
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Haoyu Yang | + 2619 word(s) | 2619 | 2021-08-20 11:13:35 | | | |
| 2 | Conner Chen | Meta information modification | 2619 | 2021-09-03 10:00:44 | | |
Video Upload Options
Integrin β4 (ITGβ4) is a class of transmembrane adhesion molecules composed of hemidesmosomes (HDs). Its unique long intracellular domain provides intricate signal transduction functions. These signal transduction effects are especially prominent in tumors. Integrin β4 is differentially expressed in various tumors, and it plays a vital role in tumor invasion, proliferation, epithelial–mesenchymal transition, and angiogenesis. In clinical practice, it is described as a diagnostic marker for the targeted treatment of cancer and will be helpful in the clinical diagnosis and treatment of tumors.
1. Function of Integrin β4
Integrins are a superfamily of cell adhesion receptors. They mainly recognize extracellular matrix ligands and cell surface ligands. They mainly act as traction receptors and can transmit and detect changes in the mechanical forces acting on the extracellular matrix [1].
Integrin β4 only binds to integrin α6 to form a heterodimer [2], and its extracellular domain usually binds to the basement membrane component laminin-332, which is the main HD component [3]. Laminin interacts with integrin α6β4 to maintain the integrity of epithelial cells, affects cell adhesion functioning, and regulates cell proliferation [4]. Integrin β4 is a transmembrane protein and plays the role of signal transduction. Therefore, integrin β4 has the ability to promote HD composition and stabilize epidermal adhesion [2].
Through the intracytoplasmic region of integrin β4, it interacts with ECM molecules to activate many intracellular signal mediators, including FAK, src, PKC, PI3K, and MAPKs, resulting in alterations in the actin cytoskeleton and gene expression [4]. For example, critical for the mechanical stability of HDs is the interaction between integrin α6β4 and plectin, which is destabilized when HD disassembly is required. Growth factors such as epidermal growth factor (EGF) can trigger HD disassembly and induce the phosphorylation of integrin β4 intracellular domains, which mediate invasion in carcinoma cells [5].
Therefore, integrin β4 can activate intracellular signal transduction in normal tissues and can reorganize the actin cell signal skeleton to maintain the integrity of epithelial cells. Recent studies found that integrin β4 promotes cell proliferation, migration, and invasion and plays a pivotal role in tumorigenesis, leading to poor prognosis and reduced survival rates [6].
2. Differential Expression of Integrin β4 in Tumors
2.1. Lung Cancer
Both quantitative mass spectrometry analysis and Western blot analysis showed that integrin β4 is abundantly present in lung cancer cells. Clinical information was available on 21 patients with a median age at diagnosis of 63 years (range: 44–79 years). Survival information was available for 20 patients. The five-year overall survival for MUC5AC-negative patients was 93% (95% confidence interval = 59–99%), compared to 67% in the MUC5AC-expressing patients (95% confidence interval = 19–90%). MUC5AC is a class of mucins that are secreted and polymerized to form gels in the airways. It is regulated at the transcriptional, posttranscriptional, and epigenetic levels, and posttranslational modifications play an important role in mucin binding and the clearance of microbes and pollutants [7]. It is overexpressed in lung cancer cells, interacts with integrin β4 to initiate FAK phosphorylation, and mediates the invasion and migration of lung cancer cells through chitosan separation of tumor stem-like cells [8][9]. TP53 mutation and integrin β4 overexpression co-occur in many aggressive malignancies, and lung squamous cell carcinoma has a higher TP53 mutation frequency; the overexpression of integrin β4 can also lead to venous infiltration and a reduction in overall survival in patients with non-small cell lung cancer [10].
2.2. Breast Cancer
The role of integrin β4 in breast cancer progression has been well established [11]. Studies showed that integrin β4 participates in the growth and branching of the breast and plays a key role in the development of the breast [12]. In transitional breast cancer cells, integrin β4 is expressed, but not in non-invasive breast cancer cells or normal breast epithelial cells [13]. The tissue of triple-negative breast cancer (TNBC) is also rich in integrin β4 [14].
Integrin β4 is used to identify more aggressive subtypes of mesenchymal cancer cells [14]. Sung et al. found that in a co-transplant mouse model, human breast cancer cell line MDA-MB-231 with cancer-associated fibroblasts (CAF) produced a larger tumor mass than ITGB4 knockdown MDA-MB-231 [15].
Integrin β4 can induce an invasive status of breast cancer cells. The acyltransferase DHHC3 transfers palmitate as palmitoyl-CoA to the cysteines of integrin β4, which induces its palmitoylation and then triggers the invasive status of breast cancer cells [16]. Cell viability and motility are enhanced when estrogen induces integrin β4 to promote the phosphorylation of AKT [17]. Integrin β4 physically interacts with the ecto domain of nectin-4, and then promotes angiogenesis in metastatic breast cancer stem cells (mBCSCs) via the Src, PI3K, AKT, and iNOS pathways [18].
