
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Chunsheng Wu | + 4215 word(s) | 4215 | 2021-09-08 04:20:18 | | | |
| 2 | Peter Tang | Meta information modification | 4215 | 2021-09-17 04:30:29 | | |
Video Upload Options
The mammalian olfactory system has an amazing ability to distinguish thousands of odorant molecules at the trace level. Scientists have made great achievements on revealing the olfactory sensing mechanisms in decades; even though many issues need addressing. Optogenetics provides a novel technical approach to solve this dilemma by utilizing light to illuminate specific part of the olfactory system; which can be used in all corners of the olfactory system for revealing the olfactory mechanism.
1. Introduction
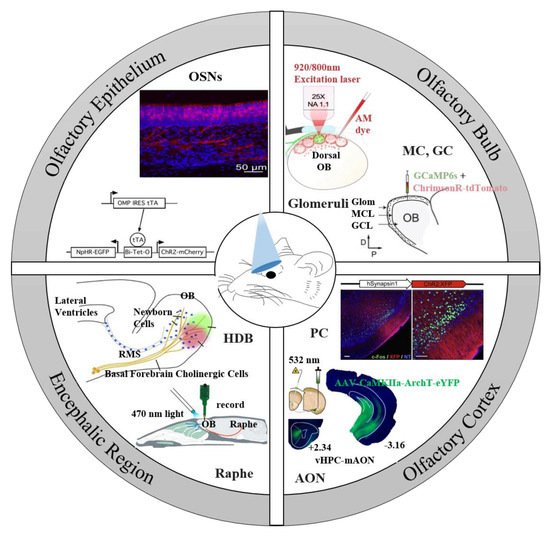
2. Optogenetic Tools for the Olfactory System
|
Level |
Expression Target |
Model Animal |
Expression Approach |
Light Delivery |
Electrophysiology Recordings |
Behavior |
Ref. |
||||
|---|---|---|---|---|---|---|---|---|---|---|---|
|
Tool |
Wavelength (nm) |
Duration (ms) |
Frequency (Hz) |
Power (mW) |
|||||||
|
GL |
Glomeruli |
OMP-ChR2-YFP transgenic mice |
Transgenic animal model |
A 470 nm LED coupled with an objective |
470 |
10 |
- |
- |
M/T cells: patch-clamp |
- |
[6] |
|
SACs |
TH-Cre mice |
Injection of AAV-ChR2 into the GL |
A solid-state laser coupled with an optical fiber |
473 |
- |
- |
100 |
M/T cells or ETCs: whole-cell patch-clamp or cell-attached and tungsten microelectrodes |
- |
[39] |
|
|
EPL |
EPL-INs |
Crh-Cre mice |
Injection of AAV-ChR2 into the OB |
A BLM-Series 473 nm blue laser system coupled with an objective |
473 |
20–40 |
EPL INs: whole-cell patch-clamp |
Olfactory associative learning training |
[40] |
||
|
EPL-INs |
CRH-Cre mice |
Injection of AAV-ChR2 into the OB |
A blue laser system guided by implanted fiber optics |
473 |
10 |
- |
30 |
MCs: whole-cell patch-clamp and extracellular recording electrodes |
Olfactory associative learning training |
[59] |
|
|
IPL |
dSACs |
Chrna2-Cre mice |
Injection of AAV-ChR2 into the IPL |
A 75 W xenon arc lamp coupled with an objective |
- |
- |
- |
- |
TCs: whole cell patch clamp |
- |
[35] |
|
GCL |
GCs |
Dlx5/6-Cre mice |
Injection of AAV-ChR2 into the OB |
A BLM-Series 473 nm blue laser system coupled with an objective |
473 |
20–40 |
GCs: whole-cell patch-clamp |
Olfactory associative learning training |
[40] |
||
|
GCs |
OMP-Cre mice |
Injection of AAV-ChR2 into the GCL |
An implanted LEDs driven with a high-power LED driver |
470 |
5 |
40 |
23 |
M/T cells: a silicon-based recording electrode and 32 channels optrode |
Habituation task; Olfactory discrimination task |
[60] |
|
|
Encephalic Region |
Expression Target |
Model Animal |
Expression Approach |
Light Delivery |
Electrophysiology Recordings |
