
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Adrian Tiron | + 2483 word(s) | 2483 | 2021-09-30 08:41:09 | | | |
| 2 | Catherine Yang | Meta information modification | 2483 | 2021-12-10 07:25:50 | | |
Video Upload Options
Hyperoxia effects on cancer biology were explored following multiple pathways, using both in vitro cancer cell cultures and in vivo tumoral animal models. Experimental data support clinical evidences demonstrating that hyperoxia, mainly if prolonged, can induce lung injury and cerebral damage, and this can be counteracted by down-modulation of Akt, by low-dose vitamin D or aspirin.
1. Background
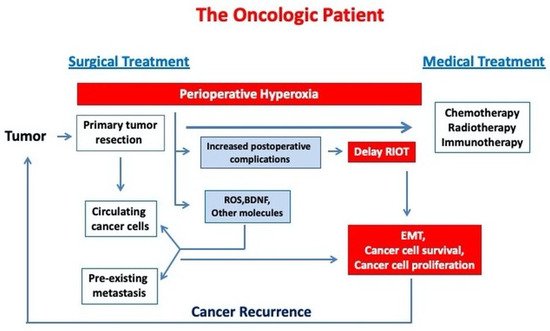
Despite the development of a more targeted and personalised multimodal treatment, cancer mortality remains high. In the USA, during 2020, a COVID-19 pandemic year, cancer was responsible for almost 600,000 casualties, representing the second leading cause of death after heart disease [1]. The main cause of death is cancer progression, the occurrence of distal metastasis or cancer recurrence, as the vast majority of patients are now successfully treated for primary tumours, with 64% of them having more than 5 years survival [2].
In cancer patients receiving general anaesthesia for surgery, increased inspiratory oxygen concentration is commonly used both intraoperatively and, for a variable time period, postoperatively, in order to prevent the potential development of hypoxemia and to ensure an adequate oxygen transport to the organs, tissues and cells. For a long time considered as a useful and harmless intervention, oxygen therapy has been under scrutiny in recent years due to the accumulation of data regarding its deleterious effects in critically ill patients [3] and many areas of acute medicine (stroke, acute myocardial infarction, cardiac arrest) [4][5][6][7][8]. In critically ill patient care, current data does not support a benefit from supranormal oxygen delivery. A meta-analysis of 19 clinical studies showed that hyperoxaemia was associated with increased in-hospital mortality [9]. A large observational multicentric study, collecting almost 300,000 arterial blood gas analysis from more than 14,000 critically ill patients, demonstrated a linear relationship between hyperoxemia and mortality, with both duration and severity adversely affecting the outcome [10]. A recent systematic review of 16,000 critically ill patients suggested that hyperoxia exposure might be associated with increased hospital mortality [11]. The British clinical practice guideline for oxygen therapy in acutely ill medical patients recommends to stop supplemental oxygen therapy when SpO 2 reaches 96% and not to start oxygen therapy at or above 93% oxygen saturation in patients with acute stroke or myocardial infarction (strong recommendations) [12].
During the perioperative care of an oncologic patient, the anaesthetist may currently choose between different types of anaesthesia techniques (general anaesthesia versus regional or neuraxial anaesthesia) and can also choose between various anaesthetic and analgesic agents (inhalation versus intravenous anaesthesia). However, oxygen, the most commonly used perioperative drug, cannot be replaced. The optimal dose and duration for oxygen therapy in surgical cancer patients is not clearly established.
2. Potential Molecular Mechanisms Exploring Hyperoxia Effects on Cancer Progression
2.1. ROS Production and Oxidative Stress
2.2. Hyperoxia and the Immune System
2.3. Angiogenesis and Epithelial Mesenchymal Transition (EMT)
2.4. Brain-Derived Neurotrophic Factor (BDNF)
2.5. Hyperoxic-Hypoxic Paradox
References
- Ahmad, F.B.; Anderson, R.N. The Leading Causes of Death in the US for 2020. JAMA 2021, 325, 1829.
- American Cancer Society. Cancer Treatment & Survivorship, Facts & Figures 2019–2021; American Cancer Society: Atlanta, GA, USA, 2019.
- Damiani, E.; Adrario, E.; Girardis, M.; Romano, R.; Pelaia, P.; Singer, M.; Donati, A. Arterial hyperoxia and mortality in critically ill patients: A systematic review and meta-analysis. Crit. Care 2014, 18, 711.