2.3. Prostate Cancer
Integrin β4 also plays a vital role in the occurrence and development of prostate cancer. Studies found that integrin β4 promotes prostate tumorigenesis by co-expressing with ErbB2 and c-Met in tumor progenitor cells [19]. A compelling body of evidence indicates that integrin β4 is a key player in prostate tumor development. Some data show a large CpG island present in the integrin β4 promoter in prostate tumor development, which mediates the differential transcription of the integrin β4 gene [20]. A study found that integrin β4 knockdown attenuated both cell migration and invasion but did not affect proliferation in castration-resistant prostate cancer cells [21]. All these results indicate that integrin β4 is closely related to the occurrence and development of prostate cancer.
2.4. Colon Cancer
Various forms of integrin α6β4 can differentially regulate intestinal epithelial cell functions under both normal and pathological conditions [17]. A study showed that two forms of integrin β4 exist in the human intestinal epithelium: a full-length 205 kDa β4A subunit expressed in differentiated enterocytes, and a novel β4A subunit that does not contain the COOH- terminal segment of the cytoplasmic domain (integrin β4A (ctd-)), which is found in undifferentiated crypt cells [22]. The integrin β4 ctd- form was identified in normal proliferative colonic cells but was found to be predominantly absent in colon cancer cells, whereas the level of the wild-type form of integrin β4, which is required for adhesion to laminin, is increased in primary tumors [23]. In addition, focal adhesion kinase (FAK) initiated by integrin β4 was found to be expressed in the nuclei of colon cancer cells, indicating that the activation of integrin β4 downstream molecules is also associated with abnormal cell proliferation [24], and other pathways such as miRNA-21-integrin β4-PDCD4 can affect cell viability [25]. When integrin β4 decreased sialylation by NEU1, it was accompanied by decreased phosphorylation of integrin followed by the attenuation of focal adhesion kinase and the Erk1/2 pathway, which led to the suppression of metastasis [26].
The expression and function of integrin β4 in the tumors listed above are described in Table 1.
Table 1. Expression level and function of integrin β4 in different cancers.
| Lung Cancer | Breast Cancer | Prostate Cancer | Colon Cancer | |
|---|---|---|---|---|
| Expression Level | ① Abundantly present ② Strongly overexpressed |
① Occurs in transitional breast cancer cells ② Not in non-invasive breast cancer cells |
Overexpressed (has a large CpG island) | ① Wild-type form is increased ② Integrin β4A(ctd-) is predominantly absent |
| Function | ① Initiates phosphorylation, invasion, and migration ② Occurs with gene mutation |
① Induces invasive status ② Enhances cell viability and motility ③ Promotes angiogenesis |
① Induces tumorigenesis ② Induces cell migration and invasion ③ Does not affect cell proliferation |
① Induces abnormal cell proliferation ② Affects the cell viability ③ Induces metastasis |
3. Integrin-β4-Mediated Cancer Progression
To explore the downstream signaling pathways of integrin β4, cBioPoral was used to detect some genes related to integrin β4. Among them, integrin β4 is strongly correlated with EGFR, which amplifies proliferative and invasive signaling. EGFR-family genes EGF, EGFR, and ErbB3 are also strongly related to integrin β4. In the work map, a number of laminin subunits (LAMA3, LAMB3, and LAMC2) connect with integrin β4. This is consistent with the finding that integrin β4 binds laminin-332 in the extracellular matrix, which indicates that integrin β4 plays a vital role in the stabilization of cells, which means that cell invasion and migration can be limited [10]. Phosphoinositide 3-kinase (PI3K) activity is stimulated by diverse oncogenes and growth factor receptors, and elevated PI3K signaling is considered a hallmark of cancer [27]; its pathway genes PIK3CA and AKT1, and other known signal partners, such as MET and FYN, etc., were found to be strongly related to integrin β4 [10].
3.1. Integrin β4 Is Associated with Tumor Invasion and Migration
Overexpression of integrin α6β4 is related to aggressive tumor behavior and poor prognosis [28]. Six hours after a single layer of epithelium was scratched, a burst of integrin β4 expression was observed, especially in cells at the edge of the wound, accompanied by an increase in chemical kinetic migration [29].
In the process of cancer progression, integrin α6β4 is released from hemidesmosomes, responds to the wounded epithelium or cancer progression, and is located in the F-actin protrusions [30][31][32][33][34]. It binds to the actin cytoskeleton, where it activates RhoA, mediating membrane ruffling, lamellae formation, and generating traction, thereby causing cell invasion and metastasis [35]. Various interactions between the extracellular domain of integrin β4 and the matrix play an important role in tumor invasion and migration.