Ref. |
||||
|---|---|---|---|---|---|---|---|---|---|---|
|
Tool |
Wavelength (nm) |
Duration (ms) |
Frequency (Hz) |
Power (mW) |
||||||
|
Basal forebrain |
HDB cholinergic neurons |
VGLUT3-Cre mice |
Injection of AAV-ChR2 into the HDB |
A 75W xenon arc lamp coupled with an objective |
- |
10–20 |
- |
- |
OB cells: patch clamp |
[61] |
|
ChAT-ChR2-EYFP transgenic mice |
Transgenic animal model |
A diode-pumped solid-state 473 nm laser coupled with an optical fiber target the HDB |
473 |
15 |
5–50 |
- |
M/T cells and brain slices: patch clamp |
[41] |
||
|
ChAT-ChR2-EYFP mice transgenic |
Transgenic animal model |
A blue light diode laser and a blue LED coupled with implanted fiber |
473 |
15 |
5–50 |
- |
- |
[32] |
||
|
HDB GABAergic neurons |
DLX5/6-Cre mice |
Injection of AAV-ChR2 into the HDB |
A blueCoolLED pE 100 coupled with an objective |
490 |
- |
- |
- |
OB cells: whole-cell |
[62] |
|
|
ChAT/GAD2-Cre mice |
Injection of AAV-ChR2/eNpHR into the HDB |
A 470 or 565 nm LED coupled with an optical fiber positioned in the OB |
470; 565 |
10000 |
- |
10;3 |
M/T cells: sixteen channel electrodes |
[63] |
||
|
Raphe nuclei |
5-HT axons |
TPH2-ChR2-YFP transgenic mice |
Transgenic animal model |
A bright light-emitting diode (LED) array coupled with a microscope |
473 |
10 |
10 |
15 |
M/T cells: tungsten electrodes and whole-cell |
[64] |
|
serotonergic cells |
Slc6a4-Cre mice |
Injection of AAV-ChR2 into DRN |
A 470 nm LED coupled with a glass fiber positioned close to the OB |
470 |
10000 |
1–10 |
OB cells: 16-channel electrode |
[31] |
||
|
serotonergic cells |
SERT-Cre mice |
Injection of AAV-ChR2 in the DRN |
A 470 nm laser coupled with an optrode lowered into the DRN |
470 |
10 |
1–30 |
- |
APC neurons: microelectrodes; |
[65] |
|
|
locus coreuleus |
noradrenergic neurons |
DBH-Cre-NpHR transgenic mice |
Transgenic animal model |
A solid-state laser coupled with an optical fiber implanted in the OB |
532 |
- |
- |
2–10 |
MCs: tetrodes |
[66] |
3. The Sensory Input from OSNs to the OB
4. How Do Activities of the Olfactory Bulb Neurons Affect Perception?
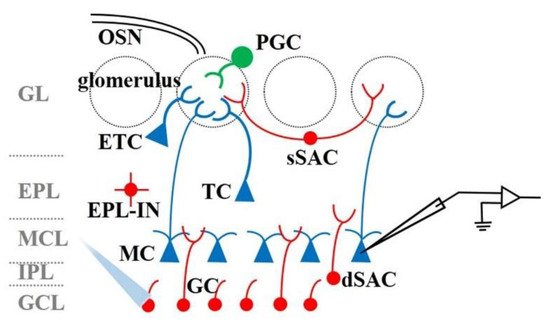
5. The Function of OB Interneurons in Odor Information Processing
References
- Giessel, A.J.; Datta, S.R. Olfactory maps, circuits and computations. Curr. Opin. Neurobiol. 2014, 24, 120–132.
- Genva, M.; Kenne Kemene, T.; Deleu, M.; Lins, L.; Fauconnier, M.-L. Is It Possible to Predict the Odor of a Molecule on the Basis of its Structure? Int. J. Mol. Sci. 2019, 20, 3018.
- Mainland, J.D.; Keller, A.; Li, Y.R.; Zhou, T.; Trimmer, C.; Snyder, L.L.; Moberly, A.H.; Adipietro, K.A.; Liu, W.L.L.; Zhuang, H.; et al. The missense of smell: Functional variability in the human odorant receptor repertoire. Nat. Neurosci. 2014, 17, 114–120.
