Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Edelson Dias | + 2106 word(s) | 2106 | 2021-09-29 05:35:04 | | | |
| 2 | Vivi Li | Meta information modification | 2106 | 2021-11-01 04:07:59 | | | | |
| 3 | Vivi Li | Meta information modification | 2106 | 2021-11-08 04:10:33 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Dias, E. Polyphenols in Humiria balsamifera (Aubl). Encyclopedia. Available online: https://encyclopedia.pub/entry/15525 (accessed on 24 May 2026).
Dias E. Polyphenols in Humiria balsamifera (Aubl). Encyclopedia. Available at: https://encyclopedia.pub/entry/15525. Accessed May 24, 2026.
Dias, Edelson. "Polyphenols in Humiria balsamifera (Aubl)" Encyclopedia, https://encyclopedia.pub/entry/15525 (accessed May 24, 2026).
Dias, E. (2021, October 29). Polyphenols in Humiria balsamifera (Aubl). In Encyclopedia. https://encyclopedia.pub/entry/15525
Dias, Edelson. "Polyphenols in Humiria balsamifera (Aubl)." Encyclopedia. Web. 29 October, 2021.
Copy Citation
Humiria balsamifera (Aubl), commonly known as “mirim”, is a plant of the Humiriaceae family, which consists of 39 species divided between eight genera: Duckesia, Endopleura, Humiria, Humiriastrum, Hylocara, Sacoglottis, Schistostemon, and Vantenea. This study aimed to characterize H. balsamifera extracts by LC-MS/MS and evaluate their antimicrobial potential through in vitro and in vivo assays.
Humiriaceae
Humiria
antimicrobial
microorganisms
flavonoids
1. Introduction
Microorganisms are naturally well-spread out in the environment, and they can easily reach surfaces people come into contact with, including food products, whether at the harvest, slaughter, processing, or even packaging. Once in contact with the food, they start their growth process by consuming nutrients and causing the product to deteriorate [1][2][3].
Bacteria, fungi, viruses, and protozoa are the main microorganisms responsible for food contamination, infecting humans through the consumption of beef, fish, poultry, eggs, unhygienic fruits and fresh produce, causing a variety of diseases [4][5]. The World Health Organization estimates that one in 10 people worldwide become ill after consuming contaminated food and about 420,000 people die each year, resulting in the loss of 33 million healthy life years (DALYs) [2][6].
Bacteria represent an added concern for health and food safety organizations, especially those able to grow at low temperatures and resist a wide range of temperature variations [7][8]. Bacterial pathogens such as Escherichia coli, Salmonella enterica, Listeria monocytogenes, and Staphylococcus aureus, among others, are responsible for several global foodborne outbreaks and cause life-threatening illnesses such as diarrheal diseases [5][9][10][11][12]. Another problem in fighting bacteria is their ability to develop resistance to conventional antimicrobials. These pathogens can use various strategies to inhibit the effects of antimicrobials, such as the production of inactivating enzymes, reduction of outer membrane permeability, efflux system, and blocking or altering the target site of antibiotics, further motivating the research focused on finding alternative ways to combat them [13][14].
In the search for new effective substances against resistant pathogens, several secondary metabolites from plants and endophytic microorganisms have shown promise [15][16]. Most of the drugs used in general today were developed based on ethnopharmacological knowledge [17][18][19], indicating that the chemistry of natural products is a big ally in the development of therapeutic agents [20][21].
The plant species Humiria balsamifera (Aubl), popularly known as “mirim”, presents interesting biological activities. The literature reports, most of all, anti-inflammatory [22][23], antimalarial [24], antioxidant [25][26], and antifungal activity [27], highlighting the therapeutic potential of this plant. Some substances isolated from this species so far have already been reported as well, such as bergenin, arjunolic acid, friedelin, lupeol, phytol, caryophyllene oxide, epoxide humulene, and trans-isolongifolanone, among others [24]. However, the antibacterial and antibiofilm activities of its derived products have not been extensively examined. Thus, this work aims to characterize and evaluate the effectiveness of H. balsamifera extracts in terms of the antimicrobial and antibiofilm activities against foodborne pathogens (Escherichia coli ATCC 25922, Listeria monocytogenes ATCC 15313, Salmonella enterica Typhimurium ATCC 14028, and Staphylococcus aureus ATCC 6538). The in vivo antimicrobial action of the most active extract was analyzed using a method based on the infection of Tenebrio molitor larvae.