- Ibanez, B.; James, S.; Agewall, S.; Antunes, M.J.; Bucciarelli-Ducci, C.; Bueno, H.; Caforio, A.L.P.; Crea, F.; Goudevenos, J.A.; Halvorsen, S.; et al. 2017 ESC Guidelines for the management of acute myocardial infarction in patients presenting with ST-segment elevation. Eur. Heart J. 2017, 39, 119–177.
- Writing Committee Members; O’Gara, P.T.; Kushner, F.G.; Ascheim, D.D.; Casey, D.E.; Chung, M.K.; De Lemos, J.A.; Ettinger, S.M.; Fang, J.C.; Fesmire, F.M.; et al. 2013 ACCF/AHA Guideline for the Management of ST-Elevation Myocardial Infarction. Circulation 2013, 127, e362–e425.
- Powers, W.J.; Rabinstein, A.A.; Ackerson, T.; Adeoye, O.M.; Bambakidis, N.C.; Becker, K.; Biller, J.; Brown, M.; Demaerschalk, B.M.; Hoh, B.; et al. 2018 Guidelines for the Early Management of Patients with Acute Ischemic Stroke: A Guideline for Healthcare Professionals from the American Heart Association/American Stroke Association. Stroke 2018, 49, e46–e99.
- Kobayashi, A.; Czlonkowska, A.; Ford, G.A.; Fonseca, A.C.; Luijckx, G.J.; Korv, J.; de la Ossa, N.P.; Price, C.; Russell, D.; Tsiskaridze, A.; et al. European Academy of Neurology and European Stroke Organization consensus statement and practical guidance for pre-hospital management of stroke. Eur. J. Neurol. 2017, 25, 425–433.
- Elmer, J.; Scutella, M.; Pullalarevu, R.; Wang, B.; Vaghasia, N.; Trzeciak, S.; Rosario-Rivera, B.L.; Guyette, F.; Rittenberger, J.C.; Dezfulian, C.; et al. The association between hyperoxia and patient outcomes after cardiac arrest: Analysis of a high-resolution database. Intensive Care Med. 2014, 41, 49–57.
- Helmerhorst, H.J.; Roos-Blom, M.J.; van Westerloo, D.J.; de Jonge, E. Association between arterial hyperoxia and outcome in subsets of critical illness: A systematic review, meta- analysis, and meta-regression of cohort studies. Crit. Care Med. 2015, 43, 1508–1519.
- Helmerhorst, H.J.F.; Arts, D.; Schultz, M.J.; Van Der Voort, P.H.J.; Abu-Hanna, A.; De Jonge, E.; Van Westerloo, D.J. Metrics of Arterial Hyperoxia and Associated Outcomes in Critical Care. Crit. Care Med. 2017, 45, 187–195.
- Chu, D.; Kim, L.H.-Y.; Young, P.; Zamiri, N.; A Almenawer, S.; Jaeschke, R.; Szczeklik, W.; Schünemann, H.J.; Neary, J.D.; Alhazzani, W. Mortality and morbidity in acutely ill adults treated with liberal versus conservative oxygen therapy (IOTA): A systematic review and meta-analysis. Lancet 2018, 391, 1693–1705.
- Siemieniuk, R.A.C.; Chu, D.; Kim, L.H.-Y.; Güell-Rous, M.-R.; Alhazzani, W.; Soccal, P.M.; Karanicolas, P.J.; Farhoumand, P.D.; Siemieniuk, J.L.K.; Satia, I.; et al. Oxygen therapy for acutely ill medical patients: A clinical practice guideline. BMJ 2018, 363, k4169.
- Helmerhorst, H.J.F.; Schultz, M.J.; van der Voort, P.H.J.; de Jonge, E.; van Westerloo, D.J. Bench-to-bedside review: The effects of hyperoxia during critical illness. Crit. Care 2015, 19, 284.
- Ottolenghi, S.; Rubino, F.M.; Sabbatini, G.; Coppola, S.; Veronese, A.; Chiumello, D.; Paroni, R. Oxidative Stress Markers to Investigate the Effects of Hyperoxia in Anesthesia. Int. J. Mol. Sci. 2019, 20, 5492.
- Semenza, G.L. Defining the role of hypoxia-inducible factor 1 in cancer biology and therapeutics. Oncogene 2009, 29, 625–634.
