Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Cristina Lanni | + 1172 word(s) | 1172 | 2021-11-29 10:01:53 | | | |
| 2 | Peter Tang | + 81 word(s) | 1253 | 2021-12-08 03:52:46 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Lanni, C. Pin1 involvement in Vascular Diseases. Encyclopedia. Available online: https://encyclopedia.pub/entry/16840 (accessed on 28 March 2026).
Lanni C. Pin1 involvement in Vascular Diseases. Encyclopedia. Available at: https://encyclopedia.pub/entry/16840. Accessed March 28, 2026.
Lanni, Cristina. "Pin1 involvement in Vascular Diseases" Encyclopedia, https://encyclopedia.pub/entry/16840 (accessed March 28, 2026).
Lanni, C. (2021, December 07). Pin1 involvement in Vascular Diseases. In Encyclopedia. https://encyclopedia.pub/entry/16840
Lanni, Cristina. "Pin1 involvement in Vascular Diseases." Encyclopedia. Web. 07 December, 2021.
Copy Citation
The vascular endothelium, the active inner layer of the blood vessel, releases a wide array of biologically active molecules acting in an autocrine or paracrine fashion, thereby controlling arterial structure and vasodilatory, thrombolytic, and vaso-protective functions. By controlling the change of the backbones of several cellular substrates, the peptidyl-prolyl cis-trans isomerase Pin1 acts as key fine-tuner and amplifier of multiple signaling pathways, thereby inducing several biological consequences, both in physiological and pathological conditions. Data from the literature indicate a prominent role of Pin1 in regulating vascular homeostasis.
Pin1
vascular endothelium
nitric oxide synthase
nitric oxide
vascular dementia
aging
1. Aging and the Vasculature: Focus on Endothelial Dysfunction
The vascular endothelium, the active inner layer of the blood vessel, releases a wide array of biologically active molecules acting in an autocrine or paracrine fashion, thereby controlling arterial structure and vasodilatory, thrombolytic, and vaso-protective functions. In particular, it regulates a number of biological functions, such as substrate exchange/transport, innate immunity, the regulation of vascular tone by balancing the production of vasodilators and vasoconstrictors, angiogenesis, and hemostasis by secreting antiplatelet and anticoagulant molecules (for a comprehensive review on the topic see [1]). Endothelial dysfunction occurs during aging [2], where an imbalance between vasodilator and vasoconstriction factors, a progressive reduction of nitric oxide (NO) bioavailability, as well as an increase in cyclooxygenase (COX)-derived vasoconstrictor factors have been reported [2][3]. In addition, a decreased expression and activity of endothelial NOS (eNOS) has been observed in older animals [4]. Moreover, endothelial dysfunction represents a risk factor for the development of several human vascular diseases, such as atherosclerosis, hypertension and stroke, peripheral arterial disease, metabolic syndrome (obesity, insulin resistance), and diabetes. The molecular mechanisms underpinning endothelial dysfunction are rather complex. Indeed, multiple mechanisms (i.e. impaired vasodilation, oxidative stress, inflammation, cell injury/death, senescence) have been reported to be involved. Notably, among them, decreased NO bioavailability represents a key hallmark of endothelial dysfunction. Thus, the severity of endothelial dysfunction has been shown to have prognostic value for cardiovascular events [5].
2. The Role of the cis-trans Isomerase Pin1 in Vascular Endothelium
Originally identified as a protein physically interacting with NIMA (never in mitosis A) mitotic kinase, Pin1 (protein interacting with NIMA-1) is a member of the evolutionarily conserved peptidyl-prolyl cis-trans isomerase (PPIase) family that, unlike all other known prolyl isomerases, mediates the isomerization of phosphorylated serine- and threonine-proline (pSer/Thr-Pro) motifs, mediating the conformational change of cellular substrates [6]. From a structural point of view, human Pin1 contains an N-terminal WW protein interaction domain (residues 1–39) and a C-terminal PPIase domain (residues 50–163), connected by a flexible linker [6] (Figure 1A). Notably, Pin1-catalyzed phosphorylation-dependent conformational changes of its substrates have been reported to control many key regulators involved in numerous cellular processes (e.g., cell growth, cell cycle progression, cellular stress response, development, apoptosis, neuronal differentiation, and immune response) [7]. In particular, it acts as a unique molecular timer regulating multiple targets at different steps of a given signaling pathway to synergistically orchestrate cellular responses [7].