2. Antimicrobial Activity Evaluation
The antimicrobial activity of Humiria balsamifera (Aubl) leaf and stem bark extracts was evaluated by the determination of their minimum inhibitory concentrations (MIC) against four foodborne bacteria species: E. coli, L. monocytogenes, S. enterica Typhimurium, and S. aureus (Table 1).
Table 1. Determination of the minimum inhibitory concentrations (MIC) of the leaf and stem bark extracts of Humiria balsamifera (Aubl).
| Bacteria Species | HSBE | EASBE | MSBE | HLE | EALE | MLE |
|---|---|---|---|---|---|---|
| E. coli ATCC 25922 | >12,500 | >12,500 | >12,500 | >12,500 | >12,500 | >12,500 |
| L. monocytogenes ATCC 6538 | >12,500 | >12,500 | >12,500 | >12,500 | 3120 | 3120 |
| S. aureus ATCC 6538 | >12,500 | >12,500 | >12,500 | 1560 | 780 | 780 |
| S. enterica Typhimurium ATCC 14028 | >12,500 | >12,500 | >12,500 | >12,500 | >12,500 | >12,500 |
Footnote: HSBE = hexane stem bark extract, EASBE = ethyl acetate stem bark extract, MSBE = methanol stem bark extract, HLE = hexane leaf extract, EALE = ethyl acetate leaf extract, and MLE = methanol leaf extract. MIC values are expressed in µg/mL.
Stem bark extracts did not exhibit antimicrobial action at any of the concentrations tested (MIC > 1250 µg/mL). However, the leaf extracts successfully inhibited S. aureus, with MIC = 780 µg/mL (EALE and MLE) and 1560 µg/mL (HLE). The EALE and the MLE also inhibited L. monocytogenes (MIC = 3120 µg/mL). The leaf extracts presented no action against the Gram-negative bacteria tested in this study.
3. Evaluation of the Antibiofilm Activity of Humiria balsamifera (Aubl) Extracts
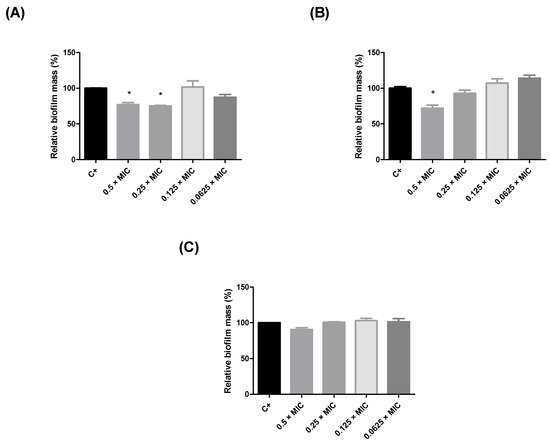
Since the leaf extracts of Humiria balsamifera presented better inhibition results against S. aureus, their antibiofilm action at subinhibitory concentrations (0.5 × MIC, 0.25 × MIC, 0.125 × MIC, and 0.0625 × MIC) was also evaluated (Figure 1). The EALE and the HLE reduced biofilm production by S. aureus by nearly 25% at concentrations higher than 390 µg/mL (Figure 1A,B). The MLE did not exhibit significant antibiofilm activity (Figure 1C). Considering the results for the antimicrobial and antibiofilm activities, the EALE was chosen to proceed to the in vivo tests, using Tenebrio molitor larvae.

Figure 1. Inhibition of the biofilm formation in Staphylococcus aureus by Humiria balsamifera (Aubl) extracts. (A) HLE = hexane leaf extract, (B) EALE = ethyl acetate leaf extract, and (C) MLE = methanol leaf extract. (*) Significant differences (p < 0.05) in relation to untreated biofilm (C +).
