
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Yannick Rossez | + 2441 word(s) | 2441 | 2021-11-15 10:24:34 | | | |
| 2 | Amina Yu | + 56 word(s) | 2497 | 2021-11-16 04:52:24 | | |
Video Upload Options
G. mellonella, also known as a wax moth, belongs to Lepidoptera order from the Pyralidae family. This moth is distributed worldwide, and is commercially available for fishing or to feed reptiles and birds, making them readily accessible. The last larval stage of this insect has been utilized as a host model to extensively study bacteria and fungi pathogenesis, including Acinetobacter baumannii
1. Introduction
Over the past decades, Acinetobacter baumannii has widely emerged as one of the major causes of highly invasive nosocomial pathogen infections in the health system [1]. Infections by this microorganism are responsible for increased morbidity and mortality, and make a huge burden to patients and hospitals [2]. As the top concerning microorganism on the global priority list ranked by the World Health Organization (WHO) [3], A. baumannii is a multi-drug resistant (MDR) bacterium which needs new drug development [4]. Therefore, the screening of the most adapted animal models for studying pathogenic mechanisms and therapeutic strategies before clinical therapies is particularly critical.
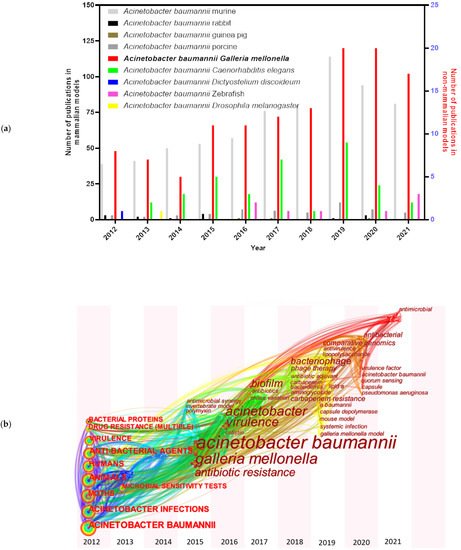
A series of animal models have been examined and established for A. baumannii studies, including mammalian and non-mammalian models. Murine models [5] are still the predominant mammalian models in A. baumannii researches, though some other mammalian models have also been tested, such as rabbits [6], guinea pigs [7], and porcine models [8] ( Figure 1 a). A. baumannii is frequently associated with pneumonia, making small rodent lung infection models well suited for these bacteria [9]. However, increasing costs and growing ethical concerns made the use of rodents more difficult [10]. Non-mammalian models, such as Galleria mellonella (greater wax moth) [11], Caenorhabditis elegans (roundworm) [12], Dictyostelium discoideum (slime mold) [13], Danio rerio (zebrafish) [14] and Drosophila melanogaster (common fruit fly) [9], are also informative to decipher virulence factors needed during host–pathogen interactions of A. baumannii . Among them, G. mellonella caterpillars have attracted more and more attention in the last ten years ( Figure 1 a). The keywords for each node distributed in time-zone visualization ( Figure 1 b) indicate an increased interest towards the G. mellonella model system. The research involving G. mellonella model mainly focused on A. baumannii pathogenicity factors (such as surface antigen proteins and efflux pump) and drug therapies.
The benefits of using G. mellonella models are numerous. G. mellonella produce a huge progeny quantity with a short life cycle, and are inexpensive because they are easy to rear without special laboratory infrastructure. The possibility of using many animals per experiment makes them eligible for high-throughput studies. The relatively large size of the larvae (12–20 mm) allows precise quantification of the inoculation, and facilitates handling for tissue extraction and histological analysis [15][16]. Importantly, there is no ethical approval requirement for research on G. mellonella [17].
Despite a large number of articles describing the feasibility and safety of G. mellonella for microbial studies [18], its value for drug-resistant microorganisms remains to be explored. In this review, we highlight why G. mellonella can be used as a model for MDR A. baumannii infection, the contributions of this model to study A. baumannii pathogenicity, and to target the most effective and prospective therapy strategies to fight A. baumannii infection.
2. G. mellonella-Based Model
G. mellonella has a rapid life cycle with four developmental stages: egg; larvae; pupa; and adult moth [19] ( Figure 2 a). Differences in temperature and humidity affect the developmental speed, with a full life cycle under favorable conditions being only 8–12 weeks [20]. The white dome-shaped eggs hatch to larvae in about 1–2 weeks at 28–34 °C [20]. The creamy-colored larvae pass through 8–10 molting stages in 5–6 weeks until cocoon development [20]. After 2–3 weeks of incubation, the reddish-brown pupa evolves into a pale cream moth [20].
Insects’ innate immune system has been well documented to protect them against infection from a broad spectrum of pathogens [21]. Genome research has shown that larvae have many homologous genes to humans, who participate in pathogen recognition and signal transduction [22]. In G. mellonella , the innate immune system is constituted by cuticle, cellular, and humoral immune defense [23].
The cellular immune system is mediated by phagocytic cells, called hemocytes, which are mainly responsible of encapsulation, nodulation, and phagocytosis [17][24] ( Figure 2 b). To date, six out of the eight types of hemocytes found in insects have been identified to be responsible of these functions in G. mellonella (plasmatocytes, granulocytes, prohemocytes, spherulocytes, coagulocytes, and oenocytoids) [15][25]. Firstly, granular cells attack the penetrated microorganisms, then, the process promotes the attachment of plasmatocytes to form a layer of cells, resulting in encapsulation and nodulation. Phagocytosis is similar to human cellular defense reactions with the participation of hemocytes [18]. The humoral immune response is highly regulated by soluble effectors, such as complement-like proteins (opsonins), melanin, and antimicrobial peptides (AMPs), which play a role in melanization, hemolymph clotting, and primary immunization [26] ( Figure 2 c).
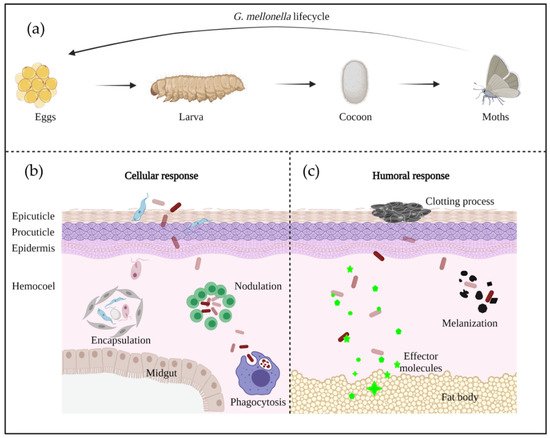
In the early stage of A. baumannii invasion, the larval immune response is activated, and struggles against A. baumannii virulence factors. If the infection is controlled by the immune system, the larvae will survive—alternatively, the larvae will continue melanization and finally die. The two different responses are dependent of the phagocytosis by hemocytes, or the melanization caused by the deposition of melanin around microorganisms [27].
