| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Jihong Liu | + 3319 word(s) | 3319 | 2020-07-20 05:16:32 | | | |
| 2 | Jihong Liu | Meta information modification | 3319 | 2020-07-26 14:51:11 | | | | |
| 3 | Camila Xu | Meta information modification | 3319 | 2020-07-30 09:47:08 | | | | |
| 4 | Camila Xu | -7 word(s) | 3312 | 2020-07-30 09:48:05 | | | | |
| 5 | Camila Xu | -162 word(s) | 3150 | 2020-10-27 08:46:35 | | |
Video Upload Options
Sensors are the core of wearable monitoring equipment. In wearable devices (such as wrist strap or chest strap), the sensor will inevitably contact with the skin.
1. Introduction
In recent years, many new health-tracking devices and smartwatches have appeared in several global electronic product consumer exhibitions. The concept of wearable devices continues to be hot, and the market demand continues to grow. According to the latest data from the International Data Corporation (IDC)[1] , global wearable device shipments grew 82.3% in the fourth quarter of 2019 to 118.9 million units. Throughout the year, global wearable device shipments reached 335.5 million in 2019, an increase of 89% compared with 2018. At present, the product forms of wearable devices mainly include smart glasses, smartwatches, smart bracelets, etc. By connecting to the Internet and combining with various kinds of software, we can provide consumers with some relevant vital signs information and keep users in touch in a way that other devices (even smartphones) cannot, especially for improving the current medical disputes between doctors and patients, and effectively configuring the short medical treatment resources play an important role. However, in practical clinical applications, wearable devices should not only ensure the accuracy of signal acquisition but also ensure comfort in the process of wearing as much as possible. By reducing the volume of implantable devices, improving their biocompatibility and endurance, combined with minimally invasive surgery, the invasive injury to the human body should be reduced as much as possible [2], which limits its development. In addition, the comfort experience of consumers is also affected by the stability of battery power supply, service life, sensor placement, power consumption, accuracy, etc.
Sensors are the core of wearable monitoring equipment. In wearable devices (such as wrist strap or chest strap), the sensor will inevitably contact with the skin. This skin contact can cause many problems, such as discomfort due to the presence of sensory nerves and steam on sweaty skin. If a rigid sensor is used, not only will the wearing comfort be affected, but also the signal error of some motion artifacts will be caused. This is because the rigid material does not have enough flexibility and adaptability [3]. Nowadays, wearable devices have low precision, high power consumption and high stiffness, which are mainly due to the characteristics of sensing elements and the need for extensive signal processing to remove noise and extract required features. Therefore, the development of new sensors is of great significance to meet the above challenges. The latest development of nano materials, flexible electronics, and intelligent textiles will bring new development opportunities for wearable devices and practical clinical applications. The new materials used in these sensors are lightweight and natural fitting, thus providing better signal quality.
2. Bioelectric Signal Monitoring
With the acceleration of people’s social life rhythm, it becomes more and more important to continuously monitor the physiological parameters of the human body. The traditional wet electrode clinical system (i.e., Ag/AgCl electrode) can obtain high-quality signals, but, in the long-term monitoring or wearable applications, the traditional wet electrode has obvious limitations. Because of the need for the participation of conductive paste, the signal quality will be reduced and the wearing comfort will be affected when the conductive paste is dry for a long time. In addition, they may cause skin irritation and allergic contact dermatitis [4]. In order to solve this limitation, there are two solutions, one is a contact electrode without conductive paste, the other is a capacitive non-contact electrode. The flexible electrode is a more reasonable and comfortable solution than the rigid ones. Flexible electrodes are generally divided into two types. One is the self-supporting electrode with excellent electrical conductivity and electrochemical activity at the same time, such as activated carbon, graphene, carbon nanotube flexible electrode, etc., which integrates fluid and active materials. The other is a flexible electrode made by loading electrochemical active material on a flexible fluid collector with high conductivity. Since charge adsorption/desorption only occurs on the electrode surface and the process is completely reversible, the electrode material will not be damaged in the electrochemical reaction process, so the supercapacitor prepared by carbon material has the advantages of high-power density, good performance, and long cycle life. The role of flexible packaging materials is to provide external protection and mechanical support. Thus, on account of the excellent mechanical properties of flexible plastic sealing materials, polyethylene terephthalate (PET) and polydimethylsiloxane (PDMS) are generally chosen. The role of the diaphragm is to prevent the electrode short circuit caused by the contact of the anode and cathode. To make the electrochemical active materials on the positive and negative electrodes fully participate in the reaction, the materials with good wettability to the electrolyte are generally selected. To make the electrochemical active materials on the anode and cathode fully participate in the reaction, the electrolyte is generally selected to have good wettability[5] . Therefore, we have reviewed the research progress of flexible bioelectrical signal sensors in recent years.
