
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Giammauro Berardi | + 2169 word(s) | 2169 | 2021-11-30 02:50:55 | | | |
| 2 | Peter Tang | Meta information modification | 2169 | 2021-12-09 02:45:43 | | |
Video Upload Options
Liver resection is recognized worldwide as a potentially curative treatment for patients with primary and secondary malignancies and resectable disease. Preoperative 3D reconstructions and printing as well as augmented reality can increase the knowledge of the specific anatomy of the case and therefore plan the surgery accordingly and tailor the procedure on the patient. Furthermore, the indocyanine green retention dye is an increasingly used tool that can nowadays improve the precision during laparoscopic hepatectomies, especially when considering anatomical resection. The use of preoperative modern imaging and intraoperative indocyanine green dye are key to successfully perform complex hepatectomies such as laparoscopic parenchymal sparing liver resections.
1. Introduction
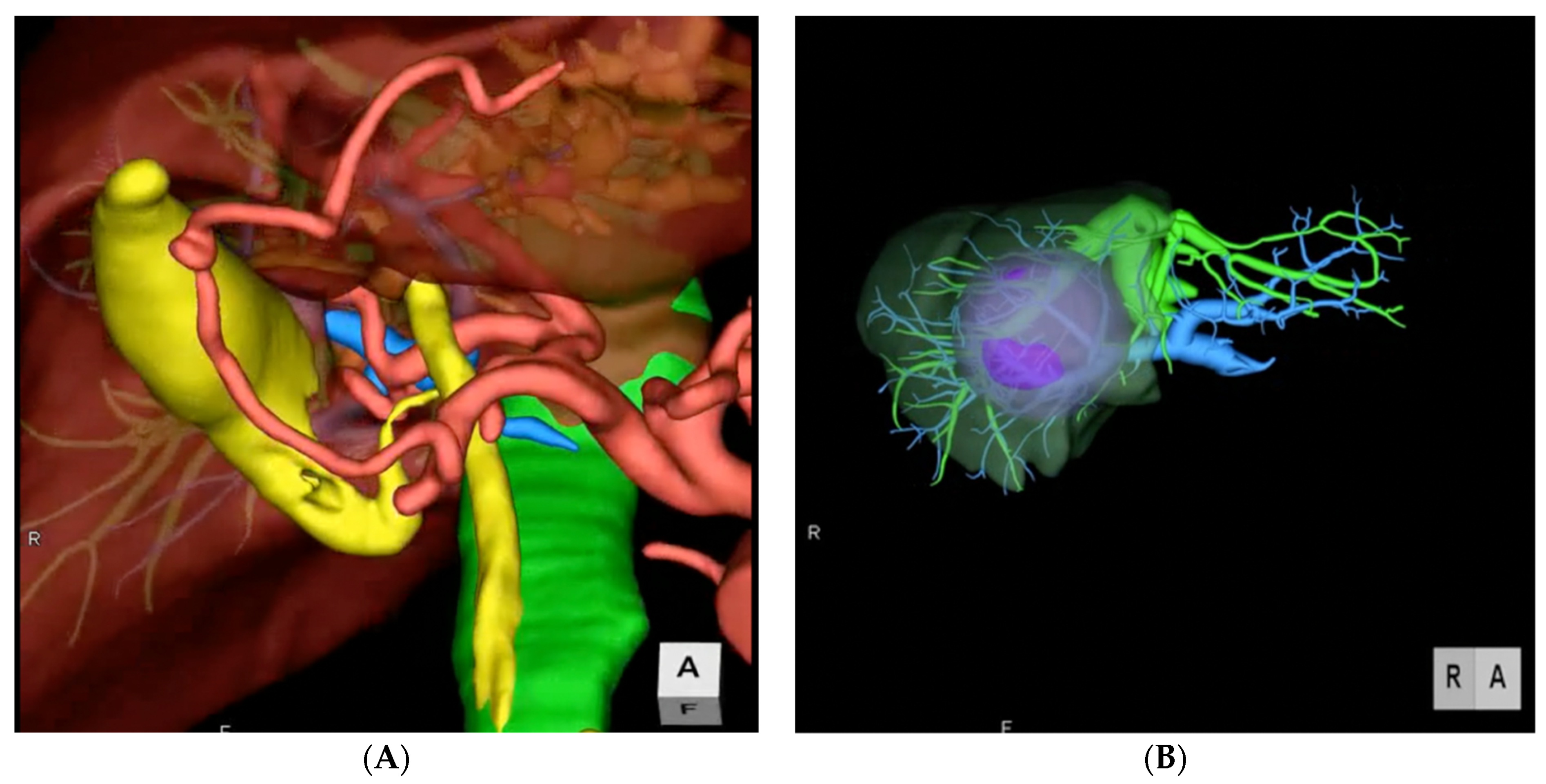
2. Preoperative Imaging
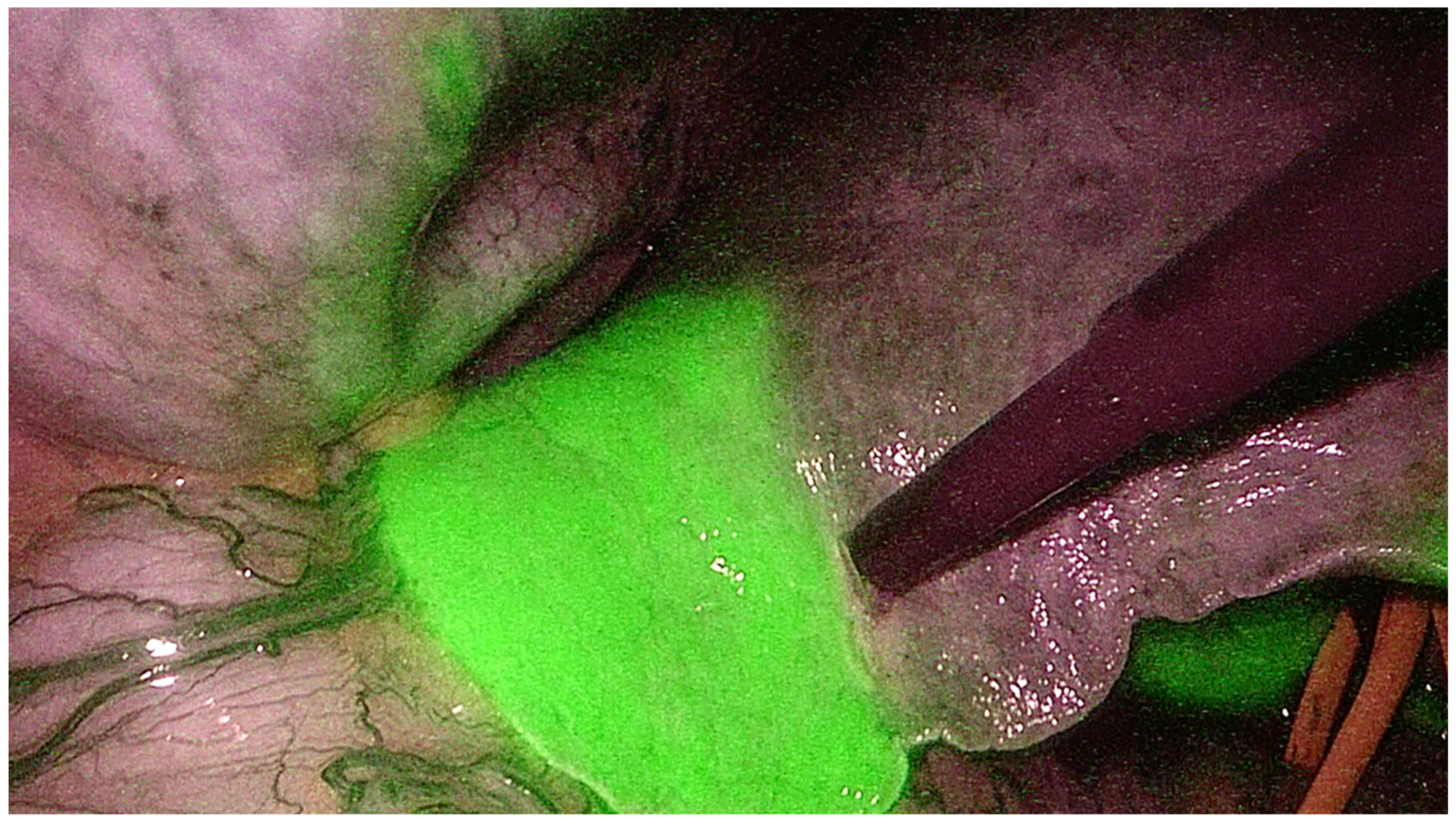
3. Indocyanine Green Dye Fluorescence
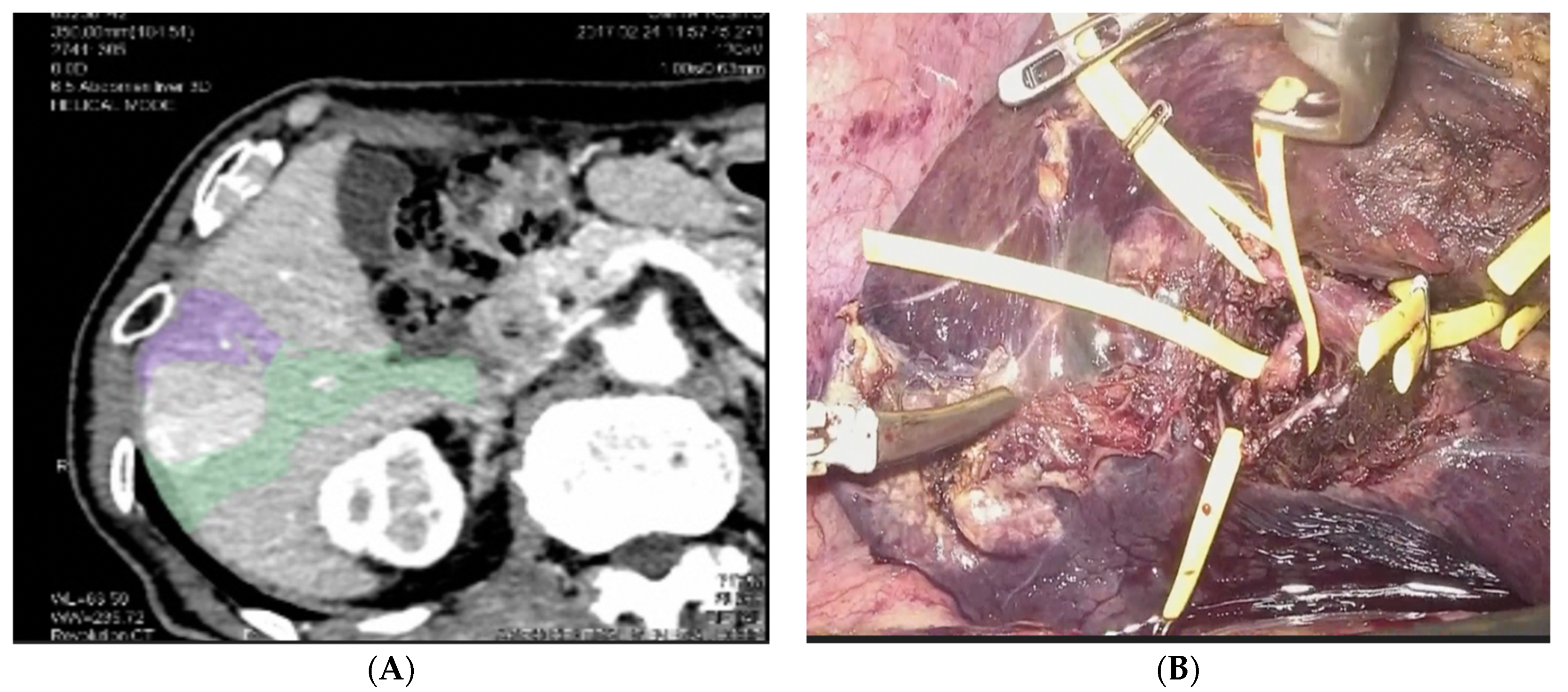
4. Laparoscopic Anatomical Parenchymal Sparing Liver Resections
References
- De Andrade, J.P.; Warner, S.G.; Fong, Y. Treatment of metastatic colorectal cancer: Innovations in surgical techniques. J. Surg. Oncol. 2019, 119, 653–659.
- European Association for the Study of the Liver. Electronic address eee, European Association for the Study of the L. EASL Clinical Practice Guidelines: Management of hepatocellular carcinoma. J. Hepatol. 2018, 69, 182–236.
- Berardi, G.; Antonelli, G.; Colasanti, M.; Meniconi, R.; Guglielmo, N.; Laurenzi, A.; Ferretti, S.; Sandri, G.B.L.; Spagnoli, A.; Moschetta, G.; et al. Association of Sarcopenia and Body Composition With Short-term Outcomes After Liver Resection for Malignant Tumors. JAMA Surg. 2020, 155, e203336.
