
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | lincui da | + 1471 word(s) | 1471 | 2021-11-03 04:10:35 | | | |
| 2 | Catherine Yang | Meta information modification | 1471 | 2021-11-17 02:33:46 | | |
Video Upload Options
The membranous extracellular matrix (ECM) decellularized from body membranes (e.g., pleura, peritoneum, and amniotic membrane, etc) retain multiple bioactive components like collagen, laminin, fibronectin, growth factors, and polysaccharide. In addition, they also possess ultrastructure features similar to that of the natural tissues with distinct advantage in high density cell seeding, the migration of repair cells from adjacent tissues, and the mass exchange between tissues. These merits make membranous ECM scaffolds extremely attractive in regenerative medicine including but not limited to skin wound healing.
1. Introduction
Body membranes are thin layers of cells or tissues covering the surface of body, the internal organs, or the body cavities. Generally, they can be classified into two categories: (1) the epithelial membranes, and (2) the connective tissue membranes [1]. The epithelial membranes, which are composed of epithelial tissue and fibrous connective tissue, can be further divided into (1) the cutaneous membrane (i.e., skin), (2) the serous membranes, such as pleura, peritoneum, and amniotic membrane, and (3) the mucous membranes [1]. Unlike epithelial membranes, connective tissue membranes (e.g., periosteum, fascia, and synovial membrane) are typically composed of cells, ground substance, and connective tissue fibers [1].
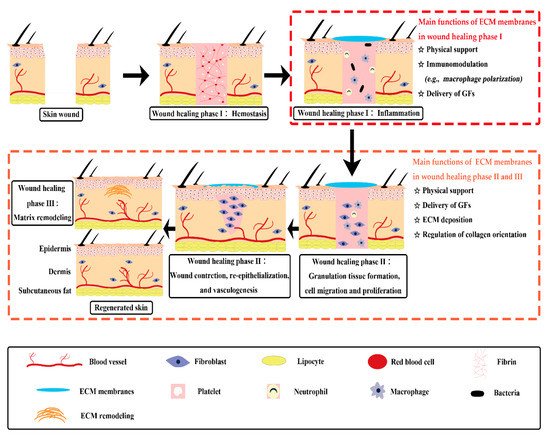
After decellularization, extracellular matrix (ECM) scaffolds obtained from different types of body membranes retain a variety of bioactive substances such as the growth factors, collagen, laminin, fibronectin, and polysaccharide. Notably, they possess ultrastructure features similar to that of the natural tissues [2][3][4]. These microporous thin films have a distinct advantage in the mass exchange between tissues [5][6]. Particularly, scaffolds in a thin planar form are favorable for high density cell seeding and the migration of repair cells from adjacent tissues [5][7]. These merits make membranous ECM scaffolds extremely attractive for skin wound healing.
Indeed, the safety and efficiency of several membranous ECM scaffolds have been verified in many clinical practices; however, some of their physicochemical properties, such as the mechanical strength and the degradation characteristics, are far from satisfactory for broad applications. This is partially due to the damage of the crosslinked networks of natural tissues during scaffold preparation, especially the use of acids, alkalis or proteases for decellularization. It is well-known that an ideal scaffold for tissue repair should possess good biocompatibility, robust bioactivity, suitable degradation, and proper mechanical properties. Therefore, attempts to endow traditional membranous ECM scaffolds with desired properties have been the focus of many researches. For instance, to meliorate the physical or chemical defects of traditional ECM scaffolds, different crosslinking methods have been developed [8]. Furthermore, diverse macromolecules, either natural or synthetic, have been used as functional additives to produce ECM-based implants [9][10][11][12][13]. In light of the functional requirements of normal wound healing, many types of biomolecules, nanoparticles, and drugs have been utilized to engineer new generations of ECM-based biomaterials, which can stimulate a specific wound healing stage or event to facilitate chronic wound healing.
2. Traditional ECM Scaffolds Derived from Body Membranes for Skin Wound Healing
2.1. Acellular Dermal Matrix (ADM)
2.2. Small Intestinal Submucosa (SIS)
2.3. Acellular Amniotic Membrane (AAM)
2.4. Other ECM Membranes
References
- Inci, I.; Norouz Dizaji, A.; Ozel, C.; Morali, U.; Dogan Guzel, F.; Avci, H. Decellularized inner body membranes for tissue engineering: A review. J. Biomater. Sci. Polym. Ed. 2020, 31, 1287–1368.
- Parveen, S.; Singh, S.P.; Panicker, M.M.; Gupta, P.K. Amniotic membrane as novel scaffold for human iPSC-derived cardiomyogenesis. In Vitro Cell Dev. Biol. Anim. 2019, 55, 1–13.