In order to improve the accuracy of signal monitoring and diagnosis and user comfort, people have made a long-term exploration and improvement on materials, power consumption, life, and wearing parts for wearable devices. Bioelectric monitoring is susceptible to noise interference, so reducing its motion artifacts, prolonging the effective monitoring time, improving biocompatibility and improving the quality of signal acquisition are the main research focuses. At present, wearable devices are mainly divided into two types: (a) non-implantable recording device, whose sensing electrode is attached to the patient’s body surface (usually the chest or the arm), which is suitable for short-term recording; (b) implantable circulatory recorder. Although subcutaneous implantation can monitor the heart rhythm for a long time, the cost of the device itself and hospitalization is considerable, including surgical risks. Table 1 lists some typical examples of wearable vital signs monitoring that we will discuss later, as well as relevant features and limitations for comparative analysis.
Table 1. Comparation for flexible sensors for vital sign monitoring.
|
Vital Signs |
Materials |
Key Features |
Limitations |
Reference |
|
|
Contact sensor |
ECG/EMG |
Ag NW/PDMS |
Anti-microbial, Eliminated motion artifacts |
Material oxidation |
|
|
ECG |
Polymer/Ag NWs electrode |
Highly stretchable, low sensing limit, and good durability |
Requires tight contact |
||
|
ECG |
Graphene, textile |
Easy to make |
High noise |
||
|
ECG |
PEDOT: PSS, LIG |
Prolonged stability, High waveform quality |
Prone to motion artifact |
||
|
ECG/EMG |
PDMS |
Scalable, less skin irritation |
Prone to motion artifact |
[3] |
|
|
ECG |
Ag/AgCl |
Wi-Fi wireless transmission |
High power consumption, short lifespan |
||
|
ECG |
Ag/AgCl |
Low power consumption, dry 3D printed electrodes |
Short battery lifespan |
||
|
EMG |
Ag, nylon plastic |
Convenient, real time processed |
Data accuracy |
||
|
Non-contact sensor |
ECG/EMG/EEG |
PS25255 EPIC |
Portability, long-term monitoring |
Poor tight contact, prone to motion |
|
|
ECG/EMG/EEG |
Flexible printed circuits (FPC) |
Flexible, no obvious power frequency noise |
Baseline drift exists |
[14,15] |
|
|
ECG |
ASOPA4002 |
Completely flexible and ultra-thin |
High power consumption |
||
|
ECG |
Silicone-based sensors |
Comfortable, noise immunization |
Short monitoring period |
||
|
ECG |
Silicone dry electrode |
Reliable, low power consumption, low cost, |
Irregular waveforms, low CR |
||
|
ECG |
PDMS-Graphene |
Textile based, high quality |
Limited stability |
||
|
ECG |
Graphene |
Soft, low cost, scalable |
Contact impedance exists |
||
|
Implant-able sensor |
Peripheral neural signals |
TiO2, silicone |
Good biocompatibility |
Unknown mechanical properties |
|
|
ECG |
PI, AU/Ti |
Flexible, robust performance |
High impedance |
||
|
ECG |
Ag/AgCl |
Low noise, good biocompatibility |
High power consumption |
3. Respiratory Rate Monitoring
With the improvement of human living standards, more and more people are very concerned about their health status, and vital signs can reflect people’s health status, so it is particularly necessary to obtain vital signs. As an important aspect of vital signs, breathing frequency detection and analysis can help people understand their own health to a certain extent. This portion mainly summarizes the research progress of the measurement sensors from the perspective of respiratory sensing, and puts forward the development trends of these aspects in the future.
Currently, available techniques for detecting breathing frequency mainly include the chest impedance method, three-dimensional acceleration-derived breathing frequency, plethysmography using piezoelectric sensors and breathing induction plethysmography [24,25,26]. At the beginning, chest impedance detection was mostly used, but this method uses electrodes on the skin to obtain signals, and these patches attached to the skin for a long time may cause irritation, so piezoelectric sensors or acceleration sensors are also used to detect the breathing rate. An accelerometer placed on the torso can measure the change in tilt angle during breathing, and then measure the helix rate. Hong et al. [27] adopted a new method based on a chest biaxial accelerometer to derive the breathing rate in static activities. Bates et al. [28] proposed a three-dimensional acceleration or obtaining the respiratory rate from a wireless sensor device, by tracking the rotation axis to obtain a regular rate of respiratory motion. Of course, some scholars have also studied the feasibility of these sensors in respiratory monitoring systems, and found that the respiratory signals collected by piezoelectric sensors or three-dimensional acceleration sensors contain a lot of motion artifacts, which will affect the accuracy of respiratory frequency measurement [26,29]. In order to make the detection method more effective, some works [30,31] focused on the wearable respiratory monitoring device based on respiratory inductance plethysmography (RIP) and used it for respiratory biofeedback training. However, since the device is difficult to embed in a wearable device, it is difficult and complicated to use in practice. In view of this issue, Chen et al. [26] proposed a wearable respiratory monitoring system based on RIP, which uses a new type of PDMS-graphene composite tensile sensor (Figure 1a,b). Compared with traditional RIP-based sensors, the sensors they proposed are more convenient and suitable for wearable products and can be directly combined with clothing as elastic bands. Moreover, the sensor showed high sensitivity during stretching, has good circulation stability in 3600 consecutive cycles and can obtain respiratory signals without interference with high accuracy and satisfactory user experience. Therefore, it has great potential for home monitoring.