4. Chemical Characterization of Humiria balsamifera (Aubl) Leaf and Stem Bark Extracts
Analyses of the leaf and stem bark extracts of Humiria balsamifera (Aubl) by HPLC-ESI-IT/MS in negative-ion mode identified 11 molecular ions (Table 2, Table 3 and Table 4). Their structures were proposed (Figure 3) based on the fragments originated from the molecular ion by multi-stage mass spectrometry (MSn). The mass spectrometry ionization source was the electrospray (ESI). The ESI source may not have ionized the compounds like steroids and triterpenes. It was possible to identify only phenolic compounds in the extracts. From the 11 identified substances, seven were flavonoids (gallocatechin, kaempferol 3-neohesperidoside, sophoricoside, quercetin 3-arabinoside, quercetin-O-rhamnoside, kaempferol-dirhamnoside, and vitexin-dirhamnoside); three were coumarins (bergenin and two derivatives: galloylbergenin and hydroxybenzoyl bergenin); and one was an oligosaccharide (maltotetraose).

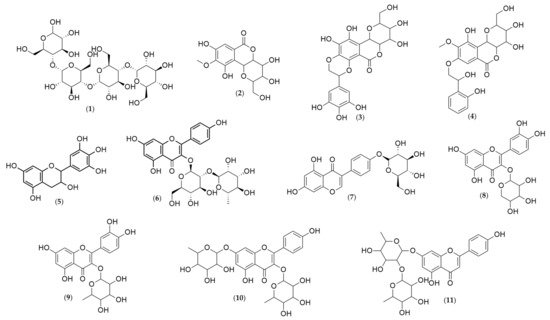
Figure 3. Chemical structures of the substances identified in the extracts of Humiria balsamifera (Aubl) by HPLC-ESI-IT/MS using the MassBank Spectral DataBase. Maltotetraose (1); Bergenin (2); Galloylbergenin (3); Hydroxybenzoyl bergenin (4); Gallocatechin (5); Kaempferol 3-O-neohesperidoside (6); Sophoricoside (7); Quercetin 3-arabinoside (8); Quercetin-O-rhamnoside (9); Kaempferol-dirhamnoside (10); and Vitexin-dirhamnoside (11).
Table 2. Identification of the substances present in the ethyl acetate stem bar extract of Humiria balsamifera (Aubl).
| RT (min) | [M-H] | MSn | Proposed Substance | Reference |
|---|---|---|---|---|
| 1.97 | 665 | 664, 305 | Maltotetraose | [28] |
| 2.85 | 327 | 249, 234, 207 | Bergenin | [29] |
| 3.58 | 479 | 327, 249, 234, 207 | Galloylbergenin | [29] |
| 4.87 | 463 | 327, 249, 234, 207 | Hydroxybenzoyl bergenin | [29] |
Table 4. Identification of the substances present in the Humiria balsamifera (Aubl) ethyl acetate leaf extract.
| RT (min) | [M-H] | MSn | Proposed Substance | Reference |
|---|---|---|---|---|
| 2.83 | 327 | 249, 234, 207, 192 | Bergenin | [29] |
| 5.27 | 431 | 311, 283, 341 | Sophoricoside | [32] |
| 6.09 | 433 | 300, 271, 243, 227 | Quercetin 3-arabinoside | [33] |
| 6.46 | 447 | 300, 271, 243 | Quercetin-O-rhamnoside | [33] |
| 7.17 | 577 | 431, 413, 341, 300 | Kaempferol-dirhamnoside | [33] |
| 8.38 | 561 | 431, 293 | Vitexin-dirhamnoside | [32] |
Maltotetraose (1) presented a molecular ion of m/z = 665, with a fragment of m/z = 664 after the loss of a proton. In the third stage of the fragmentation process, the loss of 341 Da as C12H21O11 and 18 Da as a water molecule produced a fragment of m/z = 305.
Bergenin (2) originated four ionic fragments: from an initial loss of 60 Da as C2H4O2, and 18 Da as a water molecule, resulted the fragment C12H10O6− (m/z = 249); the second one was produced from the loss of 93 Da as C2H4O2, a methyl group, and a water molecule, resulting in the ion C11H7O6− (m/z = 234); the third fragment C10H8O5− (m/z = 207) was the result of the loss of 120 Da as C4H8O4; and finally, the loss of 135 Da as C4H8O4 and a methyl group led to the fourth fragment, C9H5O5− (m/z = 192).