- Tavare, A.N.; Perry, N.J.; Benzonana, L.L.; Takata, M.; Ma, D. Cancer recurrence after surgery: Direct and indirect effects of anesthetic agents. Int. J. Cancer 2011, 130, 1237–1250.
- Sun, S.; Lee, D.; Lee, N.P.; Pu, J.K.S.; Wong, T.S.; Lui, W.M.; Fung, C.F.; Leung, G.K.K. Hyperoxia resensitizes chemoresistant human glioblastoma cells to temozolomide. J. Neuro-Oncol. 2012, 109, 467–475.
- Wang, P.; Wan, W.; Xiong, S.; Wang, J.; Zou, D.; Lan, C.; Yu, S.; Liao, B.; Feng, H.; Wu, N. HIF1α regulates glioma chemosensitivity through the transformation between differentiation and dedifferentiation in various oxygen levels. Sci. Rep. 2017, 7, 7965.
- De Bels, D.; Tillmans, F.; Corazza, F.; Bizzarri, M.; Germonpre, P.; Radermacher, P.; Orman, K.G.; Balestra, C. Hyperoxia Alters Ultrastructure and Induces Apoptosis in Leukemia Cell Lines. Biomolecules 2020, 10, 282.
- Raa, A.; Stansberg, C.; Steen, V.M.; Bjerkvig, R.; Reed, R.K.; Stuhr, L.E.B. Hyperoxia retards growth and induces apoptosis and loss of glands and blood vessels in DMBA-induced rat mammary tumors. BMC Cancer 2007, 7, 23.
- Moen, I.; Øyan, A.M.; Kalland, K.-H.; Tronstad, K.J.; Akslen, L.A.; Chekenya, M.; Sakariassen, P.; Reed, R.K.; Stuhr, L.E.B. Hyperoxic Treatment Induces Mesenchymal-to-Epithelial Transition in a Rat Adenocarcinoma Model. PLoS ONE 2009, 4, e6381.
- Qian, X.; Zhang, Q.; Shao, N.; Shan, Z.; Cheang, T.; Zhang, Z.; Su, Q.; Wang, S.; Lin, Y. Respiratory hyperoxia reverses immunosuppression by regulating myeloid-derived suppressor cells and PD-L1 expression in a triple-negative breast cancer mouse model. Am. J. Cancer Res. 2019, 9, 529–545.
- Yamamoto, N.; Oyaizu, T.; Enomoto, M.; Horie, M.; Yuasa, M.; Okawa, A.; Yagishita, K. VEGF and bFGF induction by nitric oxide is associated with hyperbaric oxygen-induced angiogenesis and muscle regeneration. Sci. Rep. 2020, 10, 2744.
- Huang, X.; Liang, P.; Jiang, B.; Zhang, P.; Yu, W.; Duan, M.; Guo, L.; Cui, X.; Huang, M.; Huang, X. Hyperbaric oxygen potentiates diabetic wound healing by promoting fibroblast cell proliferation and endothelial cell angiogenesis. Life Sci. 2020, 259, 118246.
- Chang, H.-C.; Yang, Y.-R.; Wang, R.-Y. Effects of repetitive hyperbaric oxygen therapy on neuroprotection in middle cerebral artery occlusion rats. Brain Res. 2020, 1748, 147097.
- Hatfield, S.M.; Kjaergaard, J.; Lukashev, D.; Schreiber, T.; Belikoff, B.; Abbott, R.; Sethumadhavan, S.; Philbrook, P.; Ko, K.; Cannici, R.; et al. Immunological mechanisms of the antitumor effects of supplemental oxygenation. Sci. Transl. Med. 2015, 7, 277ra30.
- Kumar, V.H.S.; Wang, H.; Nielsen, L. Adaptive immune responses are altered in adult mice following neonatal hyperoxia. Physiol. Rep. 2018, 6, e13577.
- Kiers, D.; Gerretsen, J.; Janssen, E.; John, A.; Groeneveld, R.; Van Der Hoeven, J.G.; Scheffer, G.-J.; Pickkers, P.; Kox, M. Short-term hyperoxia does not exert immunologic effects during experimental murine and human endotoxemia. Sci. Rep. 2015, 5, 17441.
- Ash, S.A.; Valchev, G.I.; Looney, M.; Ni Mhathuna, A.; Crowley, P.D.; Gallagher, H.C.; Buggy, D.J. Xenon decreases cell migration and secretion of a pro-angiogenesis factor in breast adenocarcinoma cells: Comparison with sevoflurane. Br. J. Anaesth. 2014, 113, i14–i21.
