
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Diego A. Moreno-Fernandez | + 3381 word(s) | 3381 | 2021-10-14 12:28:29 | | | |
| 2 | Diego A. Moreno-Fernandez | + 15 word(s) | 3396 | 2021-10-15 08:42:43 | | | | |
| 3 | Peter Tang | -43 word(s) | 3353 | 2021-10-15 09:35:31 | | |
Video Upload Options
Edible sprouts with germinating seeds of a few days of age are naturally rich in nutrients and other bioactive compounds. Among them, the cruciferous (Brassicaceae) sprouts stand out due to their high contents of glucosinolates (GLSs) and phenolic compounds.
1. Introduction
2. Bioactive Secondary Metabolites in Edible Cruciferous Sprouts
|
Edible Sprout |
Main Bioactive Compounds |
Main Bioactivities Associated with Sprout Consumption |
References |
|---|---|---|---|
|
Broccoli (Brassica oleracea var. Italica) |
Flavonoids Quercetin, kaempferol, and flavonol glycosides |
Cancer risk (↓) Degenerative diseases (↓) Obesity-related metabolic disorders (↓) Allergic nasal symptoms (↓) Inflammation (↓) Pain (↓) Antioxidant capacity (↑) |
|
|
Phenolic acids Chlorogenic, sinapic, and ferulic acid derivatives |
|||
|
Glucosinolates Glucoraphanin, glucoiberin, glucoraphenin, glucobrassicin, 4-hydroxyglucobrassicin, 4-methoxyglucobrassicin, and neoglucobrassicin |
|||
|
Isothiocyanates Sulphoraphane, iberin, and indole-3-carbinol |
|||
|
Radish (Raphanus sativus L.) |
Flavonoids Quercetin |
Risk of cancer (↓) Heart disease (↓) Diabetes (↓) Antioxidant capacity (↑) |
[8] |
|
Phenolic acids Ferulic, caffeic and p-coumaric acids, and derivatives |
|||
|
Glucosinolates Glucoraphenin, dehydroerucin, glucobrassicin, and 4-methoxyglucobrassicin |
|||
|
Isothiocyanates Sulforaphene, sulforaphane, and indole-3-carbinol |
|||
|
Kale (Brassica oleracea var. acephala) |
Flavonoids Quercetin and cyanidin |
Risk of cancer (↓) Heart disease (↓) Diabetes (↓) Antioxidant capacity (↑) |
[9] |
|
Phenolic acids Chlorogenic and ferulic acids |
|||
|
Glucosinolates Glucoraphanin, glucoiberin, gluconapin, gluconasturtin, progoitrin, gluconapin, gluconapoleiferin, sinigrin, glucobrassicin, 4-hydroxyglucobrassicin, 4-methoxyglucobrassicin, and neoglucobrassicin |
|||
|
Pak choi (Brassica rapa var. chinensis) |
Flavonoids Kaempferol, quercetin, and isorhamnetin glucosides |
Risk of cancer (↓) Heart disease (↓) Diabetes (↓) Antioxidant capacity (↑) |
|
|
Phenolic acids Ferulic, sinapic, caffeic, and p-coumaric acids, and derivatives |
|||
|
Glucosinolates Gluconapin, glucoalyssin, gluconasturtin, progoitrin, glucobrassicin, 4-hydroxyglucobrassicin, 4-methoxyglucobrassicin, and neoglucobrassicin |
3. Elicitation of Brassicaceae Sprouts to Enhance the Content of Bioactive (Poly)phenols and Glucosinolates
|
Raw Edible Sprout |
Elicitor Treatment |
Elicitor Classification |
Application |
Target Compound and Increase |
Reference |
|---|---|---|---|---|---|
|
Broccoli sprouts (Brassica oleracea) (7 days of growth) |
Sucrose, fructose, and glucose (146 mM) |
Biotic elicitor |
In 0.5% agar media for 5 days after sowing seeds |
Total anthocyanins (10.0%) |
[14] |
|
Broccoli sprouts (Brassica oleracea) (7 days of growth) |
Sucrose and mannitol (176 mM) |
Biotic elicitor |
Hydroponic system for 5 days after sowing seeds |
Total anthocyanins (40.0%) and phenolics (60.0%) Total glucosinolates (50.0%) |
[14] |
|
Broccoli (Brassica oleracea) (7 days of growth) |
Met (5 mM) Trp (10 mM) SA (100 μM) MeJA (25 μM) |
Biotic elicitors (Met, Trp, and plant hormones—SA and MeJA) |