- Berardi, G.; Morise, Z.; Sposito, C.; Igarashi, K.; Panetta, V.; Simonelli, I.; Kim, S.; Goh, B.K.; Kubo, S.; Tanaka, S.; et al. Development of a nomogram to predict outcome after liver resection for hepatocellular carcinoma in Child-Pugh B cirrhosis. J. Hepatol. 2020, 72, 75–84.
- Moris, D.; Tsilimigras, D.I.; Kostakis, I.D.; Ntanasis-Stathopoulos, I.; Shah, K.N.; Felekouras, E.; Pawlik, T.M. Anatomic versus non-anatomic resection for hepatocellular carcinoma: A systematic review and meta-analysis. Eur. J. Surg. Oncol. (EJSO) 2018, 44, 927–938.
- Mueller, M.; Breuer, E.; Mizuno, T.; Bartsch, F.; Ratti, F.; Benzing, C.; Ammar-Khodja, N.; Sugiura, T.; Takayashiki, T.; Hessheimer, A.; et al. Perihilar Cholangiocarcinoma–Novel Benchmark Values for Surgical and Oncological Outcomes From 24 Expert Centers. Ann. Surg. 2021, 274, 780–788.
- Sposito, C.; Busset, M.D.D.; Virdis, M.; Citterio, D.; Flores, M.; Bongini, M.; Niger, M.; Mazzaferro, V. The role of lymphadenectomy in the surgical treatment of intrahepatic cholangiocarcinoma: A review. Eur. J. Surg. Oncol. (EJSO) 2021.
- Wakabayashi, G.; Cherqui, D.; A Geller, D.; Buell, J.F.; Kaneko, H.; Han, H.S.; Asbun, H.; Oʼrourke, N.; Tanabe, M.; Koffron, A.J.; et al. Recommendations for laparoscopic liver resection: A report from the second international consensus conference held in Morioka. Ann. Surg. 2015, 261, 619–629.
- Ciria, R.; Cherqui, D.; Geller, D.A.; Briceno, J.; Wakabayashi, G. Comparative Short-term Benefits of Laparoscopic Liver Resection: 9000 Cases and Climbing. Ann. Surg. 2016, 263, 761–777.
- Cho, J.Y.; Han, H.-S.; Yoon, Y.-S.; Shin, S.-H. Feasibility of laparoscopic liver resection for tumors located in the posterosuperior segments of the liver, with a special reference to overcoming current limitations on tumor location. Surgery 2008, 144, 32–38.
- Cipriani, F.; Shelat, V.; Rawashdeh, M.; Francone, E.; Aldrighetti, L.; Takhar, A.; Armstrong, T.; Pearce, N.W.; Abu Hilal, M. Laparoscopic Parenchymal-Sparing Resections for Nonperipheral Liver Lesions, the Diamond Technique: Technical Aspects, Clinical Outcomes, and Oncologic Efficiency. J. Am. Coll. Surg. 2015, 221, 265–272.
- Coles, S.R.; Besselink, M.G.; Serin, K.R.; Alsaati, H.; Di Gioia, P.; Samim, M.; Pearce, N.W.; Abu Hilal, M. Total laparoscopic management of lesions involving liver segment 7. Surg. Endosc. 2015, 29, 3190–3195.
- Teo, J.Y.; Kam, J.H.; Chan, C.Y.; Goh, B.K.P.; Wong, J.-S.; Lee, V.T.W.; Cheow, P.C.; Chow, P.K.H.; Ooi, L.L.P.J.; Chung, A.Y.F.; et al. Laparoscopic liver resection for posterosuperior and anterolateral lesions—A comparison experience in an Asian centre. HepatoBiliary Surg. Nutr. 2015, 4, 379–390.
- Scuderi, V.; Barkhatov, L.; Montalti, R.; Ratti, F.; Cipriani, F.; Pardo, F.; Tranchart, H.; Dagher, I.; Rotellar, F.; Abu Hilal, M.; et al. Outcome after laparoscopic and open resections of posterosuperior segments of the liver. BJS 2017, 104, 751–759.
