
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | M Abdullah | + 3026 word(s) | 3026 | 2021-10-10 07:56:00 | | | |
| 2 | Conner Chen | -10 word(s) | 3016 | 2021-10-11 07:51:56 | | |
Video Upload Options
The application of epoxy adhesive is widespread in electronic packaging. Epoxy adhesives can be integrated with various types of nanoparticles for enhancing thermal conductivity. The joints with thermally conductive adhesive (TCA) are preferred for research and advances in thermal management. The rapid advancement of microelectronic technology has resulted in the progressive transformation of electronic components from isolated to highly integrated. It produces a lot of heat while they are functioning. The materials and structures used to attach the semiconductor chip to other electronic components involve the sector of electronic packaging. Conductive adhesives represent a significant advancement in electronic packaging for advanced devices, where effective heat dissipation and enough electrical conductivity are critical. The TCA joint is a crucial choice for the thermal management of the device. The advances in TCA are essential because of future demands for inexpensive and handy electronic devices. The adhesive applications have become more important due to their remarkable versatility and unparalleled compatibility. The electronic industry has indeed developed, especially in consumer electronics, which depends on conductive adhesives. It would not be possible to identify the most modern electronic devices without conductive adhesives.
1. Formulation of Thermally Conductive Adhesive and Heat Transfer Mechanism
A polymer matrix and thermally conductive filler make thermally conductive adhesives that are formed as composites. Nanoparticles are having a diameter of less than 100 nm exhibit unique physical and mechanical properties. These fillers have a large specific surface area, high surface energy, and interfacial area with the polymeric matrix [1]. Thermal conductivity of epoxy is inadequate to remove heat produced from electronic devices. Dispersing nanoparticles inside an epoxy adhesive matrix can dramatically improve the adhesive’s thermal properties [2] and the performance of adhesively bonded joints [3]. The high thermal conductive solid particles like—graphene [4], carbon nanotubes [5], carbon fiber, carbon black are the most common carbon-based fillers. Metallic (gold, copper, silver, aluminium) [6], and ceramic (aluminium nitride, boron nitride, silicon carbide, aluminium oxide) [7] fillers have been blended with matrix resin to increase the thermal conductivity. Carbon-based and metallic filler adhesive has received significant attention because of its excellent thermal conductivity and specific surface area [8]. These conductive filler particles can alter polymer matrix isolation properties by making them very heat conductive. The thermally conductive adhesive is a composite material, and it follows the same formulation process of nano-composite [9]. Most common formulation process starts with the filler’s dispersion with organic solvents. An example of graphene nanoparticle (GNP)-based nano-composite [10] is shown in Figure 1. The process until the application, i.e., (a) to (h), is maintained for all thermally conductive adhesive according to the filler type. Bubbles and voids reduce the bonding quality of the adhesive [11]. To avoid the bubble and void in the adhesive joint the vacuum degassing is important in thermally conductive adhesive production process.
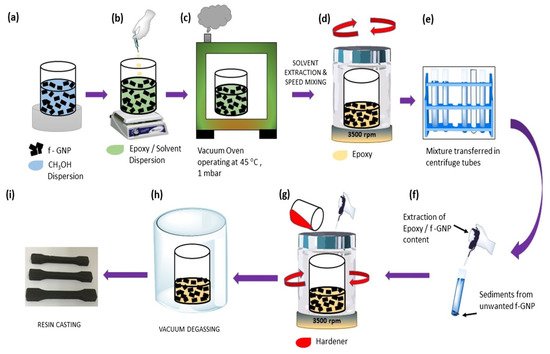
Figure 1. Schematic of the dispersion (a) to application (i) process of conductive filler in nano-composite [10].
The non-conductive properties can be transformed into conductive properties with the addition of fine conductive filler particles in polymer matrices, resulting in a continuous thermal conductive network [12]. The thermal interface material (TIM) required high thermal conductivity. The thermal conductivity of polymers ranged from 0.1 to 0.5 Wm−1 K−1 [13]. The simple form of polymer matrix is insufficient to meet the thermal conductivity requirement. Conductive filler particles increase heat conductivity while conserving polymer matrix characteristics [14]. Before integrating the epoxy with the filler, it is essential to know the thermal conductivity of the filler. A list of widely used conductive fillers is presented in Table 1.
Table 1. Thermal conductivity (TC) of materials used as conductive fillers [15][16][17][18][19].
| Material | TC (Wm−1 K−1) | Material | TC (Wm−1 K−1) |
|---|---|---|---|
| Aluminum oxide | 20–30 | Graphite | 100–400 (on plane) |
| Molybdenum | 142 | Silver | 450 |
| Tungsten | 155 | Copper | 401 |
| Nickel | 158 | Silicon carbide (SiC) | 490 |
| Aluminum | 204 | Diamond | 2000 |
| Beryllium oxide | 260 | Boron nitride | ~2000 (in-plane); ~380 (out-of-plane) |
| Carbon fiber | 260 | Multiwalled carbon nanotube (MWCNT) | ~3000 |
| Aluminum nitride (AlN) | 200–320 | Graphene | ~5300 |
| Gold | 345 | Single-walled carbon nanotube (SWCNT) | ~6000 |
Electrons and phonons are responsible to heat transfer in metal. Due to the lack of free electrons in non-metals, the phonon is responsible for heat transmission. When a polymer surface comes into contact with a heat source, heat is transferred via vibrations to the nearest atom, then to the next, and so on [20]. The formulation of thermally conductive adhesives consists of a polymer matrix, filler, and specific additives that are uniform and isotropic at the macro scale.