Daily exogenous spraying during 3, 5, and 7 days |
Met: glucoiberin, glucoraphanin, and glucoerucin (30.0%) Trp: 4-hydroxyglucobrassicin, glucobrassicin, 4-Methoxyglucobrassicin, and neoglucobrassicin (80.0%) SA: 4-hydroxyglucobrassicin, glucobrassicin, 4-Methoxyglucobrassicin, and neoglucobrassicin (30.0%) MeJA: 4-hydroxyglucobrassicin, glucobrassicin, 4-Methoxyglucobrassicin, neoglucobrassicin (50.0%) |
[15] |
|
Broccoli sprouts (Brassica oleracea) |
Sucrose (146 mM) |
Biotic elicitor |
In 0.5% agar media for 5 days after sowing |
Total GLS (2.0-fold) |
[14] |
|
Broccoli sprouts (Brassica oleracea) (7 days of growth) |
Mg (300 mg L−1) |
Abiotic elicitor |
Suplementation with MgSO4 |
Increase of total ascorbic acid contain (29.1–44.5%) |
[16] |
|
Radish sprouts (raphanistrum subsp. sativus) (12 days of growth) |
MeJA (100 μM) |
Biotic elicitor (plant hormones—MeJA) |
Treatment with MeJA in growth chamber under dark conditions |
Glucoalyssin (1.4-fold) Glucoerucin (2.0-fold) Glucotropaeolin (1.8-fold) Glucoraphasatin (1.4-fold) |
[17] |
|
Radish sprouts (raphanistrum subsp. sativus) (12 days of growth) |
MeJA (100 μM) Light |
Biotic elicitor (plant hormones—MeJA-) Abiotic elicitor |
Treatment with MeJA in growth chamber under light |
Glucoraphanin (1.5-fold) Glucoerucin (1.6-fold) Glucotropaeolin (1.3-fold) 4-hydroxyglucobrassicin (4.4-fold) Pergonidin (1.7-fold) Cyanidin (2.0-fold) |
[17] |
|
Radish sprouts (raphanistrum subsp. sativus) (7 days of growth) |
Mg (300 mg L−1) |
Abiotic elicitor |
Supplementation with MgSO4 |
Phenolic compounds (13.9–21.7%) |
[16] |
|
Radish sprouts (raphanistrum subsp. sativus) |
NaCl (100 mM) |
Abiotic elicitor |
In 0.5% agar media for 3.5 and 7.0 days after sowing |
Total phenolics (30 and 50% in 5 and 7 day-old sprouts, respectively) Total GLS (50% and 120% in 5 and 7 day-old sprouts, respectively) |
[18] |
|
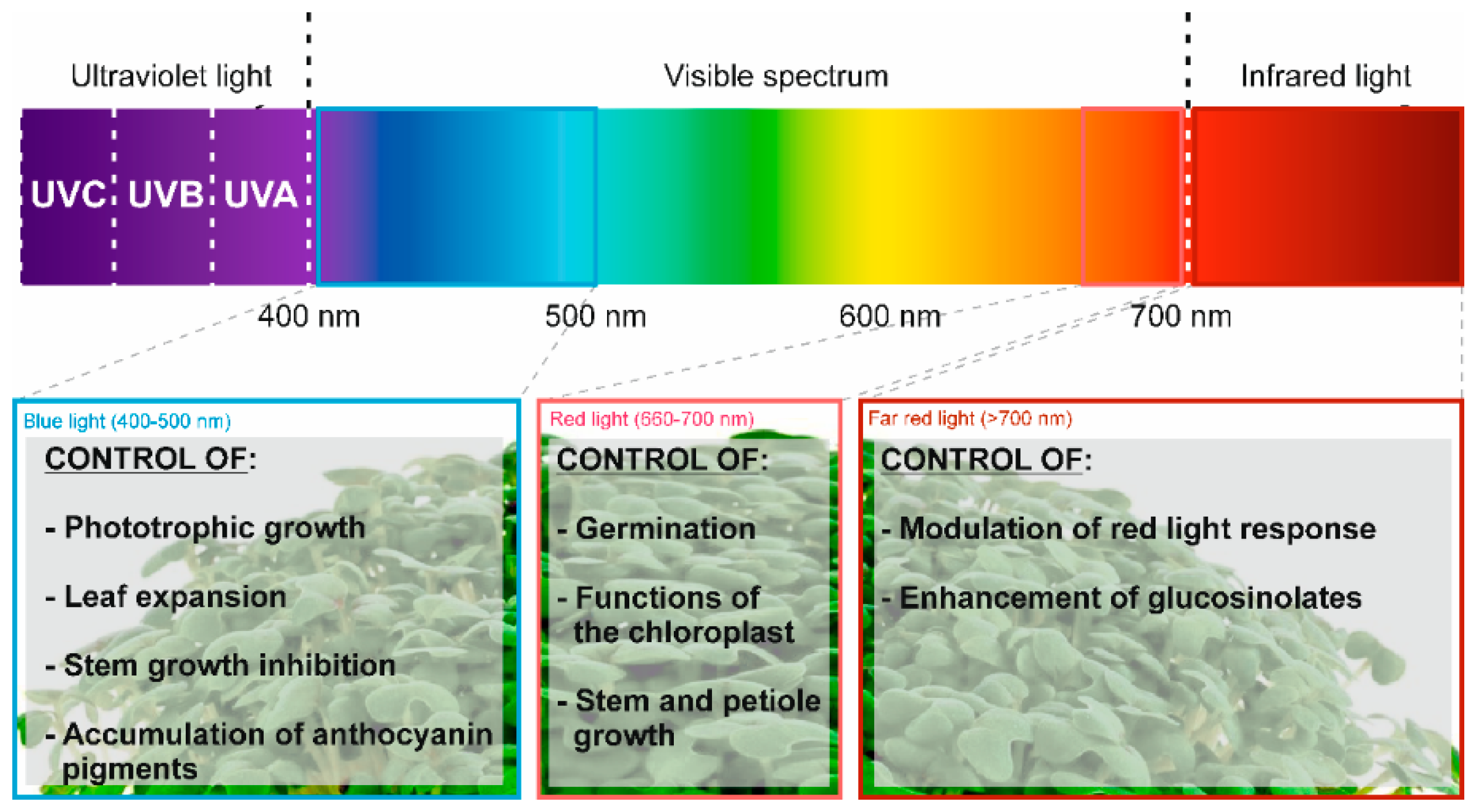
Pak Choi sprouts (rapa subsp. chinensis) |
Application of different wavelengths of LED light (white, blue, and red) |
Abiotic elicitor |
Medium of perlite for 5 days in darkness and 18 h at the different wavelengths |
Total carotenoid content (12.1% and 9.2% with white light (respect to blue and red light, respectively) |
[11] |
|
Pak Choi sprouts (rapa subsp. chinensis) |
Application of different wavelengths of LED light (white, blue, and red) |
Abiotic elicitor |
Medium of perlite for 5 days in darkness and 18 h at the different wavelengths |
Enhanced transcription of genes involved in carotenoid biosynthesis (CYP97A3, CYP97C1, βLCY, εLCY, β-OHASE1, PDS, PSY, VDE, ZEP) |
[11] |
|
Kale Sprouts (oleracea var. sabellica) |
Application of different light wavelengths (470, 660, and 730 nm) |
Abiotic elicitor |
Seeds stratified for 2 days, exposed to light for 1 h, exposed to darkness for between 1 and 3 days and later, the specific light treatment |
Total GLS content (31.7%) |
[19] |
|
Radish, Chinese kale and pak choi sprouts (3 days of growth) |
Glucose (5 g 100 mL−1) |
Biotic elicitor |
Hydroponic system for 3 days after sowing seeds |
Total phenolics (20.0%), gluconapin (150.0% and 60.0% in Chinese kale and pak choi, respectively), glucobrassicanapin (110-fold in pak choi) |
[20] |
|
Different Brassica sprouts (broccoli, turnip, and rutabaga) |
MeJA (25 μM) JA (150 μM) Sucrose (146 mM) |
Biotic elicitors (Sucrose and plant hormones—MeJA and JA) |
Sprayed for 5 days before harvest |
Total GLS (>50%, broccoli; >20.0% turnip; >100.0% rutabaga) |
