
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Jittima Luckanagul | + 4302 word(s) | 4302 | 2021-11-29 07:58:28 | | | |
| 2 | Vivi Li | Meta information modification | 4302 | 2021-12-07 08:37:59 | | | | |
| 3 | Vivi Li | Meta information modification | 4302 | 2021-12-07 08:39:30 | | |
Video Upload Options
Currently, research in nanoparticles as a drug delivery system has broadened to include their use as a delivery system for bioactive substances and a diagnostic or theranostic system. Nanogels, nanoparticles containing a high amount of water, have gained attention due to their advantages of colloidal stability, core-shell structure, and adjustable structural components. These advantages provide the potential to design and fabricate multifunctional nanosystems for various biomedical applications. Modified or functionalized polymers and some metals are components that markedly enhance the features of the nanogels, such as tunable amphiphilicity, biocompatibility, stimuli-responsiveness, or sensing moieties, leading to specificity, stability, and tracking abilities.
1. Introduction
2. Core-Shell Structure of Polymeric Nanogels
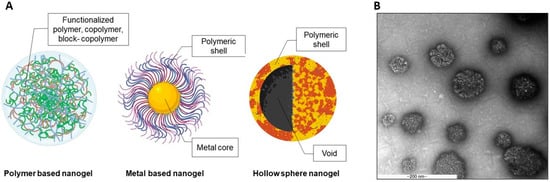
2.1. Polymer Based Polymeric Nanogels
2.2. Metal Based Polymeric Nanogels
3. Biomedical Applications
3.1. Therapeutics
3.2. Diagnotics and Theranostics
References
- Son, S.J.; Bai, X.; Lee, S.B. Inorganic hollow nanoparticles and nanotubes in nanomedicine: Part Drug/gene delivery applications. Drug Discov. Today 2007, 12, 650–656.
- Lee, Y.; Thompson, D.H. Stimuli-responsive liposomes for drug delivery. WIREs Nanomed. Nanobiotechnol. 2017, 9, e1450.
- Jamkhande, P.G.; Ghule, N.W.; Bamer, A.H.; Kalaskar, M.G. Metal nanoparticles synthesis: An overview on methods of preparation, advantages and disadvantages, and applications. J. Drug Deliv. Sci. Technol. 2019, 53, 101174.
- George, A.; Shah, P.A.; Shrivastav, P.S. Natural biodegradable polymers based nano-formulations for drug delivery: A review. Int. J. Pharmaceut. 2019, 561, 244–264.
- Sunshine, J.C.; Akanda, M.I.; Li, D.; Kozielski, K.L.; Green, J.J. Effects of base polymer hydrophobicity and end-group modification on polymeric gene delivery. Biomacromolecules 2011, 12, 3592–3600.
- Wang, Y.; Li, J.; Chen, Y.; Oupický, D. Balancing polymer hydrophobicity for ligand presentation and siRNA delivery in dual function CXCR4 inhibiting polyplexes. Biomater. Sci. 2015, 3, 1114–1123.
- Wang, C.; Zhou, D.-D.; Gan, Y.-W.; Zhang, X.-W.; Ye, Z.-M.; Zhang, J.-P. A partially fluorinated ligand for two super-hydrophobic porous coordination polymers with classic structures and increased porosities. Natl. Sci. Rev. 2020, 8.
- Liu, D.; Ma, L.; An, Y.; Li, Y.; Liu, Y.; Wang, L.; Guo, J.; Wang, J.; Zhou, J. Thermoresponsive Nanogel-Encapsulated PEDOT and HSP70 Inhibitor for Improving the Depth of the Photothermal Therapeutic Effect. Adv. Funct. Mater. 2016, 26, 4749–4759.
- Ji, Y.; Winter, L.; Navarro, L.; Ku, M.-C.; Periquito, J.S.; Pham, M.; Hoffmann, W.; Theune, L.E.; Calderón, M.; Niendorf, T. Controlled Release of Therapeutics from Thermoresponsive Nanogels: A Thermal Magnetic Resonance Feasibility Study. Cancers 2020, 12, 1380.
