Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Marcin Włodarczyk | + 1400 word(s) | 1400 | 2021-11-04 04:28:52 | | | |
| 2 | Rita Xu | Meta information modification | 1400 | 2021-11-12 10:01:46 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Włodarczyk, M. Perianal Fistula with Crohn’s Disease. Encyclopedia. Available online: https://encyclopedia.pub/entry/15931 (accessed on 26 March 2026).
Włodarczyk M. Perianal Fistula with Crohn’s Disease. Encyclopedia. Available at: https://encyclopedia.pub/entry/15931. Accessed March 26, 2026.
Włodarczyk, Marcin. "Perianal Fistula with Crohn’s Disease" Encyclopedia, https://encyclopedia.pub/entry/15931 (accessed March 26, 2026).
Włodarczyk, M. (2021, November 12). Perianal Fistula with Crohn’s Disease. In Encyclopedia. https://encyclopedia.pub/entry/15931
Włodarczyk, Marcin. "Perianal Fistula with Crohn’s Disease." Encyclopedia. Web. 12 November, 2021.
Copy Citation
Crohn’s disease (CD) is a relapsing systemic inflammatory disease that can cause persistent transmural inflammation anywhere along the gastrointestinal tract.
mesenchymal stem cells
perianal fistula
Crohn’s disease
1. Introduction
Crohn’s disease (CD) is a relapsing systemic inflammatory disease that can cause persistent transmural inflammation anywhere along the gastrointestinal tract. A substantial number of CD patients present with various anorectal pathologies including perianal abscesses, fistulas, hemorrhoids, skin tags and fissures [1]. Perianal fistulas are described as an abnormal connection between the anal canal and the perianal skin.
According to population-based studies, around one in every four CD patients will develop perianal fistula at some point in their life. Patients with colonic or rectal localization of intestinal inflammatory lesions are at an even higher risk, at about 92% [2]. Diagnosis and management of perianal Crohn’s Disease (PCD) requires an expert, multidisciplinary approach, considering that most of the perianal fistulas are defined as complex (meaning they involve the upper part of the sphincters, are complicated with a perianal abscess, or have multiple external openings or penetrate to vagina). These patients’ quality of life is considerably impaired due to persistent drainage, pain, recurrent perianal sepsis and continuous need to seek medical attention [3]. Patients with perianal fistula are also more likely to have extraintestinal manifestations of CD including arthritis, oral ulcerations and skin manifestations.
The management of PCD remains a challenge, as 37% of patients experience refractory disease. The relapse rate is estimated at around 40% in 5-year follow-ups [4]. For this reason, many patients are exposed to the necessity to repeatedly take immunosuppressive medications, which increases the risk of opportunistic infections. Furthermore, about 90% of them undergo numerous surgical interventions, becoming liable to complications, including fecal incontinence [2][4]. Even with combined pharmacological and surgical therapy, 40% of patients do not achieve remission and are left with active disease, facing the risk of undergoing proctectomy.
This perspective encouraged us to seek more desirable treatment options to provide higher therapy effectiveness with a lack of adverse effects and lower risk of incontinence. Recently, promising results using local injections with mesenchymal stem cells (MSCs) have been reported. This treatment has been proven to effectively induce as well as maintain fistula closure [5][6].
MSCs are stromal cells that have the ability to self-renew and differentiate into adipocytes, myocytes, osteocytes and chondrocytes. Moreover, they have powerful immunomodulatory effects and are able to reduce escalated inflammation, as they inhibit the proliferation and function of T, B and NK cells [6]. MSCs are present in almost all tissues; however, they are most commonly isolated from bone marrow, adipose tissue or an umbilical cord. In the last decade, physicians have been able to activate and supplement these cells to treat a variety of conditions, for instance, many autoimmune diseases. At present, multiple new studies on the use of MSCs for Multiple Sclerosis, chronic obstructive pulmonary disease, cancer, lupus and Parkinson’s are being carried out [6].
2. Mesenchymal Stem Cells Mechanism in Crohn’s Disease Perianal Fistula
To this day, the exact mechanism in which MSCs heal perianal fistulas remains unknown, as no human studies have clarified it. It likely results from their immunomodulatory properties [7]. Firstly, they migrate to the sites of injury or inflammation and directly spur tissue healing by tissue specific differentiation and the secretion of factors with the ability to promote epithelial cell proliferation and angiogenesis. MSCs help maintain an anti-inflammatory habitat by modulating the function of macrophages, lymphocytes and dendritic cells.
Regarding CD, MSCs’ ability to upregulate the CD4+ T cell subset is particularly important, as patients with CD are known to have a deficiency of those cells [8][9]. Furthermore, MSCs secrete various anti-inflammatory particles (including TGFB1, growth factors, interleukins and indoleamine 2,3-dioxygenase) and suppress M1 macrophages, cytotoxic T cells and dendritic cells [10]. They play a significant role in healing of the fistula, owing to their role in angio- and mitogenesis, as well as immunomodulatory effect [11]. They reduce inflammation in tissues around the fistula and accelerate healing. Due to its’ promising preclinical studies results, clinicians are more and more interested in CD treatment using MSCs.