[21] |
|
Radish sprouts (raphanistrum subsp. sativus) (8 days of growth) |
MeJA (25 μM) SA (100 μM) Glucose (277 mM) |
Biotic elicitors (glucose and plant hormones—MeJA and JA) |
Sprayed for 5 days before harvest |
Total GLS (20.0%) |
[21] |
4. The Challenges of Including Cruciferous Sprouts in Balanced Diets and Personalized Nutrition
|
Matrix |
Pathophysiological Condition |
Effect |
Model |
Action Mechanism Z |
Ref. |
|---|---|---|---|---|---|
|
Broccoli sprouts |
Metabolic profile |
No specific effect monitored |
Humans |
FA 14:1, FA 16:1, FA 18:1, FA 14:0, FA 16:0, FA 18:0, dehydroepiandrosterone, glutathione, cysteine, and glutamine (↑) Deoxy-uridin monophosohate (↓) |
[29] |
|
Radish sprouts |
Energy metabolism |
Decrease glucose level |
Drosophila melanogaster |
Expression of spargel (↑) |
[30] |
|
Broccoli sprouts |
Pregnancy |
Prevention of brain injury in newborns |
Rats |
Not determined |
[31] |
|
Broccoli sprouts |
Inflammation and oxidative stress |
Modulation of inflammation and vascular events |
Humans |
Not determined |
[32] |
|
Broccoli sprouts |
Inflammation in overweight population |
Anti-inflammatory activity |
Humans |
IL-6 and C-reactive protein (↓) |
[33] |
|
Broccoli sprout powder |
Diabetes |
Anti-inflammatory effect |
Humans |
C-reactive protein (↓) |
[34] |
|
Broccoli sprouts |
Hypertension |
Does not improve endothelial function of hypertension in humans |
Humans |
Not determined |
[35] |
|
Broccoli sprouts |
Hypertension |
Attenuation of oxidative stress, hypertension, and inflammation |
Rats |
Not determined |
[36] |
|
Rutabaga sprouts |
Thyroid function and iodine deficiency. Role as goitrogenic foods |
Protective effect against thyroid damage Goitrogenic activity not discarded |
Male rats |
Dietary source of iodine GPX1, GPX3, and FRAP (↓) |
[37] |
|
Broccoli sprouts |
Hepatic and renal toxicity |
Antioxidant activity |
Female rats |
Phase-II enzymes (↑) Lipid peroxidation and apoptosis (↓) |
[38] |
|
Broccoli sprouts |
Bowel habits |
Decrease in the constipation scoring system Decrease of Bifidobacterium |
Humans |
Not determined |
[39] |
|
Broccoli sprouts |
Pain assessment and analgesia |
Dose-dependent nociceptive activity |
Rats |
Agonists of central and peripheral opioid receptors |
[40] |
|
Tuscan black cabbage sprout extract |
Xenobiotic metabolism and antioxidant defense |
Improvement of the detoxification of xenebiotics |
Rats |
Induction of phase-II enzymes and boosting of the enzymatic activity of catalase, NAD(P)H:quinone reductase, glutathione reductase, and glutathione peroxidase |
[41] |
|
Japanese Radish Sprout |
Diabetes |
Decrease in plasma fructosamine, glucose, and insulin in diabetic rats |
Rats |
Not determined |
[27] |
|
Radish sprouts |
Diabetes |
Increase in blood glucose, triglycerides, total cholesterol, low-density lipoproteins, and very low density lipoproteins |
Rats |
Not determined |
[42] |
|
Broccoli sprout extracts |
Skin disorders |
Induction of phase-II response |
Mice and humans |
NQO1 enzyme activity (↑) |
[43] |
|
Broccoli sprout extracts |
Skin disorders |
Protection against inflammation, edema, and carcinogens in humans |
Humans |
Phase-II enzymes (↑) NQO1 enzyme activity (↑) |
[44] |
|
Broccoli sprout homogenate |
Physiological upper airway |
No specific effect monitored |
Humans |
Phase-II enzymes (↑) |
[45] |
|
Broccoli sprouts |
Physiological upper airway |
No specific effect monitored |
Humans |