- Taylor, D.A.; Sampaio, L.C.; Ferdous, Z.; Gobin, A.S.; Taite, L.J. Decellularized matrices in regenerative medicine. Acta Biomater. 2018, 74, 74–89.
- Cao, G.; Huang, Y.; Li, K.; Fan, Y.; Xie, H.; Li, X. Small intestinal submucosa: Superiority, limitations and solutions, and its potential to address bottlenecks in tissue repair. J. Mater. Chem. B 2019, 7, 5038–5055.
- Vernon, R.B.; Gooden, M.D.; Lara, S.L.; Wight, T.N. Native fibrillar collagen membranes of micron-scale and submicron thicknesses for cell support and perfusion. Biomaterials 2005, 26, 1109–1117.
- Cui, W.; Khan, K.M.; Ma, X.; Chen, G.; Desai, C.S. Human amniotic epithelial cells and human amniotic membrane as a vehicle for islet cell transplantation. Transplant. Proc. 2020, 52, 982–986.
- Chouhan, D.; Dey, N.; Bhardwaj, N.; Mandal, B.B. Emerging and innovative approaches for wound healing and skin regeneration: Current status and advances. Biomaterials 2019, 216, 1–19.
- Wang, M.; Li, Y.Q.; Cao, J.; Gong, M.; Zhang, Y.; Chen, X.; Tian, M.X.; Xie, H.Q. Accelerating effects of genipin-crosslinked small intestinal submucosa for defected gastric mucosa repair. J. Mater. Chem. B 2017, 5, 7059–7071.
- Hong, Y.; Huber, A.; Takanari, K.; Amoroso, N.J.; Hashizume, R.; Badylak, S.F.; Wagner, W.R. Mechanical properties and in vivo behavior of a biodegradable synthetic polymer microfiber—Extracellular matrix hydrogel biohybrid scaffold. Biomaterials 2011, 32, 3387–3394.
- Thao, N.T.T.; Lee, S.; Shin, G.R.; Kang, Y.; Choi, S.; Kim, M.S. Preparation of electrospun small intestinal submucosa/poly(caprolactone-co-lactide-co-glycolide) nanofiber sheet as a potential drug carrier. Pharmaceutics 2021, 13, 253.
- Da, L.; Gong, M.; Chen, A.; Zhang, Y.; Huang, Y.; Guo, Z.; Li, S.; Li-Ling, J.; Zhang, L.; Xie, H. Composite elastomeric polyurethane scaffolds incorporating small intestinal submucosa for soft tissue engineering. Acta Biomater. 2017, 59, 45–57.
- Fan, M.R.; Gong, M.; Da, L.C.; Bai, L.; Li, X.Q.; Chen, K.F.; Li-Ling, J.; Yang, Z.M.; Xie, H.Q. Tissue engineered esophagus scaffold constructed with porcine small intestinal submucosa and synthetic polymers. Biomed. Mater. 2014, 9, 1–12.
- Fard, M.; Akhavan-Tavakoli, M.; Khanjani, S.; Zare, S.; Edalatkhah, H.; Arasteh, S.; Mehrabani, D.; Zarnani, A.H.; Kazemnejad, S.; Shirazi, R. Bilayer amniotic membrane/nano-fibrous fibroin scaffold promotes differentiation capability of menstrual blood stem cells into keratinocyte-like cells. Mol. Biotechnol. 2018, 60, 100–110.
- Alizadeh, M.; Rezakhani, L.; khodaei, M.; Alizadeh, A. Characterization of the decellularized ovine pericardium for skin tissue engineering. J. Shahrekord Univ. Med Sci. 2020, 22, 173–180.
- Brantley, J.N.; Verla, T.D. Use of placental membranes for the treatment of chronic diabetic foot ulcers. Adv. Wound Care 2015, 4, 545–559.
- Shakouri-Motlagh, A.; Khanabdali, R.; Heath, D.E.; Kalionis, B. The application of decellularized human term fetal membranes in tissue engineering and regenerative medicine (TERM). Placenta 2017, 59, 124–130.
- Frazao, L.P.; Vieira de Castro, J.; Nogueira-Silva, C.; Neves, N.M. Decellularized human chorion membrane as a novel biomaterial for tissue regeneration. Biomolecules 2020, 10, 1208.
- Dussoyer, M.; Michopoulou, A.; Rousselle, P. Decellularized scaffolds for skin repair and regeneration. Appl. Sci. 2020, 10, 3435.
- AL-Bayati, A.H.F.; Hameed, F.M. Effect of acellular bovine pericardium and dermal matrixes on cutaneous wounds healing in male rabbits: Histopathological evaluation. J. Entomol. Zool. Stud. 2018, 6, 1976–1986.
- Da, L.C.; Huang, Y.Z.; Xie, H.Q. Progress in development of bioderived materials for dermal wound healing. Regen. Biomater. 2017, 4, 325–334.