3. Experimental Design Suitable for G. mellonella/A. baumannii Interaction
Generally, the larvae are employed at the 5th to 6th instar, at about 2–3 cm length and a weight of around 250–350 mg. The spontaneous mobility of larvae is a good indication of their viability [20][28]. For one experiment, the larvae are conventionally divided into three groups of about 10 to 20 individuals, one group inoculated with PBS, one group with bacteria sub-divided by the different conditions/strains needed, and one group without injection. In Table 1 and Table 2 , the inoculation methods, culture conditions, and larval detection indicators are listed. These different studies have described virulence factors of A. baumannii ( Table 1 ) and antimicrobial agents tested against A. baumannii ( Table 2 ) in G. mellonella .
| A. baumannii | Larva/ Group |
Larva Inoculation | Larva Incubation | Refs | |||||
|---|---|---|---|---|---|---|---|---|---|
| Pathogenicity | Strains and Mutants | Style | Volume/Larva | Concentration | Temp | Time | |||
| Virulence factors | |||||||||
| Phospholipases | Phospholipases C | ΔplcN | 20 | Injection | 10 µL | 2 × 106 CFU/mL | 37 °C | 8 days | [29][30] |
| ATCC 19606T, plc2::aph, plc1::aph-FRT, plc1::ermAM/plc2::aph | 10 | Injection | 1 × 105 CFU | 37 °C | 5 days | ||||
| Phospholipases D | ATCC 19606T, Δpld | 16 | Injection | 10 µL | 1 × 106 CFU/mL | 37 °C | 4 days | [31] | |
| Membrane proteins | Surface antigen protein 1 (SurA1) | ATCC 17978, CCGGD201101, ΔSurA1 | 20 | Injection | 20 µL | 1 × 106 CFU/mL | 37 °C | 7 days | [32] |
| Capsular polysaccharides and LOS | Capsule genes, epsA and ptk | AB5075, AB5075 epsA::Tn5, AB5075 ptk::Tn5 | - | Injection | 5 µL | 1 × 107 CFU/mL | 37 °C | 5 days | [33] |
| K locus | MDR-ZJ06, ΔgnaA | - | Injection | 10 µL | 1 × 108 CFU/mL | 37 °C | 3 days | [34] | |
| ptk gene | AB5075, Δptk | - | Injection | - | 1 × 105, 1 × 106 CFU | 37 °C | 6 days | [35] | |
| LOS | ATCC 17978, ΔlpxO, ΔlpxO::Tn7lpxO | 10 | Injection | 10 µL | 5 × 104 CFU | 37 °C | 3 days | [36] | |
| Protein secretion system | Type VI secretion system (T6SS) | DSM30011, ΔtssM | 20 | Injection | 10 µL | 1 × 105 CFU | 37 °C | - | [37][38] |
| 17978, 17978 ΔtssM | 10 | Injection | 5 µL | 106–107 CFU | 37 °C | 40–60 h | |||
| Metal acquisition systems | Iron acquisition | ATCC 19606T, basD, bauA | 30, 10 | Injection | 5 µL | 1 × 102, 1 × 105 CFU | 37 °C | 18 h/6 days | [39][40][41][42] |
| A118, ATCC 19606T, ATCC 17978 | - | Injection | - | 1 × 105 CFU | 37 °C | 6 days | |||
| ATCC 19606T, ΔbasD | 30 | Injection | 10 µL | OD600: 0.2 | 37 °C | 72 h | |||
| ATCC 19606T, entA::aph, tonB1::aph, tonB2::aacC1, tonB1::aph tonB2::aacC1 | 10 | Injection | - | 1 × 105 CFU | 37 °C | 6 days | |||
| Zinc acquisition | AB5075, znuB::Tn | - | Injection | - | 1 × 106 CFU | 37 °C | 0 h, 4 h | [35] | |
| Antimicrobial resistance | |||||||||
| β-lactamases | AB5075, ZJ06, LS01, ATCC 17978 | 10 | Injection | 10 µL | OD600: 0.1 | 37 °C | 72 h | [43] | |
| Efflux pumps | ATCC 17978, A1S | 16 | Injection | 10 µL | OD600: 0.5 | 37 °C | 96 h | [44] | |
| Permeability defects | ATCC 19606, ΔkupΔtrkΔkdp, ΔkupΔtrk | 20 | Injection | 10 µL | 1 × 106 CFU | 37 °C | 6 days | [45] | |
| Aminoglycoside modifying enzymes | AbA155 | 10 | Injection | 5 µL | 5 × 105 CFU | 37 °C | >120 h | [46] | |
| Alternation of target sites | MB_2, MB_6C, MB_23C, MB_177, MB_90, MB_119, SG3161, SG3166 | 10 | Injection | - | 1 × 105 CFU | 37 °C | 96 h | [47] | |
| Dissemination | |||||||||
| Quorum sensing | 3-hydroxy-C12-homoserine lactone | M2, aba1::Km | 16 | Injection | 10 µL | >0.5 log CFU | 37 °C | 6 days | [27] |
| abaM gene | AB5075, abaI::T26, abaM::T26 | 10 | Injection | - | 2 × 104 CFU, 2 × 105 CFU | 37 °C | 120 h | [48] | |
| Biofilm | NCTC 12156, NCTC 10303, ATCC 17978, NCTC 13302, W1, NCTC 13423, ATCC BAA-1710, NCTC 13424, ATCC BAA-1709, UKA1-UKA19 | 10 | Injection | - | 1 × 105, 1 × 106 CFU | 37 °C | 5 days | [49] | |
| Motility | ATCC 17978, 129/ddc, 277/dat | 16 | Injection | 5 µL | 3 × 105 CFU | 37 °C | 5 days | [50] | |
| Others | |||||||||
| Stress response | Reactive oxygen species (ROS) resistance | ATCC 17978, ATCC 17978 sod2343::Km, ATCC 17978 sod2343::Km pWHsod2343 | 16, 10 | Injection | 5 µL | 3 × 105 CFU, 1.5 × 106 CFU | 37 °C, −80 °C | 5 days, immediately | [51] |
| Temperature | ATCC 17978 | - | Injection | 10 µL | 1 × 106 CFU/mL | 28 °C, 37 °C | 72 h | [52] | |
| Ethanol | ATCC 19606T | 30 | Injection | - | 1 × 105 CFU | 37 °C | 6 days | [53] | |
| Phase-variable switch | AB5075 opaque, AB5075 translucent | 10 | Injection | - | 3 × 104 CFU | 37 °C | 24 h | [54][55] | |
| AB5075, ΔompR, ΔenvZ, ΔompR ΔenvZ | 30 | Injection | - | 103–104 CFU | 37 °C | 5 days | |||
| Category | A. baumannii | Treatment Type | Dose | Time | Refs | |
|---|---|---|---|---|---|---|