- Crowley, P.D.; Stuttgen, V.; O’Carroll, E.; A Ash, S.; Buggy, D.J.; Gallagher, H.C. Exposure to 60% oxygen promotes migration and upregulates angiogenesis factor secretion in breast cancer cells. Med. Gas Res. 2017, 7, 226–235.
- Dongre, A.; Weinberg, R.A. New insights into the mechanisms of epithelial–mesenchymal transition and implications for cancer. Nat. Rev. Mol. Cell Biol. 2018, 20, 69–84.
- Aiello, N.; Kang, Y. Context-dependent EMT programs in cancer metastasis. J. Exp. Med. 2019, 216, 1016–1026.
- Tadokoro, A.; Kanaji, N.; Liu, D.; Yokomise, H.; Haba, R.; Ishii, T.; Takagi, T.; Watanabe, N.; Kita, N.; Kadowaki, N.; et al. Vimentin Regulates Invasiveness and Is a Poor Prognostic Marker in Non-small Cell Lung Cancer. Anticancer Res. 2016, 36, 1545–1551.
- Du, L.; Li, J.; Lei, L.; He, H.; Chen, E.; Dong, J.; Yang, J. High Vimentin Expression Predicts a Poor Prognosis and Progression in Colorectal Cancer: A Study with Meta-Analysis and TCGA Database. BioMed Res. Int. 2018, 2018, 6387810.
- Chaw, S.Y.; Majeed, A.A.; Dalley, A.J.; Chan, A.; Stein, S.; Farah, C.S. Epithelial to mesenchymal transition EMT) biomarkers–E-cadherin, beta-catenin, APC and Vimentin–in oral squamous cell carcinogenesis and transformation. Oral Oncol. 2012, 48, 997–1006.
- Tiron, A.; Ristescu, I.; Postu, P.A.; Tiron, C.E.; Zugun-Eloae, F.; Grigoras, I. Long-Term Deleterious Effects of Short-term Hyperoxia on Cancer Progression—Is Brain-Derived Neurotrophic Factor an Important Mediator? An Experimental Study. Cancers 2020, 12, 688.
- Yao, Q.; Haxhiu, M.A.; Zaidi, S.I.; Liu, S.; Jafri, A.; Martin, R.J. Hyperoxia enhances brain-derived neurotrophic factor and tyrosine kinase B receptor expression in peribronchial smooth muscle of neonatal rats. Am. J. Physiol. Cell. Mol. Physiol. 2005, 289, L307–L314.
- Choi, J.; Kwon, H.-J.; Lee, J.-E.; Lee, Y.; Seoh, J.-Y.; Han, P.-L. Hyperoxygenation revitalizes Alzheimer’s disease pathology through the upregulation of neurotrophic factors. Aging Cell 2019, 18, e12888.
- Kujawski, S.; Słomko, J.; Morten, K.J.; Murovska, M.; Buszko, K.; Newton, J.L.; Zalewski, P. Autonomic and Cognitive Function Response to Normobaric Hyperoxia Exposure in Healthy Subjects. Preliminary Study. Medicina 2020, 56, 172.
- Paris, A.J.; Hayer, K.E.; Oved, J.H.; Avgousti, D.C.; Toulmin, S.A.; Zepp, J.A.; Zacharias, W.J.; Katzen, J.B.; Basil, M.C.; Kremp, M.M.; et al. STAT3–BDNF–TrkB signalling promotes alveolar epithelial regeneration after lung injury. Nat. Cell Biol. 2020, 22, 1197–1210.
- Lee, B.; Cao, R.; Choi, Y.-S.; Cho, H.-Y.; Rhee, A.D.; Hah, C.K.; Hoyt, K.; Obrietan, K. The CREB/CRE transcriptional pathway: Protection against oxidative stress-mediated neuronal cell death. J. Neurochem. 2009, 108, 1251–1265.
- Yoo, J.-M.; Lee, B.D.; Sok, D.-E.; Ma, J.Y.; Kim, M.R. Neuroprotective action of N-acetyl serotonin in oxidative stress-induced apoptosis through the activation of both TrkB/CREB/BDNF pathway and Akt/Nrf2/Antioxidant enzyme in neuronal cells. Redox Biol. 2017, 11, 592–599.