All these ions were also present in the spectra of the bergenin derivatives, plus one other fragment, observed in both spectra.
The fragment C14H15O9− (m/z = 327) was observed in both spectra—galloylbergenin (3) and hydroxybenzoyl bergenin (4). For galloylbergenin, this fragment was a result of the loss of 152 Da as the galloyl group. For hydroxybenzoyl bergenin, this fragment resulted from the elimination of the hydroxybenzoyl group (136 Da).
Gallocatechin (5) produced two fragment ions: C9H8O4− (m/z = 179), resulting from the loss of 126 Da as C6H6O3, and C8H8O4 (m/z = 165) from the loss of 140 Da as C7H8O3.
Kaempferol 3-O-neohesperidoside (6) also produced only two fragment ions: C21H19O10− (m/z = 431), generated by the loss of 162 Da as C6H10O5, and C21H15O9− (m/z = 411), originated by the ion C21H19O10−, after a water loss. Bergenin (2) was also identified.
Sophoricoside (7) originated three ions: C21H19O10− (m/z = 431), as a result of the loss of 90 Da as C3H6O3, C17H11O6− (m/z = 311) from the loss of 120 Da as C4H8O4−, and from C17H11O6−, the third fragment, C16H11O5− (m/z = 283), was formed as a result of the loss of 28 Da as a carbon monoxide molecule.
In the quercetin 3-arabinoside spectrum (8), four fragment ions were observed: C15H9O7− (m/z = 300) from the loss of 133 Da as C5H9O4, C14H7O6− (m/z = 271) from the loss of 162 Da as C6H10O5, and the fragments C13H7O5− (m/z = 243) and C13H7O4− (m/z = 227), resulting from the loss of a carbon monoxide and dioxide, respectively.
The fragmentation of quercetin-O-rhamnoside (9) were similar to what was observed for quercetin 3-arabinoside. The loss of a rhamnose molecule, C6H11O4 (m/z = 147 Da), led to the fragment ion C15H9O7− (m/z = 300). Then, the loss of carbon monoxide produced the fragments C14H7O6− (m/z = 271) and C13H7O5− (m/z = 243).
The fragmentation of kaempferol-dirhamnoside produced four ions. The first one, C21H19O10− (m/z = 431), was generated by the loss of a rhamnose molecule (C6H11O4, m/z = 147 Da). From the C21H19O10− fragment (m/z = 431), the loss of a water molecule produced the second fragment ion, C21H17O9− (m/z = 413), which, in turn, generated the fragment C18H13O7− (m/z = 341) by the loss of 72 Da as C3H4O2. Finally, the loss of 41 Da as C2OH produced the fragment C16H12O6− (m/z = 300).
The fragmentation of vitexin-dirhamnoside resulted in two ionic products: C21H17O9− (m/z = 413), generated by the loss of a rhamnose molecule (C6H11O4, 147 Da), and C17H9O5− (m/z = 293), produced by the loss of C4H6O3 (m/z = 102), followed by a water molecule.
5. Discussion
This research aimed to characterize and evaluate the antimicrobial potential of the extracts of Humiria balsamifera (Aubl), also known as “mirim”. This species belongs to the Humiriaceae family, and its tea is used in many Brazilian regions for its anti-inflammatory action, especially for treating uterine inflammation [22][23].
Analyses of the leaf extracts by HPLC-ESI-MS and FIA-ESI-IT/MS led to the identification of 11 substances, 10 of which had not yet been reported for this species: seven flavonoids (gallocatechin, kaempferol 3-neohesperidoside, sophoricoside, quercetin 3-arabinoside, quercetin-O-rhamnoside, kaempferol-dirhamnoside, and vitexin-dirhamnoside); one oligosaccharide (maltotetraose), bergenin; and two derivatives (galloylbergenin and hydroxybenzoyl bergenin).