| Volume/Larva | Concentration | |||||
| AMPs | ||||||
| Amphiphilic peptide zp3 | - | Post-treatment | 10 µL | 200–800 mg/kg | 30 min | [56] |
| Anti-lpxB pPNA | MDR | Post-treatment | 10 µL | 75 mg/kg | 1 h | [57] |
| PNA (RXR)4 XB | MDR | Post-treatment | 10 µL | 150/600 µM | 30 min | [58] |
| Antibiotics | ||||||
| Colistin | MDR | Post-treatment | 10 µL | 2.5 mg/kg | 30 min | [57][59][60][61][62][63] |
| - | Post-treatment | 10 µL | 2.5 mg/kg | 30 min | ||
| Clinical isolate | Post-treatment | 10 µL | 2.5 mg/kg | 2 h | ||
| Carbapenem-resistant | Post-treatment | 10 µL | 2.5 mg/kg | 2 h | ||
| Colistin-resistant | Post-treatment | 5 µL | 2.5 mg/kg | 30 ± 5 min | ||
| MDR | Post-treatment | 10 µL | 40 mg/kg | - | ||
| MDR | Post-treatment | 10 µL | 2 mg/kg | 1 h | ||
| Cefozopran | MDR | Post-treatment | 10 µL | 40 mg/kg | - | [63] |
| Ciprofloxacin | - | Post-treatment | - | 10 mg/kg | 20 min | [64] |
| Clarithromycin | MDR | Pre-treatment | 5 µL | 25 mg/kg | 2.5 h | [65] |
| Cotrimoxazole | Carbapenem-resistant | Post-treatment | 10 µL | 10 mg/kg | 2 h | [62] |
| Doripenem | Colistin-resistant | Post-treatment | 5 µL | 7.5 mg/kg | 30 ± 5 min | [66] |
| Gentamicin | - | Post-treatment | - | 8 mg/kg | 20 min | [59][64] |
| - | Post-treatment | - | 8 mg/kg | 20 min | ||
| Imipenem | MDR | Post-treatment | - | 5 mg/mL | 30 min | [67] |
| Levofloxacin | MDR | Post-treatment | 10 µL | 6.7 mg/kg | 2 h | [61] |
| Meropenem | Clinical isolate | Post-treatment | 10 µL | 4 mg/kg | 1 h | [64][68] |
| - | Post-treatment | - | 20 mg/kg | 20 min | ||
| Minocycline | MDR | Post-treatment | 10 µL | 40 mg/kg | - | [63] |
| Mitomycin C | - | Post-treatment | - | 13–16 mg/kg | 2–5 min | [69] |
| Netropsin | Clinical isolate | Post-treatment | 5 µL | 12.5 mg/L | 30 min | [70] |
| Novobiocin | MDR | Post-treatment | 10 µL | 100 mg/kg | 3 h | [71] |
| Polymyxin B | Clinical isolate | Post-treatment | 5 µL | 4 mg/L | 30 min | [63][70] |
| MDR | Post-treatment | 10 µL | 40 mg/kg | - | ||
| Rifampicin | MDR | Post-treatment | 2 µL | 2.5, 5, 10 mg/kg | 30 min | [72] |
| Sitafloxacin | MDR | Post-treatment | 10 µL | 40 mg/kg | - | [63] |
| Teicoplanin | MDR | Post-treatment | 10 µL | 10 mg/kg | 30 min | [59] |
| Telavancin | - | Post-treatment | 10 µL | 10 mg/kg | 30 min | [60] |
| Tetracycline | MDR | Post-treatment | 10 µL | 40 mg/kg | - | [63] |
| Tigecycline | MDR | Post-treatment | 10 µL | 40 mg/kg | - | [63] |
| Vancomycin | Colistin-resistant | Post-treatment | 5 µL | 15 mg/kg | 30 ± 5 min | [66] |
| Cotrimoxazole/colistin | Carbapenem-resistant | Post-treatment | 10 µL | 10 mg/kg + 2.5 mg/kg | 2 h | [62] |
| Daptomycin/colistin | MDR | Post-treatment | - | 4 mg/L + 2.5 mg/L | 2 h | [73] |
| Doripenem/Vancomycin | Colistin-resistant | Post-treatment | 5 µL | 7.5 mg/kg + 15 mg/kg | 30 ± 5 min | [66] |
| Doripenem/Vancomycin/colistin | Colistin-resistant | Post-treatment | 5 µL | 7.5 mg/kg + 15 mg/kg + 2.5 mg/kg | 30 ± 5 min | [66] |
| Levofloxacin/colistin | MDR | Post-treatment | 10 µL | 6.7 mg/kg + 2.5 mg/kg | 2 h | [61] |
| Polymyxin B/netropsin | Clinical isolate | Post-treatment | 5 µL | 4 mg/L + 12.5 mg/L | 30 min | [70] |
| Teicoplanin/colistin | MDR | Post-treatment | 10 µL | 10 mg/kg + 2.5 mg/kg | 30 min | [59] |
| Telavancin/colistin | - | Post-treatment | 10 µL | 10 mg/kg + 2.5 mg/kg | 30 min | [60] |
| Vancomycin/colistin | MDR | Post-treatment | 10 µL | 15 mg/kg + 2.5 mg/kg | 2 h | [59][74] |
| MDR | Post-treatment | 10 µL | 10 mg/kg + 2.5 mg/kg | 30 min | ||
| Others | ||||||
| Anti-lpxB pPNA/colistin | MDR | Post-treatment | 10 µL | 75 mg/kg + 2 mg/kg | 1 h | [57] |
| Bacteriophage | Carbapenem-resistant | Post-treatment | 5 µL | 1 × 1010, 1 × 109 PFU/mL | 30 min | [11][64][67][75] |
| - | Post-treatment | - | MOI ≈ 1 | 20 min | ||
| MDR | Post-treatment | 10 µL | 5.107 PFU, MOI = 100 | 30 min | ||
| Carbapenem-resistant | Post-treatment | 10 µL | 104 pfu | 30 min | ||
| Capsule depolymerase Dpo48 | Extensive drug-resistant | Pre-treatment, post-treatment | 10 µL | 50 µg/mL, 5 µg | 1 h, 5 min | [76] |
| Epicatechin | MDR | Post-treatment | - | 40 mg/kg | 30 min | [77] |
| Homodimeric Tobramycin Adjuvant/Novobiocin | MDR | Post-treatment | 10 µL | 25/50 mg/kg + 25/50 mg/kg | 3 h | [71] |
| Gallium nitrate | MDR | Post-treatment | - | 1.2 mmol/kg | 15 min | [78] |
| Gallium protoporphyrin IX | - | Simultaneously | 5 µL | 20, 40 µg/mL | - | [79] |
| Manganese (i) tricarbonyl complexes | MDR | Post-treatment | - | 5 mg/kg | 30 min | [80] |
| SCH-79797 | MDR | Simultaneously | 66.6 µg/larva | - | [81] | |
| Silver acetate | Carbapenem-resistant | Post-treatment | - | 0, 10, 20 mg/kg | 30 min | [82] |
| Theaflavin | MDR | Post-treatment | - | 20 mg/kg | 30 min | [77] |
| Theaflavin/Epicatechin | MDR | Post-treatment | - | 20 mg/kg + 40 mg/kg | 30 min | [77] |