- Shi, X.; Ma, X.; Hou, M.; Gao, Y.-E.; Bai, S.; Xiao, B.; Xue, P.; Kang, Y.; Xu, Z.; Li, C.M. pH-Responsive unimolecular micelles based on amphiphilic star-like copolymers with high drug loading for effective drug delivery and cellular imaging. J. Mater. Chem. B 2017, 5, 6847–6859.
- Liu, S.; Ono, R.J.; Yang, C.; Gao, S.; Ming Tan, J.Y.; Hedrick, J.L.; Yang, Y.Y. Dual pH-Responsive Shell-Cleavable Polycarbonate Micellar Nanoparticles for in Vivo Anticancer Drug Delivery. ACS Appl. Mater. Interfaces 2018, 10, 19355–19364.
- Cazotti, J.C.; Fritz, A.T.; Garcia-Valdez, O.; Smeets, N.M.B.; Dubé, M.A.; Cunningham, M.F. Graft modification of starch nanoparticles with pH-responsive polymers via nitroxide-mediated polymerization. J. Polym. Sci. 2020, 58, 2211–2220.
- Maiti, C.; Parida, S.; Kayal, S.; Maiti, S.; Mandal, M.; Dhara, D. Redox-responsive core-cross-linked block copolymer micelles for overcoming multidrug resistance in cancer cells. ACS Appl. Mater. Interfaces 2018, 10, 5318–5330.
- Aramoto, H.; Osaki, M.; Konishi, S.; Ueda, C.; Kobayashi, Y.; Takashima, Y.; Harada, A.; Yamaguchi, H. Redox-responsive supramolecular polymeric networks having double-threaded inclusion complexes. Chem. Sci. 2020, 11, 4322–4331.
- Vicario-de-la-Torre, M.; Forcada, J. The Potential of Stimuli-Responsive Nanogels in Drug and Active Molecule Delivery for Targeted Therapy. Gels 2017, 3, 16.
- Zhang, Y.; Dosta, P.; Conde, J.; Oliva, N.; Wang, M.; Artzi, N. Prolonged Local In Vivo Delivery of Stimuli-Responsive Nanogels That Rapidly Release Doxorubicin in Triple-Negative Breast Cancer Cells. Adv. Healthc. Mater. 2020, 9, 1901101.
- Li, S.; Zhang, T.; Xu, W.; Ding, J.; Yin, F.; Xu, J.; Sun, W.; Wang, H.; Sun, M.; Cai, Z. Sarcoma-targeting peptide-decorated polypeptide nanogel intracellularly delivers shikonin for upregulated osteosarcoma necroptosis and diminished pulmonary metastasis. Theranostics 2018, 8, 1361.
- Pan, G.; Guo, Q.; Cao, C.; Yang, H.; Li, B. Thermo-responsive molecularly imprinted nanogels for specific recognition and controlled release of proteins. Soft Matter 2013, 9, 3840–3850.
- Liu, P. 1—Redox—and pH-responsive polymeric nanocarriers. In Stimuli Responsive Polymeric Nanocarriers for Drug Deliv. Applications; Makhlouf, A.S.H., Abu-Thabit, N.Y., Eds.; Woodhead Publishing: Cambridge, UK, 2019; pp. 3–36.
- Badeau, B.A.; DeForest, C.A. Programming Stimuli-Responsive Behavior into Biomaterials. Annu. Rev. Biomed. Eng. 2019, 21, 241–265.
- Caro, C.; García-Martín, M.L.; Pernia Leal, M. Manganese-Based Nanogels as pH Switches for Magnetic Resonance Imaging. Biomacromolecules 2017, 18, 1617–1623.
- Zhang, X.; Chen, X.; Wang, H.-Y.; Jia, H.-R.; Wu, F.-G. Supramolecular Nanogel-Based Universal Drug Carriers Formed by “Soft–Hard” Co-Assembly: Accurate Cancer Diagnosis and Hypoxia-Activated Cancer Therapy. Adv. Ther. 2019, 2, 1800140.
- Wang, Y.; Zu, M.; Ma, X.; Jia, D.; Lu, Y.; Zhang, T.; Xue, P.; Kang, Y.; Xu, Z. Glutathione-Responsive Multifunctional “Trojan Horse” Nanogel as a Nanotheranostic for Combined Chemotherapy and Photodynamic Anticancer Therapy. ACS Appl. Mater. Interfaces 2020, 12, 50896–50908.