Nrf2 activity (↑) Secretory leukocyte protease inhibitor (↑) |
[46] |
|
Broccoli sprout extract |
Asthma |
Blocking the bronchoconstrictor hyperresponsiveness of some asthmatic phenotypes |
Humans |
Activity of Nrf2 regulated antioxidant and anti-inflammatory genes (↓) |
[47] |
|
Broccoli sprout extract |
Hepatic disturbances |
Improvement of liver functions and reduction of oxidative stress |
Rats |
Not determined |
[48] |
|
Broccoli sprout-based supplements |
General carcinogenic processes |
Chemopreventive effect |
Humans |
Not determined |
[49] |
|
Broccoli sprout extract |
Head and neck squamous cell carcinoma |
Chemopreventive activity of sulforaphane against carcinogen-induced oral cancer |
Mice |
Time and dose dependent induction of Nrf2 and Nrf2 target genes (NQO1 and GCLC) Dephosphorilation of pSTAT3 |
[50] |
|
Broccoli sprouts homogenate |
Sickle cell disease (hemoglobinopathy) |
Change in the gene expression levels |
Humans |
Expression of Nrf2 targets (HMOX1 and HBG1) (↑) |
[51] |
|
Broccoli sprouts |
Oxidative stress |
Improvement in cholesterol metabolism and decrease in oxidative stress |
Humans |
Not determined |
[52] |
|
Broccoli sprouts |
General carcinogenic processes |
Chemopreventive agent |
Humans |
Histone deacetylase activity (↓) |
[53] |
|
Broccoli sprouts |
Unspecific frame |
Not determined |
Humans |
Histone deacetylase activity (↓) |
[54] |
|
Broccoli sprouts |
Antimicrobial activity against Helicobacter pylori |
Reduction of Helicobacter pylori colonization in mice Enhancement of sequelae of Helicobacter pylori infection in mice and humans |
Mice and humans |
Not determined |
[55] |
|
Broccoli sprout extract |
Allergic response |
Broccoli sprouts reduce the impact of particulate pollution of allergic disease and asthma |
Humans |
Not determined |
[56] |
|
Broccoli sprout extract |
Prostate cancer |
Inconclusive |
Humans |
Not determined |
[57] |
|
Broccoli sprout and myrosinase-treated broccoli sprout extracts |
Chemoprevention of carcinogenesis processes |
Inconclusive |
Humans |
No dose response was observed for molecular targets |
[58] |
|
Broccoli sprout extract |
Psychiatric disorders |
Improvement of the cognitive function in patients affected by schizophrenia |
Humans |
Not determined |
[59] |
|
Broccoli sprout extract |
Type II diabetes |
Reduction of fasting blood glucose and glycated hemoglobin |
Mice |
(↑) Nuclear translocation of Nrf2 (↓) Glucose production and intolerance |
[60] |
|
Broccoli sprout extract |
Neurological disorder |
Inconclusive improvement of Autism symptoms |
Humans |
(↑) Gene transcription in multiple cell signaling pathways |
[61] |
|
Broccoli sprout homogenate |
Viral infections |
Enhancement of antiviral defense response |
Humans |
Modulation of natural killer cell activation Production of granzyme B by natural killer cells (↑) |
[62] |
Z FA, fatty acids; FRAP, ferric reducing activity of plasma; GCLC, glutamate-cysteine ligase catalytic subunit; GPX1, cytosolic glutathione peroxidase-1; GPX3, cytosolic glutathione peroxidase-3; HBG1, Hemoglobin subunit gamma 1; HMOX1, heme oxygenase (decycling) 1; IL-6, interleukina 6; NAD(P)H, nicotinamide adenine dinucleotide phosphate; NQO1, NAD(P)H:quinone oxidoreductase 1; TNF-α, tumor necrosis factor-alpha; Nrf2, nuclear factor erythroid 2–related factor 2; pSTAT3, signal transducer and activator of transcription-3; TSH, thyroid stimulating hormone. (↓↑) Non-significant variation, (↓) decrease, and (↑) increase.