- Xu, L.; Li, W.; Voleti, V.; Zou, D.-J.; Hillman, E.M.C.; Firestein, S. Widespread receptor-driven modulation in peripheral olfactory coding. Science 2020, 368, eaaz5390.
- Cavarretta, F.; Burton, S.D.; Igarashi, K.M.; Shepherd, G.M.; Hines, M.L.; Migliore, M. Parallel odor processing by mitral and middle tufted cells in the olfactory bulb. Sci. Rep. 2018, 8, 7625.
- Geramita, M.; Urban, N.N. Differences in Glomerular-Layer-Mediated Feedforward Inhibition onto Mitral and Tufted Cells Lead to Distinct Modes of Intensity Coding. J. Neurosci. 2017, 37, 1428–1438.
- Liu, G.; Froudarakis, E.; Patel, J.M.; Kochukov, M.Y.; Pekarek, B.; Hunt, P.J.; Patel, M.; Ung, K.; Fu, C.-H.; Jo, J.; et al. Target specific functions of EPL interneurons in olfactory circuits. Nat. Commun. 2019, 10, 3369.
- Bolding, K.A.; Franks, K.M. Recurrent cortical circuits implement concentration-invariant odor coding. Science 2018, 361.
- Otazu, G.H.; Chae, H.; Davis, M.B.; Albeanu, D.F. Cortical Feedback Decorrelates Olfactory Bulb Output in Awake Mice. Neuron 2015, 86, 1461–1477.
- Wang, C.Y.; Liu, Z.; Ng, Y.H.; Südhof, T.C. A Synaptic Circuit Required for Acquisition but Not Recall of Social Transmission of Food Preference. Neuron 2020, 107, 144–157.
- Uchida, N.; Poo, C.; Haddad, R. Coding and Transformations in the Olfactory System. Annu. Rev. Neurosci. 2014, 37, 363–385.
- Wilson, D.A.; Sullivan, R.M. Cortical processing of odor objects. Neuron 2011, 72, 506–519.
- Sohal, V.S.; Zhang, F.; Yizhar, O.; Deisseroth, K.J.N. Parvalbumin neurons and gamma rhythms enhance cortical circuit performance. Nature 2009, 459, 698–702.
- Boyden, E.S.; Zhang, F.; Bamberg, E.; Nagel, G.; Deisseroth, K. Millisecond-timescale, genetically targeted optical control of neural activity. Nat. Neurosci. 2005, 8, 1263–1268.
- Zhang, F.; Wang, L.; Brauner, M.; Liewald, J.F.; Kay, K.; Watzke, N.; Wood, P.G.; Bamberg, E.; Nagel, G.; Gottschalk, A.J.N. Multimodal fast optical interrogation of neural circuitry. Nature 2007, 446, 633–639.
- Nagel, G.; Szellas, T.; Huhn, W.; Kateriya, S.; Adeishvili, N.; Berthold, P.; Ollig, D.; Hegemann, P.; Bamberg, E. Channelrhodopsin-2, a directly light-gated cation-selective membrane channel. Proc. Natl. Acad. Sci. USA 2003, 100, 13940–13945.
- Klapoetke, N.C.; Murata, Y.; Kim, S.S.; Pulver, S.R.; Birdsey-Benson, A.; Cho, Y.K.; Morimoto, T.K.; Chuong, A.S.; Carpenter, E.J.; Tian, Z.; et al. Independent optical excitation of distinct neural populations. Nat. Methods 2014, 11, 338–346.
- Chaigneau, E.; Ronzitti, E.; Gajowa, M.A.; Soler-Llavina, G.J.; Tanese, D.; Brureau, A.Y.B.; Papagiakoumou, E.; Zeng, H.; Emiliani, V. Two-Photon Holographic Stimulation of ReaChR. Front. Cell. Neurosci. 2016, 10, 234.
- Watanabe, H.; Sano, H.; Chiken, S.; Kobayashi, K.; Fukata, Y.; Fukata, M.; Mushiake, H.; Nambu, A. Forelimb movements evoked by optogenetic stimulation of the macaque motor cortex. Nat. Commun 2020, 11, 3253.