- Zhang, H.; Ba, S.; Lee, J.Y.; Xie, J.; Loh, T.-P.; Li, T. Cancer Biomarker-Triggered Disintegrable DNA Nanogels for Intelligent Drug Delivery. Nano Lett. 2020, 20, 8399–8407.
- Cazares-Cortes, E.; Espinosa, A.; Guigner, J.-M.; Michel, A.; Griffete, N.; Wilhelm, C.; Ménager, C. Doxorubicin Intracellular Remote Release from Biocompatible Oligo(ethylene glycol) Methyl Ether Methacrylate-Based Magnetic Nanogels Triggered by Magnetic Hyperthermia. ACS Appl. Mater. Interfaces 2017, 9, 25775–25788.
- Khan, A.R.; Yang, X.; Du, X.; Yang, H.; Liu, Y.; Khan, A.Q.; Zhai, G. Chondroitin sulfate derived theranostic and therapeutic nanocarriers for tumor-targeted drug delivery. Carbohydr. Polym. 2020, 233, 115837.
- Fusco, L.; Gazzi, A.; Peng, G.; Shin, Y.; Vranic, S.; Bedognetti, D.; Vitale, F.; Yilmazer, A.; Feng, X.; Fadeel, B.; et al. Graphene and other 2D materials: A multidisciplinary analysis to uncover the hidden potential as cancer theranostics. Theranostics 2020, 10, 5435–5488.
- He, F.; Zhang, Y.; Li, J.; Liu, S.; Chi, Z.; Xu, J. Preparation and properties of multi-responsive semi-IPN hydrogel modified magnetic nanoparticles as drug carrier. J. Control. Rel. 2011, 152, e119–e121.
- Gonzalez-Urias, A.; Zapata-Gonzalez, I.; Licea-Claverie, A.; Licea-Navarro, A.F.; Bernaldez-Sarabia, J.; Cervantes-Luevano, K. Cationic versus anionic core-shell nanogels for transport of cisplatin to lung cancer cells. Colloids Surf. B Biointerfaces 2019, 182, 110365.
- Amanlou, N.; Parsa, M.; Rostamizadeh, K.; Sadighian, S.; Moghaddam, F. Enhanced cytotoxic activity of curcumin on cancer cell lines by incorporating into gold/chitosan nanogels. Mater. Chem. Phys. 2019, 226, 151–157.
- Qiu, M.; Wu, H.; Cao, L.; Shi, B.; He, X.; Geng, H.; Mao, X.; Yang, P.; Jiang, Z. Metal–Organic Nanogel with Sulfonated Three-Dimensional Continuous Channels as a Proton Conductor. ACS Appl. Mater. Interfaces 2020, 12, 19788–19796.
- Tang, J.; Cui, X.; Caranasos, T.G.; Hensley, M.T.; Vandergriff, A.C.; Hartanto, Y.; Shen, D.; Zhang, H.; Zhang, J.; Cheng, K. Heart Repair Using Nanogel-Encapsulated Human Cardiac Stem Cells in Mice and Pigs with Myocardial Infarction. ACS Nano. 2017, 11, 9738–9749.
- Shoueir, K.R.; Sarhan, A.A.; Atta, A.M.; Akl, M.A. Macrogel and nanogel networks based on crosslinked poly (vinyl alcohol) for adsorption of methylene blue from aqua system. Environ. NanoTechnol. Monitor. Manag. 2016, 5, 62–73.
- Lou, S.; Gao, S.; Wang, W.; Zhang, M.; Zhang, J.; Wang, C.; Li, C.; Kong, D.; Zhao, Q. Galactose-functionalized multi-responsive nanogels for hepatoma-targeted drug delivery. Nanoscale 2015, 7, 3137–3146.
- Liechty, W.B.; Scheuerle, R.L.; Vela Ramirez, J.E.; Peppas, N.A. Uptake and function of membrane-destabilizing cationic nanogels for intracellular drug delivery. Bioeng. Transl. Med. 2019, 4, 17–29.
- Liechty, W.B.; Scheuerle, R.L.; Peppas, N.A. Tunable, responsive nanogels containing t-butyl methacrylate and 2-(t-butylamino)ethyl methacrylate. Polymer 2013, 54, 3784–3795.