The thermal conductivity of the polymer matrix is less than 0.5 Wm−1 K−1. Thermal conductivity is improved by incorporating a highly thermally conductive filler into the insulating polymer matrix. The filler has a significant influence on the thermal conductivity of polymer composites. In the case of the filler network, it can speed heat transmission along the continuous and integrated filler network with less phonon scattering. Significantly, optimizing the filler structure (including size and aspect ratio) and dispersion can improve thermal conductivity by arranging the filler in a thermally conductive network within the polymer matrix [21]. Therefore, heat transmission across TCA occurs when filler particles establish a conductive path. A general heat transfer mechanism of TCA is shown in Figure 2.
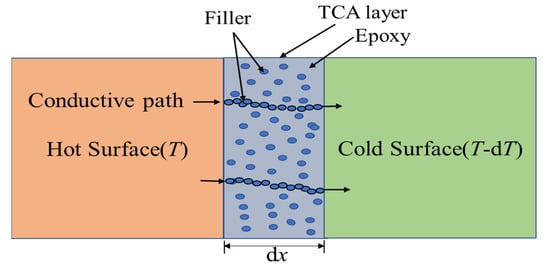
Figure 2. Heat conduction mechanism of TCA.
The basic principle of heat conductance through a thermally conductive adhesive layer dx (Figure 2), also known as Fourier’s law [22], states that the rate of heat transfer through a material is proportional to the negative gradient in the temperature and to the area at right angles to that gradient through which the heat flows and can be written as follows:

where Q is the quantity of heat energy (J), t is time (s), λ is a thermal conductivity (Wm−1 K−1), dT/dx is the temperature gradient in the heat flow direction (K/m), x is the distance along the direction of heat flow (m), A is the area of the cross-section (m2). The thermal conductivity is given by:
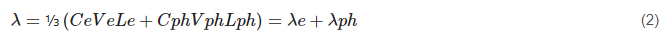
where Ce and Cph are the heat capacities per unit volume (J/m3 K) of electrons and phonons, respectively, Ve and Vph are their root-mean-square velocities and Le, Lph are their mean free paths. The thermal conductivity of electron type λe is dominant for metals, and one can roughly assume that:

In such a case, the ratio of thermal conductivity λe and electrical conductivity σe, according to Wiedemann–Franz’s law, can be expressed as:

where T is temperature (K) and L is the Lorenz constant, the theoretical value of which is 2.44 × 10–8 W·Ω/K2. The transport of heat in nonmetals occurs mainly by phonons. For insulators, thermal conductivity increases linearly with temperature, and the efficiency of phonon heat conduction is size dependent. In nanostructures, the thermal conductivity of a phonon type λph may be drastically different than in macrostructures. When the size of a nanostructure approaches or exceeds the mean-free path of a phonon, phonons collide with the border more frequently than in bulk materials. This extra collision mechanism increases the resistance to heat transfer, hence lowering the effective thermal conductivity of thin films, wires, nanotubes, and other nanoparticles [9].
2. Adhesives with Improved Conductivity
Epoxy is inferior in heat conductivity. However, research in electronic packaging is improving the heat conductivity of nanoelectronics epoxy adhesion. The conductivity range of TCAs is reported as 1–30 Wm−1 K−1 for sufficient heat transfer [23]. The thermal conductivity of epoxy has been increased using ceramic [24], metallic [25], and carbon-based [26] conductive fillers to increase the thermal conductivity of the epoxy, which are described below.
2.1. Ceramic-Based Fillers
Chip power density is a significant factor in the performance of electronic devices. TCAs minimize the thermal resistance between the electronic devices’ cooling medium and the outside environment. A ceramic substance is used to insulate to prevent electrical shorts [27] and enhance electronic equipment’s effectiveness [28]. Highly thermally conductive ceramic fillers have been added in epoxy adhesive systems, such as boron nitride (BN), aluminium nitride (AlN), alumina (Al2O3), and silicon carbide (SiC) particles. Figure 3 shows various types of thermally conductive ceramic used in TCAs.

Figure 3. Commonly used thermally conductive ceramics filler.
2.1.1. Boron Nitride
Boron nitride (BN) has the same number of boron (B) and nitrogen (N) atoms and is isoelectronic to carbon structures [29]. These particles have received considerable interest because of their excellent characteristics in all aspects. It is also referred to as “white graphene” because of its honeycomb composition. It has an insulating property with a bandgap of 5.2 eV and possesses considerable thermal conductivity (experimentally determined 360 Wm−1 K−1 and theoretically 2000 Wm−1 K−1 [30]. The findings are outstanding since the dielectric characteristics and the anti-oxidation resistance were found to be excellent. Electronic devices require better thermal management to match the rising demand for BN and epoxy mixed composites. The hexagonal form of BN (h-BN) [31] is comparable to graphite, the cubic form (c-BN) is like diamond [32], and the amorphous form (a-BN) is similar to amorphous carbon [33]. Several BN/epoxy composites have been created. However, a hybrid BN solution was unable to be found since the interfacial thermal resistance was too high [34]. BN nanoplates filler affects thermal conductivity of the composite with the increase in filler concentrations. It is found that the 2D-BN nanoplates filler at 14 wt% with silicone can reduce the mechanical strength, but the thermal conductivity is 30% better than the without filler sample. The reduction in the mechanical strength can be due to agglomeration of BN nanoparticles, whereas the increase in the thermal conductivity is due to the acceleration of phonons transmission by the BN nanoplates [35][36].
C. Xiaoa et al. [37] used hollow boron nitride microbeads (BNMB) at 65.6 vol% for improving thermal conductivity of epoxy resin. The result shows that the maximum thermal conductivity reached 17.61 W/m−1 K−1 in-plane direction and 5.08 W/m−1 K−1 for out-plane direction of BN.