- Hacioglu, G.; Senturk, A.; Ince, I.; Alver, A. Assessment of oxidative stress parameters of brain-derived neurotrophic factor heterozygous mice in acute stress model. Iran. J. Basic Med. Sci. 2016, 19, 388–393.
- Jiang, J.M.; Zhou, C.F.; Gao, S.L.; Tian, Y.; Wang, C.Y.; Wang, L.; Gu, H.F.; Tang, X.Q. BDNF-TrkB pathway mediates neuroprotection of hydrogen sulfide against formaldehyde-induced toxicity to PC12 cells. PLoS ONE 2015, 10, e0119478.
- Wu, C.L.; Chen, S.D.; Yin, J.H.; Hwang, C.S.; Yang, D.I. Nuclear factor-kappaB-dependent sestrin2 induction mediates the antioxidant effects of BDNF against mitochondrial inhibition in rat cortical neurons. Mol. Neurobiol. 2016, 53, 4126–4142.
- Boyadjieva, N.I.; Sarkar, D.K. Cyclic Adenosine Monophosphate and Brain-Derived Neurotrophic Factor Decreased Oxidative Stress and Apoptosis in Developing Hypothalamic Neuronal Cells: Role of Microglia. Alcohol. Clin. Exp. Res. 2013, 37, 1370–1379.
- Huang, Y.Z.; McNamara, J.O. Neuroprotective Effects of Reactive Oxygen Species Mediated by BDNF-Independent Activation of TrkB. J. Neurosci. 2012, 32, 15521–15532.
- Usui, T.; Naruo, A.; Okada, M.; Hayabe, Y.; Yamawaki, H. Brain-derived neurotrophic factor promotes angiogenic tube formation through generation of oxidative stress in human vascular endothelial cells. Acta Physiol. 2014, 211, 385–394.
- Song, X.; Zhou, B.; Zhang, P.; Lei, D.; Wang, Y.; Yao, G.; Hayashi, T.; Xia, M.; Tashiro, S.-I.; Onodera, S.; et al. Protective Effect of Silibinin on Learning and Memory Impairment in LPS-Treated Rats via ROS–BDNF–TrkB Pathway. Neurochem. Res. 2016, 41, 1662–1672.
- Jia, S.; Wang, W.; Hu, Z.; Shan, C.; Wang, L.; Wu, B.; Yang, Z.; Yang, X.; Lei, D. BDNF mediated TrkB activation contributes to the EMT progression and the poor prognosis in human salivary adenoid cystic carcinoma. Oral Oncol. 2015, 51, 64–70.
- Kim, M.S.; Lee, W.S.; Jeong, J.; Kim, S.J.; Jin, W. Induction of metastatic potential by TrkB via activation of IL6/JAK2/STAT3 and PI3K/AKT signaling in breast cancer. Oncotarget 2015, 6, 40158.
- Kawamura, K.; Kawamura, N.; Okamoto, N.; Manabe, M. Suppression of choriocarcinoma invasion and metastasis following blockade of BDNF /TrkB signaling. Cancer Med. 2013, 2, 849–861.
- Götz, R.; Sendtner, M. Cooperation of Tyrosine Kinase Receptor TrkB and Epidermal Growth Factor Receptor Signaling Enhances Migration and Dispersal of Lung Tumor Cells. PLoS ONE 2014, 9, e100944.
- Tanaka, K.; Okugawa, Y.; Toiyama, Y.; Inoue, Y.; Saigusa, S.; Kawamura, M.; Araki, T.; Uchida, K.; Mohri, Y.; Kusunoki, M. Brain-Derived Neurotrophic Factor (BDNF)-Induced Tropomyosin-Related Kinase B (Trk B) Signaling Is a Potential Therapeutic Target for Peritoneal Carcinomatosis Arising from Colorectal Cancer. PLoS ONE 2014, 9, e96410.
- Polakowski, N.; Terol, M.J.; Hoang, K.; Nash, I.; Laverdure, S.; Gazon, H.; Belrose, G.; Mesnard, J.-M.; Césaire, R.; Peloponese, J.-M.; et al. HBZ Stimulates Brain-Derived Neurotrophic Factor/TrkB Autocrine/Paracrine Signaling To Promote Survival of Human T-Cell Leukemia Virus Type 1-Infected T Cells. J. Virol. 2014, 88, 13482–13494.