- Tsung-Hsuan, W.; Bertasi, G.; Elfahar, M. The use of dermacell® in fingertip injury. J. Clin. Case Rep. Images 2019, 1, 14–22.
- Ariganello, M.B.; Simionescu, D.T.; Labow, R.S.; Lee, J.M. Macrophage differentiation and polarization on a decellularized pericardial biomaterial. Biomaterials 2011, 32, 439–449.
- Dziki, J.L.; Wang, D.S.; Pineda, C.; Sicari, B.M.; Rausch, T.; Badylak, S.F. Solubilized extracellular matrix bioscaffolds derived from diverse source tissues differentially influence macrophage phenotype. J. Biomed. Mater. Res. Part A 2017, 105, 138–147.
- Tan, J.; Zhang, Q.Y.; Huang, L.P.; Huang, K.; Xie, H.Q. Decellularized scaffold and its elicited immune response towards the host: The underlying mechanism and means of immunomodulatory modification. Biomater. Sci. 2021, 9, 4803–4820.
- Qing, C. The molecular biology in wound healing & non-healing wound. Chin. J. Traumatol. 2017, 20, 189–193.
- Liu, C.; Sun, J. A porcine acellular dermal matrix induces human fibroblasts to secrete hyaluronic acid by activating JAK2/STAT3 signalling. RSC Adv. 2020, 10, 18959–18969.
- Cui, H.; Chai, Y.; Yu, Y. Progress in developing decellularized bioscaffolds for enhancing skin construction. J. Biomed. Mater. Res. Part A 2019, 107, 1849–1859.
- Cazzell, S.; Moyer, P.M.; Samsell, B.; Dorsch, K.; McLean, J.; Moore, M.A. A prospective, multicenter, single-arm clinical trial for treatment of complex diabetic foot ulcers with deep exposure using acellular dermal matrix. Adv. Skin Wound Care 2019, 32, 409–415.
- Lewis, R.E.; Towery, E.A.; Bhat, S.G.; Ward, A.J.; Heidel, R.E.; Bielak, K.M.; Simpson, H.E.; McLoughlin, J.M.; Lewis, J.M. Human acellular dermal matrix is a viable alternative to autologous skin graft in patients with cutaneous malignancy. Am. Surg. 2019, 85, 1056–1060.
- Newton, D.J.; Khan, F.; Belch, J.J.; Mitchell, M.R.; Leese, G.P. Blood flow changes in diabetic foot ulcers treated with dermal replacement therapy. J. Foot Ankle Surg. 2002, 41, 233–237.
- Holl, J.; Kowalewski, C.; Zimek, Z.; Fiedor, P.; Kaminski, A.; Oldak, T.; Moniuszko, M.; Eljaszewicz, A. Chronic diabetic wounds and their treatment with skin substitutes. Cells 2021, 10, 655.
- Alvaro-Afonso, F.J.; Garcia-Alvarez, Y.; Lazaro-Martinez, J.L.; Kakagia, D.; Papanas, N. Advances in dermoepidermal skin substitutes for diabetic foot ulcers. Curr. Vasc. Pharmacol. 2020, 18, 182–192.
- Ayaz, M.; Najafi, A.; Karami, M.Y. Thin split thickness skin grafting on human acellular dermal matrix scaffold for the treatment of deep burn wounds. Int. J. Organ. Transplant. Med. 2021, 12, 44–51.
- Reyzelman, A.; Crews, R.T.; Moore, J.C.; Moore, L.; Mukker, J.S.; Offutt, S.; Tallis, A.T.W.B.; Vayser, D.; Winters, C.; Armstrong, D.G. Clinical effectiveness of an acellular dermal regenerative tissue matrix compared to standard wound management in healing diabetic foot ulcers: A prospective, randomised, multicentre study. Int. Wound J. 2009, 6, 196–208.
- Reyzelman, A.M.; Bazarov, I. Human acellular dermal wound matrix for treatment of DFU: Literature review and analysis. J. Wound Care 2015, 24, 128–134.
- Asodiya, F.A.; Kumar, V.; Vora, S.D.; Singh, V.K.; Fefar, D.T.; Gajera, H.P. Preparation, characterization, and xenotransplantation of the caprine acellular dermal matrix. Xenotransplantation 2020, 27, 1–12.
- Zhang, Z.; Lv, L.; Mamat, M.; Chen, Z.; Liu, L.; Wang, Z. Xenogenic (porcine) acellular dermal matrix is useful for the wound healing of severely damaged extremities. Exp. Ther. Med. 2014, 7, 621–624.
- Kim, T.H.; Park, J.H.; Jeong, H.G.; Wee, S.Y. The utility of novel fish-skin derived acellular dermal matrix (Kerecis) as a wound dressing material. J. Wound Manag. Res. 2021, 17, 39–47.