2.1.2. Alumina
Alumina (Al2O3) is used in electronics packaging as a low-cost filler. It has high thermal conductivity (30 Wm−1 K−1) and electrical insulation properties. Because of these unique properties become an essential filer in the thermal management of electronic devices [38]. It has been used in light-emitting diode (LED) packaging to avoid voltage drop, short circuits, or noise reduction. Alumina nanoparticles can significantly improve the mechanical properties of epoxy adhesives and interfacial wettability with an aluminum substrate [39]. A mixture of Bisphenol-F epoxy resin with 80% of 30 μm and 20% of 5 μm spherical Alumina (S-Al2O3) particle can increase thermal conductivity up to 1.364 Wm−1 K−1 [40].
A study by Mai et al. [41] reported that epoxy adhesive with Alumina contained hybrid filler can improve the thermal conductivity of the composite materials. Another experimental and simulation work was performed using Al2O3 and BN in the epoxy polymer (EP) matrix. The thermal conductivity of the BN/Al2O3/EP composite is higher than that of the Al2O3/EP composite. The thermal conductivity increases from 2.77 Wm−1 K−1 to 3.35 Wm−1 K−1 for the composite without BN loading (shown in Figure 4). Graphene coated alumina was used as a thermal conductivity enhancer in epoxy composite and was found to be a potential filler [42].
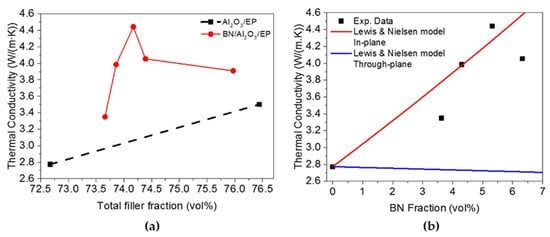
Figure 4. (a) Thermal conductivity vs. total filler fraction graph (b) Experimental and simulated data comparison graph [41].
2.1.3. Aluminum Nitride
Aluminum nitride (AlN) particles are a promising filler material in electronics packaging. The size of the AlN filler determines the characteristics of AlN-filled epoxy composites [43]. The thermal conductivity, based on AlN’s particle size, ranges from 200 to 320 Wm−1 K−1 at room temperature. The maximum amount of particle in epoxy composite does not ensure the highest thermal conductivity. Research result shows that 1 wt% nano-AlN sample has superior electrical insulation and thermal conductivity among the pure epoxy, 0.5 wt%, 1 wt% and 2 wt% of AlN/epoxy solution [44]. However, magnetic-aligned AlN/epoxy composite at low filler content can effectively generate thermal transport channels and enhance thermal conductivity from 0.915 Wm−1 K−1 to 1.754 Wm−1 K−1.
The thermal conductivity of adhesive is mainly determined by the filler heat transfer capacity, density of thermal network, as well interfacial thermal resistance. Thus, the formation of effective thermal flow 3-D percolating network through synergistic effect in matrix is a crucial criterion, dominating the thermal conductivity. Yuan et al. [45] worked on different sized (5 µm, 2 µm and, 50 nm) AlN- with graphite and graphene oxide(GO) as a hybrid filler to observe the improvement of thermal conductivity of composite. The results demonstrate that large particles of AlN with epoxy are more heat conductive than small particles. Similarly, GO can improve the thermal conductivity of epoxy resin more effectively than natural graphite. In the case of a single filler, adding 70 wt% 5 µm-AlN particles to the epoxy resulted in the maximum conductivity which is 10.8 times that of pure epoxy (shown in Figure 5).
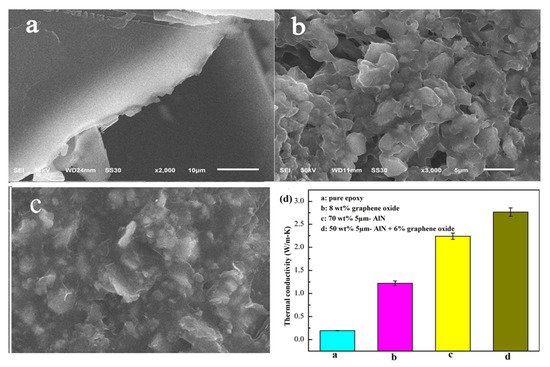
Figure 5. SEM images of epoxy adhesives containing (a) only epoxy resin, (b) epoxy with AlN particles, (c) epoxy with AlN and GO hybrid filler and (d) Thermal conductivity of different test samples [45].
The heat conductivity of epoxy adhesive containing GO is more effective than natural graphite. For single fillers, 10.8 times higher than pure epoxy is the most excellent conductivity AlN’s thermal expansion coefficient (CTE) is low. It is relatively low-cost, non-toxic, and can provide a stable crystalline structure [46].
2.1.4. Silicon Carbide
The researchers are interested in the Silicon Carbide Filler (SiC) as it has greater hardness and strength, good resistance to corrosion and oxidation. SiC has high intrinsic thermal conductivity (490 Wm−1 K−1), more than three times higher than silicon and 10 times higher than gallium arsenide and sapphire [47]. Nanowire form of SiC is a primarily familiar shape for dispersing with epoxy. Most of the recent works are focused on hybridization with SiC nanowire. According to Dianyu Shen et al. [48], the thermal conductivity of 3 wt% SiC nanowire with epoxy is 0.449 Wm−1 K−1, which is 1.06 times higher than plain epoxy. Another experimental output found thermal conductivity 0.43 Wm−1 K−1 at 3.91 vol% of SiC nanowire epoxy composite [49]. BN and SiC hybrid filler with vertical alignment was examined, and thermal conductivity enhancement was found [50][51]. Carbon fibre (CF) was also used with SiC to improve the heat transfer properties and saw satisfactory improvement [52]. A summary of research output from recently published works on ceramic fillers are given in Table 2.