Integrin β4 can promote the invasiveness of certain malignant tumors, such as squamous cell carcinoma, breast cancer, and gastric cancer, through the MAPK and NF-κB activation pathways [34][36][37][38]. Li et al. used bioinformatics analysis and found approximately 70 pathways significantly dysregulated when integrin β4 expression was high. The MAPK pathway and propanoate metabolism were found to be located in the network center of these pathways [39]. Integrin β4 does not trigger any pathway, but it can bind to different proteins, including human leukocyte antigen-1(HLA-1) [40], epidermal growth factor receptor EGFR [5], extracellular matrix protein ECM1 [41], etc. Human leukocyte antigen-1 (HLA-1) binds to the integrin β4 subunit and then mediates the activation downstream. When its extracellular domain is stimulated, phosphorylation of Src kinase is induced and focal adhesion kinase (FAK) starts, then activating PI3K/AKt and Erk. These enzymes can mediate the occurrence of tumor cell migration [40]. Integrin β4 EGFR/Src signaling mediates the tyrosine phosphorylation of integrin β4, and then recruits FAK to integrin β4, leading to FAK activation and signal transduction, thereby activating the AKT signaling pathway and promoting tumor invasion [5][42][43]. The integrin β4/FAK/SOX2/HIF-1α signaling pathway also regulates the metastasis of tumor cells [41]; integrin β4 overexpression triggers increases in the expression of matrix metalloproteinase-2 (MMP-2) and MMP-9 through the ERK1/2 pathway, the degradation of the extracellular matrix, and the invasion ability of tumor cells [44][45] (Figure 1).
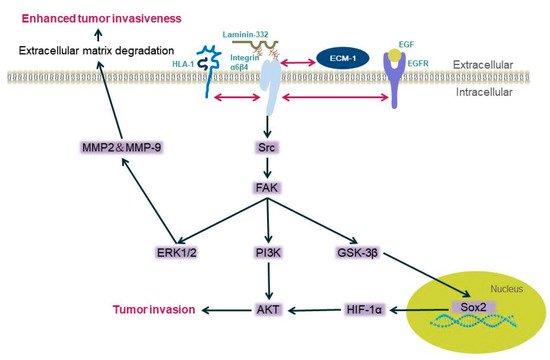
Figure 1. Diagram of the mechanism of integrin β4 mediating tumor cell invasion. Integrin α6β4 binds with laminin-332, and when the pathways are activated, the binding between laminin and α6β4 is not stable enough, and cells are more likely to be invasive (the double-headed arrow in the figure represents the interaction between the two proteins).
The extracellular domain of integrin β4 has potential glycosylation sites. A report showed that N-glycan deletion on integrin β4 impaired integrin-β4-dependent cancer cell migration, invasion, and growth in vitro and diminished tumorigenesis in vivo, which means that N-glycosylation is important in cancer progression. It activates the intracellular PI3K pathway to promote cell invasion and migration [6] and Ca2+/PKC; the Sp1/CaM and Akt/GSK-3β/Snail1 signaling pathways are also activated when glucosamine triggers integrin β4/plectin complex reduction [46].
Certain endocrine substances can also affect tumor cell invasion. For example, the N-terminal truncated subtype of the p63 transcription factor ΔNp63 acts as a transcription factor for integrin β4, and estrogen can activate estrogen receptor α (ERα) transcription, thereby inducing ΔNp63 expression; then, ΔNp63 induces the expression of integrin β4, which leads to the phosphorylation of AKT and enhances cell viability and motility [17]. Changes in integrin β4 in cancer can also mediate the inhibition of miR-29a through a TOR-dependent mechanism, thereby mediating the increase in its downstream signaling molecule SPARC, which is a secreted glycoprotein that results in tumor cells becoming aggressive phenotypes [47].
The invasive and metastatic phenotype promoted by integrin α6β4 signaling is mediated by the phosphorylation of the cytoplasmic tail of the integrin β4 subunit [48]. As mentioned above, integrin β4 is involved in the formation of HDs. HDs promote the stable adhesion of basal epithelial cells to basement membranes (BMs). The key to the mechanical stability of HDs is the interaction between integrin α6β4 and plectin. Integrin α6β4 binds to skeletal connexin and to keratin filaments, stabilizing the adhesion of cells to the epidermal basement membrane [49]. When damage occurs, causing the HD structure to disassemble, the interaction becomes unstable. Growth factors such as epidermal growth factor (EGF) can trigger HD disassembly and induce the phosphorylation of integrin β4 intracellular domains; serine phosphorylation seems to be the main mechanism regulating HD instability [5][43]. Usually, tyrosine kinase mediates the phosphorylation process [40][50]. Disruption of the α6β4–plectin interaction through phosphorylation of the β4 subunit results in a reduction in the adhesive strength of keratinocytes to laminin-332 and the dissolution of HDs, resulting in enhanced cell motility [49].