References
- Gan, R.-Y.; Lui, W.-Y.; Wu, K.; Chan, C.-L.; Dai, S.-H.; Sui, Z.-Q.; Corke, H. Bioactive compounds and bioactivities of germinated edible seeds and sprouts: An updated review. Trends Food Sci. Technol. 2017, 59, 1–14.
- Moreno, D.A.; Perez-Balibrea, S.; Garcia-Viguera, C. Phytochemical quality and bioactivity of edible sprouts. Nat. Prod. Commun. 2006, 11, 1037–1048.
- Baenas, N.; Ferreres, F.; García-Viguera, C.; Moreno, D.A. Radish sprouts—Characterization and elicitation of novel varieties rich in anthocyanins. Food Res. Int. 2015, 69, 305–312.
- Conzatti, A.; Telles da Silva Fróes, F.C.; Schweigert Perry, I.D.; Guerini de Souza, C. Clinical and molecular evidence of the consumption of broccoli, glucoraphanin and sulforaphane in humans. Nutr. Hosp. 2015, 31, 559–569.
- Baenas, N.; Gómez-Jodar, I.; Moreno, D.A.; García-Viguera, C.; Periago, P.M. Broccoli and radish sprouts are safe and rich in bioactive phytochemicals. Postharvest Boil. Technol. 2017, 127, 60–67.
- Gagné, F. Chapter 6—Oxidative Stress. In Biochemical Ecotoxicology; Gagné, F., Ed.; Academic Press: Oxford, UK, 2014; pp. 103–115.
- Wang, C.; Wang, C. Anti-nociceptive and anti-inflammatory actions of sulforaphane in chronic constriction injury-induced neuropathic pain mice. Inflammopharmacology 2016, 25, 99–106.
- Li, R.; Zhu, Y. The primary active components, antioxidant properties, and differential metabolite profiles of radish sprouts (Raphanus sativus L.) upon domestic storage: Analysis of nutritional quality. J. Sci. Food Agric. 2018, 98, 5853–5860.
- Jeon, J.; Kim, J.K.; Kim, H.; Kim, Y.J.; Park, Y.J.; Kim, S.J.; Kim, C.; Park, S.U. Transcriptome analysis and metabolic profiling of green and red kale (Brassica oleracea var. acephala) seedlings. Food Chem. 2018, 241, 7–13.
- Liang, X.; Lee, H.W.; Li, Z.; Lu, Y.; Zou, L.; Ong, C.N. Simultaneous Quantification of 22 Glucosinolates in 12 Brassicaceae Vegetables by Hydrophilic Interaction Chromatography–Tandem Mass Spectrometry. ACS Omega 2018, 3, 15546–15553.
- Frede, K.; Schreiner, M.; Zrenner, R.; Graefe, J.; Baldermann, S. Carotenoid biosynthesis of pak choi (Brassica rapa ssp. chinensis) sprouts grown under different light-emitting diodes during the diurnal course. Photochem. Photobiol. Sci. 2018, 17, 1289–1300.
- Baenas, N.; Villaño, D.; García-Viguera, C.; Moreno, D.A. Optimizing elicitation and seed priming to enrich broccoli and radish sprouts in glucosinolates. Food Chem. 2016, 204, 314–319.
- Baenas, N.; García-Viguera, C.; Moreno, A.D. Elicitation: A Tool for Enriching the Bioactive Composition of Foods. Molecules 2014, 19, 13541.
- Guo, R.; Yuan, G.; Wang, Q. Sucrose enhances the accumulation of anthocyanins and glucosinolates in broccoli sprouts. Food Chem. 2011, 129, 1080–1087.