Table 2. Summary of the recently published works on ceramic fillers.
| Filler | Conditions/Process | TC (Wm−1 K−1) | References |
|---|---|---|---|
| BN | Platelet-shaped Boron Nitride(BN) particles | 3.5 | [53] |
| BN | At 70 wt% functionalized and mix with epoxy resin | 2.8 | [29] |
| BN | Admicellar-treated BN particles. | 2.7 | [54] |
| BN | 30 wt% of BN particles modified by 3-aminopropyl triethoxysilane | 1.178 | [55] |
| BN | Hexagonal BN/epoxy composites at 44 vol% (densely packed and vertically aligned). | 9 | [56] |
| BN | Hexagonal, cubic, and conglomerated -BN. | 2.91, 3.95, and 10.1 | [57] |
| BN | Hexagonal boron nitride laminates | 20 | [58] |
| BN | Untreated and OTAB-treated BN/epoxy composites. | 1.9 and 3.4 | [59] |
| BN | 88 wt% of BN loading. | 32.5 | [60] |
| AlN | 58.4 vol% of large-sized Aluminum nitride (AlN) with small-sized Al2O3 | 2.842 and 3.4 | [61] |
| AlN | 29 wt% of MWCNTs/AlN | 1.04 | [62] |
| AlN | 20 vol% AlN particles (magnetically aligned) | 1.8 | [63] |
| AlN | 50 wt% of 5 μm-AlN particles and 6 wt% of GO | 2.77 | [45] |
| AlN | 67 vol% of AlN particles (large-sized silane-coated). | 14 | [64] |
| AlN | Cycloaliphatic epoxy/trimethacrylate system | 0.47 | [65] |
| AlN | At 47 vol% nano-whiskers AlN | 4.2 | [66] |
| Al2O3 | At 80 wt% of Alumina (Al2O3)/epoxy, filled with 5 wt% of graphene oxide (GO) and 5 wt% of Al(OH)3-coated GO | 3.5 and 3.1 | [67] |
| Al2O3 | Al2O3/GFRP (amino group grafted) | 1.07 | [68] |
| Al2O3 | At 60 vol% of micron-sized alumina | 4.3 | [69] |
| SiC | Magnetically aligned BN and Silicon Carbide (SiC) filler system | 5.77 | [50] |
| SiC | Nano-sized SiC particles with triethylenetetramine (TETA) functionalized MWCNTs, (at 30% vol%) | 2.00 | [70] |
| SiC | At 20 vol% of SiC particles (magnetically aligned Fe3O4 coated) | 1.681 | [71] |
2.2. Metallic Fillers
Metal nanoparticles (NPs) have recently gained popularity due to their unique properties such as low melting temperature and high diffusion coefficient [72]. Research has shown that epoxy conductive adhesives enhance their heat conductivity when integrated with metal fillers. Sphere, fibre, granules, or flakes may be metallic particles. The optimal shape can be such that the filler levels of the surrounding metallic parts are the lowest crucial, well interacted, and the best matrix-resin adhesion can be found. These criteria are suitable for metallic flakes because of their high aspect ratio [73]. However, polymers loaded with metal are required with the required reinforcement. The amount of filler may be readily changed to adjust thermal conductivity. The material for heat sinking requires a low coefficient of thermal expansion (CTE) since the semiconductor chips have low CTE content. Thus, it has good thermal conductivity, and low CTE needed for thermally conductive material. Cu is usually not utilized in TCAs. Because Cu is thermally good yet has a high CTE content, Figure 6 shows some metal fillers used to manufacture TCAs.

Figure 6. Commonly used thermally conductive metallic filler.
Copper nanoparticle paste is used for bonding chips and different metallic substrates at low temperatures. The result shows that good bonds are mainly achieved with Cu and Au surfaces [74]. It provided good thermal conductivity and shear strength (20 MPa) when sintered at 350 °C [75]. The thermal conductivity of the composite was three times higher than pure epoxy at 15 wt% of MWCNT, and 34 wt% of copper nanoparticles [76], TiO2 coated copper nanowires 0.2–1.12 Wm−1 K−1 [77] and 2.59 Wm−1 K−1 [78] were found for a length of up to ~40 μm and diameter of ~20 nm.
Silver (Ag) particles exhibit maximum thermal conductivity among functional metallic fillers. Silver nanoparticles have a vast application in catalysis, conduction, antibacterial and electrical devices [79]. Silver nanoparticles have different forms and sizes to obtain a high heat conductivity and decrease total product costs. Silver’s thermal conductivity is 450 Wm−1 K−1, although its conductivity improvement with an epoxy adhesive is lower. Silver/epoxy self-constructed nano-structured networks led to two times better thermal conductivity than plain epoxy [80]. Ag particle at 25.1 vol% of Ag- decorated BN nanosheets provides 3.06 Wm−1 K−1 [81] and silver-decorated MWCNT/epoxy adhesive 0.88 Wm−1 K−1 [82]. Ni decorated MWCNTs as a reinforcement provides 0.30 Wm−1 K−1 [83]. The best part of Ag nano particle is that it improves the thermal conductivity and the shear strength of the adhesive joint [84], which is essential for the electronic packaging reliability [85]. Additionally, some composites of silver-coated copper, silver-coated reduced, graphene oxide nanoparticles, and graphene nanosheets embedded in the epoxy resin provide good thermal and mechanical properties [86]. Another research observed the thermal conductivity of epoxy adhesive filled by eight different filler- ZnO powders, BN powders, Al2O3 powders, graphite flake, Al powders, Cu powders, diamond powders and Ag powder. The results indicate that each sample is capable of significantly increasing the thermal conductivity of the epoxy resin. The highest thermal conductivity (1.68 Wm−1 K−1) was obtained in the graphite-epoxy adhesive at 44.3 wt%. Meanwhile, the layer-shaped filler and sharp corner-shaped fillers are preferable for improving the thermal conductivity of epoxy resin [87]. The morphologies of the eight fillers samples after spray-gold treatment were observed by SEM images (shown in Figure 7).