| Bacteriophage/Ciprofloxacin | - | Post-treatment | - | MOI ≈ 1 + 10 mg/kg | 20 min | [64] |
| Bacteriophage/Gentamicin | - | Post-treatment | - | MOI ≈ 1 + 8 mg/kg | 20 min | [64] |
| Bacteriophage/Meropenem | - | Post-treatment | - | MOI ≈ 1 + 20 mg/kg | 20 min | [64] |
| Endolysin/colistin | - | Post-treatment | 10 µL | 25 µg/mL + 1/4 MIC | 1 h | [83] |
Notes: MDR: multi-drug resistant. Pre-treatment/post-treatment: the antimicrobial agents were added before/after the A. baumannii infection. MOI: multiplicity of infection. CFU: colony forming unit. Time: the period between the first and second injection.
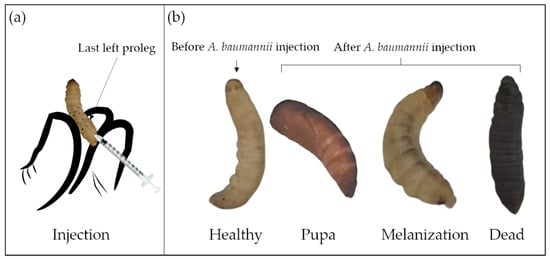
After 24 h of starvation at room temperature, three inoculation methods have been described to work with G. mellonella : topical application [84]; force-feeding [85]; and injection [11]. For A. baumannii infection, only the injection method into the hemocoel of the larval cuticle of the last left proleg [27] has been used ( Figure 3 a). For drug treatment, the correct timing of drug administration is also important, commonly within 3 h after A. baumannii injection. In some studies, drug application before or simultaneously with A. baumannii infection has been reported, but such cases are rare [76][78][81]. Compared to the two other methods, the injection has the advantage to accurately deliver the inoculum, and is therefore more reproducible [27]. However, the control group, injected only with buffer or medium, is crucial to ensure that the death of larvae is not caused by trauma or solvents.
G. mellonella larvae can be maintained at different temperatures after injection, between 15 °C to over 37 °C [86]. In order to better understand the interaction between the host and the pathogen in an environment closer to the mammalian organism, 37 °C is the most employed temperature for A. baumannii infection [16]. The viability, motility, and virulence of A. baumannii at 28 °C [52] and 30 °C [27] were also studied in order to assess the adaptability of the different clinical strains’ response to environmental changes. The incubation duration inside the larvae usually varies from few hours to few days. Experiments suggest that too short periods (<4 h) are not conducive to an accurate evaluation of A. baumannii virulence or drug efficacy. Conversely, after too long (>8 days) time periods, the larvae metamorphose into moths.
The G. mellonella larvae assessments could be larval mobility [77], mortality/survival rate [59], histological analysis [11], and bacterial numbers recovered after incubation [51]. Table 3 introduces the health index scoring system to evaluate the larval health status, including larvae mobility, cocoon formation, melanization, and survival [86]. The movement, observed by touching and the melanization, visible by naked eyes, are keys to distinguish the larval morbidity after A. baumannii infection ( Figure 3 b) [77]. Though the A. baumannii virulence overcomes the larval immune system over time, the larval movement gradually decreases, and the melanization progresses gradually. Complete melanization indicates death. Mortality/survival rate is the most monitored indicator, which directly reflects A. baumannii virulence. The survival percentage, usually characterized by the Kaplan–Meier curve, is investigated every 24 h [87]. Histological analyses are essential for studying host–defense mechanisms and pathogen infection pathways. A rare study associated with tissue damage, fat body, and muscle layer melanization has been reported for A. baumannii infected larvae [11].
| Category | Description | Score |
|---|---|---|
| Activity | No movement | 0 |
| Minimal movement on stimulation | 1 | |
| Move when stimulated | 2 | |
| Move without stimulation | 3 | |
| Cocoon formation | No cocoon | 0 |
| Partial cocoon | 0.5 | |
| Full cocoon | 1 | |
| Melanization | Black larvae | 0 |
| Black spots on brown larvae | 1 | |
| ≥3 spots on beige larvae | 2 | |
| <3 spots on beige larvae | 3 | |
| No melanization | 4 | |
| Survival | Dead | 0 |
| Alive | 2 |
References
- Dijkshoorn, L.; Nemec, A.; Seifert, H. An increasing threat in hospitals: Multidrug-resistant Acinetobacter baumannii. Nat. Rev. Microbiol. 2007, 5, 939–951.
- Patamatamkul, S.; Klungboonkrong, V.; Praisarnti, P.; Jirakiat, K. A case-control study of community-acquired Acinetobacter baumannii pneumonia and melioidosis pneumonia in northeast Thailand: An emerging fatal disease with unique clinical features. Diagn. Microbiol. Infect. Dis. 2017, 87, 79–86.
- Willyard, C. The drug-resistant bacteria that pose the greatest health threats. Nature 2017, 543, 15.
- Moubareck, C.A.; Halat, D.H. Insights into Acinetobacter baumannii: A review of microbiological, virulence, and resistance traits in a threatening nosocomial pathogen. Antibiotics 2020, 9, 119.