- Tamura, K.; Takeda, M.; Setsuie, R.; Tsubota, T.; Hirabayashi, T.; Miyamoto, K.; Miyashita, Y. Conversion of object identity to object-general semantic value in the primate temporal cortex. Science 2017, 357, 687–692.
- Hardt, O.; Nadel, L. Systems consolidation revisited, but not revised: The promise and limits of optogenetics in the study of memory. Neurosci. Lett. 2018, 680, 54–59.
- Parnaudeau, S.; Bolkan, S.S.; Kellendonk, C.J.B.P. The Mediodorsal Thalamus: An Essential Partner of the Prefrontal Cortex for Cognition. Biol. Psychiatry 2017, 83, 648–656.
- Cheng, Z.; Cui, R.; Ge, T.; Yang, W.; Li, B. Optogenetics: What it has uncovered in potential pathways of depression. Pharmacol. Res. 2020, 152, 104596.
- Jarrin, S.; Finn, D.P.J.N.; Reviews, B. Optogenetics and its application in pain and anxiety research. Neurosci. Biobehav. Rev. 2019, 105, 200–211.
- Grimaud, J.; Lledo, P.M. Illuminating odors: When optogenetics brings to light unexpected olfactory abilities. Learn. Mem. 2016, 23, 249–254.
- Gire, D.H.; Franks, K.M.; Zak, J.D.; Tanaka, K.F.; Whitesell, J.D.; Mulligan, A.A.; Hen, R.; Schoppa, N.E. Mitral cells in the olfactory bulb are mainly excited through a multistep signaling path. J. Neurosci. 2012, 32, 2964–2975.
- Braubach, O.A.-O.; Tombaz, T.; Geiller, T.; Homma, R.; Bozza, T.A.-O.; Cohen, L.B.; Choi, Y. Sparsened neuronal activity in an optogenetically activated olfactory glomerulus. Sci. Rep. 2018, 8, 1–17.
- Gill, J.V.; Lerman, G.M.; Zhao, H.; Stetler, B.J.; Rinberg, D.; Shoham, S. Precise Holographic Manipulation of Olfactory Circuits Reveals Coding Features Determining Perceptual Detection. Neuron 2020, 108, 382–393.
- Choi, G.B.; Stettler, D.D.; Kallman, B.R.; Bhaskar, S.T.; Fleischmann, A.; Axel, R. Driving Opposing Behaviors with Ensembles of Piriform Neurons. Cell 2011, 146, 1004–1015.
- Aqrabawi, A.J.; Kim, J.C. Hippocampal projections to the anterior olfactory nucleus differentially convey spatiotemporal information during episodic odour memory. Nat. Commun. 2018, 9, 2735.
- Brunert, D.; Tsuno, Y.; Rothermel, M.; Shipley, M.T.; Wachowiak, M. Cell-Type-Specific Modulation of Sensory Responses in Olfactory Bulb Circuits by Serotonergic Projections from the Raphe Nuclei. J. Neurosci. 2016, 36, 6820.
- Nitenson, A.S.; Nieves, G.M.; Poeta, D.L.; Bahar, R.; Rachofsky, C.; Mandairon, N.; Bath, K.G. Acetylcholine Regulates Olfactory Perceptual Learning through Effects on Adult Neurogenesis. iScience 2019, 22, 544–556.
- Rost, B.R.; Schneider-Warme, F.; Schmitz, D.; Hegemann, P. Optogenetic Tools for Subcellular Applications in Neuroscience. Neuron 2017, 96, 572–603.
- McCarthy, E.A.; Kunkhyen, T.; Korzan, W.J.; Naik, A.; Maqsudlu, A.; Cherry, J.A.; Baum, M.J. A comparison of the effects of male pheromone priming and optogenetic inhibition of accessory olfactory bulb forebrain inputs on the sexual behavior of estrous female mice. Horm. Behav. 2017, 89, 104–112.
- Burton, S.D.; Larocca, G.; Liu, A.; Cheetham, C.E.J.; Urban, N.N. Olfactory Bulb Deep Short-Axon Cells Mediate Widespread Inhibition of Tufted Cell Apical Dendrites. J. Neurosci. 2017, 37, 1117–1138.