Figure 7. SEM images of fillers sample as powder form. (a) ZnO, (b) BN, (c) Al2O3, (d) graphite flake, (e) Al (f) Cu (g) diamond and, (h) Ag powders [87].
References
- Jouyandeh, M.; Paran, S.M.R.; Jannesari, A.; Puglia, D.; Saeb, M.R. Protocol for nonisothermal cure analysis of thermoset composites. Prog. Org. Coat. 2019, 131, 333–339.
- Ahmadi, Z. Nanostructured epoxy adhesives: A review. Prog. Org. Coat. 2019, 135, 449–453.
- Jouyandeh, M.; Rahmati, N.; Movahedifar, E.; Hadavand, B.S.; Karami, Z.; Ghaffari, M.; Taheri, P.; Bakhshandeh, E.; Vahabi, H.; Ganjali, M.R.; et al. Properties of nano-Fe3O4 incorporated epoxy coatings from Cure Index perspective. Prog. Org. Coat. 2019, 133, 220–228.
- Kim, J.; Yim, B.; Kim, J.; Kim, J. The effects of functionalized graphene nanosheets on the thermal and mechanical properties of epoxy composites for anisotropic conductive adhesives (ACAs). Microelectron. Reliab. 2012, 52, 595–602.
- Zhao, J.; Du, F.; Zhou, X.; Cui, W.; Wang, X.; Zhu, H.; Xie, X. Thermal conductive and electrical properties of polyurethane/hyperbranched poly (urea-urethane)-grafted multi-walled carbon nanotube composites. Compos. Part B Eng. 2011, 42, 2111–2116.
- Adeniyi, A.G.; Ighalo, J.O.; Abdulkareem, S.A. Al, Fe and Cu waste metallic particles in conductive polystyrene composites. Int. J. Sustain. Eng. 2021, 14, 893–898.
- Huang, X.; Iizuka, T.; Jiang, P.; Ohki, Y.; Tanaka, T. Role of Interface on the Thermal Conductivity of Highly Filled Dielectric Epoxy/AlN Composites. J. Phys. Chem. C 2012, 116, 13629–13639.
- Huang, X.; Jiang, P.; Tanaka, T. A Review of Dielectric Polymer Composites with High Thermal. IEEE Electr. Insul. Mag. 2011, 27, 8–16.
- Felba, J. Thermally conductive adhesives in electronics. Adv. Adhes. Electron. Mater. Prop. Appl. 2011, 15–52.
- Domun, N.; Hadavinia, H.; Zhang, T.; Liaghat, G.; Vahid, S.; Spacie, C.; Paton, K.R.; Sainsbury, T. Improving the fracture toughness properties of epoxy using graphene nanoplatelets at low filler content. Nanocomposites 2017, 3, 85–96.
- Zhang, J.; Cheng, X.; Zhang, J.; Guo, X.; Huang, W. Effect of curing condition on bonding quality of scarf-repaired composite laminates. Chin. J. Aeronaut. 2020, 33, 2257–2267.
- Lee, J.-H.; Koh, C.Y.; Singer, J.P.; Jeon, S.-J.; Maldovan, M.; Stein, O.; Thomas, E.L. 25th Anniversary Article: Ordered Polymer Structures for the Engineering of Photons and Phonons. Adv. Mater. 2014, 26, 532–569.
- Liu, Y.; Wu, K.; Lu, M.; Jiao, E.; Zhang, H.; Shi, J.; Lu, M. Highly thermal conductivity and flame retardant flexible graphene/MXene paper based on an optimized interface and nacre laminated structure. Compos. Part A Appl. Sci. Manuf. 2021, 141, 106227.
- Messina, E.; Leone, N.; Foti, A.; Di Marco, G.; Riccucci, C.; Di Carlo, G.; Di Maggio, F.; Cassata, A.; Gargano, L.; D’Andrea, C.; et al. Double-wall nanotubes and graphene nanoplatelets for hybrid conductive adhesives with enhanced thermal and electrical conductivity. ACS Appl. Mater. Interfaces 2016, 8, 23244–23259.
- Singh, A.K.; Panda, B.P.; Mohanty, S.; Nayak, S.K.; Gupta, M.K. Synergistic effect of hybrid graphene and boron nitride on the cure kinetics and thermal conductivity of epoxy adhesives. Polym. Adv. Technol. 2017, 28, 1851–1864.
- Chung, D.D.L. Review: Electrical application of carbon materials. J. Mater. Sci. 2004, 39, 2645–2661.
- Luo, R.; Liu, T.; Li, J.; Zhang, H.; Chen, Z.; Tian, G. Thermophysical properties of carbon/carbon composites and physical mechanism of thermal expansion and thermal conductivity. Carbon 2004, 42, 2887–2895.
- Chung, D.D.L. Applied Materials Science; CRC Press: Boca Raton, FL, USA, 2001; ISBN 9781420040975.
- Román-Manso, B.; Chevillotte, Y.; Osendi, M.I.; Belmonte, M.; Miranzo, P. Thermal conductivity of silicon carbide composites with highly oriented graphene nanoplatelets. J. Eur. Ceram. Soc. 2016, 36, 3987–3993.
- Maldovan, M. Sound and heat revolutions in phononics. Nature 2013, 503, 209–217.
- Zhang, Y.; Heo, Y.J.; Son, Y.R.; In, I.; An, K.H.; Kim, B.J.; Park, S.J. Recent advanced thermal interfacial materials: A review of conducting mechanisms and parameters of carbon materials. Carbon 2019, 142, 445–460.