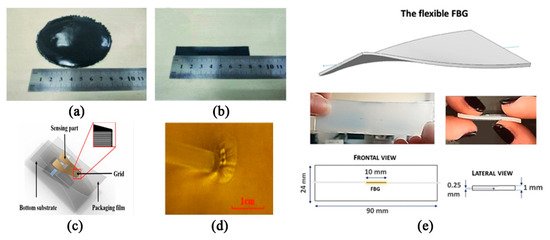
Figure 1. Respiratory monitoring sensors: (a) PDMS-graphene compound prototype after the manufacture process and (b) a tensile sensor after cutting [26] © 2019 IEEE; (c) schematic diagram of a flexible strain sensor with the inset showing the grid part and (d) a sensor attached to the skin [33] © 2016 IEEE; (e) the flexible fiber-based Bragg grating (FBG) proposed in this work [38] © 2019 IEEE.
Further works are done to optimizing the flexibility and portability for wearable products. Fan et al. [32] proposed a friction electric full textile sensor array with high pressure sensitivity and comfort. The fabricated triboelectric all-textile sensor array (TATSA) can be directly integrated into different parts of the fabric. These parts correspond to the pulse wave at the neck, wrist, fingertip, and ankle, as well as the abdomen and chest breathing waves. This study provides a comfortable, effective, and user-friendly method for measuring human pulse and respiration. In order to meet the biocompatibility of the monitoring device as much as possible, Chen et al. [33] proposed to attach biocompatible and ultra-flexible inorganic strain sensors to the skin to monitor vital signs (mainly pulse and respiratory rate) for a long time (Figure 1c,d). Considering that there are few measurements of breathing rate during exercise, Yamamoto et al. [34] developed a non-invasive strain sensor providing real-time measurements of respiration during exercise. The precision accuracy of the experimental results is high, which also indicates that it may be a useful clinical practice. In addition, fiber optic sensors can be used in any environment because they are not affected by electromagnetic interference, and have received the attention of some scholars in recent years. Kawamura, M. et al. [35] proposed a new vital sign sensing method in 2011, which uses fiber Bragg grating sensors to simultaneously monitor pulse rate and respiration rate. Dziuda, Ł. et al. [36] proposed to use optical strain sensors based on fiber Bragg gratings to monitor the patient’s breathing and cardiac activity during magnetic resonance imaging (MRI) investigations in 2007. Fajkus, M. et al. [37] describes an original simple, low-cost MR fully compatible and safe fiber-optic breathing sensor (FOBS), which can be used for respiratory triggering and monitoring the respiratory frequency in the MR environment. In addition to this, there is a growing demand for strain sensors that can be embedded in wearable devices for a variety of potential applications. Breathing and heart rate monitoring focused on chest wall displacement have promoted the development of strain sensors based on fiber-based Bragg grating (FBG) combined with polymer [38]. The geometric characteristics of flexible fiber gratings and some images related to the sensing elements are shown below (Figure 1e). Of course, the detection accuracy of this optical fiber-based strain sensor needs further experimental evaluation, but it is not difficult to see that the optical fiber-based sensor detection device is a trend for future development.
4. Temperature Monitoring
Body temperature can largely reflect people’s physical condition. The body temperature of normal people is relatively constant, which is maintained between 36–37 °C and has nothing to do with the surrounding environment. Normal body temperature is a necessary condition to ensure normal metabolism and life activities. Therefore, in order to monitor body temperature in real time, a variety of flexible temperature sensors have been developed. Generally speaking, there are two ways to measure body temperature, thermistor, and thermoelectric effect.