- Choy, J.M.C.; Suzuki, N.; Shima, Y.; Budisantoso, T.; Nelson, S.B.; Bekkers, J.M. Optogenetic Mapping of Intracortical Circuits Originating from Semilunar Cells in the Piriform Cortex. Cereb. Cortex 2015, 27, 589–601.
- Markopoulos, F.; Rokni, D.; Gire, D.H.; Murthy, V.N. Functional Properties of Cortical Feedback Projections to the Olfactory Bulb. Neuron 2012, 76, 1175–1188.
- Alonso, M.; Lepousez, G.; Wagner, S.; Bardy, C.; Gabellec, M.M.; Torquet, N.; Lledo, P.M. Activation of adult-born neurons facilitates learning and memory. Nat. Neurosci. 2012, 15, 897–904.
- Liu, S.; Puche, A.C.; Shipley, M.T. The Interglomerular Circuit Potently Inhibits Olfactory Bulb Output Neurons by Both Direct and Indirect Pathways. J. Neurosci. 2016, 36, 9604–9617.
- Huang, L.; Ung, K.; Garcia, I.; Quast, K.B.; Cordiner, K.; Saggau, P.; Arenkiel, B.R. Task Learning Promotes Plasticity of Interneuron Connectivity Maps in the Olfactory Bulb. J. Neurosci. 2016, 36, 8856–8871.
- Ma, M.; Luo, M. Optogenetic Activation of Basal Forebrain Cholinergic Neurons Modulates Neuronal Excitability and Sensory Responses in the Main Olfactory Bulb. J. Neurosci. 2012, 32, 10105–10116.
- Arenkiel, B.R.; Peca, J.; Davison, I.G.; Feliciano, C.; Deisseroth, K.; Augustine, G.J.; Ehlers, M.D.; Feng, G. In vivo light-induced activation of neural circuitry in transgenic mice expressing channelrhodopsin-2. Neuron 2007, 54, 205–218.
- Zhao, S.; Ting, J.T.; Atallah, H.E.; Qiu, L.; Tan, J.; Gloss, B.; Augustine, G.J.; Deisseroth, K.; Luo, M.; Graybiel, A.M.; et al. Cell type–specific channelrhodopsin-2 transgenic mice for optogenetic dissection of neural circuitry function. Nat. Methods 2011, 8, 745–752.
- Dhawale, A.K.; Hagiwara, A.; Bhalla, U.S.; Murthy, V.N.; Albeanu, D.F. Non-redundant odor coding by sister mitral cells revealed by light addressable glomeruli in the mouse. Nat. Neurosci. 2010, 13, 1404–1412.
- Smear, M.C.; Shusterman, R.; Oconnor, R.P.; Bozza, T.C.; Rinberg, D. Perception of sniff phase in mouse olfaction. Nature 2011, 479, 397–400.
- Smear, M.C.; Resulaj, A.; Zhang, J.; Bozza, T.C.; Rinberg, D. Multiple perceptible signals from a single olfactory glomerulus. Nat. Neurosci. 2013, 16, 1687–1691.
- Luna, V.M.; Morozov, A. Input-specific excitation of olfactory cortex microcircuits. Front. Neural Circuits 2012, 6, 69.
- Gunaydin, L.A.; Yizhar, O.; Berndt, A.; Sohal, V.S.; Deisseroth, K.; Hegemann, P. Ultrafast optogenetic control. Nat. Neurosci. 2010, 13, 387–392.
- Murata, K.; Kinoshita, T.; Fukazawa, Y.; Kobayashi, K.; Yamanaka, A.; Hikida, T.; Manabe, H.; Yamaguchi, M. Opposing Roles of Dopamine Receptor D1- and D2-Expressing Neurons in the Anteromedial Olfactory Tubercle in Acquisition of Place Preference in Mice. Front. Behav. Neurosci. 2019, 13, 50.
- Aqrabawi, A.J.; Kim, J.C. Olfactory memory representations are stored in the anterior olfactory nucleus. Nat. Commun. 2020, 11, 1246.