3. Mesenchymal Stem Cells Application in Perianal Crohn’s Disease
Regrettably, there is no clear surgical protocol for the administration of stem cells in PCD therapy. To start with, there are no studies comparing side by side the use of adipose- vs. bone-marrow-derived MSCs. Adipose-derived stem cells are usually preferred because they are much easier to harvest with the use of liposuction. Furthermore, they are known to have a higher replication rate and longer proliferation in culture [5][12]. They might also induce a stronger immunomodulatory effect, due to the fact they have higher secretion levels of cytokines, including TGFB1 and IL-6 [8].
MSCs are typically used locally, to avoid side effects and help keep cells in direct contact with inflamed tissue [13]. In that case, either autologous or allogenic cells can be used. During treatment with the use of autologous cells, extraction and application happens in the course of the same procedure [12][14]. Thus, MSCs are injected without previous expansion, which results in their low dose [15].
According to a meta-analysis based on four clinical trials involving different doses of stem cell therapy ranging from 1 × 107 to 9 × 107 MSCs per mL, the dose of 2 × 107 and 3 × 107 cells/mL were characterized by a highest rate of fistula healing in Crohn’s patients [16]. However, the mentioned study is strongly limited by a low number of included patients (n = 31). Due to the lack of other studies based on a larger group, it is difficult to propose specific guidelines regarding the dosage of MSCs. Therefore, it is crucial to continue such studies.
However, allogenic cells are endowed with excellent homogeneity [17]. They might not create antibodies, which would be clinically relevant. Cells are previously expanded and frozen. It only takes 4 days to defrost them and prepare for administration. Allogenic MSCs are also considered particularly beneficial in CD.
To date, no definite conclusion about an optimal dosage has been made. According to Molendijk et al.’s study, the best results are achieved with the use of 30 million cells [18]. The most commonly used concentration contains 5 × 106 MSCs per mL; however, no studies have directly compared the outcome determined by the use of different MSCs concentrations.
Preoperatively, each patient should be thoroughly examined, to prepare a precise description of the fistula tract. It is necessary to estimate the needed MSCs volume. A magnetic resonance imaging or endorectal ultrasonography (ERUS) can be found helpful to provide information concerning the type, location and presence of branches of the fistula.
Prior to surgery, any active infection should be controlled by antibiotics, and any abscesses greater than 2 cm should be incised and drained. Fistula biopsy should also be considered to rule out any type of local neoplasia, since MSCs might promote tumor growth. There is no reason to discontinue systemic treatment for the CD.
As far as anesthesia is concerned, most frequently used local anesthetics such as lidocaine can cause a cytotoxic effect to the MSCs when in direct contact. For that reason, local anesthesia ought to be avoided. If necessary, the recommended approach is to perform a pudendal block. This concern should also be kept in mind if the MSCs are obtained by liposuction. During this procedure, the use of local anesthetics is discouraged, as they weaken the MSCs’ immunomodulatory properties and viability.
The antiseptic used to prepare the surgical site must not be harmful to the cells. Thus, the use of alcoholic, povidone solutions or hydrogen peroxide is discouraged. Rather, it is advised to use octenidine, chlorhexidine or normal saline.
The next step is to identify the internal fistula opening using a probe, or by injecting normal saline through the external orifice. To create an optimal environment for healing, the fistula tract needs to be mechanically debrided using curette and irrigated with saline. Afterwards, the internal opening needs to be securely closed with the use of an absorbable suture.
MSCs can be delivered in direct injections, united with fibrin glue or impregnated on a fistula plug. It is believed that healing rates are higher when scaffolding material is used, due to the fact that glue or plug help maintain MSCs at the desirable location. On the other hand, a randomized clinical trial involving 200 patients comparing the efficiency of treatments using MSCs, MScs mixed with fibrin glue or fibrin glue alone proved there are no significant differences in the healing rate with the use of these three methods. In Figure 1 we have provided a general overview regarding MCSc application process.
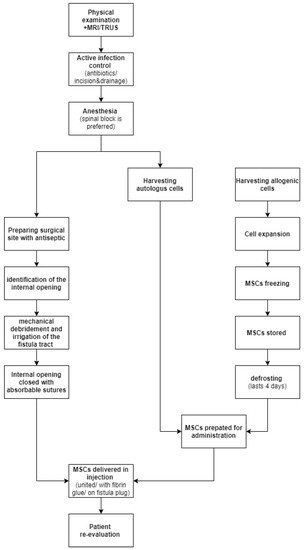
Figure 1. MSCs application process.
References
- Baumgart, D.C.; Sandborn, W.J. Crohn’s disease. Lancet 2012, 380, 1590–1605.
- Pogacnik, J.S.; Salgado, G. Perianal Crohn’s Disease. Clin. Colon Rectal Surg. 2019, 32, 377–385.
- Multidisciplinary Team (MDT) Approach to Diagnosis & Management of Perianal Crohn’s Disease | Crohn’s & Colitis Foundation. Available online: https://www.crohnscolitisfoundation.org/clinical-pearls/mdt-perianal-crohns-disease (accessed on 30 June 2021).
