
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Yedi Herdiana | + 2861 word(s) | 2861 | 2021-08-31 10:53:16 | | | |
| 2 | Vivi Li | Meta information modification | 2861 | 2021-09-03 09:41:10 | | | | |
| 3 | Conner Chen | Meta information modification | 2861 | 2021-09-22 03:15:42 | | |
Video Upload Options
α-Mangostin (AMG) is a potent anticancer xanthone that was discovered in mangosteen (Garcinia mangostana Linn.). AMG possesses the highest opportunity for chemopreventive and chemotherapeutic therapy. AMG inhibits every step in the process of carcinogenesis. AMG suppressed multiple breast cancer (BC) cell proliferation and apoptosis by decreasing the creation of cancerous compounds. Accumulating BC abnormalities and their associated molecular signaling pathways promotes novel treatment strategies. Chemotherapy is a commonly used treatment; due to the possibility of unpleasant side effects and multidrug resistance, there has been substantial progress in searching for alternative solutions, including the use of plant-derived natural chemicals. Due to the limitations of conventional cancer therapy, nanotechnology provides hope for effective and efficient cancer diagnosis and treatment. Nanotechnology enables the delivery of nanoparticles and increased solubility of drugs and drug targeting, resulting in increased cytotoxicity and cell death during BC treatment.
1. Introduction
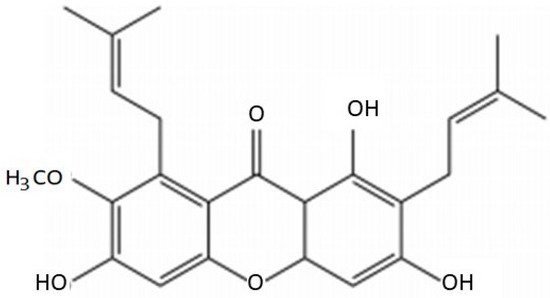
2. Chemical Characteristics of AMG
3. The Activity of AMG in Breast Cancer
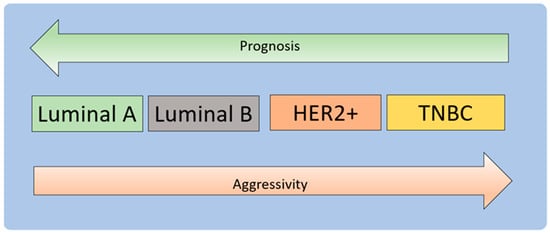
3.1. Molecular Development of Breast Cancer
3.2. Mechanisms of Molecules Drug Resistance (MDR)
3.3. Cytotoxicity
3.4. Mechanisms of Cell Death
4. Nanotechnology of AMG in Breast Cancer
References
- Ziai, J.; Gilbert, H.N.; Foreman, O.; Eastham-Anderson, J.; Chu, F.; Huseni, M.; Kim, J.M. CD8+ T cell infiltration in breast and colon cancer: A histologic and statistical analysis. PLoS ONE 2018, 13, e0190158.
- Van Oost, V. Study of alpha mangostin as a chemoprotective agent for breast cancer via activation of the p53 pathway. Elaia 2018, 2, 16.
- Shan, T.; Ma, Q.; Guo, K.; Liu, J.; Li, W.; Wang, F.; Wu, E. Xanthones from Mangosteen Extracts as Natural Chemopreventive Agents: Potential Anticancer Drugs. Curr. Mol. Med. 2011, 11, 666–677.
- Baktiar Laskar, Y.; Meitei Lourembam, R.; Behari Mazumder, P. Herbal Remedies for Breast Cancer Prevention and Treatment. Med. Plants - Use Prev. Treat. Dis. 2020, 1–22.
- Ullah, I.; Khalil, A.T.; Ali, M.; Iqbal, J.; Ali, W.; Alarifi, S.; Shinwari, Z.K. Green-Synthesized Silver Nanoparticles Induced Apoptotic Cell Death in MCF-7 Breast Cancer Cells by Generating Reactive Oxygen Species and Activating Caspase 3 and 9 Enzyme Activities. Oxid. Med. Cell. Longev. 2020, 2020.