- McConnell, M.J.; Actis, L.; Pachón, J. Acinetobacter baumannii: Human infections, factors contributing to pathogenesis and animal models. FEMS Microbiol. Rev. 2013, 37, 130–155.
- Spicer, P.P.; Shah, S.R.; Henslee, A.M.; Watson, B.M.; Kinard, L.A.; Kretlow, J.D.; Bevil, K.; Kattchee, L.; Bennett, G.N.; Demian, N.; et al. Evaluation of antibiotic releasing porous polymethylmethacrylate space maintainers in an infected composite tissue defect model. Acta Biomater. 2013, 9, 8832–8839.
- Bernabeu-Wittel, M.; Pichardo, C.; García-Curiel, A.; Pachón-Ibáñez, M.E.; Ibáñez-Martínez, J.; Jiménez-Mejías, M.E.; Pachón, J. Pharmacokinetic/pharmacodynamic assessment of the in-vivo efficacy of imipenem alone or in combination with amikacin for the treatment of experimental multiresistant Acinetobacter baumannii pneumonia. Clin. Microbiol. Infect. 2005, 11, 319–325.
- Zurawski, D.V.; Black, C.C.; Alamneh, Y.A.; Biggemann, L.; Banerjee, J.; Thompson, M.G.; Wise, M.C.; Honnold, C.L.; Kim, R.K.; Paranavitana, C.; et al. A Porcine Wound Model of Acinetobacter baumannii Infection. Adv. Wound Care 2019, 8, 14–27.
- Cerqueira, G.M.; Peleg, A.Y. Insights into Acinetobacter baumannii pathogenicity. IUBMB Life 2011, 63, 1055–1060.
- Morris, F.C.; Dexter, C.; Kostoulias, X.; Uddin, M.I.; Peleg, A.Y. The Mechanisms of Disease Caused by Acinetobacter baumannii. Front. Microbiol. 2019, 10, 1601.
- Jeon, J.; Park, J.H.; Yong, D. Efficacy of bacteriophage treatment against carbapenem-resistant Acinetobacter baumannii in Galleria mellonella larvae and a mouse model of acute pneumonia. BMC Microbiol. 2019, 19, 70.
- Scott, E.; Holden-Dye, L.; O’Connor, V.; Wand, M.E. Intra Strain Variation of the Effects of Gram-Negative ESKAPE Pathogens on Intestinal Colonization, Host Viability, and Host Response in the Model Organism Caenorhabditis elegans. Front. Microbiol. 2020, 10, 3113.
- Iwashkiw, J.A.; Seper, A.; Weber, B.S.; Scott, N.E.; Vinogradov, E.; Stratilo, C.; Reiz, B.; Cordwell, S.J.; Whittal, R.; Schild, S.; et al. Identification of a general O-linked protein glycosylation system in Acinetobacter baumannii and its role in virulence and biofilm formation. PLoS Pathog. 2012, 8, e1002758.
- Roy, R.; You, R.I.; Der Lin, M.; Lin, N.T. Mutation of the carboxy-terminal processing protease in Acinetobacter baumannii affects motility, leads to loss of membrane integrity, and reduces virulence. Pathogens 2020, 9, 322.
- Pereira, M.F.; Rossi, C.C.; Da Silva, G.C.; Rosa, J.N.; Bazzolli, D.M.S. Galleria mellonella as an infection model: An in-depth look at why it works and practical considerations for successful application. Pathog. Dis. 2020, 78, ftaa056.
- Cook, S.M.; McArthur, J.D. Developing Galleria mellonella as a model host for human pathogens. Virulence 2013, 4, 350–353.
- Tsai, C.J.Y.; Loh, J.M.S.; Proft, T. Galleria mellonella infection models for the study of bacterial diseases and for antimicrobial drug testing. Virulence 2016, 7, 214–229.
- Singkum, P.; Suwanmanee, S.; Pumeesat, P.; Luplertlop, N. A powerful in vivo alternative model in scientific research: Galleria mellonella. Acta Microbiol. Immunol. Hung. 2019, 66, 31–55.
- Jorjão, A.L.; Oliveira, L.D.; Scorzoni, L.; Figueiredo-Godoi, L.M.A.; Prata, M.C.A.; Jorge, A.O.C.; Junqueira, J.C. From moths to caterpillars: Ideal conditions for Galleria mellonella rearing for in vivo microbiological studies. Virulence 2018, 9, 383–389.
- Firacative, C.; Khan, A.; Duan, S.; Ferreira-Paim, K.; Leemon, D.; Meyer, W. Rearing and maintenance of Galleria mellonella and its application to study fungal virulence. J. Fungi 2020, 6, 130.
- Strand, M.R. The insect cellular immune response. Insect Sci. 2008, 15, 1–14.
- Mikulak, E.; Gliniewicz, A.; Przygodzka, M.; Solecka, J. Galleria mellonella L. as model organism used in biomedical and other studies. Przegl. Epidemiol. 2018, 72, 57–73.
- Kavanagh, K.; Reeves, E.P. Exploiting the potential of insects for in vivo pathogenicity testing of microbial pathogens. FEMS Microbiol. Rev. 2004, 28, 101–112.
- Tojo, S.; Naganuma, F.; Arakawa, K.; Yokoo, S. Involvement of both granular cells and plasmatocytes in phagocytic reactions in the greater wax moth, Galleria mellonella. J. Insect Physiol. 2000, 46, 1129–1135.
- Neuwirth, M. The structure of the hemocytes of Galleria mellonella (Lepidoptera). J. Morphol. 1973, 139, 105–123.
- Pereira, T.C.; de Barros, P.P.; de Oliveira Fugisaki, L.R.; Rossoni, R.D.; Ribeiro, F.d.C.; de Menezes, R.T.; Junqueira, J.C.; Scorzoni, L. Recent advances in the use of Galleria mellonella model to study immune responses against human pathogens. J. Fungi 2018, 4, 128.
- Peleg, A.Y.; Jara, S.; Monga, D.; Eliopoulos, G.M.; Moellering, R.C.; Mylonakis, E. Galleria mellonella as a model system to study Acinetobacter baumannii pathogenesis and therapeutics. Antimicrob. Agents Chemother. 2009, 53, 2605–2609.
- Andrea, A.; Krogfelt, K.A.; Jenssen, H. Methods and challenges of using the greater wax moth (Galleria mellonella) as a model organism in antimicrobial compound discovery. Microorganisms 2019, 7, 85.