- Lin, J.Y.; Knutsen, P.M.; Muller, A.; Kleinfeld, D.; Tsien, R.Y. ReaChR: A red-shifted variant of channelrhodopsin enables deep transcranial optogenetic excitation. Nat. Neurosci. 2013, 16, 1499–1508.
- Inagaki, H.K.; Jung, Y.; Hoopfer, E.D.; Wong, A.M.; Mishra, N.; Lin, J.Y.; Tsien, R.Y.; Anderson, D.J. Optogenetic control of Drosophila using a red-shifted channelrhodopsin reveals experience-dependent influences on courtship. Nat. Methods 2014, 11, 325–332.
- Inada, K.; Tsuchimoto, Y.; Kazama, H. Origins of Cell-Type-Specific Olfactory Processing in the Drosophila Mushroom Body Circuit. Neuron 2017, 95, 357–367.e354.
- Guo, H.; Kunwar, K.; Smith, D. Odorant Receptor Sensitivity Modulation in Drosophila. J. Neurosci. 2017, 37, 9465.
- Fukunaga, I.; Herb, J.T.; Kollo, M.; Boyden, E.S.; Schaefer, A.T. Independent control of gamma and theta activity by distinct interneuron networks in the olfactory bulb. Nat. Neurosci. 2014, 17, 1208–1216.
- Gradinaru, V.; Zhang, F.; Ramakrishnan, C.; Mattis, J.; Prakash, R.; Diester, I.; Goshen, I.; Thompson, K.R.; Deisseroth, K. Molecular and Cellular Approaches for Diversifying and Extending Optogenetics. Cell 2010, 141, 154–165.
- Midroit, M.; Chalençon, L.; Renier, N.; Milton, A.; Thevenet, M.; Sacquet, J.; Breton, M.; Forest, J.; Noury, N.; Richard, M.; et al. Neural processing of the reward value of pleasant odorants. Curr. Biol. 2021, 31, 1592–1605.e1599.
- Kermen, F.; Midroit, M.; Kuczewski, N.; Forest, J.; Thévenet, M.; Sacquet, J.; Benetollo, C.; Richard, M.; Didier, A.; Mandairon, N. Topographical representation of odor hedonics in the olfactory bulb. Nat. Neurosci. 2016, 19, 876–878.
- Huang, L.; Garcia, I.; Jen, H.; Arenkiel, B.R. Reciprocal connectivity between mitral cells and external plexiform layer interneurons in the mouse olfactory bulb. Front. Neural Circuits 2013, 7, 32.
- Gschwend, O.; Abraham, N.M.; Lagier, S.; Begnaud, F.; Rodriguez, I.; Carleton, A. Neuronal pattern separation in the olfactory bulb improves odor discrimination learning. Nat. Neurosci. 2015, 18, 1474–1482.
- Case, D.T.; Burton, S.D.; Gedeon, J.Y.; Williams, S.P.G.; Urban, N.N.; Seal, R.P. Layer- and cell type-selective co-transmission by a basal forebrain cholinergic projection to the olfactory bulb. Nat. Commun. 2017, 8, 652.
- Diez, A.S.; Najac, M.; De Saint Jan, D. Basal forebrain GABAergic innervation of olfactory bulb periglomerular interneurons. J. Physiol. 2019, 597, 2547–2563.
- Böhm, E.; Brunert, D.; Rothermel, M. Input dependent modulation of olfactory bulb activity by HDB GABAergic projections. Sci. Rep. 2020, 10, 10696.
- Kapoor, V.; Provost, A.C.; Agarwal, P.; Murthy, V.N. Activation of raphe nuclei triggers rapid and distinct effects on parallel olfactory bulb output channels. Nat. Neurosci. 2016, 19, 271–282.
- Lottem, E.; Lorincz, M.L.; Mainen, Z.F. Optogenetic Activation of Dorsal Raphe Serotonin Neurons Rapidly Inhibits Spontaneous but Not Odor-Evoked Activity in Olfactory Cortex. J. Neurosci. 2016, 36, 7–18.
- Ramirezgordillo, D.; Ma, M.; Restrepo, D. Precision of Classification of Odorant Value by the Power of Olfactory Bulb Oscillations Is Altered by Optogenetic Silencing of Local Adrenergic Innervation. Front. Cell. Neurosci. 2018, 12, 48.