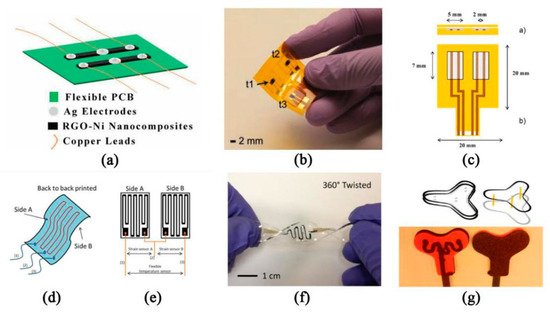
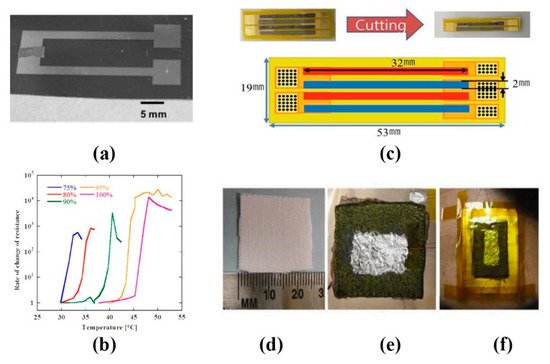
The thermistor temperature sensor can be divided into positive temperature coefficient thermistor (PTC) and negative temperature coefficient thermistor (NTC) [39]. They show different resistance values at different temperatures. The higher the temperature is, the higher the resistance of PTC is, and the lower the resistance of NTC is. The temperature coefficient of resistance (TCR) is an important index of sensitivity of resistance temperature sensor, which indicates the relative change of resistance value when the temperature changes 1 °C. Kedambaimoole, V. et al. [40] printed Graphene-Nickel (Ni) nanocomposite film on flexible PCB by screen printing, which is a new method to fabricate temperature sensor arrays. The calculated sensitivity response of the sensor is about 2.455 Ω/K, with TCR about −2.635 × 10−3 Ω/K. The sensor manufacturing process is simple, can be mass-produced on PCB, and easy to integrate with electronic devices (Figure 2a). Khan et al. [41] printed NiO/PSBR composite on top of gold electrodes by stencil printing and obtained a high sensitivity thermistor with a temperature coefficient of about −5.84% K−1 and a material constant of about 4330 K (Figure 2b). Giuliani et al. [42] adopted a new polystyrene-based ionomer/Multiwalled carbon nanotubes (MWCNT) nanocomposite, which can be made into a small and highly repeatable temperature sensor in the range of 20–40 °C at a low cost (Figure 2c). Wang et al. [43] impregnated the graphene dispersed by sodium alginate solution into the sodium alginate matrix in the dip-coating method, and prepared a flexible temperature sensor (FTS) suitable for human skin, which has good repeatability and stability. In addition, it can eliminate the interference of strain and humidity and distinguish the subtle difference in temperature. In order to further make the flexible temperature sensor easier to stretch, so that the resistance will not produce large error due to the movement of the body, Ali et al. [44] proposed a novel differential temperature sensor (DTS) made of the ink-jet material printer (Figure 2d,e). Compared with the simple resistance temperature detector (RTD), the sensor can measure the temperature of a flexible or curved surface with minimum error. The sensor is ideal for measuring the temperature of a flexible substrate. Yan et al. [45] fabricated a stretched graphene thermistor with high tensile properties by photolithography filtration (Figure 2f). They embed the detection channel and electrode into the elastomer matrix completely, then obtain the equipment that can maintain the function even in the state of high tension. This provides a broad prospect for the application of a wearable temperature sensor.
Figure 2. Thermistor temperature sensors: (a) schematic diagram of graphene–nickel nanocomposite temperature sensor on a flexible PCB [40] © 2017 IEEE; (b) picture of a screen-printed thermistor composed of nickel oxide (NiO) nanoparticles and PSBR adhesive on a Kapton-PI substrate. The four thermistors were labeled t1, t2, t3 and t4, respectively [41]; (c) profile and top view of the sensor [42]; (d) DTS layout diagram and (e) DTS connection diagram [44] © 2019 IEEE; (f) actual image of the stretchable graphene thermistor [45]; (g) the design structure of the Y-shaped sensor, showing the location of the three pairs of thermistors and the usage of the three heat flows [47] © 2018 IEEE.
Most of these studies are based on temperature measurements of the surface of the human skin, but sometimes we need to know the core temperature of the human body. Core body temperature (CBT) refers to the internal temperature of the human body, such as the working temperature of liver, brain, and heart. However, previous measurement methods include direct measurement with esophageal and rectal probes or estimation with mercury and infrared sensors [46]. These measurements are not suitable for all patients, and are not continuous, with errors. In order to estimate the core temperature continuously, Atallah et al. [47] proposed a foam-based Y-shaped sensor with flexible electronic components. The prototype of the sensor is composed of a foam layer, a closed-cell polyethylene foam insulation layer, and thermistor (Figure 2g). With 7.7 min as the average heating time, 0.10 °C for an average error of forehead, 6.9 min as the average heating time of mastoid area, and 0.03 °C as the average error, it can be said to be very suitable for hospital application.