Notably, Pin1 deregulation has been associated with a number of pathological conditions, ranging from cancers [8] to age-related and neurodegenerative diseases[9]. Pin1 has been found to act as a critical driver of vascular cell proliferation, apoptosis, and inflammation, with implication in many cardiovascular diseases (e.g., atherosclerosis, coronary restenosis, and cardiac hypertrophy) [10][11][12]. Notably, Pin1 has been reported to regulate the degradation of the inducible nitric oxide synthase (iNOS) in murine aortic endothelial cells upon induction by lipopolysaccharide and IFN-γ [13], as well as to interact with eNOS in a phosphorylation-dependent manner (Figure 1B) , thereby suppressing eNOS activity, similar to the tonic suppression of eNOS activity by caveolin-1 [14].
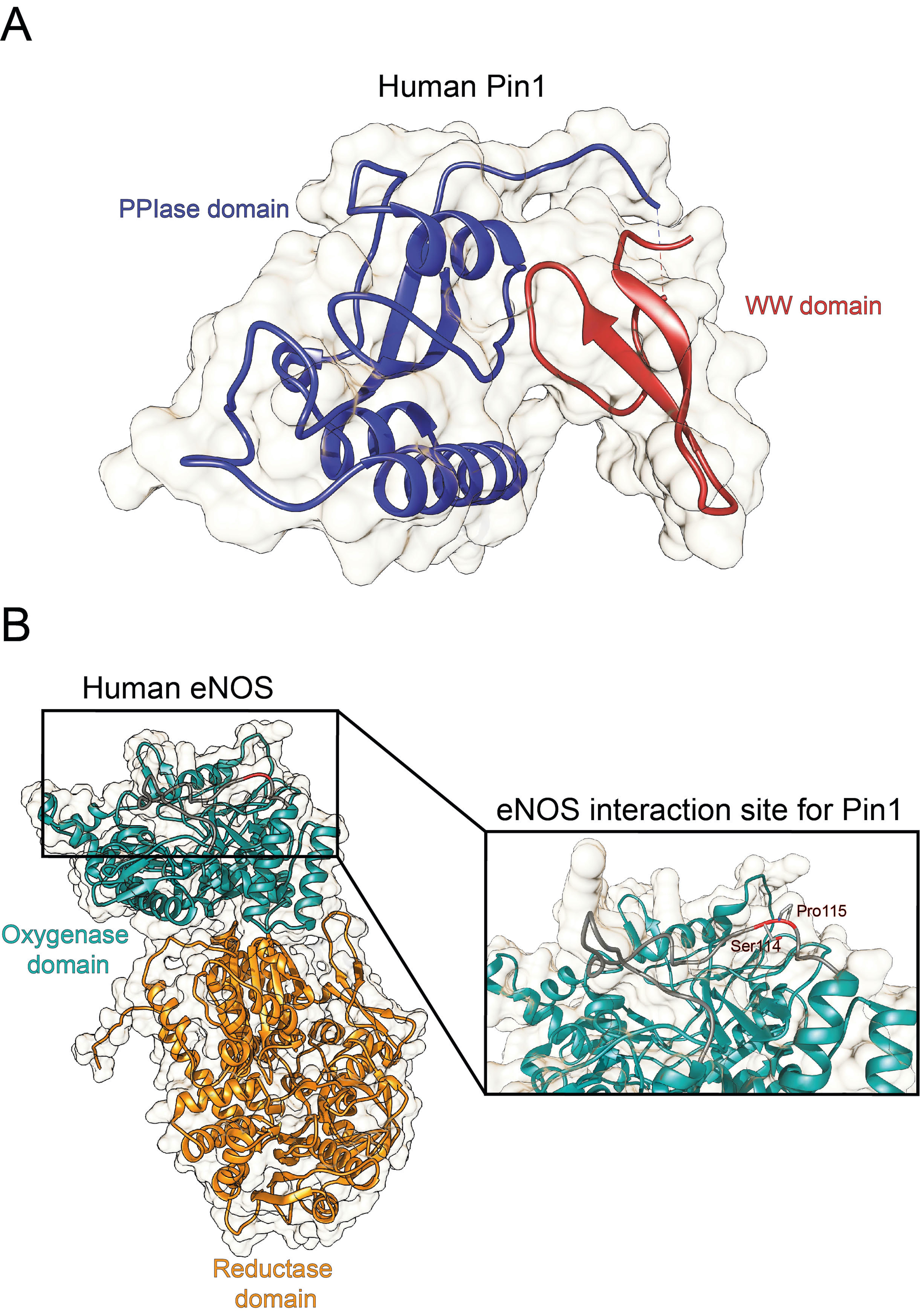
Figure 1. Structure of human Pin1 and human eNOS. (A) Ribbon representation of the full structure of human Pin1 (PDBID 1PIN; DOI: 10.1016/s0092-8674(00)80273-1). The PPIase and the WW domain are colored in blue and red, respectively. (B) Ribbon representation of the full structure of human eNOS. The structure was recovered from Alphafold protein structure database [15]. The oxygenase and the reductase domains are colored in light green and in orange, respectively. The eNOS Ser114-Pro115 residues are reported in the inset.