- Horvat, S.; Yu, Y.; Bojte, S.; Tessmer, I.; Lowman, D.W.; Ma, Z.; Williams, D.L.; Beilhack, A.; Albrecht, K.; Groll, J. Engineering Nanogels for Drug Delivery to Pathogenic Fungi Aspergillus fumigatus by Tuning Polymer Amphiphilicity. Biomacromolecules 2020, 21, 3112–3121.
- Costa, D.; Valente, A.J.; Queiroz, J. Plasmid DNA nanogels as photoresponsive materials for multifunctional bio-applications. J. Biotechnol. 2015, 202, 98–104.
- Schmid, A.J.; Dubbert, J.; Rudov, A.A.; Pedersen, J.S.; Lindner, P.; Karg, M.; Potemkin, I.I.; Richtering, W. Multi-Shell Hollow Nanogels with Responsive Shell Permeability. Sci. Rep. 2016, 6, 22736.
- Yasun, E.; Gandhi, S.; Choudhury, S.; Mohammadinejad, R.; Benyettou, F.; Gozubenli, N.; Arami, H. Hollow micro and nanostructures for therapeutic and imaging applications. J. Drug Deliv. Sci. Technol. 2020, 60.
- Osorio-Blanco, E.R.; Rancan, F.; Klossek, A.; Nissen, J.H.; Hoffmann, L.; Bergueiro, J.; Riedel, S.; Vogt, A.; Ruhl, E.; Calderon, M. Polyglycerol-Based Thermoresponsive Nanocapsules Induce Skin Hydration and Serve as a Skin Penetration Enhancer. ACS Appl. Mater. Interfaces 2020, 12, 30136–30144.
- Wang, Z.; Li, S.; Zhang, M.; Ma, Y.; Liu, Y.; Gao, W.; Zhang, J.; Gu, Y. Laser-Triggered Small Interfering RNA Releasing Gold Nanoshells against Heat Shock Protein for Sensitized Photothermal Therapy. Adv. Sci. 2017, 4, 1600327.
- Han, H.; Li, L.; Tian, Y.; Wang, Y.; Ye, Z.; Yang, Q.; Wang, Y.; von Klitzing, R.; Guo, X. Spherical polyelectrolyte nanogels as templates to prepare hollow silica nanocarriers: Observation by small angle X-ray scattering and TEM. RSC Adv. 2017, 7, 47877–47885.
- Ranzoni, A.; Cooper, M.A. Chapter One—The Growing Influence of Nanotechnology in Our Lives. In Micro and NanoTechnol. in Vaccine Development; Skwarczynski, M., Toth, I., Eds.; William Andrew Publishing: Norwich, NY, USA, 2017; pp. 1–20.
- Roberts, J.R.; Ritter, D.W.; McShane, M.J. A design full of holes: Functional nanofilm-coated microdomains in alginate hydrogels. J. Mater. Chem. B 2013, 1, 3195–3201.
- Pan, H.M.; Yu, H.; Guigas, G.; Fery, A.; Weiss, M.; Patzel, V.; Trau, D. Engineering and Design of Polymeric Shells: Inwards Interweaving Polymers as Multilayer Nanofilm, Immobilization Matrix, or Chromatography Resins. ACS Appl. Mater. Interfaces 2017, 9, 5447–5456.
- Bornhoeft, L.R.; Biswas, A.; McShane, M.J. Composite hydrogels with engineered microdomains for optical glucose sensing at low oxygen conditions. Biosensors 2017, 7, 8.
- Choi, D.; Heo, J.; Park, J.H.; Jo, Y.; Jeong, H.; Chang, M.; Choi, J.; Hong, J. Nano-film coatings onto collagen hydrogels with desired drug release. J. Ind. Eng. Chem. 2016, 36, 326–333.
- Kim, M.; Kim, H.; Lee, Y.-s.; Lee, S.; Kim, S.-E.; Lee, U.-J.; Jung, S.; Park, C.-G.; Hong, J.; Doh, J.; et al. Novel enzymatic cross-linking–based hydrogel nanofilm caging system on pancreatic β cell spheroid for long-term blood glucose regulation. Sci. Adv. 2021, 7, eabf7832.
- Lu, S.; Neoh, K.G.; Huang, C.; Shi, Z.; Kang, E.T. Polyacrylamide hybrid nanogels for targeted cancer chemotherapy via co-delivery of gold nanoparticles and MTX. J. Colloid Interface Sci. 2013, 412, 46–55.