Thermoelectric sensors can convert temperature changes into changes in resistance, permeability or electromotive force. These changes in electrical parameters can be expressed as temperature variations through appropriate measurement circuits. Tien et al. [48] fabricated flexible pyroelectric OFET devices with piezo-and-pyroelectric nanocomposite gate dielectrics formed by a mixture of P(VDF-TrFE) and BaTiO3 nanoparticles, which can effectively distinguish the temperature effect and strain effect of the sensor. Using the co-evaporation method, Lee et al. [49] fabricated thermoelectric (TE) films of n-type bismuth telluride and p-type antimony telluride on PCB (Figure 3c). The sensitivity of the flexible TE temperature sensor was up to 192.70 μVK−1. Yang et al. [50] developed a flexible thermoelectric nanogenerator (TENG) based on TE-nanowire/poly (3-hexyl thiophene) (P3HT) polymer composite as a thermoelectric material (Figure 3d–f). The sensor can be self-powered with body temperature as the energy source, with response time of 17 s, reset time of 9 s, and detection sensitivity of 0.15 k. Tatsuya Nakamura et al. [51] proposed a thin, flexible, polymer PTC sensor with a wide temperature measurement range (25–45 °C). They discussed the characteristics of this new type of sensor from three aspects: the characteristics of single polymer PTC sensor, the characteristics of multi polymer PTC sensor and the thickness characteristic (Figure 3a,b). Compared with the traditional polymer PTC, this polymer PTC is more suitable for wearable temperature sensors and medical devices, and its wide temperature measurement range can also help people measure the temperature of various parts of the body in different environments.
Figure 3. Thermoelectric sensors: (a) polymer positive temperature coefficient (PTC) sensor; (b) the picture shows the variation of resistance temperature characteristics of the single polymer PTC sensor. The reaction temperature of the sensor varies with the content of octadecyl acrylate from 75% to 100% [51] © 2016 IEEE; (c) the size of the flexible TE device [49]; (d) fixed-area fabrics; (e) devices fabricated on flexible Kapton substrate and (f) the device attached to the human body [50].
References
- Shirer, M.Shipments of Wearable Devices Reach 118.9 Million Units in the Fourth Quarter and 336.5 Million for 2019, According to IDC . IDC. Retrieved 2020-7-25
- Hua, Q.; Sun, J.; Liu, H.; Bao, R.; Yu, R.; Zhai, J.; Pan, C.; Wang, Z.L.; Skin-inspired highly stretchable and conformable matrix networks for multifunctional sensing. Nat. Commun 2018, 9, 1-11.
- Fayyaz Shahandashti, P.; Pourkheyrollah, H.; Jahanshahi, A.; Ghafoorifard, H.; Highly conformable stretchable dry electrodes based on inexpensive flex substrate for long-term biopotential (EMG/ECG) monitoring. Sens. Actuators Phys. 2019, 295, 678–686, 10.1016/j.sna.2019.06.041.
- Sun, Y.; Yu, X.B.; Capacitive biopotential measurement for electrophysiological signal acquisition: A review. IEEE Sens. J. 2016, 16, 2832–2853.
- Zhang, L. Design, Fabrication and Performance Study of the Flexible Supercapacitor Electrode. Ph.D. Thesis, Shenzhen Institutes of Advanced Technology, Chinese Academy of Sciences, Shenzhen, China, 2020.
- Myers, A.C.; Huang, H.; Zhu, Y.; Wearable silver nanowire dry electrodes for electrophysiological sensing. RSC Adv. 2015, 5, 11627–11632., 5, 11627–11632.
- Zhang, Y.; He, P.; Luo, M.; Xu, X.; Dai, G.; Yang, J.; Highly stretchable polymer/silver nanowires composite sensor for human health monitoring. . Nano Res. 2020, 13, 919–926, doi:10.1007/s12274-020-2730-z..
- Lam, C.L.; Saleh, S.M.; Yudin, M.B.M.; Harun, F.K.; Sriprachuabwong, C.; Tuantranont, A.; Wicaksono, D.H. Graphene Ink-Coated Cotton Fabric-Based Flexible Electrode for Electrocardiography. In Proceedings of the 2017 5th International Conference on Instrumentation, Communications, Information Technology, and Biomedical Engineering (ICICI-BME), Bandung, Indonesia, 6–7 November 2017; pp. 73–75.
- Zahed, M.A.; Das, P.S.; Maharjan, P.; Barman, S.C.; Sharifuzzaman, M.; Yoon, S.H.; Park, J.Y.; Flexible and robust dry electrodes based on electroconductive polymer spray-coated 3D porous graphene for long-term electrocardiogram signal monitoring system. Carbon 2020, 165, 26–36.