- Park, I.S.; Cho, J.H.; Han, Y.; Lee, K.W.; Song, Y.S. Targeting Cancer Stem Cells with Phytoceuticals for Cancer Therapy; Elsevier Inc.: Amsterdam, The Netherlands, 2020; ISBN 9780128161517.
- Bonafè, F.; Pazzini, C.; Marchionni, S.; Guarnieri, C.; Muscari, C. Complete Disaggregation of MCF-7-derived Breast Tumour Spheroids with Very Low Concentrations of α -Mangostin Loaded in CD44 Thioaptamer-tagged Nanoparticles. Int. J. Med. Sci. 2019, 16, 33–41.
- Zhang, K.J.; Gu, Q.L.; Yang, K.; Ming, X.J.; Wang, J.X. Anticarcinogenic Effects of α -Mangostin: A Review. Planta Med. 2017, 83, 188–202.
- Scolamiero, G.; Pazzini, C.; Bonafè, F.; Guarnieri, C.; Muscari, C. Effects of α-mangostin on viability, growth and cohesion of multicellular spheroids derived from human breast cancer cell lines. Int. J. Med. Sci. 2018, 15, 23–30.
- Chen, G.; Li, Y.; Wang, W.; Deng, L. Bioactivity and pharmacological properties of α-mangostin from the mangosteen fruit: A review. Expert Opin. Ther. Pat. 2018, 28, 415–427.
- Minute, L.; Teijeira, A.; Sanchez-Paulete, A.R.; Ochoa, M.C.; Alvarez, M.; Otano, I.; Etxeberrria, I.; Bolaños, E.; Azpilikueta, A.; Garasa, S.; et al. Cellular cytotoxicity is a form of immunogenic cell death. J. Immunother. Cancer 2020, 8.
- Escobar-Sánchez, M.L.; Sandoval-Ramírez, J.; Sánchez-Sánchez, L. Steroidal Saponins and Cell Death in Cancer Chapter. In Cell Death—Autophagy, Apoptosis and Necrosis; INTECH: London, UK, 2015; Volume 32, pp. 331–352.
- Ahmad, J.; Wahab, R.; Siddiqui, M.A.; Saquib, Q.; Al-Khedhairy, A.A. Cytotoxicity and cell death induced by engineered nanostructures (quantum dots and nanoparticles) in human cell lines. J. Biol. Inorg. Chem. 2020, 25, 325–338.
- Yang, Y.; Du, X.; Wang, Q.; Liu, J.; Zhang, E.; Sai, L.; Peng, C.; Lavin, M.F.; Yeo, A.J.; Yang, X.; et al. Mechanism of cell death induced by silica nanoparticles in hepatocyte cells is by apoptosis. Int. J. Mol. Med. 2019, 44, 903–912.
- Ke, X.; Shen, L. Molecular targeted therapy of cancer: The progress and future prospect. Front. Lab. Med. 2017, 1, 69–75.
- Khan, I.; Saeed, K.; Khan, I. Nanoparticles: Properties, applications and toxicities. Arab. J. Chem. 2019, 12, 908–931.
- Muchtaridi, M.; Wijaya, C.A. Anticancer potential of α-mangostin. Asian J. Pharm. Clin. Res. 2017, 10, 440–445.
- Klein-Júnior, L.C.; Campos, A.; Niero, R.; Corrêa, R.; Vander Heyden, Y.; Filho, V.C. Xanthones and Cancer: From Natural Sources to Mechanisms of Action. Chem. Biodivers. 2020, 17.
- Ryu, H.W.; Curtis-Long, M.J.; Jung, S.; Jin, Y.M.; Cho, J.K.; Ryu, Y.B.; Lee, W.S.; Park, K.H. Xanthones with neuraminidase inhibitory activity from the seedcases of Garcinia mangostana. Bioorganic Med. Chem. 2010, 18, 6258–6264.
- Asasutjarit, R.; Meesomboon, T.; Adulheem, P.; Kittiwisut, S.; Sookdee, P.; Samosornsuk, W.; Fuongfuchat, A. Physicochemical properties of alpha-mangostin loaded nanomeulsions prepared by ultrasonication technique. Heliyon 2019, 5, e02465.