- Kareem, S.M.; Al-Kadmy, I.M.S.; Al-Kaabi, M.H.; Aziz, S.N.; Ahmad, M. Acinetobacter baumannii virulence is enhanced by the combined presence of virulence factors genes phospholipase C (plcN) and elastase (lasB). Microb. Pathog. 2017, 110, 568–572.
- Fiester, S.E.; Arivett, B.A.; Schmidt, R.E.; Beckett, A.C.; Ticak, T.; Carrier, M.V.; Ghosh, R.; Ohneck, E.J.; Metz, M.L.; Jeffries, M.K.S.; et al. Iron-Regulated phospholipase C Activity contributes to the cytolytic activity and virulence of Acinetobacter baumannii. PLoS ONE 2016, 11, e0167068C.
- Stahl, J.; Bergmann, H.; Göttig, S.; Ebersberger, I.; Averhoff, B. Acinetobacter baumannii virulence is mediated by the concerted action of three phospholipases D. PLoS ONE 2015, 10, e0138360.
- Liu, D.; Liu, Z.S.; Hu, P.; Cai, L.; Fu, B.Q.; Li, Y.S.; Lu, S.Y.; Liu, N.N.; Ma, X.L.; Chi, D.; et al. Characterization of surface antigen protein 1 (SurA1) from Acinetobacter baumannii and its role in virulence and fitness. Vet. Microbiol. 2016, 186, 126–138.
- Regeimbal, J.M.; Jacobs, A.C.; Corey, B.W.; Henry, M.S.; Thompson, M.G.; Pavlicek, R.L.; Quinones, J.; Hannah, R.M.; Ghebremedhin, M.; Crane, N.J.; et al. Personalized therapeutic cocktail of wild environmental phages rescues mice from Acinetobacter baumannii wound infections. Antimicrob. Agents Chemother. 2016, 60, 5806–5816.
- Xu, Q.; Chen, T.; Yan, B.; Zhang, L.; Pi, B.; Yang, Y.; Zhang, L.; Zhou, Z.; Ji, S.; Leptihn, S.; et al. Dual Role of gnaA in Antibiotic Resistance and Virulence in Acinetobacter baumannii. Antimicrob. Agents Chemother. 2019, 63, e00694-19.
- Gebhardt, M.J.; Gallagher, L.A.; Jacobson, R.K.; Usacheva, E.A.; Peterson, L.R.; Zurawski, D.V.; Shuman, H.A. Joint transcriptional control of virulence and resistance to antibiotic and environmental stress in Acinetobacter baumannii. MBio 2015, 6, e01660-15.
- Bartholomew, T.L.; Kidd, T.J.; Pessoa, J.S.; Álvarez, R.C.; Bengoechea, J.A. 2-Hydroxylation of Acinetobacter baumannii Lipid a Contributes To Virulence. Infect. Immun. 2019, 87, e00066-19.
- Repizo, G.D.; Gagné, S.; Foucault-Grunenwald, M.L.; Borges, V.; Charpentier, X.; Limansky, A.S.; Gomes, J.P.; Viale, A.M.; Salcedo, S.P. Differential role of the T6SS in Acinetobacter baumannii virulence. PLoS ONE 2015, 10, e0138265.
- Weber, B.S.; Miyata, S.T.; Iwashkiw, J.A.; Mortensen, B.L.; Skaar, E.P.; Pukatzki, S.; Feldman, M.F. Genomic and Functional Analysis of the Type VI Secretion System in Acinetobacter. PLoS ONE 2013, 8, e55142.
- Gaddy, J.A.; Actis, L.A.; Arivett, B.A.; Mcconnell, M.J.; Rafael, L.R.; Pachón, J. Role of Acinetobactin-mediated iron acquisition functions in the interaction of Acinetobacter baumannii strain ATCC 19606T with human lung epithelial cells, Galleria mellonella caterpillars, and mice. Infect. Immun. 2012, 80, 1015–1024.
- Ramirez, M.S.; Penwell, W.F.; Traglia, G.M.; Zimbler, D.L.; Gaddy, J.A.; Nikolaidis, N.; Arivett, B.A.; Adams, M.D.; Bonomo, R.A.; Actis, L.A.; et al. Identification of Potential Virulence Factors in the Model Strain Acinetobacter baumannii A118. Front. Microbiol. 2019, 10, 1599.
- Fleming, I.D.; Krezalek, M.A.; Belogortseva, N.; Zaborin, A.; Defazio, J.; Chandrasekar, L.; Actis, L.A.; Zaborina, O.; Alverdy, J.C. Modeling Acinetobacter baumannii wound infections. J. Trauma Acute Care Surg. 2017, 82, 557–565.
- Zimbler, D.L.; Arivett, B.A.; Beckett, A.C.; Menke, S.M.; Actis, L.A. Functional features of TonB energy transduction systems of Acinetobacter baumannii. Infect. Immun. 2013, 81, 3382–3394.
- Zhou, H.; Larkin, P.M.K.; Huang, J.; Yao, Y.; Zhu, B.; Yang, Q.; Hua, X.; Zhou, J.; Yang, S.; Yu, Y. Discovery of a novel hypervirulent Acinetobacter baumannii strain in a case of community-acquired pneumonia. Infect. Drug Resist. 2020, 13, 1147–1153.
- Pérez-Varela, M.; Corral, J.; Aranda, J.; Barbé, J. Roles of efflux pumps from different superfamilies in the surface-associated motility and virulence of Acinetobacter baumannii ATCC 17978. Antimicrob. Agents Chemother. 2019, 63, e02190-18.
- König, P.; Averhoff, B.; Müller, V. K+ and its role in virulence of Acinetobacter baumannii. Int. J. Med. Microbiol. 2021, 311, 151516.
- Lopez, C.; Arivett, B.A.; Actis, L.A.; Tolmasky, M.E. Inhibition of AAC(6′)-Ib-mediated resistance to amikacin in Acinetobacter baumannii by an antisense peptide-conjugated 2′,4′-bridged nucleic acid-NC-DNA hybrid oligomer. Antimicrob. Agents Chemother. 2015, 59, 5798–5803.
- Gerson, S.; Betts, J.W.; Lucaßen, K.; Nodari, C.S.; Wille, J.; Josten, M.; Göttig, S.; Nowak, J.; Stefanik, D.; Roca, I.; et al. Investigation of Novel pmrB and eptA Mutations in Isogenic Acinetobacter baumannii Isolates Associated with Colistin Resistance and Increased Virulence In Vivo. Antimicrob. Agents Chemother. 2019, 63, e01586-18.