- Villegas, A.; McEneaney, D.; Escalona, O. Arm-ECG Wireless Sensor System for Wearable Long-Term Surveillance of Heart Arrhythmias. Electronics 2019, 8, 1300.
- Beach, C.; Krachunov, S.; Pope, J.; Fafoutis, X.; Piechocki, R.J.; Craddock, I.; Casson, A.J. An Ultra Low Power Personalizable Wrist Worn ECG Monitor Integrated With IoT Infrastructure. IEEE Access 2018, 6, 44010–44021, doi:10.1109/access.2018.2864675.
- Kosmyna, N.; Morris, C.; Sarawgi, U.; Nguyen, T.; Maes, P. AttentivU: A Wearable Pair of EEG and EOG Glasses for Real-Time Physiological Processing. In Proceedings of the 2019 IEEE 16th International Conference on Wearable and Implantable Body Sensor Networks (BSN), Chicago, IL, USA, 19–22 May 2019; pp. 1–4.
- Li, X.; Sun, Y. NCMB-Button: A Wearable Non-contact System for Long-Term Multiple Biopotential Monitoring. In Proceedings of the 2017 IEEE/ACM International Conference on Connected Health: Applications, Systems and Engineering Technologies (CHASE), Philadelphia, PA, USA, 17–19 July 2017; pp. 348–355.
- Liu, S.; Liu, X.; Jiang, Y.; Wang, X.; Huang, P.; Wang, H.; Zhu, M.; Tan, J.; Li, P.; Lin, C.; et al. Flexible Non-contact Electrodes for Bioelectrical Signal Monitoring. In Proceedings of the 2018 40th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Honolulu, HI, USA 18–21 July 2018; pp. 4305–4308.
- Wang, X.; Liu, S.; Zhu, M.; Liu, Z.; Jiang, Y.; Wang, D.; Li, P.; Samuel, O.W.; Wu, W.; Chen, S.; et al. Performance of Flexible Non-contact Electrodes in Bioelectrical Signal Measurements. In Proceedings of the 2019 IEEE International Conference on Real-time Computing and Robotics (RCAR), Irkutsk, Russia, 4–9 August 2019; pp. 175–179.
- Gao, Y.; Soman, V.V.; Lombardi, J.P.; Rajbhandari, P.P.; Dhakal, T.P.; Wilson, D.; Poliks, M.; Ghose, K.; Turner, J.N.; Jin, Z. Heart Monitor Using Flexible Capacitive ECG Electrodes. IEEE Trans. Instrum. Meas. 2019. doi:10.1109/tim.2019.2949320.
- Christian, S.; François, P.; Marina, S.; Pascal, F.-P.; Gilles, O.H.; Franck, M.; Jean-François, S.; Isabelle, N.; Louis, B.; Karine, R.; et al. A Novel Wearable Device for Continuous Ambulatory ECG Recording: Proof of Concept and Assessment of Signal Quality. Biosensors 2019, 9, 17.
- Wang, L.-H.; Zhang, W.; Guan, M.-H.; Jiang, S.-Y.; Fan, M.-H.; Abu, P.A.R.; Chen, C.-A.; Chen, S.-L. A Low-Power High-Data-Transmission Multi-Lead ECG Acquisition Sensor System. Sensors 2019, 19, 4996.
- Chen, H.; Bao, S.; Lu, C.; Wang, L.; Ma, J.; Wang, P.; Lu, H.; Shu, F.; Oetomo, S.B.; Chen, W. Design of an Integrated Wearable Multi-Sensor Platform Based on Flexible Materials for Neonatal Monitoring. IEEE Access 2020, 8, 23732–23747, doi:10.1109/access.2020.2970469.
- Yapici, M.K.; Alkhidir, T.E. Intelligent Medical Garments with Graphene-Functionalized Smart-Cloth ECG Sensors. Sensors 2017, 17, 875.
- Al-Othman, A.; Alatoom, A.; Farooq, A.; Al-Sayah, M.; Al-Nashash, H. Novel flexible implantable electrodes based on conductive polymers and Titanium dioxide. In Proceedings of the 2018 IEEE 4th Middle East Conference on Biomedical Engineering (MECBME), Gammarth, Tunis, 28–30 March 2018; pp. 30–33.
- Lee, S.M.; Byeon, H.J.; Kim, B.H.; Lee, J.; Jeong, J.Y.; Lee, J.H.; Moon, J.-H.; Park, C.; Choi, H.; Lee, S.-H.; et al. Flexible and implantable capacitive microelectrode for bio-potential acquisition. Biochip. J. 2017, 11, 153–163, doi:10.1007/s13206-017-1304-y.