- Sitje, T.S.; Grøndahl, E.C.; Sørensen, J.A. Clinical innovation: Fish-derived wound product for cutaneous wounds. Wounds Int. 2018, 9, 44–50.
- Parmaksiz, M.; Dogan, A.; Odabas, S.; Elcin, A.E.; Elcin, Y.M. Clinical applications of decellularized extracellular matrices for tissue engineering and regenerative medicine. Biomed. Mater. 2016, 11, 1–14.
- Magden, G.K.; Vural, C.; Bayrak, B.Y.; Ozdogan, C.Y.; Kenar, H. Composite sponges from sheep decellularized small intestinal submucosa for treatment of diabetic wounds. J. Biomater. Appl. 2021, 36, 1–15.
- Andree, B.; Bar, A.; Haverich, A.; Hilfiker, A. Small intestinal submucosa segments as matrix for tissue engineering: Review. Tissue Eng. Part B Rev. 2013, 19, 279–291.
- Lucich, E.A.; Rendon, J.L.; Valerio, I.L. Advances in addressing full-thickness skin defects: A review of dermal and epidermal substitutes. Regen. Med. 2018, 13, 443–456.
- Brown, B.N.; Londono, R.; Tottey, S.; Zhang, L.; Kukla, K.A.; Wolf, M.T.; Daly, K.A.; Reing, J.E.; Badylak, S.F. Macrophage phenotype as a predictor of constructive remodeling following the implantation of biologically derived surgical mesh materials. Acta Biomater. 2012, 8, 978–987.
- Badylak, S.F.; Valentin, J.E.; Ravindra, A.K.; McCabe, G.P.; Stewart-Akers, A.M. Macrophage phenotype as a determinant of biologic scaffold remodeling. Tissue Eng. Part A 2008, 14, 1835–1842.
- Shi, L.; Ramsay, S.; Ermis, R.; Carson, D. In vitro and in vivo studies on matrix metalloproteinases interacting with small intestine submucosa wound matrix. Int. Wound J. 2012, 9, 44–53.
- Krishnaswamy, V.R.; Mintz, D.; Sagi, I. Matrix metalloproteinases: The sculptors of chronic cutaneous wounds. Biochim. Biophys. Acta Mol. Cell. Res. 2017, 1864, 2220–2227.
- Lazaro, J.L.; Lzzo, V.; Meaume, S.; Davies, A.H.; Lobmann, R.; Uccioli, L. Elevated levels of matrix metalloproteinases and chronic wound healing_an updated review of clinical evidence. J. Wound Care 2016, 25, 1–11.
- Brown-Etris, M.; Milne, C.T.; Hodde, J.P. An extracellular matrix graft (Oasis® wound matrix) for treating fullthickness pressure ulcers: A randomized clinical trial. J. Tissue Viability 2019, 28, 21–26.
- Hodde, J.P.; Hiles, M.C.; Metzger, D.W. Characterization of the local wound environment following treatment of chronic leg ulcers with SIS wound matrix. J. Tissue Viability 2020, 29, 42–47.
- Xue, S.L.; Liu, K.; Parolini, O.; Wang, Y.; Deng, L.; Huang, Y.C. Human acellular amniotic membrane implantation for lower third nasal reconstruction: A promising therapy to promote wound healing. Burn. Trauma 2018, 6, 1–8.
- Farhadihosseinabadi, B.; Farahani, M.; Tayebi, T.; Jafari, A.; Biniazan, F.; Modaresifar, K.; Moravvej, H.; Bahrami, S.; Redl, H.; Tayebi, L.; et al. Amniotic membrane and its epithelial and mesenchymal stem cells as an appropriate source for skin tissue engineering and regenerative medicine. Artif. Cells Nanomed. Biotechnol. 2018, 46, 431–440.
- Dadkhah Tehrani, F.; Firouzeh, A.; Shabani, I.; Shabani, A. A review on modifications of amniotic membrane for biomedical applications. Front. Bioeng. Biotechnol. 2020, 8, 1–25.
- Liang, X.; Zhou, L.; Yan, J. Amniotic membrane for treating skin graft donor sites: A systematic review and meta-analysis. Burns 2020, 46, 621–629.
- Su, Y.N.; Zhao, D.Y.; Li, Y.H.; Yu, T.Q.; Sun, H.; Wu, X.Y.; Zhou, X.Q.; Li, J. Human amniotic membrane allograft, a novel treatment for chronic diabetic foot ulcers: A systematic review and meta-analysis of randomised controlled trials. Int. Wound J. 2020, 17, 1–12.
- Negron, L.; Lun, S.; May, B.C. Ovine forestomach matrix biomaterial is a broad spectrum inhibitor of matrix metalloproteinases and neutrophil elastase. Int. Wound J. 2014, 11, 392–397.