- Lin, C.-Y.; Hung, S.-Y.; Chen, H.-T.; Tsou, H.-K.; Fong, Y.-C.; Wang, S.-W.; Tang, C.-H. Brain-derived neurotrophic factor increases vascular endothelial growth factor expression and enhances angiogenesis in human chondrosarcoma cells. Biochem. Pharmacol. 2014, 91, 522–533.
- Huang, S.-M.; Lin, C.; Lin, H.-Y.; Chiu, C.-M.; Fang, C.-W.; Liao, K.-F.; Chen, D.-R.; Yeh, W.-L. Brain-derived neurotrophic factor regulates cell motility in human colon cancer. Endocr.-Relat. Cancer 2015, 22, 455–464.
- Nakamura, K.; Martin, K.C.; Jackson, J.K.; Beppu, K.; Woo, C.-W.; Thiele, C.J. Brain-Derived Neurotrophic Factor Activation of TrkB Induces Vascular Endothelial Growth Factor Expression via Hypoxia-Inducible Factor-1α in Neuroblastoma Cells. Cancer Res. 2006, 66, 4249–4255.
- Terraneo, L.; Virgili, E.; Caretti, A.; Bianciardi, P.; Samaja, M. In vivo hyperoxia induces hypoxia-inducible factor-1α overexpression in LNCaP tumors without affecting the tumor growth rate. Int. J. Biochem. Cell Biol. 2014, 51, 65–74.
- Smit, M.A.; Geiger, T.R.; Song, J.Y.; Gitelman, I.; Peeper, D.S. A Twist-Snail axis critical for TrkB-induced epithelial-mesenchymal transition-like transformation, anoikis resistance, and metastasis. Mol. Cell Biol. 2009, 29, 3722–3737.
- Bao, W.; Qiu, H.; Yang, T.; Luo, X.; Zhang, H.; Wan, X. Upregulation of TrkB Promotes Epithelial-Mesenchymal Transition and Anoikis Resistance in Endometrial Carcinoma. PLoS ONE 2013, 8, e70616.
- Yuan, Y.; Ye, H.; Ren, Q. Proliferative role of BDNF/TrkB signaling is associated with anoikis resistance in cervical cancer. Oncol. Rep. 2018, 40, 621–634.
- Li, T.; Yu, Y.; Song, Y.; Li, X.; Lan, D.; Zhang, P.; Xiao, Y.; Xing, Y. Activation of BDNF/TrkB pathway promotes prostate cancer progression via induction of epithelial-mesenchymal transition and anoikis resistance. FASEB J. 2020, 34, 9087–9101.
- Parekh, A.; Das, S.; Parida, S.; Das, C.K.; Dutta, D.; Mallick, S.K.; Wu, P.-H.; Kumar, B.N.P.; Bharti, R.; Dey, G.; et al. Multi-nucleated cells use ROS to induce breast cancer chemo-resistance in vitro and in vivo. Oncogene 2018, 37, 4546–4561.
- Barker, H.E.; Paget, J.T.E.; Khan, A.; Harrington, K. The tumour microenvironment after radiotherapy: Mechanisms of resistance and recurrence. Nat. Rev. Cancer 2015, 15, 409–425.
- Jarosz-Biej, M.; Smolarczyk, R.; Cichoń, T.; Kułach, N. Tumor microenvironment as a “game changer” in cancer radiotherapy. Int. J. Mol. Sci. 2019, 20, 3212.
- Agliano, A.; Balarajah, G.; Ciobota, D.M.; Sidhu, J.; Clarke, P.; Jones, C.; Workman, P.; Leach, M.O.; Al-Saffar, N.M. Pediatric and adult glioblastoma radiosensitization induced by PI3K/mTOR inhibition causes early metabolic alterations detected by nuclear magnetic resonance spectroscopy. Oncotarget 2017, 8, 47969–47983.
- Meng, C.; He, Z.; Xing, D. Low-level laser therapy rescues dendrite atrophy via upregulating BDNF expression: Implications for Alzheimer’s disease. J. Neurosci. 2013, 33, 13505–13517.
- Cui, M.; Xiao, H.; Li, Y.; Dong, J.; Luo, D.; Li, H.; Feng, G.; Wang, H.; Fan, S. Total abdominal irradiation exposure impairs cognitive function involving miR-34a-5p/BDNF axis. Biochim. Biophys. Acta (BBA)-Mol. Basis Dis. 2017, 1863, 2333–2341.
- Amir, H.; Shai, E. The Hyperoxic-Hypoxic Paradox. Biomolecules 2020, 10, 958.