- Wu, W.; Shen, J.; Banerjee, P.; Zhou, S. Water-dispersible multifunctional hybrid nanogels for combined curcumin and photothermal therapy. BioMaterials 2011, 32, 598–609.
- Xue, Y.; Xia, X.; Yu, B.; Tao, L.; Wang, Q.; Huang, S.W.; Yu, F. Selenylsulfide Bond-Launched Reduction-Responsive Superparamagnetic Nanogel Combined of Acid-Responsiveness for Achievement of Efficient Therapy with Low Side Effect. ACS Appl. Mater. Interfaces 2017, 9, 30253–30257.
- Gao, X.; Zhuang, X.; Tian, C.; Liu, H.; Lai, W.-F.; Wang, Z.; Yang, X.; Chen, L.; Rogach, A.L. A copper nanocluster incorporated nanogel: Confinement-assisted emission enhancement for zinc ion detection in living cells. Sens. Actuat. B Chem. 2020, 307, 127626.
- Jiang, G.; Park, K.; Kim, J.; Kim, K.S.; Hahn, S.K. Target Specific Intracellular Delivery of siRNA/PEI−HA Complex by Receptor Mediated Endocytosis. Molec. Pharmaceut. 2009, 6, 727–737.
- Nagahama, K.; Sano, Y.; Kumano, T. Anticancer drug-based multifunctional nanogels through self-assembly of dextran-curcumin conjugates toward cancer theranostics. Bioorg. Med. Chem. Lett. 2015, 25, 2519–2522.
- Maxwell, T.; Banu, T.; Price, E.; Tharkur, J.; Campos, M.G.; Gesquiere, A.; Santra, S. Non-Cytotoxic Quantum Dot-Chitosan Nanogel Biosensing Probe for Potential Cancer Targeting Agent. NanoMater. 2015, 5, 2359–2379.
- The global challenge of cancer. Nat. Cancer 2020, 1, 1–2.
- Martin, T.A.; Ye, L.; Sanders, A.J.; Lane, J.; Jiang, W.G. Cancer invasion and metastasis: Molecular and cellular perspective. In Madame Curie BioSci. Database ; Landes Bioscience: Austin, TX, USA, 2013.
- Tan, L.; He, C.; Chu, X.; Chu, Y.; Ding, Y. Charge-reversal ZnO-based nanospheres for stimuli-responsive release of multiple agents towards synergistic cancer therapy. Chem. Eng. J. 2020, 395.
- Liu, Y.; Zhao, N.; Xu, F.J. pH-Responsive Degradable Dextran-Quantum Dot Nanohybrids for Enhanced Gene Delivery. ACS Appl. Mater. Interfaces 2019, 11, 34707–34716.
- Gui, R.; Wang, Y.; Sun, J. Embedding fluorescent mesoporous silica nanoparticles into biocompatible nanogels for tumor cell imaging and thermo/pH-sensitive in vitro drug release. Colloids Surf. B Biointerfaces 2014, 116, 518–525.
- Nasr, F.H.; Khoee, S. Design, characterization and in vitro evaluation of novel shell crosslinked poly(butylene adipate)-co-N-succinyl chitosan nanogels containing loteprednol etabonate: A new system for therapeutic effect enhancement via controlled drug delivery. Eur. J. Med. Chem. 2015, 102, 132–142.
- Wei, X.; Xiong, H.; He, S.; Wang, Y.; Zhou, D.; Jing, X.; Huang, Y. A facile way to prepare functionalized dextran nanogels for conjugation of hemoglobin. Colloids Surf. B Biointerfaces 2017, 155, 440–448.
- Li, D.; Chen, Y.; Mastrobattista, E.; van Nostrum, C.F.; Hennink, W.E.; Vermonden, T. Reduction-Sensitive Polymer-Shell-Coated Nanogels for Intracellular Delivery of Antigens. ACS Biomater. Sci. Eng. 2017, 3, 42–48.
- Jin, H.; Tan, H.; Zhao, L.; Sun, W.; Zhu, L.; Sun, Y.; Hao, H.; Xing, H.; Liu, L.; Qu, X.; et al. Ultrasound-triggered thrombolysis using urokinase-loaded nanogels. Int. J. Pharm. 2012, 434, 384–390.