- Lee, J.-H. Miniaturized Human Insertable Cardiac Monitoring System with Wireless Power Transmission Technique. J. Sens. 2016, 2016, 1–7.
- Liu, G.Z.; Huang, B.Y.; Wang, L. A wearable respiratory biofeedback system based on generalized body sensor network. Telemed. J. E-Health Off. J. Am. Telemed. Assoc. 2011, 17, 348–357.
- Liu, G.-Z.; Wu, D.; Mei, Z.-Y.; Zhu, Q.-S.; Wang, L. Automatic detection of respiratory rate from electrocardiogram, respiration induced plethysmography and 3D acceleration signals. J. Cent. South. Univ. 2013, 20, 2423–2431, doi:10.1007/s11771-013-1752-z.
- Chen, H.; Bao, S.; Ma, J.; Wang, P.; Lu, H.; Oetomo, S.B.; Chen, W. A Wearable Daily Respiration Monitoring System Using PDMS-graphene Compound Tensile Sensor for Adult. In Proceedings of the 2019 41st Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Berlin, Germany, 23–27 July 2019; pp. 1269–1273.
- Hung, P.D.; Bonnet, S.; Guillemaud, R.; Castelli, E.; Yen, P.T.N. Estimation of respiratory waveform using an accelerometer. In Proceedings of the 2008 5th IEEE International Symposium on Biomedical Imaging: From Nano to Macro, Paris, France, 14–17 May 2008; pp. 1493–1496.
- Bates, A.; Ling, M.J.; Mann, J.; Arvind, D.K. Respiratory Rate and Flow Waveform Estimation from Tri-axial Accelerometer Data. In Proceedings of the 2010 International Conference on Body Sensor Networks, Singapore, 7–9 June 2010; pp. 144–150.
- Vertens, J.; Fischer, F.; Heyde, C.; Höflinger, F.; Zhang, R.; Reindl, L.; Gollhofer, A. Measuring Respiration and Heart Rate Using Two Acceleration Sensors on a Fully Embedded Platform. In Proceedings of the 3rd International Congress on Sport Sciences Research and Technology Support (icSPORTS 2015), Lisbon, Portugal, 15–17 November 2015, doi:10.5220/0005604000150023.
- Mack, D.C.; Patrie, J.T.; Suratt, P.M.; Felder, R.A.; Alwan, M. Development and Preliminary Validation of Heart Rate and Breathing Rate Detection Using a Passive, Ballistocardiography-Based Sleep Monitoring System. IEEE Trans. Inf. Technol. Biomed. 2009, 13, 111–120, doi:10.1109/titb.2008.2007194.
- Zhang, Z.; Wang, W.; Wang, B.; Wu, H.; Liu, H.; Zhang, Y. A Prototype of Wearable Respiration Biofeedback Platform and Its Preliminary Evaluation on Cardiovascular Variability. In Proceedings of the 2009 3rd International Conference on Bioinformatics and Biomedical Engineering, Beijing, China, 11–13 June 2009; pp. 1–4.
- Fan, W.; He, Q.; Meng, K.; Tan, X.; Zhou, Z.; Zhang, G.; Yang, J.; Wang, Z.L. Machine-knitted washable sensor array textile for precise epidermal physiological signal monitoring. Sci. Adv. 2020, 6, eaay2840, doi:10.1126/sciadv.aay2840.
- Chen, Y.; Lu, B.; Feng, X. Biocompatible and Ultra-Flexible Inorganic Strain Sensors Attached to Skin for Long-Term Vital Signs Monitoring. IEEE Electron. Device Lett. 2016, 37, 496–499, doi:10.1109/led.2016.2536036.
- Yamamoto, A.; Nakamoto, H.; Bessho, Y.; Watanabe, Y.; Oki, Y.; Ono, K.; Fujimoto, Y.; Terada, T.; Ishikawa, A. Monitoring respiratory rates with a wearable system using a stretchable strain sensor during moderate exercise. Med. Biol. Eng. Comput. 2019, 57, 2741–2756, doi:10.1007/s11517-019-02062-2.
- Kawamura, M.; Ishizawa, H.; Sato, S.; Koyama, S. Application to vital signs by Fiber Bragg Grating sensing. In Proceedings of the SICE Annual Conference 2011, Tokyo, Japan, 13–18 September 2011; pp. 2702–2704.
- Dziuda, Ł.; Krej, M.; Skibniewski, F.W. Fiber Bragg Grating Strain Sensor Incorporated to Monitor Patient Vital Signs During MRI. IEEE Sens. J. 2013, 13, 4986–4991, doi:10.1109/jsen.2013.2279160.