- López-Martín, M.; Dubern, J.F.; Alexander, M.R.; Williams, P. Abam regulates quorum sensing, biofilm formation, and virulence in Acinetobacter baumannii. J. Bacteriol. 2021, 203, e00635-20.
- Wand, M.E.; Bock, L.J.; Turton, J.F.; Nugent, P.G.; Mark Sutton, J. Acinetobacter baumannii virulence is enhanced in Galleria mellonella following biofilm adaptation. J. Med. Microbiol. 2012, 61, 470–477.
- Skiebe, E.; de Berardinis, V.; Morczinek, P.; Kerrinnes, T.; Faber, F.; Lepka, D.; Hammer, B.; Zimmermann, O.; Ziesing, S.; Wichelhaus, T.A.; et al. Surface-associated motility, a common trait of clinical isolates of Acinetobacter baumannii, depends on 1,3-diaminopropane. Int. J. Med. Microbiol. 2012, 302, 117–128.
- Heindorf, M.; Kadari, M.; Heider, C.; Skiebe, E.; Wilharm, G. Impact of Acinetobacter baumannii superoxide dismutase on motility, virulence, oxidative stress resistance and susceptibility to antibiotics. PLoS ONE 2014, 9, e101033.
- Malaka De Silva, P.; Chong, P.; Fernando, D.M.; Westmacott, G.; Kumara, A. Effect of incubation temperature on antibiotic resistance and virulence factors of Acinetobacter baumannii ATCC 17978. Antimicrob. Agents Chemother. 2018, 62, e01514-17.
- Nwugo, C.C.; Arivett, B.A.; Zimbler, D.L.; Gaddy, J.A.; Richards, A.M.; Actis, L.A. Effect of Ethanol on Differential Protein Production and Expression of Potential Virulence Functions in the Opportunistic Pathogen Acinetobacter baumannii. PLoS ONE 2012, 7, e51936.
- Tipton, K.A.; Dimitrova, D.; Rather, P.N. Phase-variable control of multiple phenotypes in Acinetobacter baumannii strain AB5075. J. Bacteriol. 2015, 197, 2593–2599.
- Tipton, K.A.; Rather, P.N. An ompRenvZ two-component system ortholog regulates phase variation, osmotic tolerance, motility, and virulence in Acinetobacter baumannii strain AB5075. J. Bacteriol. 2017, 199, e00705-16.
- Zeng, P.; Yi, L.; Xu, J.; Gao, W.; Xu, C.; Chen, S.; Chan, K.F.; Wong, K.Y. Investigation of antibiofilm activity, antibacterial activity, and mechanistic studies of an amphiphilic peptide against Acinetobacter baumannii. Biochim. Biophys. Acta—Biomembr. 2021, 1863, 183600.
- Martínez-Guitián, M.; Vázquez-Ucha, J.C.; Álvarez-Fraga, L.; Conde-Pérez, K.; Bou, G.; Poza, M.; Beceiro, A. Antisense inhibition of lpxB gene expression in Acinetobacter baumannii by peptide-PNA conjugates and synergy with colistin. J. Antimicrob. Chemother. 2020, 75, 51–59.
- Rose, M.; Lapuebla, A.; Landman, D.; Quale, J. In Vitro and in Vivo Activity of a Novel Antisense Peptide Nucleic Acid Compound Against Multidrug-Resistant Acinetobacter baumannii. Microb. Drug Resist. 2019, 25, 961–965.
- Hornsey, M.; Wareham, D.W. In vivo efficacy of glycopeptide-colistin combination therapies in a Galleria mellonella model of Acinetobacter baumannii infection. Antimicrob. Agents Chemother. 2011, 55, 3534–3537.
- Hornsey, M.; Phee, L.; Longshaw, C.; Wareham, D.W. In vivo efficacy of telavancin/colistin combination therapy in a Galleria mellonella model of Acinetobacter baumannii infection. Int. J. Antimicrob. Agents 2013, 41, 285–287.
- Wei, W.; Yang, H.; Hu, L.; Ye, Y.; Li, J. Activity of levofloxacin in combination with colistin against Acinetobacter baumannii: In vitro and in a Galleria mellonella model. J. Microbiol. Immunol. Infect. 2017, 50, 821–830.
- Khalil, M.A.F.; Moawad, S.S.; Hefzy, E.M. In vivo activity of co-trimoxazole combined with colistin against Acinetobacter baumannii producing oxa-23 in a Galleria mellonella model. J. Med. Microbiol. 2019, 68, 52–59.
- Nishida, S.; Ono, Y. Comparative analysis of the pathogenicity between multidrug-resistant Acinetobacter baumannii clinical isolates: Isolation of highly pathogenic multidrug-resistant A. baumannii and experimental therapeutics with fourth-generation cephalosporin cefozopran. Infect. Drug Resist. 2018, 11, 1715–1722.
- Grygorcewicz, B.; Roszak, M.; Golec, P.; Śleboda-taront, D.; Łubowska, N.; Górska, M.; Jursa-kulesza, J.; Rakoczy, R.; Wojciuk, B.; Dołęgowska, B. Antibiotics act with vb_abap_agc01 phage against Acinetobacter baumannii in human heat-inactivated plasma blood and Galleria mellonella models. Int. J. Mol. Sci. 2020, 21, 4390.
- Martin, S.E.; Melander, R.J.; Brackett, C.M.; Scott, A.J.; Chandler, C.E.; Nguyen, C.M.; Minrovic, B.M.; Harrill, S.E.; Ernst, R.K.; Manoil, C.; et al. Small Molecule Potentiation of Gram-Positive Selective Antibiotics against Acinetobacter baumannii. ACS Infect. Dis. 2019, 5, 1223–1230.
- O’Hara, J.A.; Ambe, L.A.; Casella, L.G.; Townsend, B.M.; Pelletier, M.R.; Ernst, R.K.; Shanks, R.M.Q.; Doi, Y. Activities of vancomycin-containing regimens against colistin-resistant Acinetobacter baumannii clinical strains. Antimicrob. Agents Chemother. 2013, 57, 2103–2108.
- Leshkasheli, L.; Kutateladze, M.; Balarjishvili, N.; Bolkvadze, D.; Save, J.; Oechslin, F.; Que, Y.-A.; Resch, G. Efficacy of newly isolated and highly potent bacteriophages in a mouse model of extensively drug-resistant Acinetobacter baumannii bacteraemia. J. Glob. Antimicrob. Resist. 2019, 19, 255–261.
- Tietgen, M.; Leukert, L.; Sommer, J.; Kramer, J.S.; Brunst, S.; Wittig, I.; Proschak, E.; Göttig, S. Characterization of the novel OXA-213-like β-lactamase OXA-822 from Acinetobacter calcoaceticus. J. Antimicrob. Chemother. 2021, 76, 626–634.