The substances present in the chemical composition of H. balsamifera tell a lot about the species. Flavonoids, according to the characterization presented in this study, are the most abundant class of compounds. These substances exhibit high bioactive potential and present anti-ulcer, antioxidant, anti-inflammatory, anti-allergic, antitumor, antiviral, antifungal, and antidiabetic activities [34][35][36][37].
Antimicrobial tests with flavonoids have received increasing attention in recent years, since these compounds are synthesized by plants in response to various types of stress, including microbial infections [38][39][40]. Researchers are also interested in how flavonoids are able to exhibit antibacterial activity through mechanisms different from conventional drugs, hindering the development of microbial resistance [41][42].
During this study, the H. balsamifera extracts were subjected to three tests for the evaluation of their antimicrobial potential against different bacteria strains. By the end of the in vitro tests, the extracts which presented the best results were selected for the in vivo anti-infective assay using T. molitor larvae. Our results showed that the H. balsamifera ethyl acetate leaf extract (EALE) showed efficacy against S. aureus, one of the most resistant pathogens in existence, in all three tests (MIC, antibiofilm potential, and the tests in vivo). The efficacy of this extract is believed to be due to the flavonoids present in its composition. Flavonoids are well-known in the literature, as other polyphenols, to be able to inhibit microbial growth through several mechanisms, such as the inhibition of ATP synthesis in the electron transport chain, inhibition of nucleic acid synthesis, inhibition of the efflux pump, inhibition of biofilm formation, inhibition of virulence factors, inhibition of quorum sensing, membrane disruption, inhibitors of bacterial toxins, and inhibition of cell envelope synthesis [41][43][44].
Bergenin, identified in the three extracts analyzed, has already been reported in H. balsamifera [24] and other two species from the Humiriaceae family: Endopleura uchi and Sacoglottis gabonensis [45][46]. This isocoumarin and its derivatives, such as the identified flavonoids, can be directly related to the antimicrobial activity of the extracts against S. aureus. A recent study showed that six synthetic derivatives of bergenin obtained by Williamson synthesis inhibited S. aureus growth, especially 8,10-dihexyl-bergenin and 8,10-didecyl-bergenin, which presented the most promising MIC value: 3.12 µg/mL [47].
References
- Gonelimali, F.D.; Lin, J.; Miao, W.; Xuan, J.; Charles, F.; Chen, M.; Hatab, S.R. Antimicrobial Properties and Mechanism of Action of Some Plant Extracts Against Food Pathogens and Spoilage Microorganisms. Front. Microbiol. 2018, 9, 1–9.
- Franz, C.M.A.P.; Den Besten, H.M.W.; Böhnlein, C.; Gareis, M.; Zwietering, M.H.; Fusco, V. Microbial food safety in the 21st century: Emerging challenges and foodborne pathogenic bactéria. Trends Food Sci. Technol. 2018, 81, 155–158.
- Parlapani, F.F.; Mallouchos, A.; Haroutounian, S.A.; Boziaris, I.S. Volatile organic compounds of microbial and non-microbial origin produced on model fish substrate un-inoculated and inoculated with gilt-head sea bream spoilage bacteria. LWT Food Sci. Technol. 2018, 78, 54–62.
- Lianou, A.; Panagou, E.Z.; Nychas, G.-J.E. Microbiological spoilage of foods and beverages. In The Stability and Shelf Life of Food, 2nd ed.; Subramaniam, P., Ed.; Woodhead Publishing: Cambridge, UK, 2016; pp. 3–42.
- Nahar, S.; Jeong, H.L.; Kim, Y.; Ha, A.J.; Roy, P.K.; Park, S.H.; Ashrafudoulla, M.; Mizan, M.F.R.; Ha, S. Inhibitory effects of Flavourzyme on biofilm formation, quorum sensing, and virulence genes of foodborne pathogens Salmonella Typhimurium and Escherichia coli. Food Res. Int. 2021, 147, 110461.
- WHO (World Health Organisation). WHO Estimates of the Global Burden of Foodborne Diseases Foodborne Diseases Burden Epidemiology Reference Group 2007–2015. 2015. Available online: https://www.who.int/en/news-room/fact-sheets/detail/food-safety (accessed on 1 June 2021).