- Pérez-Balibrea, S.; Moreno, D.A.; García-Viguera, C. Improving the phytochemical composition of broccoli sprouts by elicitation. Food Chem. 2011, 129, 35–44.
- Przybysz, A.; Wrochna, M.; Małecka-Przybysz, M.; Gawrońska, H.; Gawroński, S.W. The effects of Mg enrichment of vegetable sprouts on Mg concentration, yield and ROS generation. J. Sci. Food Agric. 2016, 96, 3469–3476.
- Al-Dhabi, N.A.; Arasu, M.V.; Kim, S.J.; RomijUddin, M.; Park, W.T.; Lee, S.Y.; Park, S.U. Methyl Jasmonate- and Light-Induced Glucosinolate and Anthocyanin Biosynthesis in Radish Seedlings. Nat. Prod. Commun. 2015, 10, 1211–1214.
- Yuan, G.; Wang, X.; Guo, R.; Wang, Q. Effect of salt stress on phenolic compounds, glucosinolates, myrosinase and antioxidant activity in radish sprouts. Food Chem. 2010, 121, 1014–1019.
- Carvalho, S.D.; Folta, K.M. Sequential light programs shape kale (Brassica napus) sprout appearance and alter metabolic and nutrient content. Hortic. Res. 2014, 1, 8.
- Wei, J.; Miao, H.; Wang, Q. Effect of glucose on glucosinolates, antioxidants and metabolic enzymes in Brassica sprouts. Sci. Hortic. 2011, 129, 535–540.
- Baenas, N.; García-Viguera, C.; Moreno, D.A. Biotic Elicitors Effectively Increase the Glucosinolates Content in Brassicaceae Sprouts. J. Agric. Food Chem. 2014, 62, 1881–1889.
- Park, W.T.; Kim, Y.B.; Seo, J.M.; Kim, S.-J.; Chung, E.; Lee, J.-H.; Park, S.U. Accumulation of Anthocyanin and Associated Gene Expression in Radish Sprouts Exposed to Light and Methyl Jasmonate. J. Agric. Food Chem. 2013, 61, 4127–4132.
- Guo, R.; Yuan, G.; Wang, Q. Effect of sucrose and mannitol on the accumulation of health-promoting compounds and the activity of metabolic enzymes in broccoli sprouts. Sci. Hortic. 2011, 128, 159–165.
- Gupta, S.C.; Kim, J.H.; Prasad, S.; Aggarwal, B.B. Regulation of survival, proliferation, invasion, angiogenesis, and metastasis of tumor cells through modulation of inflammatory pathways by nutraceuticals. Cancer Metastasis Rev. 2010, 29, 405–434.
- Surh, Y.J. Cancer chemoprevention with dietary phytochemicals. Nat. Rev. Cancer 2003, 3, 768–780.
- Banihani, S.A. Radish (Raphanus sativus) and Diabetes. Nutrients 2017, 9, 1014.
- Taniguchi, H.; Kobayashi-Hattori, K.; Tenmyo, C.; Kamei, T.; Uda, Y.; Sugita-Konishi, Y.; Oishi, Y.; Takita, T. Effect of Japanese radish (Raphanus sativus) sprout (Kaiware-daikon) on carbohydrate and lipid metabolisms in normal and streptozotocin-induced diabetic rats. Phytother. Res. 2006, 20, 274–278.
- Rescigno, T.; Tecce, M.F.; Capasso, A. Protective and restorative effects of nutrients and phytochemicals. Open Biochem. J. 2018, 12, 46–64.
- Housley, L.; Magana, A.A.; Hsu, A.; Beaver, L.M.; Wong, C.P.; Stevens, J.F.; Choi, J.; Jiang, Y.; Bella, D.; Williams, D.E.; et al. Untargeted Metabolomic Screen Reveals Changes in Human Plasma Metabolite Profiles Following Consumption of Fresh Broccoli Sprouts. Mol. Nutr. Food Res. 2018, 62, 1700665.
- Baenas, N.; Piegholdt, S.; Schloesser, A.; Moreno, D.A.; García-Viguera, C.; Rimbach, G.; Wagner, A.E. Metabolic Activity of Radish Sprouts Derived Isothiocyanates in Drosophila melanogaster. Int. J. Mol. Sci. 2016, 17, 251.
- Black, A.M.; Armstrong, E.A.; Scott, O.; Juurlink, B.J.H.; Yager, J.Y. Broccoli sprout supplementation during pregnancy prevents brain injury in the newborn rat following placental insufficiency. Behav. Brain Res. 2015, 291, 289–298.
- Medina, S.; Domínguez-Perles, R.; Moreno, D.A.; García-Viguera, C.; Ferreres, F.; Gil, J.I.; Gil-Izquierdo, Á. The intake of broccoli sprouts modulates the inflammatory and vascular prostanoids but not the oxidative stress-related isoprostanes in healthy humans. Food Chem. 2015, 173, 1187–1194.