- Pérez-Soto, E.; Estanislao-Gómez, C.C.; Pérez-Ishiwara, D.G.; Ramirez-Celis, C.; Gómez-García, M.d.C. Cytotoxic Effect and Mechanisms from Some Plant-Derived Compounds in Breast Cancer. In Cytotoxicity—Definition, Identification, and Cytotoxic Compounds; IntechOpen: London, UK, 2019; pp. 1–20.
- Demïrel, M.A.; Süntar, İ. The Role of Secondary Metabolites on Gynecologic Cancer Therapy: Some Pathways and Mechanisms. Turkish J. Pharm. Sci. 2017, 14, 324–334.
- Shin, S.A.; Moon, S.Y.; Kim, W.Y.; Paek, S.M.; Park, H.H.; Lee, C.S. Structure-Based Classification and Anti-Cancer Effects of Plant Metabolites. Int. J. Mol. Sci. 2018, 19, 2651.
- Ramakrishna, W.; Kumari, A.; Rahman, N.; Mandave, P. Anticancer Activities of Plant Secondary Metabolites: Rice Callus Suspension Culture as a New Paradigm. Rice Sci. 2021, 28, 13–30.
- Beutler, J.A. Natural Products as a Foundation for Drug Discovery. Curr. Protoc. Pharmacol. 2019, 86, e67.
- Lahlou, M. The Success of Natural Products in Drug Discovery. Pharmacol Pharm. 2013, 04, 17–31.
- Feng, Y.; Spezia, M.; Huang, S.; Yuan, C.; Zeng, Z.; Zhang, L.; Ji, X.; Liu, W.; Huang, B.; Luo, W.; et al. Breast cancer development and progression: Risk factors, cancer stem cells, signaling pathways, genomics, and molecular pathogenesis. Genes Dis. 2018, 5, 77–106.
- Marzaimi, I.N.; Aizat, W.M. Current Review on Mangosteen Usages in Antiinflammation and Other Related Disorders, 2nd ed.; Elsevier Inc: Amsterdam, The Netherlands, 2019; ISBN 9780128138205.
- Helm, K.; Beyreis, M.; Mayr, C.; Ritter, M.; Jakab, M.; Kiesslich, T.; Plaetzer, K. In Vitro Cell Death Discrimination and Screening Method by Simple and Cost-Effective Viability Analysis. Cell. Physiol. Biochem. 2017, 41, 1011–1019.
- Lang, G.T.; Jiang, Y.Z.; Shi, J.X.; Yang, F.; Li, X.G.; Pei, Y.C.; Zhang, C.H.; Ma, D.; Xiao, Y.; Hu, P.C.; et al. Characterization of the genomic landscape and actionable mutations in Chinese breast cancers by clinical sequencing. Nat. Commun. 2020, 11, 1–13.
- Zubair, M.; Wang, S.; Ali, N. Advanced Approaches to Breast Cancer Classification and Diagnosis. Front. Pharmacol. 2021, 11, 1–24.
- Dieleman, S.; Aarnoutse, R.; Ziemons, J.; Kooreman, L.; Boleij, A.; Smidt, M. Exploring the Potential of Breast Microbiota as Biomarker for Breast Cancer and Therapeutic Response. Am. J. Pathol. 2021, 191, 968–982.
- Szymiczek, A.; Lone, A.; Akbari, M.R. Molecular intrinsic versus clinical subtyping in breast cancer: A comprehensive review. Clin. Genet. 2021, 99, 613–637.
- Amirkhani Namagerdi, A.; d’Angelo, D.; Ciani, F.; Iannuzzi, C.A.; Napolitano, F.; Avallone, L.; De Laurentiis, M.; Giordano, A. Triple-Negative Breast Cancer Comparison with Canine Mammary Tumors from Light Microscopy to Molecular Pathology. Front. Oncol. 2020, 10, 1–15.
- Dai, X.; Li, T.; Bai, Z.; Yang, Y.; Liu, X.; Zhan, J.; Shi, B. Breast cancer intrinsic subtype classification, clinical use and future trends. Am. J. Cancer Res. 2015, 5, 2929–2943.
- Zhang, M.H.; Man, H.T.; Zhao, X.D.; Dong, N.; Ma, S.L. Estrogen receptor-positive breast cancer molecular signatures and therapeutic potentials (Review). Biomed. Reports 2014, 2, 41–52.