- Moyer, C.L.; Morita, R.Y. Psychrophiles and psychrotrophs. In Encyclopedia of Life Sciences; John Wiley & Sons Ltd.: Chichester, UK, 2007; pp. 1–6.
- Wei, Q.; Wang, X.; Sun, D.; Pu, H. Rapid detection and control of psychrotrophic microorganisms in cold storage foods: A review. Trends Food Sci. Technol. 2019, 86, 453–464.
- Kirk, M.D.; Ângulo, F.J.; Havelaar, A.H.; Black, R.E. Diarrhoeal disease in children due to contaminated food. Bull. World Health Organ. 2017, 95, 233–234.
- Anderson, C.J.; Kendall, M.M. Salmonella typhimurium Strategies for Host Adaptation. Front. Microbiol. 2017, 8, 1983.
- Neri, D.; Antoci, S.; Iannetti, L.; Ciorba, A.B.; D’Aurelio, R.; Del Matto, I.; Di Leonardo, M.; Giovannini, A.; Prencipe, V.A.; Pomilio, F.; et al. EU and US control measures on Listeria monocytogenes and Salmonella spp. in certain ready-to-eat meat products: An equivalence study. Food Control 2019, 96, 98–103.
- Shahid, A.H.; Nazir, K.H.M.; El Zowalaty, M.E.; Kabir, A.; Sarker, S.A.; Siddique, M.P.; Ashour, H.M. Molecular detection of vancomycin resistance and methicillin resistance in Staphylococcus aureus from food processing environments. One Health 2021, 100276, 1–14.
- Bello, A.; Dingle, T.C. What’s That Resistance Mechanism? Understanding Genetic Determinants of Gram-Negative Bacterial Resistance. Clin. Microbiol. Newsl. 2018, 40, 165–174.
- Holmes, A.H.; Moore, L.S.P.; Sundsfjord, A.; Steinbakk, M.; Regmi, S.; Karkey, A.; Guerin, P.J.; Piddock, L.J.V. Understanding the mechanisms and drivers of antimicrobial resistance. Lancet 2016, 387, 176–187.
- Newman, D.J.; Cragg, G.M. Natural Products as Sources of New Drugs over the Nearly Four Decades from 01/1981 to 09/2019. J. Nat. Prod. 2020, 83, 770–803.
- Chandra, H.; Bishnoi, P.; Yadav, A.; Patni, B.; Mishra, A.P.; Nautiyal, A.R. Antimicrobial Resistance and the Alternative Resources with Special Emphasis on Plant-Based Antimicrobials—A Review. Plants 2017, 6, 16.
- Da Silva, L.C.N.; Da Silva, M.V.; Correia, M.T.S. New Frontiers in the Search of Antimicrobials Agents from Natural Products. Front. Microbiol. 2017, 8, 210.
- Valli, M.; Bolzani, V.S. Natural Products: Perspectives and Challenges for use of Brazilian Plant Species in the Bioeconomy. An. Acad. Bras. Cienc. 2019, 91, e20190208.
- Rodrigues, T.; Reker, D.; Schneider, P.; Schneider, G. Counting on natural products for drug design. Nat. Chem. 2016, 8, 531–541.
- Imai, Y.; Meyer, K.J.; Iinishi, A. A new antibiotic selectively kills Gram-negative pathogens. Nature 2019, 576, 459–464.
- Melander, R.J.; Basak, A.K.; Melander, C. Natural products as inspiration for the development of bacterial antibiofilm agents. Nat. Prod. Rep. 2020, 11.
- Carneiro, D.B.; Barboza, M.S.L.; Menezes, M.P. Plantas nativas úteis na Vila dos Pescadores da Reserva Extrativista Marinha Caeté-Taperaçu, Pará, Brasil. Acta Botânica Bras. 2010, 24, 1027–1033.
- Santos, M.G.; Fevereiro, P.C.A.; Reis, G.L.; Barcelos, J.I. Recursos vegetais da restinga de Carapebus, Rio de Janeiro, Brasil. J. Neotrop. Biol. 2009, 6, 35–54.