- Dagher, I.; Gayet, B.; Tzanis, D.; Tranchart, H.; Fuks, D.; Soubrane, O.; Han, H.-S.; Kim, K.-H.; Cherqui, D.; O’Rourke, N.; et al. International experience for laparoscopic major liver resection. J. Hepato-Biliary-Pancreat. Sci. 2014, 21, 732–736.
- Rossler, F.; Sapisochin, G.; Song, G.; Lin, Y.H.; Simpson, M.A.; Hasegawa, K.; Laurenzi, A.; Cabús, S.S.; Nunez, M.I.; Gatti, A. Defining Benchmarks for Major Liver Surgery: A multicenter Analysis of 5202 Living Liver Donors. Ann. Surg. 2016, 264, 492–500.
- Nicolau, S.; Soler, L.; Mutter, D.; Marescaux, J. Augmented reality in laparoscopic surgical oncology. Surg. Oncol. 2011, 20, 189–201.
- Gavriilidis, P.; Edwin, B.; Pelanis, E.; Hidalgo, E.; De’Angelis, N.; Memeo, R.; Aldrighetti, L.; Sutcliffe, R.P. Navigated liver surgery: State of the art and future perspectives. Hepatobiliary Pancreat. Dis. Int. 2021.
- Zeng, N.; Tao, H.; Fang, C.; Fan, Y.; Xiang, N.; Yang, J.; Zhu, W.; Liu, J.; Guan, T.; Fang, C.; et al. Individualized preoperative planning using three-dimensional modeling for Bismuth and Corlette type III hilar cholangiocarcinoma. World J. Surg. Oncol. 2016, 14, 44.
- Palomar, R.; Cheikh, F.A.; Edwin, B.; Fretland, Å.; Beghdadi, A.; Elle, O.J. A novel method for planning liver resections using deformable Bézier surfaces and distance maps. Comput. Methods Programs Biomed. 2017, 144, 135–145.
- Saito, Y.; Sugimoto, M.; Imura, S.; Morine, Y.; Ikemoto, T.; Iwahashi, S.; Yamada, S.; Shimada, M. Intraoperative 3D Hologram Support With Mixed Reality Techniques in Liver Surgery. Ann. Surg. 2020, 271, e4–e7.
- Boni, L.; David, G.; Mangano, A.; Dionigi, G.; Rausei, S.; Spampatti, S.; Cassinotti, E.; Fingerhut, A. Clinical applications of indocyanine green (ICG) enhanced fluorescence in laparoscopic surgery. Surg. Endosc. 2015, 29, 2046–2055.
- Alander, J.T.; Kaartinen, I.; Laakso, A.; Pätilä, T.; Spillmann, T.; Tuchin, V.; Venermo, M.; Välisuo, P. A Review of Indocyanine Green Fluorescent Imaging in Surgery. Int. J. Biomed. Imaging 2012, 2012, 1–26.
- Ishizawa, T.; Fukushima, N.; Shibahara, J.; Masuda, K.; Tamura, S.; Aoki, T.; Hasegawa, K.; Beck, Y.; Fukayama, M.; Kokudo, N. Real-time identification of liver cancers by using indocyanine green fluorescent imaging. Cancer 2009, 115, 2491–2504.
- Berardi, G.; Wakabayashi, G.; Igarashi, K.; Ozaki, T.; Toyota, N.; Tsuchiya, A.; Nishikawa, K. Full Laparoscopic Anatomical Segment 8 Resection for Hepatocellular Carcinoma Using the Glissonian Approach with Indocyanine Green Dye Fluorescence. Ann. Surg. Oncol. 2019, 26, 2577–2578.
- Esposito, C.; Coppola, V.; Del Conte, F.; Cerulo, M.; Esposito, G.; Farina, A.; Crocetto, F.; Castagnetti, M.; Settimi, A.; Escolino, M. Near-Infrared fluorescence imaging using indocyanine green (ICG): Emerging applications in pediatric urology. J. Pediatr. Urol. 2020, 16, 700–707.
- Esposito, C.; Settimi, A.; Del Conte, F.; Cerulo, M.; Coppola, V.; Farina, A.; Crocetto, F.; Ricciardi, E.; Esposito, G.; Escolino, M. Image-Guided Pediatric Surgery Using Indocyanine Green (ICG) Fluorescence in Laparoscopic and Robotic Surgery. Front. Pediatr. 2020, 8, 314.