3.2. Integrin β4 Is Associated with Tumor Cell Proliferation
A study showed that the proliferation of tumor cells is promoted at a relatively low concentration when integrin β4 interacts with netrins, which are secretory molecules of laminin [51]. For example, in glioblastoma, netrin-4 expression was downregulated, thus reducing its inhibitory effect on cell proliferation. In parallel, the expression of integrin β4 is upregulated, which sensitizes the cells to low concentrations of netrin-4 for maintaining cell proliferation [52]. Triple-negative breast cancer cells overexpressing integrin β4 provide integrin β4 to cancer-related fibroblasts (CAF) through exosomes, which induces the phosphorylation of c-JUN or AMPK in CAF. As a result, the proliferation of breast cancer cells is enhanced [15].
3.3. Integrin β4 Is Related to Epithelial–Mesenchymal Transition
Epithelial–mesenchymal transition (EMT) is an important change in cell phenotype. It frees epithelial cells from the structural constraints imposed by the tissue structure. Its core elements include the dislocation of desmosomes and a reduction in intercellular adhesion. More importantly, after EMT, cells show an increase in motor potential. Therefore, EMT is a fundamental process that underlies development and cancer [53]. Studies showed that EMT is closely related to the invasion and metastasis of pancreatic cancer, colon cancer, breast cancer, and other tumors [54][55].
TGF-β1 is an important molecule in the initiation of EMT [56]. It is now well accepted that the loss of E-cadherin, EMT marker α-SMA, and connective tissue growth factor (CTGF) expression are key events in the EMT process. Moreover, it was reported that by stimulating human renal proximal tubular epithelial (HK-2) cells with TGF-β1, the expression of E-cadherin was downregulated and the expression of α-SMA and CTGF were upregulated in a dose-dependent manner. Additionally, stimulating TGF-β1 can increase the expression of integrin β4 [57]. It can control the expression of the integrin β4 subunit and can activate ErbB2- and FAK-dependent clusters through a pathway triggered by epithelial growth factor-dependent phosphorylation, thereby inducing crosstalk between integrin β4 and growth factor receptors [56][57][58][59][60][61]. Takaoka revealed that the expression of integrin β4 is closely related to the methylation of β4 promoter [62], which contains CpG islands, and it can be regulated dynamically during the EMT of mammary gland cells triggered by TGF-β [63].
Moreover, enhanced ECM1 expression facilitates gene expression levels associated with EMT. ECM1 directly interacts with integrin β4 and activates the ITGB4/FAK/SOX2/HIF-1α signal pathway mentioned above [41].
Additionally, a zinc-finger transcription factor, containing KRAB and SCAN domains, ZKSCAN3, directly binds to the integrin β4 promoter and enhances its expression; then, integrin β4 triggers FAK to activate the AKT signaling pathway to promote EMT progression in hepatocellular carcinoma [64].
3.4. Integrin β4 Is Associated with Angiogenesis
Tumor angiogenesis is an important condition for tumorigenesis and is crucial for tumor growth [65][66]. Integrin β4 can induce angiogenesis through two different pathways: NF-κB and phosphorylated Erk nuclear translocation, and the Src, PI3K, AKT, and iNOs pathways [67]. For example, if nectin-4 is expressed on the surface of metastatic breast cancer cells, its extracellular domain falls off and is released into the microenvironment. It combines with integrin β4 expressed on the surface of human umbilical vein endothelial cells to activate Src, PI3K, AKT, and iNOs. This induces angiogenesis [18].
Dysfunction of the vascular endothelial cell (EC) barrier is regarded as a key feature of tumor angiogenesis, and lipid rafts receptor c-Met is reported to mediate increases in EC barrier integrity. According to Yulia Ephstein et al., integrin β4 is an essential participant in this process [68]. There are other studies showing that integrin α6β4 induces endothelial cell migration and invasion by promoting the nuclear translocation of P-ERK and NF-κB, which promotes the branching of medium- and small-sized vessels from β4+ to β4- microvessels. This indicates that integrin α6β4 signaling promotes the onset of the invasive phase of pathological angiogenesis [67].
References
- Takada, Y.; Ye, X.; Simon, S. The integrins. Genome Biol. 2007, 8, 215.
- Chi, Y.X.; Xiang, Y.; Qin, X.Q. The research progress of integrin beta4. Sheng Li Xue Bao 2018, 70, 504–510.
- Kariya, Y.; Gu, J. Roles of Integrin alpha6beta4 Glycosylation in Cancer. Cancers 2017, 9, 79.
- Miyazaki, K. Laminin-5 (laminin-332): Unique biological activity and role in tumor growth and invasion. Cancer Sci. 2006, 97, 91–98.
- Margadant, C.; Frijns, E.; Wilhelmsen, K.; Sonnenberg, A. Regulation of hemidesmosome disassembly by growth factor receptors. Curr. Opin. Cell Biol. 2008, 20, 589–596.