- Fajkus, M.; Nedoma, J.; Martinek, R.; Brablik, J.; Vanus, J.; Novak, M.; Zabka, S.; Vasinek, V.; Hanzlikova, P.; Vojtisek, L. MR Fully Compatible and Safe FBG Breathing Sensor: A Practical Solution for Respiratory Triggering. IEEE Access 2019, 7, 123013–123025, doi:10.1109/access.2019.2937624.
- Presti, D.L.; Massaroni, C.; D’Abbraccio, J.; Massari, L.; Caponero, M.; Longo, U.G.; Formica, D.; Oddo, C.M.; Schena, E. Wearable System Based on Flexible FBG for Respiratory and Cardiac Monitoring. IEEE Sens. J. 2019, 19, 7391–7398, doi:10.1109/jsen.2019.2916320.
- Gu, Y.; Zhang, T.; Chen, H.; Wang, F.; Pu, Y.; Gao, C.; Li, S. Mini Review on Flexible and Wearable Electronics for Monitoring Human Health Information. Nanoscale Res. Lett. 2019, 14, 263, doi:10.1186/s11671-019-3084-x.
- Kedambaimoole, V.; Neella, N.; Gaddam, V.; Rajanna, K.; Nayak, M.M. Graphene-Nickel composite films on flexible PCB for temperature monitoring. In Proceedings of the 2017 IEEE 12th International Conference on Nano/Micro Engineered and Molecular Systems (NEMS), Los Angeles, CA, USA, 9–12 April 2017; pp. 173–176.
- Khan, Y.; Garg, M.; Gui, Q.; Schadt, M.; Gaikwad, A.; Han, D.; Yamamoto, N.A.D.; Hart, P.; Welte, R.; Wilson, W. Flexible Hybrid Electronics: Direct Interfacing of Soft and Hard Electronics for Wearable Health Monitoring. Adv. Funct. Mater. 2016, 26, 8764–8775.
- Giuliani, A.; Placidi, M.; Francesco, F.D.; Pucci, A. A new polystyrene-based ionomer/MWCNT nanocomposite for wearable skin temperature sensors. React. Funct. Polym. 2014, 76, 57–62.
- Wang, F.; Jiang, J.; Sun, F.; Sun, L.; Wang, T.; Liu, Y.; Li, M. Flexible wearable graphene/alginate composite non-woven fabric temperature sensor with high sensitivity and anti-interference. Cellulose 2020, 27, 2369–2380, doi:10.1007/s10570-019-02951-7.
- Ali, S.; Bae, J.; Bermak, A. A flexible differential temperature sensor for wearable electronics applications. In Proceedings of the 2019 IEEE International Conference on Flexible and Printable Sensors and Systems (FLEPS), Glasgow, UK, 8–10 July 2019; pp. 1–3.
- Yan, C.; Wang, J.; Lee, P.S. Stretchable Graphene Thermistor with Tunable Thermal Index. ACS Nano 2015, 9, 2130–2137.
- Bonzi, M.; Fiorelli, E.M.; Solbiati, M.; Montano, N. Accuracy of Peripheral Thermometers for Estimating Temperature. Ann. Intern. Med. 2016, 165, 73.
- Atallah, L.; Ciuhu, C.; Wang, C.; Bongers, E.; Blom, T.; Paulussen, I.; Noordergraaf, G. An ergonomic wearable core body temperature sensor. In Proceedings of the 2018 IEEE 15th International Conference on Wearable and Implantable Body Sensor Networks (BSN), Las Vegas, NV, USA, 4–7 March 2018; pp. 70–73.
- Tien, N.T.; Jeon, S.; Kim, D.-I.; Trung, T.Q.; Jang, M.; Hwang, B.-U.; Byun, K.-E.; Bae, J.; Lee, E.; Tok, J.B.H.; et al. A Flexible Bimodal Sensor Array for Simultaneous Sensing of Pressure and Temperature. Adv. Mater. 2014, 26, 796–804, doi:10.1002/adma.201302869.
- Lee, S.H.; Shen, H.; Han, S. Flexible Thermoelectric Module Using Bi-Te and Sb-Te Thin Films for Temperature Sensors. J. Electron. Mater. 2019, 48, 5464–5470, doi:10.1007/s11664-019-07235-1.
- Yang, Y.; Lin, Z.-H.; Hou, T.; Zhang, F.; Wang, Z.L. Nanowire-composite based flexible thermoelectric nanogenerators and self-powered temperature sensors. Nano Res. 2012, 5, 888–895.
- Nakamura, T.; Yokota, T.; Terakawa, Y.; Reeder, J.; Voit, W.; Someya, T.; Sekino, M. Development of flexible and wide-range polymer-based temperature sensor for human bodies. In Proceedings of the 2016 IEEE-EMBS International Conference on Biomedical and Health Informatics (BHI), Las Vegas, NV, USA, 24–27 February 2016; pp. 485–488.