- Lopez-Chillon, M.T.; Carazo-Diaz, C.; Prieto-Merino, D.; Zafrilla, P.; Moreno, D.A.; Villano, D. Effects of long-term consumption of broccoli sprouts on inflammatory markers in overweight subjects. Clin. Nutr. 2018.
- Mirmiran, P.; Bahadoran, Z.; Hosseinpanah, F.; Keyzad, A.; Azizi, F. Effects of broccoli sprout with high sulforaphane concentration on inflammatory markers in type 2 diabetic patients: A randomized double-blind placebo-controlled clinical trial. J. Funct. Foods 2012, 4, 837–841.
- Christiansen, B.; Bellostas Muguerza, N.; Petersen, A.M.; Kveiborg, B.; Madsen, C.R.; Thomas, H.; Ihlemann, N.; Sorensen, J.C.; Kober, L.; Sorensen, H.; et al. Ingestion of broccoli sprouts does not improve endothelial function in humans with hypertension. PLoS ONE 2010, 5, e12461.
- Wu, L.; Noyan Ashraf, M.H.; Facci, M.; Wang, R.; Paterson, P.G.; Ferrie, A.; Juurlink, B.H.J. Dietary approach to attenuate oxidative stress, hypertension, and inflammation in the cardiovascular system. Proc. Natl. Acad. Sci. USA 2004, 101, 7094–7099.
- Paśko, P.; Okoń, K.; Krośniak, M.; Prochownik, E.; Żmudzki, P.; Kryczyk-Kozioł, J.; Zagrodzki, P. Interaction between iodine and glucosinolates in rutabaga sprouts and selected biomarkers of thyroid function in male rats. J. Trace Elem. Med. Boil. 2018, 46, 110–116.
- Sharma, D.; Sangha, G.K. Antioxidative effects of aqueous extract of broccoli sprouts against Triazophos induced hepatic and renal toxicity in female Wistar rats. J. Appl. Biomed. 2018, 16, 100–110.
- Yanaka, A. Daily intake of broccoli sprouts normalizes bowel habits in human healthy subjects. J. Clin. Biochem. Nutr. 2018, 62, 75–82.
- Baenas, N.; Gonzalez-Trujano, M.E.; Guadarrama-Enriquez, O.; Pellicer, F.; Garcia-Viguera, C.; Moreno, D.A. Broccoli sprouts in analgesia—Preclinical in vivo studies. Food Funct. 2017, 8, 167–176.
- Melega, S.; Canistro, D.; Pagnotta, E.; Iori, R.; Sapone, A.; Paolini, M. Effect of sprout extract from Tuscan black cabbage on xenobiotic-metabolizing and antioxidant enzymes in rat liver. Mutat. Res. Genet. Toxicol. Environ. Mutagen. 2013, 751, 45–51.
- Aly, A.A.T.; Fayed, S.A.; Ahmed, A.M.; El Rahim, E.A. Effect of Egyptian Radish and Clover Sprouts on Blood Sugar and Lipid Metabolisms in Diabetic Rats. Glob. J. Biotechnol. Biochem. 2015, 10, 16–21.
- Dinkova-Kostova, A.T.; Fahey, J.W.; Wade, K.L.; Jenkins, S.N.; Shapiro, T.A.; Fuchs, E.J.; Kerns, M.L.; Talalay, P. Induction of the phase 2 response in mouse and human skin by sulforaphane-containing broccoli sprout extracts. Cancer Epidemiol. Biomark. Prev. 2007, 16, 847–851.
- Talalay, P.; Fahey, J.W.; Healy, Z.R.; Wehage, S.L.; Benedict, A.L.; Min, C.; Dinkova-Kostova, A.T. Sulforaphane mobilizes cellular defenses that protect skin against damage by UV radiation. Proc. Natl. Acad. Sci. USA 2007, 104, 17500–17505.
- Riedl, M.A.; Saxon, A.; Diaz-Sanchez, D. Oral sulforaphane increases Phase II antioxidant enzymes in the human upper airway. Clin. Immunol. 2009, 130, 244–251.
- Meyer, M.; Kesic, M.J.; Clarke, J.; Ho, E.; Simmen, R.C.; Diaz-Sanchez, D.; Noah, T.L.; Jaspers, I. Sulforaphane induces SLPI secretion in the nasal mucosa. Respir. Med. 2013, 107, 472–475.
- Brown, R.H.; Reynolds, C.; Brooker, A.; Talalay, P.; Fahey, J.W. Sulforaphane improves the bronchoprotective response in asthmatics through Nrf2-mediated gene pathways. Respir. Res. 2015, 16, 106.
- Kikuchi, M.; Ushida, Y.; Shiozawa, H.; Umeda, R.; Tsuruya, K.; Aoki, Y.; Suganuma, H.; Nishizaki, Y. Sulforaphane-rich broccoli sprout extract improves hepatic abnormalities in male subjects. World J. Gastroenterol. 2015, 21, 12457–12467.