- Teng, Y.; Jin, H.; Nan, D.; Li, M.; Fan, C.; Liu, Y.; Lv, P.; Cui, W.; Sun, Y.; Hao, H.; et al. In vivo evaluation of urokinase-loaded hollow nanogels for sonothrombolysis on suture embolization-induced acute ischemic stroke rat model. Bioact. Mater. 2018, 3, 102–109.
- Mihalko, E.P.; Sandry, M.; Mininni, N.; Nellenbach, K.; Deal, H.; Daniele, M.; Ghadimi, K.; Levy, J.H.; Brown, A.C. Fibrin-modulating nanogels for treatment of disseminated intravascular coagulation. Blood Adv. 2021, 5, 613–627.
- Zhao, H.; Zhao, Y.; Xu, J.; Feng, X.; Liu, G.; Zhao, Y.; Yang, X. Programmable co-assembly of various drugs with temperature sensitive nanogels for optimizing combination chemotherapy. Chem. Eng. J. 2020, 398.
- Zhu, J.; Li, F.; Wang, X.; Yu, J.; Wu, D. Hyaluronic Acid and Polyethylene Glycol Hybrid Hydrogel Encapsulating Nanogel with Hemostasis and Sustainable Antibacterial Property for Wound Healing. ACS Appl Mater. Interfaces 2018.
- Wu, W.; Mitra, N.; Yan, E.C.; Zhou, S. Multifunctional hybrid nanogel for integration of optical glucose sensing and self-regulated insulin release at physiological pH. Am. Chem. Soc. 2010, 4, 9.
- Kolouchova, K.; Sedlacek, O.; Jirak, D.; Babuka, D.; Blahut, J.; Kotek, J.; Vit, M.; Trousil, J.; Konefal, R.; Janouskova, O.; et al. Self-Assembled Thermoresponsive Polymeric Nanogels for (19)F MR Imaging. Biomacromolecules 2018, 19, 3515–3524.
- Rejinold, N.S.; Ranjusha, R.; Balakrishnan, A.; Mohammed, N.; Jayakumar, R. Gold–chitin–manganese dioxide ternary composite nanogels for radio frequency assisted cancer therapy. RSC Adv. 2014, 4.
- Zan, M.; Li, J.; Huang, M.; Lin, S.; Luo, D.; Luo, S.; Ge, Z. Near-infrared light-triggered drug release nanogels for combined photothermal-chemotherapy of cancer. Biomater. Sci. 2015, 3, 1147–1156.
- Kimura, A.; Jo, J.I.; Yoshida, F.; Hong, Z.; Tabata, Y.; Sumiyoshi, A.; Taguchi, M.; Aoki, I. Ultra-small size gelatin nanogel as a blood brain barrier impermeable contrast agent for magnetic resonance imaging. Acta Biomater. 2021.
- Wu, W.; Zhou, T.; Berliner, A.; Banerjee, P.; Zhou, S. Smart Core−Shell Hybrid Nanogels with Ag Nanoparticle Core for Cancer Cell Imaging and Gel Shell for pH-Regulated Drug Delivery. Chem. Mater. 2010, 22, 1966–1976.
- Jia, X.; Han, Y.; Pei, M.; Zhao, X.; Tian, K.; Zhou, T.; Liu, P. Multi-functionalized hyaluronic acid nanogels crosslinked with carbon dots as dual receptor-mediated targeting tumor theranostics. Carbohydr. Polym. 2016, 152, 391–397.
- Khatun, Z.; Nurunnabi, M.; Nafiujjaman, M.; Reeck, G.R.; Khan, H.A.; Cho, K.J.; Lee, Y.K. A hyaluronic acid nanogel for photo-chemo theranostics of lung cancer with simultaneous light-responsive controlled release of doxorubicin. Nanoscale 2015, 7, 10680–10689.
- Peng, N.; Ding, X.; Wang, Z.; Cheng, Y.; Gong, Z.; Xu, X.; Gao, X.; Cai, Q.; Huang, S.; Liu, Y. Novel dual responsive alginate-based magnetic nanogels for onco-theranostics. Carbohydr. Polym. 2019, 204, 32–41.