- Kariya, Y.; Oyama, M.; Hashimoto, Y.; Gu, J.; Kariya, Y. beta4-Integrin/PI3K Signaling Promotes Tumor Progression through the Galectin-3-N-Glycan Complex. Mol. Cancer Res. 2018, 16, 1024–1034.
- Ma, J.; Rubin, B.K.; Voynow, J.A. Mucins, Mucus, and Goblet Cells. Chest 2018, 154, 169–176.
- Yen, C.H.; Li, S.T.; Cheng, N.C.; Ji, Y.R.; Wang, J.H.; Young, T.H. Label-free platform on pH-responsive chitosan: Adhesive heterogeneity for cancer stem-like cell isolation from A549 cells via integrin beta4. Carbohydr. Polym. 2020, 239, 116168.
- Lakshmanan, I.; Rachagani, S.; Hauke, R.; Krishn, S.R.; Paknikar, S.; Seshacharyulu, P.; Karmakar, S.; Nimmakayala, R.K.; Kaushik, G.; Johansson, S.L.; et al. MUC5AC interactions with integrin beta4 enhances the migration of lung cancer cells through FAK signaling. Oncogene 2016, 35, 4112–4121.
- Stewart, R.L.; West, D.; Wang, C.; Weiss, H.L.; Gal, T.; Durbin, E.B.; O’Connor, W.; Chen, M.; O’Connor, K.L. Elevated integrin alpha6beta4 expression is associated with venous invasion and decreased overall survival in non-small cell lung cancer. Hum. Pathol. 2016, 54, 174–183.
- Soung, Y.H.; Ford, S.; Yan, C.; Chung, J. The Role of Arrestin Domain-Containing 3 in Regulating Endocytic Recycling and Extracellular Vesicle Sorting of Integrin beta4 in Breast Cancer. Cancers 2018, 10, 507.
- Li, J.; Sun, H.; Feltri, M.L.; Mercurio, A.M. Integrin beta4 regulation of PTHrP underlies its contribution to mammary gland development. Dev. Biol. 2015, 407, 313–320.
- Hoshino, A.; Costa-Silva, B.; Shen, T.-L.; Rodrigues, G.; Hashimoto, A.; Mark, M.T.; Molina, H.; Kohsaka, S.; Di Giannatale, A.; Ceder, S.; et al. Tumour exosome integrins determine organotropic metastasis. Nature 2015, 527, 329–335.
- Bierie, B.; Pierce, S.E.; Kroeger, C.; Stover, D.G.; Pattabiraman, D.R.; Thiru, P.; Donaher, J.L.; Reinhardt, F.; Chaffer, C.L.; Keckesova, Z.; et al. Integrin-beta4 identifies cancer stem cell-enriched populations of partially mesenchymal carcinoma cells. Proc. Natl. Acad. Sci. USA 2017, 114, E2337–E2346.
- Sung, J.S.; Kang, C.W.; Kang, S.; Jang, Y.; Chae, Y.C.; Gil Kim, B.; Cho, N.H. ITGB4-mediated metabolic reprogramming of cancer-associated fibroblasts. Oncogene 2020, 39, 664–676.
- Coleman, D.T.; Soung, Y.H.; Surh, Y.J.; Cardelli, J.A.; Chung, J. Curcumin Prevents Palmitoylation of Integrin beta4 in Breast Cancer Cells. PLoS ONE 2015, 10, e0125399.
- Ho, J.Y.; Chang, F.W.; Huang, F.S.; Liu, J.M.; Liu, Y.P.; Chen, S.P.; Liu, Y.L.; Cheng, K.C.; Yu, C.P.; Hsu, R.J. Estrogen Enhances the Cell Viability and Motility of Breast Cancer Cells through the ERalpha-DeltaNp63-Integrin beta4 Signaling Pathway. PLoS ONE 2016, 11, e0148301.
- Siddharth, S.; Nayak, A.; Das, S.; Nayak, D.; Panda, J.; Wyatt, M.D.; Kundu, C.N. The soluble nectin-4 ecto-domain promotes breast cancer induced angiogenesis via endothelial Integrin-beta4. Int. J. Biochem. Cell Biol. 2018, 102, 151–160.
- Yoshioka, T.; Otero, J.; Chen, Y.; Kim, Y.M.; Koutcher, J.A.; Satagopan, J.; Reuter, V.; Carver, B.; De Stanchina, E.; Enomoto, K.; et al. beta4 Integrin signaling induces expansion of prostate tumor progenitors. J. Clin. Investig. 2013, 123, 682–699.
- Wilkinson, E.J.; Woodworth, A.M.; Parker, M.; Phillips, J.L.; Malley, R.C.; Dickinson, J.L.; Holloway, A.F. Epigenetic regulation of the ITGB4 gene in prostate cancer. Exp. Cell Res. 2020, 392, 112055.