- Chen, L.; Shen, Y.; Yi, H.; Liu, Z.; Song, Q. Mathematical modeling of heat transfer in GO-doped reinforce polymer for anti-/deicing of wind turbines. Int. Commun. Heat Mass Transf. 2021, 123, 105235.
- Zhang, H.; Zhang, X.; Fang, Z.; Huang, Y.; Xu, H.; Liu, Y.; Wu, D.; Zhuang, J.; Sun, J. Recent advances in preparation, mechanisms, and applications of thermally conductive polymer composites: A review. J. Compos. Sci. 2020, 4, 180.
- Wu, J.; Zhang, Y.; Gong, Y.; Wang, K.; Chen, Y.; Song, X.; Lin, J.; Shen, B.; He, S.; Bian, X. Analysis of the Electrical and Thermal Properties for Magnetic Fe3O4-Coated SiC-Filled Epoxy Composites. Polymers 2021, 13, 3028.
- Mora, M.; Amaveda, H.; Porta-Velilla, L.; de la Fuente, G.F.; Martínez, E.; Angurel, L.A. Improved Copper–Epoxy Adhesion by Laser Micro- and Nano-Structuring of Copper Surface for Thermal Applications. Polymers 2021, 13, 1721.
- Naresh, K.; Khan, K.A.; Umer, R. Experimental Characterization and Modeling Multifunctional Properties of Epoxy/Graphene Oxide Nanocomposites. Polymers 2021, 13, 2831.
- Turner, R.C.; Fuierer, P.A.; Newnham, R.E.; Shrout, T.R. Materials for high temperature acoustic and vibration sensors: A review. Appl. Acoust. 1994, 41, 299–324.
- Zweben, C. Advances in composite materials for thermal management in electronic packaging. Jom 1998, 50, 47–51.
- Guerra, V.; Wan, C.; McNally, T. Thermal conductivity of 2D nano-structured boron nitride (BN) and its composites with polymers. Prog. Mater. Sci. 2019, 100, 170–186.
- Jo, I.; Pettes, M.T.; Kim, J.; Watanabe, K.; Taniguchi, T.; Yao, Z.; Shi, L. Thermal conductivity and phonon transport in suspended few-layer hexagonal boron nitride. Nano Lett. 2013, 13, 550–554.
- Geng, D.; Dong, J.; Kee Ang, L.; Ding, F.; Yang, H.Y. In situ epitaxial engineering of graphene and h-BN lateral heterostructure with a tunable morphology comprising h-BN domains. NPG Asia Mater. 2019, 11, 56.
- Shen, W.; Shen, S.; Liu, S.; Li, H.; Gan, Z.; Zhang, Q. Monolayer Cubic Boron Nitride Terminated Diamond (111) Surfaces for Quantum Sensing and Electron Emission Applications. ACS Appl. Mater. Interfaces 2020, 12, 33336–33345.
- Liu, Y.; Pan, Y.; Yin, D.; Song, S.; Lin, L.; Qi, X.; Zhang, M.; Yao, J. Large-scale generation and characterization of amorphous boron nitride and its mechanical properties in atomistic simulations. J. Non-Cryst. Solids 2021, 558, 120664.
- Zhang, Y.; Gao, W.; Li, Y.; Zhao, D.; Yin, H. Hybrid fillers of hexagonal and cubic boron nitride in epoxy composites for thermal management applications. RSC Adv. 2019, 9, 7388–7399.
- Khan, J.; Momin, S.A.; Mariatti, M. A review on advanced carbon-based thermal interface materials for electronic devices. Carbon 2020, 168, 65–112.
- Liu, P.; Li, L.; Wang, L.; Huang, T.; Yao, Y.; Xu, W. Effects of 2D boron nitride (BN) nanoplates filler on the thermal, electrical, mechanical and dielectric properties of high temperature vulcanized silicone rubber for composite insulators. J. Alloys Compd. 2019, 774, 396–404.
- Xiao, C.; Tang, Y.; Chen, L.; Zhang, X.; Zheng, K.; Tian, X. Preparation of highly thermally conductive epoxy resin composites via hollow boron nitride microbeads with segregated structure. Compos. Part A Appl. Sci. Manuf. 2019, 121, 330–340.
- Sim, L.C.; Lee, C.K.; Ramanan, S.R.; Ismail, H.; Seetharamu, K.N. Cure characteristics, mechanical and thermal properties of Al2O3 and ZnO reinforced silicone rubber. Polym.-Plast. Technol. Eng. 2006, 45, 301–307.
- Dorigato, A.; Pegoretti, A. The role of alumina nanoparticles in epoxy adhesives. J. Nanop. Res. 2011, 13, 2429–2441.
- Chen, C.; Xue, Y.; Li, X.; Wen, Y.; Liu, J.; Xue, Z.; Shi, D.; Zhou, X.; Xie, X.; Mai, Y.W. High-performance epoxy/binary spherical alumina composite as underfill material for electronic packaging. Compos. Part A Appl. Sci. Manuf. 2019, 118, 67–74.
- Mai, V.D.; Lee, D., II; Park, J.H.; Lee, D.S. Rheological properties and thermal conductivity of epoxy resins filled with a mixture of alumina and boron nitride. Polymers 2019, 11, 597.
- Kim, K.T.; Dao, T.D.; Jeong, H.M.; Anjanapura, R.V.; Aminabhavi, T.M. Graphene coated with alumina and its utilization as a thermal conductivity enhancer for alumina sphere/thermoplastic polyurethane composite. Mater. Chem. Phys. 2015, 153, 291–300.
- Bae, J.W.; Kim, W.; Cho, S.H.; Lee, S.H. Properties of AlN-filled epoxy molding compounds by the effects of filler size distribution. J. Mater. Sci. 2000, 35, 5907–5913.