- Matutino, A.; Joy, A.A.; Brezden-Masley, C.; Chia, S.; Verma, S. Hormone receptor-positive, HER2-negative metastatic breast cancer: Redrawing the lines. Curr. Oncol. 2018, 25, S131–S141.
- Sadeghi, F.; Asgari, M.; Matloubi, M.; Ranjbar, M.; Karkhaneh Yousefi, N.; Azari, T.; Zaki-Dizaji, M. Molecular contribution of BRCA1 and BRCA2 to genome instability in breast cancer patients: Review of radiosensitivity assays. Biol. Proced. Online 2020, 22, 1–28.
- Gorodetska, I.; Kozeretska, I.; Dubrovska, A. BRCA genes: The role in genome stability, cancer stemness and therapy resistance. J. Cancer 2019, 10, 2109–2127.
- Hedau, S.; Batra, M.; Singh, U.R.; Bharti, A.C.; Ray, A.; Das, B.C. Expression of BRCA1 and BRCA2 proteins and their correlation with clinical staging in breast cancer. J. Cancer Res. Ther. 2015, 11, 158–163.
- Blandino, G.; Di Agostino, S. New therapeutic strategies to treat human cancers expressing mutant p53 proteins. J. Exp. Clin. Cancer Res. 2018, 37, 1–13.
- Zilfou, J.T.; Lowe, S.W. Tumor suppressive functions of p53. Cold Spring Harb. Perspect. Biol. 2009, 1, 1–12.
- Tamaki, T.; Naomoto, Y.; Kimura, S.; Kawashima, R.; Shirakawa, Y.; Shigemitsu, K.; Yamatsuji, T.; Haisa, M.; Gunduz, M.; Tanaka, N. Apoptosis in normal tissues induced by anti-cancer drugs. J. Int. Med. Res. 2003, 31, 6–16.
- Wang, Z.; Sun, Y. Targeting p53 for novel anticancer therapy. Transl. Oncol. 2010, 3, 1–12.
- Chang, P.Y.; Peng, S.F.; Lee, C.Y.; Lu, C.C.; Tsai, S.C.; Shieh, T.M.; Wu, T.S.; Tu, M.G.; Chen, M.Y.; Yang, J.S. Curcumin-loaded nanoparticles induce apoptotic cell death through regulation of the function of MDR1 and reactive oxygen species in cisplatin-resistant CAR human oral cancer cells. Int. J. Oncol. 2013, 43, 1141–1150.
- Macus, T.K.; Liu, Z.; Wei, Y.; Lin-Lee, Y.C.; Tatebe, S.; Mills, G.B.; Unate, H. Induction of human MDR1 gene expression by 2-acetylaminofluorene is mediated by effectors of the phosphoinositide 3-kinase pathway that activate NF-κB signaling. Oncogene 2002, 21, 1945–1954.
- Robinson, K.; Tiriveedhi, V. Perplexing Role of P-Glycoprotein in Tumor Microenvironment. Front. Oncol. 2020, 10, 1–10.
- Zhang, E.; Xing, R.; Liu, S.; Qin, Y.; Li, K.; Li, P. Advances in chitosan-based nanoparticles for oncotherapy. Carbohydr. Polym. 2019, 222, 115004.
- Wu, C.P.; Hsiao, S.H.; Murakami, M.; Lu, Y.J.; Li, Y.Q.; Huang, Y.H.; Hung, T.H.; Ambudkar, S.V.; Wu, Y.S. Alpha-Mangostin Reverses Multidrug Resistance by Attenuating the Function of the Multidrug Resistance-Linked ABCG2 Transporter. Mol. Pharm. 2017, 14, 2805–2814.
- Stojak, M.; Mazur, L.; Opydo-Chanek, M.; Ukawska, M.; Oszczapowicz, I. In vitro induction of apoptosis and necrosis by new derivatives of daunorubicin. Anticancer Res. 2013, 33, 4439–4444.
- Kasko, A.M. Polymeric Biomaterials with Engineered Degradation. J. Polym. Sci. Part A 2013, 3531–3566.