- Kawakami, K.; Fujita, Y.; Kato, T.; Mizutani, K.; Kameyama, K.; Tsumoto, H.; Miura, Y.; Deguchi, T.; Ito, M. Integrin beta4 and vinculin contained in exosomes are potential markers for progression of prostate cancer asso-ciated with taxane-resistance. Int. J. Oncol. 2015, 47, 384–390.
- Basora, N.; Herring-Gillam, F.E.; Boudreau, F.; Perreault, N.; Pageot, L.P.; Simoneau, M.; Bouatrouss, Y.; Beaulieu, J.F. Expression of functionally distinct variants of the beta(4)A integrin subunit in relation to the differentiation state in human intestinal cells. J. Biol. Chem. 1999, 274, 29819–29825.
- Ni, H.; Dydensborg, A.B.; Herring, F.E.; Basora, N.; Gagné, D.; Vachon, P.H.; Beaulieu, J.-F. Upregulation of a functional form of the beta4 integrin subunit in colorectal cancers correlates with c-Myc expression. Oncogene 2005, 24, 6820–6829.
- Tanaka, N.; Ohno, Y.; Nobuhisa, T.; Takaoka, M.; Sirmali, M.; Nakajima, M.; Gunduz, M.; Shirakawa, Y.; Okawa, T.; Naomoto, Y.; et al. Localization of FAK is related with colorectal carcinogenesis. Int. J. Oncol. 2008, 32, 791–796.
- Lü, Y.; Han, B.; Yu, H.; Cui, Z.; Li, Z.; Wang, J. Berberine regulates the microRNA-21-ITGBeta4-PDCD4 axis and inhibits colon cancer viability. Oncol. Lett. 2018, 15, 5971–5976.
- Uemura, T.; Shiozaki, K.; Yamaguchi, K.; Miyazaki, S.; Satomi, S.; Kato, K.; Sakuraba, H.; Miyagi, T. Contribution of sialidase NEU1 to suppression of metastasis of human colon cancer cells through desialylation of integrin beta4. Oncogene 2009, 28, 1218–1229.
- Fruman, D.A.; Chiu, H.; Hopkins, B.D.; Bagrodia, S.; Cantley, L.C.; Abraham, R.T. The PI3K Pathway in Human Disease. Cell 2017, 170, 605–635.
- Draheim, K.M.; Chen, H.B.; Tao, Q.; Moore, N.; Roche, M.; Lyle, S. ARRDC3 suppresses breast cancer progression by negatively regulating integrin beta4. Oncogene 2010, 29, 5032–5047.
- Elaimy, A.L.; Wang, M.; Sheel, A.; Brown, C.W.; Walker, M.R.; Amante, J.J.; Xue, W.; Chan, A.; Baer, C.E.; Goel, H.L.; et al. Real-time imaging of integrin beta4 dynamics using a reporter cell line generated by Crispr/Cas9 genome editing. J. Cell. Sci. 2019, 132, jcs231241.
- Lotz, M.M.; Nusrat, A.; Madara, J.L.; Ezzell, R.; Wewer, U.M.; Mercurio, A.M. Intestinal epithelial restitution. Involvement of specific laminin isoforms and integrin laminin receptors in wound closure of a transformed model epithelium. Am. J. Pathol. 1997, 150, 747–760.
- O’Connor, K.L.; Nguyen, B.K.; Mercurio, A.M. RhoA function in lamellae formation and migration is regulated by the alpha6beta4 integrin and cAMP metabolism. J. Cell Biol. 2000, 148, 253–258.
- Rabinovitz, I.; Mercurio, A.M. The integrin alpha6beta4 functions in carcinoma cell migration on laminin-1 by mediating the formation and stabilization of actin-containing motility structures. J. Cell Biol. 1997, 139, 1873–1884.
- Rabinovitz, I.; Toker, A.; Mercurio, A.M. Protein kinase C-dependent mobilization of the alpha6beta4 integrin from hemidesmo-somes and its association with actin-rich cell protrusions drive the chemotactic migration of carcinoma cells. J. Cell Biol. 1999, 146, 1147–1160.
- Shaw, L.M.; Rabinovitz, I.; Wang, H.H.; Toker, A.; Mercurio, A.M. Activation of phosphoinositide 3-OH kinase by the alpha6beta4 integrin promotes carcinoma invasion. Cell 1997, 91, 949–960.
- O’Connor, K.; Chen, M. Dynamic functions of RhoA in tumor cell migration and invasion. Small GTPases 2013, 4, 141–147.
- Lipscomb, E.A.; Mercurio, A.M. Mobilization and activation of a signaling competent alpha6beta4integrin underlies its contribution to carcinoma progression. Cancer Metastasis Rev. 2005, 24, 413–423.