- Dai, C.; Chen, X.; Jiang, T.; Paramane, A.; Tanaka, Y. Improvement of electrical and material properties of epoxy resin/ aluminum nitride nanocomposites for packaging materials. Polym. Test. 2020, 86, 106502.
- Yuan, W.; Xiao, Q.; Li, L.; Xu, T. Thermal conductivity of epoxy adhesive enhanced by hybrid graphene oxide/AlN particles. Appl. Therm. Eng. 2016, 106, 1067–1074.
- Choi, S.; Im, H.; Kim, J. The thermal conductivity of embedded nano-aluminum nitride-doped multi-walled carbon nanotubes in epoxy composites containing micro-aluminum nitride particles. Nanotechnology 2012, 23, 065303.
- Alim, M.A.; Abdullah, M.Z.; Aziz, M.S.A.; Kamarudin, R.; Irawan, A.P.; Siahaan, E. Experimental study on luminous intensity of white LEDs of different configurations. IOP Conf. Ser. Mater. Sci. Eng. 2020, 1007, 012145.
- Shen, D.; Zhan, Z.; Liu, Z.; Cao, Y.; Zhou, L.; Liu, Y.; Dai, W.; Nishimura, K.; Li, C.; Lin, C.-T.; et al. Enhanced thermal conductivity of epoxy composites filled with silicon carbide nanowires. Sci. Rep. 2017, 7, 2606.
- Xiao, C.; Chen, L.; Tang, Y.; Zhang, X.; Zheng, K.; Tian, X. Enhanced thermal conductivity of silicon carbide nanowires (SiCw)/epoxy resin composite with segregated structure. Compos. Part A Appl. Sci. Manuf. 2019, 116, 98–105.
- Kim, K.; Ju, H.; Kim, J. Vertical particle alignment of boron nitride and silicon carbide binary filler system for thermal conductivity enhancement. Compos. Sci. Technol. 2016, 123, 99–105.
- Xiao, C.; Guo, Y.; Tang, Y.; Ding, J.; Zhang, X.; Zheng, K.; Tian, X. Epoxy composite with significantly improved thermal conductivity by constructing a vertically aligned three-dimensional network of silicon carbide nanowires/boron nitride nanosheets. Compos. Part B Eng. 2020, 187, 107855.
- Mun, S.Y.; Lim, H.M.; Lee, D.J. Thermal conductivity of a silicon carbide/pitch-based carbon fiber-epoxy composite. Thermochim. Acta 2015, 619, 16–19.
- Li, K.S.; Wang, Q. Thermal conductivity of filled polymer composites. Gaofenzi Cailiao Kexue Yu Gongcheng/Polym. Mater. Sci. Eng. 2002, 18, 10–15.
- Xiao, M.; Du, B.X. Review of high thermal conductivity polymer dielectrics for electrical insulation. High Volt. 2016, 1, 34–42.
- Hou, J.; Li, G.; Yang, N.; Qin, L.; Grami, M.E.; Zhang, Q.; Wang, N.; Qu, X. Preparation and characterization of surface modified boron nitride epoxy composites with enhanced thermal conductivity. RSC Adv. 2014, 4, 44282–44290.
- Yu, C.; Zhang, J.; Li, Z.; Tian, W.; Wang, L.; Luo, J.; Li, Q.; Fan, X.; Yao, Y. Enhanced through-plane thermal conductivity of boron nitride/epoxy composites. Compos. Part A Appl. Sci. Manuf. 2017, 98, 25–31.
- Wang, Z.; Iizuka, T.; Kozako, M.; Ohki, Y.; Tanaka, T. Development of epoxy/BN composites with high thermal conductivity and sufficient dielectric breakdown strength part I—Sample preparations and thermal conductivity. IEEE Trans. Dielectr. Electr. Insul. 2011, 18, 1963–1972.
- Zheng, J.C.; Zhang, L.; Kretinin, A.V.; Morozov, S.V.; Wang, Y.B.; Wang, T.; Li, X.; Ren, F.; Zhang, J.; Lu, C.Y.; et al. High thermal conductivity of hexagonal boron nitride laminates. 2D Mater. 2016, 3, 011004.
- Wattanakul, K.; Manuspiya, H.; Yanumet, N. The adsorption of cationic surfactants on BN surface: Its effects on the thermal conductivity and mechanical properties of BN-epoxy composite. Colloids Surf. A Physicochem. Eng. Asp. 2010, 369, 203–210.
- Ishida, H.; Rimdusit, S. Very high thermal conductivity obtained by boron nitride-filled polybenzoxazine. Thermochim. Acta 1998, 320, 177–186.
- Choi, S.; Kim, J. Thermal conductivity of epoxy composites with a binary-particle system of aluminum oxide and aluminum nitride fillers. Compos. Part B Eng. 2013, 51, 140–147.
- Ma, A.-J.; Chen, W.; Hou, Y. Enhanced Thermal Conductivity of Epoxy Composites with MWCNTs/AlN Hybrid Filler. Polym.-Plast. Technol. Eng. 2012, 51, 1578–1582.
- Kim, K.; Kim, J. Magnetic aligned AlN/epoxy composite for thermal conductivity enhancement at low filler content. Compos. Part B Eng. 2016, 93, 67–74.
- Hsieh, C.Y.; Chung, S.L. High thermal conductivity epoxy molding compound filled with a combustion synthesized AIN powder. J. Appl. Polym. Sci. 2006, 102, 4734–4740.
- Yu, J.H.; Duan, J.K.; Peng, W.Y.; Wang, L.C.; Peng, P.; Jiang, P.K. Influence of nano-ALN particles on thermal conductivity, thermal stability and cure behavior of cycloaliphatic epoxy/trimethacrylate system. Express Polym. Lett. 2011, 5, 132–141.