- Shahneh, F.Z.; Valiyari, S.; Azadmehr, A.; Hajiaghaee, R.; Yaripour, S.; Bandehagh, A.; Baradaran, B. Inhibition of growth and induction of apoptosis in fibrosarcoma cell lines by echinophora platyloba DC: In vitro analysis. Adv. Pharmacol. Sci. 2013, 2013.
- Anilkumar, T.V.; Sarraf, C.E.; Hunt, T.; Alison, M.R. The nature of cytotoxic drug _ induced cell death in murine intestinal crypts. Br. J. Cancer 1992, 65, 552–558.
- Moghadam, S.A.; Rezania, V.; Tuszynski, J.A. Cell death and survival due to cytotoxic exposure modelled as a two-state Ising system. R. Soc. Open Sci. 2020, 7.
- Orrenius, S.; Nicotera, P.; Zhivotovsky, B. Cell death mechanisms and their implications in toxicology. Toxicol. Sci. 2011, 119, 3–19.
- Chiangjong, W.; Chutipongtanate, S.; Hongeng, S. Anticancer peptide: Physicochemical property, functional aspect and trend in clinical application (Review). Int. J. Oncol. 2020, 57, 678–696.
- Shaw, P.; Kumar, N.; Hammerschmid, D.; Privat-Maldonado, A.; Dewilde, S.; Bogaerts, A. Synergistic Effects of Melittin and Plasma Treatment: A Promising Approach for Cancer Therapy. Cancers 2019, 11, 1109.
- Istifli, E.S.; Hüsunet, M.T.; Ila, H.B. Cell Division, Cytotoxicity, and the Assays Used in the Detection of Cytotoxicity. In Cytotoxicity—Definition, Identification, and Cytotoxic Compounds; Istifli, E.S., Ila, H.B., Eds.; IntechOpen: London, UK, 2019; pp. 1–120. ISBN 9781789847543.
- Dasari, A.; Choi, J.-S.; Berdis, A.J. Chapter 7—Chemotherapeutic Intervention by Inhibiting DNA Polymerases A2—Kelley, Mark R., 2nd ed.; Elsevier Inc.: Amsterdam, The Netherlands, 2016; ISBN 978-0-12-803582-5.
- Nappi, A.J.; Ottaviani, E. Cytotoxicity and cytotoxic molecules in invertebrates. BioEssays 2000, 22, 469–480.
- Mohammadinejad, R.; Moosavi, M.A.; Tavakol, S.; Vardar, D.Ö.; Hosseini, A.; Rahmati, M.; Dini, L.; Hussain, S.; Mandegary, A.; Klionsky, D.J. Necrotic, apoptotic and autophagic cell fates triggered by nanoparticles. Autophagy 2019, 15, 4–33.
- Lemasters, J.J. Molecular mechanisms of cell death. Mol. Pathol. Mol. Basis Hum. Dis. 2018, 1–24.
- Cogo, F.; Poreba, M.; Rut, W.; Groborz, K.; Smyth, P.; Johnston, M.C.; Williams, R.; Longley, D.B.; Burden, R.E.; Salvesen, G.S.; et al. Development of an advanced nanoformulation for the intracellular delivery of a caspase-3 selective activity-based probe. Nanoscale 2019, 11, 742–751.
- Elmore, S. Apoptosis: A Review of Programmed Cell Death. Toxicol. Pathol. 2007, 35, 495–516.
- Jin, Z.; El-Deiry, W.S. Overview of cell death signaling pathways. Cancer Biol. Ther. 2005, 4, 147–171.
- Redza-Dutordoir, M.; Averill-Bates, D.A. Activation of apoptosis signalling pathways by reactive oxygen species. Biochim. Biophys. Acta-Mol. Cell Res. 2016, 1863, 2977–2992.
- Jung, S.; Jeong, H.; Yu, S.W. Autophagy as a decisive process for cell death. Exp. Mol. Med. 2020, 52, 921–930.
- Coleman, W.B.; Tsongalis, G.J. Moleculor Pathology; Elsevier Inc.: Amsterdam, Netherlands, 2017.
- Chahal, R.; Nanda, A.; Akkol, E.K.; Sobarzo-sánchez, E.; Arya, A.; Kaushik, D.; Dutt, R.; Bhardwaj, R.; Rahman, M.H.; Mittal, V. Ageratum conyzoides L. And its secondary metabolites in the management of different fungal pathogens. Molecules 2021, 26, 2933.