- Ushida, Y.S.; Suganuma, H.; Yanaka, A. Low-Dose of the Sulforaphane Precursor Glucoraphanin as a Dietary Supplement Induces Chemoprotective Enzymes in Humans. Food Nutr. Sci. 2015, 6, 1603–1612.
- Bauman, J.E.; Zang, Y.; Sen, M.; Li, C.; Wang, L.; Egner, P.A.; Fahey, J.W.; Normolle, D.P.; Grandis, J.R.; Kensler, T.W.; et al. Prevention of Carcinogen-Induced Oral Cancer by Sulforaphane. Cancer Prev. Res. 2016, 9, 547–557.
- Doss, J.F.; Jonassaint, J.C.; Garrett, M.E.; Ashley-Koch, A.E.; Telen, M.J.; Chi, J.T. Phase 1 Study of a Sulforaphane-Containing Broccoli Sprout Homogenate for Sickle Cell Disease. PLoS ONE 2016, 11, e0152895.
- Murashima, M.; Watanabe, S.; Zhuo, X.G.; Uehara, M.; Kurashige, A. Phase 1 study of multiple biomarkers for metabolism and oxidative stress after one-week intake of broccoli sprouts. Biofactors 2004, 22, 271–275.
- Myzak, M.C.; Tong, P.; Dashwood, W.M.; Dashwood, R.H.; Ho, E. Sulforaphane retards the growth of human PC-3 xenografts and inhibits HDAC activity in human subjects. Exp. Biol. Med. 2007, 232, 227–234.
- Clarke, J.D.; Riedl, K.; Bella, D.; Schwartz, S.J.; Stevens, J.F.; Ho, E. Comparison of isothiocyanate metabolite levels and histone deacetylase activity in human subjects consuming broccoli sprouts or broccoli supplement. J. Agric. Food Chem. 2011, 59, 10955–10963.
- Yanaka, A.; Fahey, J.W.; Fukumoto, A.; Nakayama, M.; Inoue, S.; Zhang, S.; Tauchi, M.; Suzuki, H.; Hyodo, I.; Yamamoto, M. Dietary sulforaphane-rich broccoli sprouts reduce colonization and attenuate gastritis in Helicobacter pylori-infected mice and humans. Cancer Prev. Res. 2009, 2, 353–360.
- Heber, D.; Li, Z.; Garcia-Lloret, M.; Wong, A.M.; Lee, T.Y.; Thames, G.; Krak, M.; Zhang, Y.; Nel, A. Sulforaphane-rich broccoli sprout extract attenuates nasal allergic response to diesel exhaust particles. Food Funct. 2014, 5, 35–41.
- Alumkal, J.J.; Slottke, R.; Schwartzman, J.; Cherala, G.; Munar, M.; Graff, J.N.; Beer, T.M.; Ryan, C.W.; Koop, D.R.; Gibbs, A.; et al. A phase II study of sulforaphane-rich broccoli sprout extracts in men with recurrent prostate cancer. Investig. New Drugs 2015, 33, 480–489.
- Atwell, L.L.; Zhang, Z.; Mori, M.; Farris, P.; Vetto, J.T.; Naik, A.M.; Oh, K.Y.; Thuillier, P.; Ho, E.; Shannon, J. Sulforaphane Bioavailability and Chemopreventive Activity in Women Scheduled for Breast Biopsy. Cancer Prev. Res. 2015, 8, 1184–1191.
- Shiina, A.; Kanahara, N.; Sasaki, T.; Oda, Y.; Hashimoto, T.; Hasegawa, T.; Yoshida, T.; Iyo, M.; Hashimoto, K. An Open Study of Sulforaphane-rich Broccoli Sprout Extract in Patients with Schizophrenia. Clin. Psychopharmacol. Neurosci. 2015, 13, 62–67.
- Axelsson, A.S.; Tubbs, E.; Mecham, B.; Chacko, S.; Nenonen, H.A.; Tang, Y.; Fahey, J.W.; Derry, J.M.J.; Wollheim, C.B.; Wierup, N.; et al. Sulforaphane reduces hepatic glucose production and improves glucose control in patients with type 2 diabetes. Sci. Transl. Med. 2017, 9, eaah4477.
- Singh, K.; Connors, S.L.; Macklin, E.A.; Smith, K.D.; Fahey, J.W.; Talalay, P.; Zimmerman, A.W. Sulforaphane treatment of autism spectrum disorder (ASD). Proc. Natl. Acad. Sci. USA 2014, 111, 15550–15555.
- Muller, L.; Meyer, M.; Bauer, R.N.; Zhou, H.; Zhang, H.; Jones, S.; Robinette, C.; Noah, T.L.; Jaspers, I. Effect of Broccoli Sprouts and Live Attenuated Influenza Virus on Peripheral Blood Natural Killer Cells: A Randomized, Double-Blind Study. PLoS ONE 2016, 11, e0147742.