- Silva, T.B.C.; Alves, V.L.; Mendonça, L.V.H.; Conserva, L.M.; Rocha, E.M.M.; Andrade, E.H.A.; Lemos, R.P.L. Chemical Constituents and Preliminary Antimalarial Activity of Humiria balsamifera. Pharm. Biol. 2004, 42, 94–97.
- Paz, S.A.; Marques, G.E.C. Formulação de Hidratante com Extratos de Humiria balsamifera (Aubl A. St. Hil) com Ação Antioxidante. In Cadernos de Agroecologia; Anais do XI Congresso Brasileiro de Agroecologia; ABA—Associação Brasileira de Agroecologia: Rio de Janeiro, Brasil, 2020; Volume 15, p. 2. Available online: http://cadernos.aba-agroecologia.org.br/index.php/cadernos/issue/view/4 (accessed on 1 May 2021).
- Dutra, A.R.; Sutilli, F.K.; Muzitano, M.F.; Guimarães, D.O.; Souza, R.O.; Leal, I.C. Microwave-assisted extraction of metabolites from Humiria balsamifera leaves: A rapid and efficient methodology for antioxidant constituents obtainment. Planta Med. 2013, 79.
- Lima, C.N.S.; Farias, F.A.C.; Marques, G.E.C. Atividade Antimicrobiana in vitro dos Extratos de Humiria balsamífera (Aulb) A. St. Hil. Frente a Cepas Bacaterianas e Fúngicas. In Mirim (Humiria balsamifera (Aubl.): Riqueza da Biodiversidade Maranhense, 1st ed.; Luís, S., Ed.; IFMA: São Luís, Brazil, 2019; Volume 1, pp. 99–107. Available online: https://editora.ifma.edu.br/livraria-edifma-2019/ (accessed on 2 May 2021).
- Tüting, W.; Adden, R.; Mischnick, P. Fragmentation pattern of regioselectively O-methylated maltooligosaccharides in electrospray ionisation-mass spectrometry/collision induced dissociation. Int. J. Mass Spectrom. 2004, 232, 107–115.
- Song, H.; Wang, J.; Zhang, R.; Liu, X.; Yuan, G.; Wei, C.; Zhao, W.; Li, R.; Wang, B.; Guo, R. In vivo metabolism study of bergenin in rats by HPLC-QTOF mass spectrometry. Biomed. Chromatogr. 2013, 27, 1398–1405.
- Callemien, D.; Collin, S. Use of RP-HPLC-ESI (–)—MS/MS to Differentiate Various Proanthocyanidin Isomers in Lager Beer Extracts. J. Am. Soc. Brew. Chem. 2008, 66, 109–115.
- March, R.E.; Miao, X. A fragmentation study of kaempferol using electrospray quadrupole time-of-flight mass spectrometry at high mass resolution. Int. J. Mass Spectrom. 2004, 231, 157–167.
- Ablajan, K. A study of characteristic fragmentation of isoflavonoids by using negative ion ESI-MSn. Int. J. Mass Spectrom. 2011, 46, 77–84.
- Ćirić, A.; Prosen, H.; Jelikić-Stankov, M.; Đurđević, P. Evaluation of matrix effect in determination of some bioflavonoids in food samples by LC–MS/MS method. Talanta 2012, 99, 780–790.
- Havsteen, B.H. The biochemistry and medical significance of the flavonoids. Pharmacol. Ther. 2002, 96, 67–202.
- Ng, K.R.; Lyu, X.; Mark, R.; Chen, W.N. Antimicrobial and antioxidant activities of phenolic metabolites from flavonoid-producing yeast: Potential as natural food preservatives. Food Chem. 2019, 270, 123–129.
- Lalani, S.; Poh, C.L. Flavonoids as Antiviral Agents for Enterovirus A71 (EV-A71). Viruses 2020, 12, 184.
- Sun, H.; Wang, D.; Song, X. Natural Prenylchalconaringenins and Prenylnaringenins as Antidiabetic Agents: α-Glucosidase and α-Amylase Inhibition and in Vivo Antihyperglycemic and Antihyperlipidemic Effects. J. Agric. Food Chem. 2017, 65, 1574–1581.