- Panes, J.; Reinisch, W.; Rupniewska, E.; Khan, S.; Forns, J.; Khalid, J.M.; Bojic, D.; Patel, H. Burden and outcomes for complex perianal fistulas in Crohn’s disease: Systematic review. World J. Gastroenterol. 2018, 24, 4821–4834.
- Herreros, M.D.; Garcia-Olmo, D.; Guadalajara, H.; Georgiev-Hristov, T.; Brandariz, L.; Garcia-Arranz, M. Stem cell therapy: A compassionate use program in perianal fistula. Stem Cells Int. 2019, 2019, 6132340.
- Ding, D.C.; Shyu, W.C.; Lin, S.Z. Mesenchymal stem cells. Cell Transplant. 2011, 20, 5–14.
- Tozer, P.J.; Lung, P.; Lobo, A.J.; Sebastian, S.; Brown, S.R.; Hart, A.L.; Fearnhead, N.; Adegbola, S.O.; Heywood, N.; Hind, D.; et al. Review article: Pathogenesis of Crohn’s perianal fistula—Understanding factors impacting on success and failure of treatment strategies. Aliment. Pharmacol. Ther. 2018, 48, 260–269.
- Bernardi, L.; Dos Santos, C.H.M.; Pinheiro, V.A.Z.; Oliveira, R.J.; Antoniolli-Silva, A.C.M.B. Transplantation of adipose-derived mesenchymal stem cells in refractory crohn’s disease: Systematic review. Arq. Bras. Cir. Dig. 2019, 32, e1465.
- Verstockt, B.; Ferrante, M.; Vermeire, S.; Van Assche, G. New treatment options for inflammatory bowel diseases. J. Gastroenterol. 2018, 53, 585–590.
- Hoogduijn, M.J.; Lombardo, E. Mesenchymal Stromal Cells Anno 2019: Dawn of the Therapeutic Era? Concise Review. Stem Cells Transl. Med. 2019, 8, 1126–1134.
- Carvello, M.; Lightner, A.; Yamamoto, T.; Kotze, P.G.; Spinelli, A. Mesenchymal Stem Cells for Perianal Crohn’s Disease. Cells 2019, 8, 764.
- Georgiev-Hristov, T.; Guadalajara, H.; Herreros, M.D.; Lightner, A.L.; Dozois, E.J.; García-Arranz, M.; García-Olmo, D. A Step-By-Step Surgical Protocol for the Treatment of Perianal Fistula with Adipose-Derived Mesenchymal Stem Cells. J. Gastrointest. Surg. 2018, 22, 2003–2012.
- Banasiewicz, T.; Eder, P.; Rydzewska, G.; Reguła, J.; Dobrowolska, A.; Durlik, M.; Wallner, G. Position of the expert group on the current practice and prospects for the treatment of complex perirectal fistulas in the course of Crohn’s disease. Polish J. Surg. 2019, 91, 1–9.
- Herreros, M.D.; Garcia-Arranz, M.; Guadalajara, H.; De-La-Quintana, P.; Garcia-Olmo, D. Autologous expanded adipose-derived stem cells for the treatment of complex cryptoglandular perianal fistulas: A phase III randomized clinical trial (FATT 1: Fistula advanced therapy trial 1) and long-term evaluation. Dis. Colon Rectum 2012, 55, 762–772.
- Ciccocioppo, R.; Klersy, C.; Leffler, D.A.; Rogers, R.; Bennett, D.; Corazza, G.R. Systematic review with meta-analysis: Safety and efficacy of local injections of mesenchymal stem cells in perianal fistulas. JGH Open 2019, 3, 249–260.
- Cao, Y.; Su, Q.; Zhang, B.; Shen, F.; Li, S. Efficacy of stem cells therapy for Crohn’s fistula: A meta-analysis and systematic review. Stem Cell Res. Ther. 2021, 12, 32.
- Avivar-Valderas, A.; Martín-Martín, C.; Ramírez, C.; Del Río, B.; Menta, R.; Mancheño-Corvo, P.; Ortiz-Virumbrales, M.; Herrero-Méndez, Á.; Panés, J.; García-Olmo, D.; et al. Dissecting allo-sensitization after local administration of human allogeneic adipose mesenchymal stem cells in perianal fistulas of Crohn’s disease patients. Front. Immunol. 2019, 10, 1244.
- Molendijk, I.; Bonsing, B.A.; Roelofs, H.; Peeters, K.C.M.J.; Wasser, M.N.J.M.; Dijkstra, G.; Van Der Woude, C.J.; Duijvestein, M.; Veenendaal, R.A.; Zwaginga, J.J.; et al. Allogeneic Bone Marrow-Derived Mesenchymal Stromal Cells Promote Healing of Refractory Perianal Fistulas in Patients With Crohn’s Disease. Gastroenterology 2015, 149, 918–927.e6.
More
Information
Subjects:
Surgery
Contributor
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
863
Revisions:
2 times
(View History)
Update Date:
12 Nov 2021
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