- Wang, X.; Teh, C.S.C.; Ishizawa, T.; Aoki, T.; Cavallucci, D.; Lee, S.-Y.; Panganiban, K.M.; Perini, M.V.; Shah, S.R.; Wang, H.; et al. Consensus Guidelines for the Use of Fluorescence Imaging in Hepatobiliary Surgery. Ann. Surg. 2021, 274, 97–106.
- Kaibori, M.; Ishizaki, M.; Matsui, K.; Kwon, A.H. Intraoperative indocyanine green fluorescent imaging for prevention of bile leakage after hepatic resection. Surgery 2011, 150, 91–98.
- Felli, E.; Ishizawa, T.; Cherkaoui, Z.; Diana, M.; Tripon, S.; Baumert, T.F.; Schuster, C.; Pessaux, P. Laparoscopic anatomical liver resection for malignancies using positive or negative staining technique with intraoperative indocyanine green-fluorescence imaging. HPB 2021, 23, 1647–1655.
- Makuuchi, M.; Hasegawa, H.; Yamazaki, S. Ultrasonically guided subsegmentectomy. Surg. Gynecol. Obstet. 1985, 161, 346–350.
- Cho, A.; Asano, T.; Yamamoto, H.; Nagata, M.; Takiguchi, N.; Kainuma, O.; Souda, H.; Gunji, H.; Miyazaki, A.; Nojima, H.; et al. Laparoscopy-assisted hepatic lobectomy using hilar Glissonean pedicle transection. Surg. Endosc. 2007, 21, 1466–1468.
- Berardi, G.; Igarashi, K.; Li, C.J.; Ozaki, T.; Mishima, K.; Nakajima, K.; Honda, M.; Wakabayashi, G. Parenchymal Sparing Anatomical Liver Resections With Full Laparoscopic Approach: Description of Technique and Short-term Results. Ann. Surg. 2021, 273, 785–791.
- Wakabayashi, T.; Cacciaguerra, A.B.; Ciria, R.; Ariizumi, S.; Durán, M.; Golse, N.; Ogiso, S.; Abe, Y.; Aoki, T.; Hatano, E.; et al. Landmarks to identify segmental borders of the liver: A review prepared for PAM-HBP expert consensus meeting 2021. J. Hepato-Biliary-Pancreat. Sci. 2021.
- Billingsley, K.G.; Jarnagin, W.R.; Fong, Y.; Blumgart, L.H. Segment-oriented hepatic resection in the management of malignant neoplasms of the liver. J. Am. Coll. Surg. 1998, 187, 471–481.
- Hasegawa, K.; Kokudo, N.; Imamura, H.; Matsuyama, Y.; Aoki, T.; Minagawa, M.; Sano, K.; Sugawara, Y.; Takayama, T.; Makuuchi, M. Prognostic Impact of Anatomic Resection for Hepatocellular Carcinoma. Ann. Surg. 2005, 242, 252–259.
- Cho, J.Y.; Han, H.-S.; Choi, Y.; Yoon, Y.-S.; Kim, S.; Choi, J.K.; Jang, J.S.; Kwon, S.U.; Kim, H. Association of Remnant Liver Ischemia with Early Recurrence and Poor Survival After Liver Resection in Patients With Hepatocellular Carcinoma. JAMA Surg. 2017, 152, 386–392.
- Yamashita, S.; Venkatesan, A.M.; Mizuno, T.; Aloia, T.A.; Chun, Y.S.; Lee, J.E.; Vauthey, J.-N.; Conrad, C. Remnant Liver Ischemia as a Prognostic Factor for Cancer-Specific Survival After Resection of Colorectal Liver Metastases. JAMA Surg. 2017, 152, e172986.
- Couinaud, C.M. A simplified method for controlled left hepatectomy. Surgery 1985, 97, 358–361.
- Takasaki, K.; Kobayashi, S.; Tanaka, S.; Saito, A.; Yamamoto, M.; Hanyu, F. Highly anatomically systematized hepatic resection with Glissonean sheath code transection at the hepatic hilus. Int. Surg. 1990, 75, 73–77.
- Takasaki, K. Glissonean pedicle transection method for hepatic resection: A new concept of liver segmentation. J. Hepato-Biliary-Pancreat. Sci. 1998, 5, 286–291.
- Wakabayashi, G. From Louisville to Morioka: Where is now MILS? Updates Surg. 2015, 67, 101–104.