- Mori, K.; Sakano, H. How is the olfactory map formed and interpreted in the mammalian brain? Annu Rev Neurosci. 2011, 34, 467–499.
- Murthy, V.N. Olfactory maps in the brain. Annu Rev Neurosci. 2011, 34, 233–258.
- Najac, M.; De Saint Jan, D.; Reguero, L.; Grandes, P.; Charpak, S. Monosynaptic and polysynaptic feed-forward inputs to mitral cells from olfactory sensory neurons. J. Neurosci. 2011, 31, 8722–8729.
- Shmuel, R.; Secundo, L.; Haddad, R. Strong, weak and neuron type dependent lateral inhibition in the olfactory bulb. Sci Rep. 2019, 9, 1602.
- Vaaga, C.E.; Westbrook, G.L. Parallel processing of afferent olfactory sensory information. J. Physiol. 2016, 594, 6715–6732.
- Genovese, F.; Thews, M.; Mohrlen, F.; Frings, S. Properties of an optogenetic model for olfactory stimulation. J. Physiol. 2016, 594, 3501–3516.
- Mombaerts, P. Axonal wiring in the mouse olfactory system. Annu. Rev. Cell Dev. Biol. 2006, 22, 713–737.
- Vassar, R.; Chao, S.K.; Sitcheran, R.; Nuñez, J.M.; Vosshall, L.B.; Axel, R. Topographic organization of sensory projections to the olfactory bulb. Cell 1994, 79, 981–991.
- Iwata, R.; Kiyonari, H.; Imai, T. Mechanosensory-Based Phase Coding of Odor Identity in the Olfactory Bulb. Neuron 2017, 96, 1139–1152.e1137.
- Malnic, B.; Hirono, J.; Sato, T.; Buck, L.B. Combinatorial receptor codes for odors. Cell 1999, 96, 713–723.
- Zhou, Z.; Belluscio, L. Coding odorant concentration through activation timing between the medial and lateral olfactory bulb. Cell Rep. 2012, 2, 1143–1150.
- Grabe, V.; Sachse, S. Fundamental principles of the olfactory code. Biosystems 2018, 164, 94–101.
- Renou, M.; Party, V.; Rouyar, A.; Anton, S. Olfactory signal coding in an odor background. Biosystems 2015, 136, 35–45.
- Rebello, M.R.; McTavish, T.S.; Willhite, D.C.; Short, S.M.; Shepherd, G.M.; Verhagen, J.V. Perception of odors linked to precise timing in the olfactory system. PLoS Biol. 2014, 12, e1002021.
- Li, A.; Gire, D.H.; Bozza, T.C.; Restrepo, D. Precise Detection of Direct Glomerular Input Duration by the Olfactory Bulb. J. Neurosci. 2014, 34, 16058–16064.
- Wilson, C.D.; Serrano, G.O.; Koulakov, A.A.; Rinberg, D. A primacy code for odor identity. Nat. Commun. 2017, 8, 1477.
- Chong, E.; Moroni, M.; Wilson, C.; Shoham, S.; Panzeri, S.; Rinberg, D. Manipulating synthetic optogenetic odors reveals the coding logic of olfactory perception. Science 2020, 368.
- Mardinly, A.R.; Oldenburg, I.A.; Pégard, N.C.; Sridharan, S.; Lyall, E.H.; Chesnov, K.; Brohawn, S.G.; Waller, L.; Adesnik, H. Precise multimodal optical control of neural ensemble activity. Nat. Neurosci. 2018, 21, 881–893.
- Chen, I.W.; Ronzitti, E.; Lee, B.R.; Daigle, T.L.; Dalkara, D.; Zeng, H.; Emiliani, V.; Papagiakoumou, E. In Vivo Submillisecond Two-Photon Optogenetics with Temporally Focused Patterned Light. J. Neurosci. Off. J. Soc. Neurosci. 2019, 39, 3484–3497.
- Carey, R.M.; Verhagen, J.V.; Wesson, D.W.; Pírez, N.; Wachowiak, M. Temporal structure of receptor neuron input to the olfactory bulb imaged in behaving rats. J. Neurophysiol. 2009, 101, 1073–1088.