- Nikolopoulos, S.N.; Blaikie, P.; Yoshioka, T.; Guo, W.; Puri, C.; Tacchetti, C.; Giancotti, F.G. Targeted deletion of the integrin beta4 signaling domain suppresses laminin-5-dependent nuclear entry of mitogen-activated protein kinases and NF-kappaB, causing defects in epidermal growth and migration. Mol. Cell Biol. 2005, 25, 6090–6102.
- Yoon, S.O.; Shin, S.; Lipscomb, E.A. A novel mechanism for integrin-mediated ras activation in breast carcinoma cells: The al-pha6beta4 integrin regulates ErbB2 translation and transactivates epidermal growth factor receptor/ErbB2 signaling. Cancer Res. 2006, 66, 2732–2739.
- Li, J.; Luo, M.; Ou, H.; Liu, X.; Kang, X.; Yin, W. Integrin beta4 promotes invasion and anoikis resistance of papillary thyroid carcinoma and is consistently overexpressed in lymphovascular tumor thrombus. J. Cancer 2019, 10, 6635–6648.
- Zhang, X.; Rozengurt, E.; Reed, E.F. HLA class I molecules partner with integrin beta4 to stimulate endothelial cell proliferation and migration. Sci. Signal 2010, 3, ra85.
- Gan, L.; Meng, J.; Xu, M.; Liu, M.; Qi, Y.; Tan, C.; Wang, Y.; Zhang, P.; Weng, W.; Sheng, W.; et al. Extracellular matrix protein 1 promotes cell metastasis and glucose metabolism by inducing integrin be-ta4/FAK/SOX2/HIF-1alpha signaling pathway in gastric cancer. Oncogene 2018, 37, 744–755.
- Leng, C.; Zhang, Z.-G.; Chen, W.-X.; Luo, H.-P.; Song, J.; Dong, W.; Zhu, X.-R.; Chen, X.-P.; Liang, H.-F.; Zhang, B.-X. An integrin beta4-EGFR unit promotes hepatocellular carcinoma lung metastases by enhancing anchorage independence through activation of FAK–AKT pathway. Cancer Lett. 2016, 376, 188–196.
- Tai, Y.L.; Chu, P.Y.; Lai, I.R.; Wang, M.Y.; Tseng, H.Y.; Guan, J.L.; Liou, J.Y.; Shen, T.L. An EGFR/Src-dependent beta4 integrin/FAK complex contributes to malignancy of breast cancer. Sci. Rep. 2015, 5, 16408.
- Zhu, J.; Wu, Y.-N.; Zhang, W.; Zhang, X.-M.; Ding, X.; Li, H.-Q.; Geng, M.; Xie, Z.-Q.; Wu, H.-M. Monocarboxylate Transporter 4 Facilitates Cell Proliferation and Migration and Is Associated with Poor Prognosis in Oral Squamous Cell Carcinoma Patients. PLoS ONE 2014, 9, e87904.
- Marchese, V.; Juarez, J.; Patel, P.; Hutter-Lobo, D. Density-dependent ERK MAPK expression regulates MMP-9 and influences growth. Mol. Cell. Biochem. 2019, 456, 115–122.
- Jeon, J.H.; Suh, H.N.; Kim, M.O.; Han, H.J. Glucosamine-induced reduction of integrin beta4 and plectin complex stimulates migration and proliferation in mouse embryonic stem cells. Stem. Cells Dev. 2013, 22, 2975–2989.
- Gerson, K.D.; Shearstone, J.R.; Maddula, V.K.; Seligmann, B.E.; Mercurio, A.M. Integrin beta4 regulates SPARC protein to promote invasion. J. Biol. Chem. 2012, 287, 9835–9844.
- Stewart, R.L.; O’Connor, K.L. Clinical significance of the integrin alpha6beta4 in human malignancies. Lab. Investig. 2015, 95, 976–986.
- Te Molder, L.; Sonnenberg, A. PKD2 and RSK1 Regulate Integrin beta4 Phosphorylation at Threonine 1736. PLoS ONE 2015, 10, e0143357.
- Chen, W.; Sammani, S.; Mitra, S.; Ma, S.F.; Garcia, J.G.; Jacobson, J.R. Critical role for integrin-beta4 in the attenuation of murine acute lung injury by simvastatin. Am. J. Physiol. Lung Cell Mol. Physiol. 2012, 303, L279–L285.
- Cirulli, V.; Yebra, M. Netrins: Beyond the brain. Nat. Rev. Mol. Cell Biol. 2007, 8, 296–306.
- Hu, Y.; Ylivinkka, I.; Chen, P.; Li, L.; Hautaniemi, S.; Nyman, T.A.; Keski-Oja, J.; Hyytiäinen, M. Netrin-4 promotes glioblastoma cell proliferation through integrin beta4 signaling. Neoplasia 2012, 14, 219–227.