- Samy, R.P.; Gopalakrishnakone, P. Therapeutic Potential of Plants as Anti-microbials for Drug Discovery. Evid. Based Complementary Altern. Med. 2010, 7, 283–294.
- Buer, C.S.; Imin, N.; Djordjevic, M.A. Flavonoids: New roles for old molecules. J. Integr. Plant Biol. 2010, 52, 98–111.
- Petrussa, E.; Braidot, E.; Zancani, M.; Peresson, C.; Bertolini, A.; Patui, S.; Vianello, A. Plant flavonoids-biosynthesis, transport and involvement in stress responses. Int. J. Mol. Med. Sci. 2013, 14, 14950–14973.
- Biharee, A.; Sharma, A.; Kumar, A.; Jaitak, V. Antimicrobial flavonoids as a potential substitute for overcoming antimicrobial resistance. Fitoterapia 2020, 146, 104720.
- Cushnie, T.P.T.; Lamb, A.J. Antimicrobial activity of flavonoids. Int. J. Antimicrob. Agents 2005, 26, 343–356.
- Solnier, J.; Martin, L.; Bhakta, S.; Bucar, F. Flavonoids as Novel Efflux Pump Inhibitors and Antimicrobials Against Both Environmental and Pathogenic Intracellular Mycobacterial Species. Molecules 2020, 25, 734.
- Araya-Cloutier, C.; Vincken, J.; Ederen, R.V.; Den Besten, H.M.W.; Gruppen, H. Rapid membrane permeabilization of Listeria monocytogenes and Escherichia coli induced by antibacterial prenylated phenolic compounds from legumes. Food Chem. 2018, 240, 147–155.
- Silva, S.L.; Oliveira, V.G.; Yano, T.; Nunomura, R.C.S. Atividade antimicrobiana da bergenina de Endopleura uchi (Huber) Cuatrec. Acta Amaz. 2009, 39, 187–191.
- Maduka, H.C.C.; Okoye, Z.S.C.; Eje, A. The influence of Sacoglottis gabonensis stem bark extract and its isolate bergenin, Nigerian alcoholic beverage additives, on the metabolic and haematological side effects of 2,4-dinitrophenyl hydrazine-induced tissue damage. Vasc. Pharmacol. 2003, 39, 317–324.
- Silva Neto, O.C.; Teodoro, M.T.F.; Nascimento, B.O. Bergenin of Peltophorum dubium (Fabaceae) Roots and Its Bioactive Semi-Synthetic Derivatives. J. Braz. Chem. Soc. 2020, 31, 2644–2650.
- Nunomura, R.C.S.; Oliveira, V.G.; Silva, S.L.; Nunomura, S.M. Characterization of bergenin in Endopleura uchi bark and its anti-inflammatory activity. J. Braz. Chem. Soc. 2009, 20, 1060–1064.
- Shah, M.R.; Arfan, M.; Amin, H. Synthesis of new bergenin derivatives as potent inhibitors of inflammatory mediators NO and TNF-α. Bioorganic Med. Chem. Lett. 2012, 22, 2744–2747.
- Oliveira, C.M.; Nonao, F.B.; Lima, F.O. Antinociceptive Properties of Bergenin. J. Nat. Prod. 2011, 74, 2062–2068.
- Singh, J.; Kumar, A.; Sharma, A. Antianxiety activity guided isolation and characterization of bergenin from Caesalpinia digyna Rottler roots. J. Ethnopharmacol. 2017, 195, 182–187.
- Uddin, G.; Sadat, A.; Siddiqui, B.S. Comparative antioxidant and antiplasmodial activities of 11-O-galloylbergenin and bergenin isolated from Bergenia ligulata. Trop. Biomed. 2014, 31, 143–148.
- Muniz, M.P.; Nunomura, S.M.; Lima, E.S.; Lima, A.S.; Almeida, P.D.O.; Nunomura, R.C.S. Quantification of bergenin, antioxidant activity and nitric oxide inhibition from bark, leaf and twig of Endopleura uchi. Química Nova 2020, 43, 413–418.
More
Information
Subjects:
Chemistry, Organic
Contributor
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
968
Revisions:
3 times
(View History)
Update Date:
08 Nov 2021
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