However, despite sparse evidence showing the implication of Pin1 in vascular homeostasis, little is known about Pin1 implication in cerebrovascular homeostasis, both in physiological and pathological contexts. As an example, Pin1 distribution and intracellular compartmentalization in the cerebrovascular system remains unknown. In the following sections, we will discuss evidence from the literature indicating the role of Pin1 in the regulation of vasculature homeostasis, by specifically dissecting its molecular interactions with key intracellular players, such as NOS, as well as the implication of Pin1 in vascular pathology. Moreover, we will discuss the hypothesis that Pin1 may serve a pivotal role in the function of vasculature, mainly focusing on its effects on the vascular endothelium. This latter, which is at direct contact with blood and affected by various vascular risk factors, represents a critical target and key player in vascular diseases.
Therefore, understanding the role of Pin1 in the vascular homeostasis and specifically, its interplay with NOS activity, is crucial in terms of finding a new possible therapeutic player and/or target in vascular pathologies, such as hypertension, diabetes, small and large vessel diseases, and vascular dementia.
3. Pin1 Implication in Vascular Diseases
Based on evidence indicating a key role of Pin1 in regulating NO production and vascular homeostasis, alteration of Pin1 levels and/or isomerase activity may be involved in the pathogenesis of vascular pathologies, such as hypertension and diabetes. In this regard, several studies described a prominent role of Pin1 in vascular changes associated to diabetes, where endothelial dysfunction represents an initial process in its vascular manifestations [16]. Paneni et al. demonstrated that treatment of human aortic endothelial cells (HAECs) with high glucose concentration for 72 hours induced an increase in Pin1 mRNA and protein levels [12]. In particular, methylation of Pin1 promoter, which typically represses gene transcription, was substantially decreased during hyperglycemia, suggesting that this epigenetic modification may contribute to Pin1 upregulation [12]. Moreover, during hyperglycemia, Pin1 has been shown to interact with eNOS at Ser116 phosphorylation site, thereby promoting eNOS interaction with caveolin-1, an important repressor of eNOS catalytic activity in the endothelium and subsequently reducing NO availability [12]. Interestingly, diabetic Pin1−/− mice were protected against mitochondrial oxidative stress, endothelial dysfunction, and vascular inflammation. In accordance with such evidence, pharmacological inhibition of Pin1 by juglone attenuated endothelial dysfunction, oxidative stress, as well as inflammatory processes, in streptozotocin-induced diabetic mice compared to vehicle-treated animals [17]. Noteworthy, although these data suggest that Pin1 may hinder NO production by repressing eNOS activity during hyperglycemia, it is unknown whether such negative regulation may be ascribed either to a direct effect of Pin1 on eNOS activity or to the increased association of eNOS with caveolin-1 or to a combination of these two effects.
Moreover, high glucose levels not only triggered Pin1 expression, but also enhanced its enzymatic activity. Consistently, an observational study conducted in patients with type 2 diabetes mellitus showed that Pin1 expression levels and activity were increased in the peripheral blood monocytes of the subjects compared to those of age-matched healthy controls and correlated with glycemic markers (i.e., elevated fasting plasma glucose (FPG) and HbA1C (hemoglobin A1C) levels) [18]. Experiments performed in type 2 diabetic (T2D) mice and in vascular smooth muscle cells exposed to T2D-ressembling conditions also showed that Pin1 levels were increased in hyperglycemic conditions [18]. Finally, Zhang et al. confirmed the notion that high glucose levels up-regulate Pin1, by reporting a substantial elevation in circulatory Pin1 levels in diabetic mice compared to wild-type mice. Such increase was reversed with exposure to juglone [19].
References
- Xu, S.; Ilyas, I.; Little, P.J.; Li, H.; Kamato, D.; Zheng, X.; Luo, S.; Li, Z.; Liu, P.; Han, J.; et al. Endothelial Dysfunction in Atherosclerotic Cardiovascular Diseases and Beyond: From Mechanism to Pharmacotherapies. Pharmacol. Rev. 2021, 73, 924–967.
- Celermajer, D.S.; Sorensen, K.E.; Spiegelhalter, D.J.; Georgakopoulos, D.; Robinson, J.; Deanfield, J.E. Aging is associated with endothelial dysfunction in healthy men years before the age-related decline in women. J. Am. Coll. Cardiol. 1994, 24, 471–476.
- Herrera, M.D.; Mingorance, C.; Rodríguez-Rodríguez, R.; Alvarez de Sotomayor, M. Endothelial dysfunction and aging: An update. Ageing Res. Rev. 2010, 9, 142–152.
