vaccines. Front. Oncol. 2019, 9, 104.
- Filippakis, H.; Spandidos, D.A.; Sourvinos, G. Herpesviruses: Hijacking the Ras signaling pathway. Biochim. Biophys. Acta 2010, 1803, 777–785.
- Cai, Q.; Xiao, B.; Si, H.; Cervini, A.; Gao, J.; Lu, J.; Upadhyay, S.K.; Verma, S.C.; Robertson, E.S. Kaposi’s sarcoma herpesvirus upregulates Aurora A expression to promote p53 phosphorylation and ubiquitylation. PLoS Pathog. 2012, 8, e1002566.
- Frazer, I.H. The actinic keratosis virome: Can we prevent squamous cell carcinoma with a vaccine? Curr. Probl. Dermatol. 2015, 46, 28–35.
- Cassler, N.M.; Merrill, D.; Bichakjian, C.K.; Brownell, I. Merkel Cell Carcinoma Therapeutic Update. Curr. Treat. Options Oncol. 2016, 17, 36.
- Harms, P.W.; Harms, K.L.; Moore, P.S.; DeCaprio, J.A.; Nghiem, P.; Wong, M.K.K.; Brownell, I.; International Workshop on Merkel Cell Carcinoma Research (IWMCC) Working Group. The biology and treatment of Merkel cell carcinoma: Current understanding and research priorities. Nat. Rev. Clin. Oncol. 2018, 15, 763–776.
- White, M.K.; Khalili, K. Polyomaviruses and human cancer: Molecular mechanisms underlying patterns of tumorigenesis. Virology 2004, 324, 1–16.
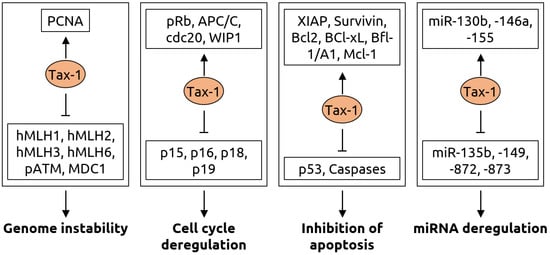
- Mohanty, S.; Harhaj, E.W. Mechanisms of Oncogenesis by HTLV-1 Tax. Pathogens 2020, 9, 543.
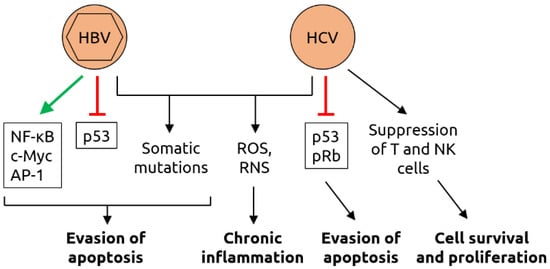
- Torresi, J.; Tran, B.M.; Christiansen, D.; Earnest-Silveira, L.; Schwab, R.H.M.; Vincan, E. HBV-related hepatocarcinogenesis: The role of signalling pathways and innovative ex vivo research models. BMC Cancer 2019, 19, 707.
- McGivern, D.R.; Lemon, S.M. Virus-specific mechanisms of carcinogenesis in hepatitis C virus associated liver cancer. Oncogene 2011, 30, 1969–1983.
- Karpiński, T.M. The Microbiota and Pancreatic Cancer. Gastroenterol. Clin. North Am. 2019, 48, 447–464.
- Matijašić, M.; Meštrović, T.; Paljetak, H.Č.; Perić, M.; Barešić, A.; Verbanac, D. Gut Microbiota beyond Bacteria-Mycobiome, Virome, Archaeome, and Eukaryotic Parasites in IBD. Int. J. Mol. Sci. 2020, 21, 2668.
- Gilbert, J.A.; Blaser, M.J.; Caporaso, J.G.; Jansson, J.K.; Lynch, S.V.; Knight, R. Current understanding of the human microbiome. Nat. Med. 2018, 24, 392–400.
- Carding, S.R.; Davis, N.; Hoyles, L. Review article: The human intestinal virome in health and disease. Aliment Pharmacol. Ther. 2017, 46, 800–815.
- Carding, S.; Verbeke, K.; Vipond, D.T.; Corfe, B.M.; Owen, L.J. Dysbiosis of the gut microbiota in disease. Microb. Ecol. Health Dis. 2015, 26, 26191.
- Tisza, M.J.; Buck, C.B. A catalog of tens of thousands of viruses from human metagenomes reveals hidden associations with chronic diseases. Proc. Natl. Acad. Sci. USA 2021, 118, e2023202118.
- Suttle, C.A. Marine viruses—Major players in the global ecosystem. Nat. Rev. Microbiol. 2007, 5, 801–812.
- Roux, S.; Hallam, S.J.; Woyke, T.; Sullivan, M.B. Viral dark matter and virus-host interactions resolved from publicly available microbial genomes. eLife 2015, 4, e08490.
- Hannigan, G.D.; Duhaime, M.B.; Ruffin, M.T., 4th; Koumpouras, C.C.; Schloss, P.D. Diagnostic Potential and Interactive Dynamics of the Colorectal Cancer Virome. mBio 2018, 9, e02248-18.
- Handley, S.A.; Devkota, S. Going Viral: A Novel Role for Bacteriophage in Colorectal Cancer. mBio 2019, 10, e02626-18.
- Flynn, K.J.; Baxter, N.T.; Schloss, P.D. Metabolic and Community Synergy of Oral Bacteria in Colorectal Cancer. mSphere 2016, 1, e00102-16.
- Norman, J.M.; Handley, S.A.; Baldridge, M.T.; Droit, L.; Liu, C.Y.; Keller, B.C.; Kambal, A.; Monaco, C.L.; Zhao, G.; Fleshner, P.; et al. Disease-specific alterations in the enteric virome in inflammatory bowel disease. Cell 2015, 160, 447–460.