- Shi, Z.; Radwan, M.; Kirihara, S.; Miyamoto, Y.; Jin, Z. Enhanced thermal conductivity of polymer composites filled with three-dimensional brushlike AlN nanowhiskers. Appl. Phys. Lett. 2009, 95, 176–179.
- Heo, Y.; Im, H.; Kim, J.; Kim, J. The influence of Al(OH)3-coated graphene oxide on improved thermal conductivity and maintained electrical resistivity of Al2O3/epoxy composites. J. Nanop. Res. 2012, 14, 1–10.
- Wan, Y.J.; Li, G.; Yao, Y.M.; Zeng, X.L.; Zhu, P.L.; Sun, R. Recent advances in polymer-based electronic packaging materials. Compos. Commun. 2020, 19, 154–167.
- Kozako, M.; Okazaki, Y.; Hikita, M.; Tanaka, T. Preparation and evaluation of epoxy composite insulating materials toward high thermal conductivity. In Proceedings of the 2010 10th IEEE International Conference on Solid Dielectrics, Potsdam, Germany, 4–9 July 2010; pp. 1–4.
- Yang, K.; Gu, M. Enhanced thermal conductivity of epoxy nanocomposites filled with hybrid filler system of triethylenetetramine-functionalized multi-walled carbon nanotube/silane-modified nano-sized silicon carbide. Compos. Part A Appl. Sci. Manuf. 2010, 41, 215–221.
- Kim, K.; Kim, M.; Kim, J.; Kim, J. Magnetic filler alignment of paramagnetic Fe3O4 coated SiC/epoxy composite for thermal conductivity improvement. Ceram. Int. 2015, 41, 12280–12287.
- Yan, J. A Review of Sintering-Bonding Technology Using Ag Nanoparticles for Electronic Packaging. Nanomaterials 2021, 11, 927.
- Janzen, J. On the critical conductive filler loading in antistatic composites. J. Appl. Phys. 1975, 46, 966–969.
- Kim, J.; Lee, B.; Koo, J.M.; Gan, C.L. Copper nanoparticle paste on different metallic substrates for low temperature bonded interconnection. In Proceedings of the 2017 IEEE 19th Electronics Packaging Technology Conference (EPTC), Singapore, 6–9 December 2017; pp. 1–4.
- Li, G.; Fana, J.; Liao, S.; Zhua, P.; Zhang, B.; Zhao, T.; Sun, R.; Wong, C.P. Low temperature sintering of dendritic cu based pastes for power semiconductor device interconnection. In Proceedings of the 2019 IEEE 69th Electronic Components and Technology Conference (ECTC), Las Vegas, NV, USA, 28–31 May 2019; pp. 81–86.
- Zhang, P.; Li, Q.; Xuan, Y. Thermal contact resistance of epoxy composites incorporated with nano-copper particles and the multi-walled carbon nanotubes. Compos. Part A Appl. Sci. Manuf. 2014, 57, 1196.
- Ahn, K.; Kim, K.; Kim, J. Thermal conductivity and electric properties of epoxy composites filled with TiO2-coated copper nanowire. Polymer 2015, 76, 313–320.
- Chen, W.; Wang, Z.; Zhi, C.; Zhang, W. High thermal conductivity and temperature probing of copper nanowire/upconversion nanoparticles/epoxy composite. Compos. Sci. Technol. 2016, 130, 63–69.
- Fortunati, E.; D’Angelo, F.; Martino, S.; Orlacchio, A.; Kenny, J.M.; Armentano, I. Carbon nanotubes and silver nanoparticles for multifunctional conductive biopolymer composites. Carbon 2011, 49, 2370–2379.
- Choi, J.R.; Rhee, K.Y.; Park, S.J. Influence of electrolessly silver-plated multi-walled carbon nanotubes on thermal conductivity of epoxy matrix nanocomposites. Compos. Part B Eng. 2015, 80, 379–384.
- Wang, F.; Zeng, X.; Yao, Y.; Sun, R.; Xu, J.; Wong, C.P. Silver Nanoparticle-Deposited Boron Nitride Nanosheets as Fillers for Polymeric Composites with High Thermal Conductivity. Sci. Rep. 2016, 6, 19394.
- Singh, A.K.; Panda, B.P.; Mohanty, S.; Nayak, S.K.; Gupta, M.K. Study on metal decorated oxidized multiwalled carbon nanotube (MWCNT)—Epoxy adhesive for thermal conductivity applications. J. Mater. Sci. Mater. Electron. 2017, 28, 8908–8920.
- Choi, J.; Sil, Y.; Park, S. Journal of Industrial and Engineering Chemistry A study on thermal conductivity of electroless Ni—B plated multi-walled carbon nanotubes-reinforced composites. J. Ind. Eng. Chem. 2014, 20, 3421–3424.
- Ekrem, M.; Ataberk, N.; Avcı, A.; Akdemir, A. Improving electrical and mechanical properties of a conductive nano adhesive. J. Adhes. Sci. Technol. 2017, 31, 699–712.
- Jiang, H.; Moon, K.S.; Lu, J.; Wong, C.P. Conductivity enhancement of nano silver-filled conductive adhesives by particle surface functionalization. J. Electron. Mater. 2005, 34, 1432–1439.
- Peighambardoust, S.J.; Rikhtegar, H.; Mohammadzadeh Pakdel, P.; Mirmohseni, A. Electrically conductive epoxy-based nanocomposite adhesives loaded with silver-coated copper and silver-coated reduced graphene oxide nanoparticles. Polym. Adv. Technol. 2019, 30, 1996–2004.
- Fu, Y.X.; He, Z.X.; Mo, D.C.; Lu, S.S. Thermal conductivity enhancement with different fillers for epoxy resin adhesives. Appl. Therm. Eng. 2014, 66, 493–498.