- Smith, A.R.; Visioli, F.; Hagen, T.M. Plasma membrane-associated endothelial nitric oxide synthase and activity in aging rat aortic vascular endothelia markedly decline with age. Arch. Biochem. Biophys. 2006, 454, 100–105.
- Widlansky, M.E.; Gokce, N.; Keaney, J.F.J.; Vita, J.A. The clinical implications of endothelial dysfunction. J. Am. Coll. Cardiol. 2003, 42, 1149–1160.
- Lu, K.P.; Hanes, S.D.; Hunter, T. A human peptidyl-prolyl isomerase essential for regulation of mitosis. Nature 1996, 380, 544–547.
- Lu, K.P.; Zhou, X.Z. The prolyl isomerase PIN1: A pivotal new twist in phosphorylation signalling and disease. Nat. Rev. Mol. Cell Biol. 2007, 8, 904–916.
- Yeh, E.S.; Means, A.R. PIN1, the cell cycle and cancer. Nat. Rev. Cancer 2007, 7, 381–388.
- Fagiani, F.; Govoni, S.; Racchi, M.; Lanni, C. The Peptidyl-prolyl Isomerase Pin1 in Neuronal Signaling: From Neurodevelopment to Neurodegeneration. Mol. Neurobiol. 2021, 58, 1062–1073.
- Liu, M.; Yu, P.; Jiang, H.; Yang, X.; Zhao, J.; Zou, Y.; Ge, J. The Essential Role of Pin1 via NF-κB Signaling in Vascular Inflammation and Atherosclerosis in ApoE(-/-) Mice. Int. J. Mol. Sci. 2017, 18, 644.
- Kurakula, K.; Hagdorn, Q.A.J.; van der Feen, D.E.; Vonk Noordegraaf, A.; Ten Dijke, P.; de Boer, R.A.; Bogaard, H.J.; Goumans, M.J.; Berger, R.M.F. Inhibition of the prolyl isomerase Pin1 improves endothelial function and attenuates vascular remodelling in pulmonary hypertension by inhibiting TGF-β signalling. Angiogenesis 2021, 1–14.
- Paneni, F.; Costantino, S.; Castello, L.; Battista, R.; Capretti, G.; Chiandotto, S.; D’Amario, D.; Scavone, G.; Villano, A.; Rustighi, A.; et al. Targeting prolyl-isomerase Pin1 prevents mitochondrial oxidative stress and vascular dysfunction: Insights in patients with diabetes. Eur. Heart J. 2015, 36, 817–828.
- Liu, T.; Huang, Y.; Likhotvorik, R.I.; Keshvara, L.; Hoyt, D.G. Protein Never in Mitosis Gene A Interacting-1 (PIN1) regulates degradation of inducible nitric oxide synthase in endothelial cells. Am. J. Physiol. Cell Physiol. 2008, 295, C819–C827.
- Ruan, L.; Torres, C.M.; Qian, J.; Chen, F.; Mintz, J.D.; Stepp, D.W.; Fulton, D.; Venema, R.C. Pin1 prolyl isomerase regulates endothelial nitric oxide synthase. Arterioscler. Thromb. Vasc. Biol. 2011, 31, 392–398.
- Jumper, J.; Evans, R.; Pritzel, A.; Green, T.; Figurnov, M.; Ronneberger, O.; Tunyasuvunakool, K.; Bates, R.; Žídek, A.; Potapenko, A.; et al. Highly accurate protein structure prediction with AlphaFold. Nature 2021, 596, 583–589.
- Deedwania, P.C. Diabetes is a vascular disease: The role of endothelial dysfunction in pathophysiology of cardiovascular disease in diabetes. Cardiol. Clin. 2004, 22, 505–509.
- Costantino, S.; Paneni, F.; Lüscher, T.F.; Cosentino, F. Pin1 inhibitor Juglone prevents diabetic vascular dysfunction. Int. J. Cardiol. 2016, 203, 702–707.
- Lv, L.; Zhang, J.; Zhang, L.; Xue, G.; Wang, P.; Meng, Q.; Liang, W. Essential role of Pin1 via STAT3 signalling and mitochondria-dependent pathways in restenosis in type 2 diabetes. J. Cell. Mol. Med. 2013, 17, 989–1005.
- Zhang, M.; Lin, L.; Xu, C.; Chai, D.; Peng, F.; Lin, J. VDR Agonist Prevents Diabetic Endothelial Dysfunction through Inhibition of Prolyl Isomerase-1-Mediated Mitochondrial Oxidative Stress and Inflammation. Oxid. Med. Cell. Longev. 2018, 2018, 1714896.
More
Information
Subjects:
Biology
Contributor
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
897
Entry Collection:
Neurodegeneration
Revisions:
2 times
(View History)
Update Date:
08 Dec 2021
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