- Chang, C.; Yang, X.; Pursell, B.; Mercurio, A.M. Id2 complexes with the SNAG domain of Snai1 inhibiting Snai1-mediated repression of integrin beta4. Mol. Cell Biol. 2013, 33, 3795–3804.
- Hugo, H.; Ackland, L.; Blick, T.; Lawrence, M.G.; Clements, J.; Williams, E.D.; Thompson, E.W. Epithelial—mesenchymal and mesenchymal—epithelial transitions in carcinoma progression. J. Cell. Physiol. 2007, 213, 374–383.
- Farahani, E.; Patra, H.K.; Jangamreddy, J.R.; Rashedi, I.; Kawalec, M.; Pariti, R.K.R.; Batakis, P.; Wiechec, E. Cell adhesion molecules and their relation to (cancer) cell stemness. Carcinog. 2014, 35, 747–759.
- Chen, X.F.; Zhang, H.J.; Wang, H.B.; Zhu, J.; Zhou, W.Y.; Zhang, H.; Zhao, M.C.; Su, J.M.; Gao, W.; Zhang, L. Transforming growth factor-beta1 induces epithelial-to-mesenchymal transition in human lung cancer cells via PI3K/Akt and MEK/Erk1/2 signaling pathways. Mol. Biol. Rep. 2012, 39, 3549–3556.
- Wang, Q.; Wang, Y.; Huang, X.; Liang, W.; Xiong, Z.; Xiong, Z. Integrin beta4 in EMT: An implication of renal diseases. Int. J. Clin. Exp. Med. 2015, 8, 6967–6976.
- Liu, H.; Radisky, D.C.; Yang, D.; Xu, R.; Radisky, E.S.; Bissell, M.J.; Bishop, J.M. MYC suppresses cancer metastasis by direct transcriptional silencing of alphav and beta3 integrin subunits. Nat. Cell. Biol. 2012, 14, 567–574.
- Giannelli, G.; Villa, E.; Lahn, M. Targeting transforming growth factor (TGF)-betaRI inhibits activation of beta1 integrin and blocks vascular invasion in hepatocellular carcinoma. Hepatology 2009, 49, 839–850.
- Yeh, Y.Y.; Chiao, C.C.; Kuo, W.Y.; Hsiao, Y.C.; Chen, Y.J.; Wei, Y.Y. TGF-beta1 increases motility and alphavbeta3 integrin up-regulation via PI3K, Akt and NF-kappaB-dependent pathway in human chondrosarcoma cells. Biochem. Pharmacol. 2008, 75, 1292–1301.
- Zhang, Y.J.; Tian, Z.L.; Yu, X.Y.; Zhao, X.X.; Yao, L. Activation of integrin beta1-focal adhesion kinase-RasGTP pathway plays a critical role in TGF beta1-induced po-docyte injury. Cell Signal. 2013, 25, 2769–2779.
- Takaoka, A.S.; Yamada, T.; Gotoh, M.; Kanai, Y.; Imai, K.; Hirohashi, S. Cloning and characterization of the human beta4-integrin gene promoter and enhancers. J. Biol. Chem. 1998, 273, 33848–33855.
- Yang, X.; Pursell, B.; Lu, S.; Chang, T.K.; Mercurio, A.M. Regulation of beta 4-integrin expression by epigenetic modifications in the mammary gland and during the epitheli-al-to-mesenchymal transition. J. Cell Sci. 2009, 122, 2473–2480.
- Li, J.; Hao, N.; Han, J.; Zhang, M.; Li, X.; Yang, N. ZKSCAN3 drives tumor metastasis via integrin beta4/FAK/AKT mediated epithelial-mesenchymal transition in hepato-cellular carcinoma. Cancer Cell. Int. 2020, 20, 216.
- Bergers, G.; Hanahan, D.; Coussens, L.M. Angiogenesis and apoptosis are cellular parameters of neoplastic progression in transgenic mouse models of tumorigenesis. Int. J. Dev. Biol. 1998, 42, 995–1002.
- Kaur, B.; Khwaja, F.W.; Severson, E.A.; Matheny, S.L.; Brat, D.J.; Van Meir, E.G. Hypoxia and the hypoxia-inducible-factor pathway in glioma growth and angiogenesis. Neuro-Oncology 2005, 7, 134–153.
- Nikolopoulos, S.N.; Blaikie, P.; Yoshioka, T.; Guo, W.; Giancotti, F.G. Integrin beta4 signaling promotes tumor angiogenesis. Cancer Cell 2004, 6, 471–483.
- Ephstein, Y.; Singleton, P.A.; Chen, W.; Wang, L.; Salgia, R.; Kanteti, P.; Dudek, S.M.; Garcia, J.G.; Jacobson, J.R. Critical role of S1PR1 and integrin beta4 in HGF/c-Met-mediated increases in vascular integrity. J. Biol. Chem. 2013, 288, 2191–2200.