- Cruz-Muñiz, M.Y.; López-Jacome, L.E.; Hernández-Durán, M.; Franco-Cendejas, R.; Licona-Limón, P.; Ramos-Balderas, J.L.; Martinéz-Vázquez, M.; Belmont-Díaz, J.A.; Wood, T.K.; García-Contreras, R. Repurposing the anticancer drug mitomycin C for the treatment of persistent Acinetobacter baumannii infections. Int. J. Antimicrob. Agents 2017, 49, 88–92.
- Chung, J.H.; Bhat, A.; Kim, C.J.; Yong, D.; Ryu, C.M. Combination therapy with polymyxin B and netropsin against clinical isolates of multidrug-resistant Acinetobacter baumannii. Sci. Rep. 2016, 6, 28168.
- Idowu, T.; Ammeter, D.; Rossong, H.; Zhanel, G.G.; Schweizer, F. Homodimeric Tobramycin Adjuvant Repurposes Novobiocin as an Effective Antibacterial Agent against Gram-Negative Bacteria. J. Med. Chem. 2019, 62, 9103–9115.
- Jacobs, A.C.; Thompson, M.G.; Black, C.C.; Kessler, J.L.; Clark, L.P.; McQueary, C.N.; Gancz, H.Y.; Corey, B.W.; Moon, J.K.; Si, Y.; et al. AB5075, a highly virulent isolate of Acinetobacter baumannii, as a model strain for the evaluation of pathogenesis and antimicrobial treatments. MBio 2014, 5, e01076-14.
- Yang, H.; Chen, G.; Hu, L.; Liu, Y.; Cheng, J.; Li, H.; Ye, Y.; Li, J. In vivo activity of daptomycin/colistin combination therapy in a Galleria mellonella model of Acinetobacter baumannii infection. Int. J. Antimicrob. Agents 2015, 45, 188–191.
- Yang, H.; Lv, N.; Hu, L.; Liu, Y.; Cheng, J.; Ye, Y.; Li, J. In vivo activity of vancomycin combined with colistin against multidrug-resistant trains of Acinetobacter baumannii in a Galleria mellonella model. Infect. Dis. 2016, 48, 189–194.
- Zhou, W.; Feng, Y.; Zong, Z. Two new lytic bacteriophages of the Myoviridae family against carbapenem-resistant Acinetobacter baumannii. Front. Microbiol. 2018, 9, 850.
- Liu, Y.; Leung, S.S.Y.; Guo, Y.; Zhao, L.; Jiang, N.; Mi, L.; Li, P.; Wang, C.; Qin, Y.; Mi, Z.; et al. Corrigendum: The Capsule Depolymerase Dpo48 Rescues Galleria mellonella and Mice From Acinetobacter baumannii Systemic Infections. Front. Microbiol. 2019, 10, 545.
- Betts, J.W.; Hornsey, M.; Wareham, D.W.; La Ragione, R.M. In vitro and In vivo Activity of Theaflavin–Epicatechin Combinations versus Multidrug-Resistant Acinetobacter baumannii. Infect. Dis. Ther. 2017, 6, 435–442.
- Antunes, L.C.S.; Imperi, F.; Minandri, F.; Visca, P. In Vitro and In Vivo antimicrobial activities of gallium nitrate against multidrug-resistant Acinetobacter baumannii. Antimicrob. Agents Chemother. 2012, 56, 5961–5970.
- Arivett, B.A.; Fiester, S.E.; Ohneck, E.J.; Penwell, W.F.; Kaufman, C.M.; Relich, R.F.; Actis, L.A. Antimicrobial activity of gallium protoporphyrin IX against Acinetobacter baumannii strains displaying different antibiotic resistance phenotypes. Antimicrob. Agents Chemother. 2015, 59, 7657–7665.
- Güntzel, P.; Nagel, C.; Weigelt, J.; Betts, J.W.; Pattrick, C.A.; Southam, H.M.; La Ragione, R.M.; Poole, R.K.; Schatzschneider, U. Biological activity of manganese(i) tricarbonyl complexes on multidrug-resistant Gram-negative bacteria: From functional studies to: In vivo activity in Galleria mellonella. Metallomics 2019, 11, 2033–2042.
- Martin, J.K.; Wilson, M.Z.; Moore, G.M.; Sheehan, J.P.; Mateus, A.; Li, S.H.J.; Bratton, B.P.; Kim, H.; Rabinowitz, J.D.; Typas, A.; et al. A dual-mechanism antibiotic targets Gram-negative bacteria and avoids drug resistance. bioRxiv 2020.
- Mannix-Fisher, E.; McLean, S. The antimicrobial activity of silver acetate against Acinetobacter baumannii in a Galleria mellonella infection model. PeerJ 2021, 9, e11196.
- Blasco, L.; Ambroa, A.; Trastoy, R.; Bleriot, I.; Moscoso, M.; Fernández-Garcia, L.; Perez-Nadales, E.; Fernández-Cuenca, F.; Torre-Cisneros, J.; Oteo-Iglesias, J.; et al. In vitro and in vivo efficacy of combinations of colistin and different endolysins against clinical strains of multi-drug resistant pathogens. Sci. Rep. 2020, 10, 7163.
- Scully, L.R.; Bidochka, M.J. Serial passage of the opportunistic pathogen Aspergillus flavus through an insect host yields decreased saprobic capacity. Can. J. Microbiol. 2005, 51, 185–189.
- Fedhila, S.; Buisson, C.; Dussurget, O.; Serror, P.; Glomski, I.J.; Liehl, P.; Lereclus, D.; Nielsen-LeRoux, C. Comparative analysis of the virulence of invertebrate and mammalian pathogenic bacteria in the oral insect infection model Galleria mellonella. J. Invertebr. Pathol. 2010, 103, 24–29.
- Ramarao, N.; Nielsen-Leroux, C.; Lereclus, D. The insect Galleria mellonella as a powerful infection model to investigate bacterial pathogenesis. J. Vis. Exp. 2012, 70, 4392.
- Kumar Goel, M.; Khanna, P.; Kishore, J. Understanding survival analysis: Kaplan-Meier estimate. Int. J. Ayurveda Res. 2010, 1, 274–278.
- Wolf, D.M.; Vazirani, V.V.; Arkin, A.P. A microbial modified prisoner’s dilemma game: How frequency-dependent selection can lead to random phase variation. J. Theor. Biol. 2005, 234, 255–262.