- Larayetan, R.; Ololade, Z.S.; Ogunmola, O.O.; Ladokun, A. Phytochemical Constituents, Antioxidant, Cytotoxicity, Antimicrobial, Antitrypanosomal, and Antimalarial Potentials of the Crude Extracts of Callistemon citrinus. Evidence-based Complement. Altern. Med. 2019, 2019.
- Hu, X.; Wu, T.; Bao, Y.; Zhang, Z. Nanotechnology based therapeutic modality to boost anti-tumor immunity and collapse tumor defense. J. Control. Release 2017, 256, 26–45.
- Iqbal, J.; Abbasi, B.A.; Batool, R.; Mahmood, T.; Ali, B.; Khalil, A.T.; Kanwal, S.; Shah, S.A.; Ahmad, R. Potential phytocompounds for developing breast cancer therapeutics: Nature’s healing touch. Eur. J. Pharmacol. 2018, 827, 125–148.
- Ivanova, N.; Gugleva, V.; Dobreva, M.; Pehlivanov, I.; Stefanov, S.; Andonova, V. Novel Applications of Nanoparticles in Nature and Building Materials. In Novel Nanomaterials; IntechOpen: London, UK, 2016; Volume i, p. 13.
- Baig, N.; Kammakakam, I.; Falath, W.; Kammakakam, I. Nanomaterials: A review of synthesis methods, properties, recent progress, and challenges. Mater. Adv. 2021, 2, 1821–1871.
- Chenthamara, D.; Subramaniam, S.; Ramakrishnan, S.G.; Krishnaswamy, S.; Essa, M.M.; Lin, F.; Qoronfleh, M.W. Therapeutic efficacy of nanoparticles and routes of administration. Biomater. Res. 2019, 23, 1–29.
- Grewal, I.K.; Singh, S.; Arora, S.; Sharma, N. Polymeric nanoparticles for breast cancer therapy: A comprehensive review. Biointerface Res. Appl. Chem. 2021, 11, 11151–11171.
- Bashir, S.; Aamir, M.; Sarfaraz, R.M.; Hussain, Z.; Sarwer, M.U.; Mahmood, A.; Akram, M.R.; Qaisar, M.N. Fabrication, characterization and in vitro release kinetics of tofacitinib-encapsulated polymeric nanoparticles: A promising implication in the treatment of rheumatoid arthritis. Int. J. Polym. Mater. Polym. Biomater. 2021, 70, 449–458.
- Paliwal, R.; Babu, R.J.; Palakurthi, S. Nanomedicine Scale-up Technologies: Feasibilities and Challenges. Ageing Int. 2014, 15, 1527–1534.
- Avramović, N.; Mandić, B.; Savić-Radojević, A.; Simić, T. Polymeric nanocarriers of drug delivery systems in cancer therapy. Pharmaceutics 2020, 12, 298.
- Bertoni, S.; Passerini, N.; Albertini, B. Nanomaterials for Oral Drug Administration; Elsevier Inc.: Amsterdam, The Netherlands, 2020; ISBN 9780128180389.
- Gagliardi, A.; Giuliano, E.; Venkateswararao, E.; Fresta, M. Biodegradable Polymeric Nanoparticles for Drug Delivery to Solid Tumors. Front. Pharmacol. 2021, 12, 1–24.
- Fathi, M.; Abdolahinia, E.D.; Barar, J.; Omidi, Y. Smart stimuli-responsive biopolymeric nanomedicines for targeted therapy of solid tumors. Nanomedicine 2020, 15, 2171–2200.
- Aisha, A.F.A.; Abdulmajid, A.M.S.; Ismail, Z.; Alrokayan, S.A.; Abu-Salah, K.M. Development of Polymeric Nanoparticles of Garcinia mangostana Xanthones in Eudragit RL100/RS100 for Anti-Colon Cancer Drug Delivery. J. Nanomater. 2015, 2015, 1–12.
- Yu, T.; Huang, X.; Liu, J.; Fu, Q.; Wang, B.; Qian, Z. Polymeric nanoparticles encapsulating ␣ -mangostin inhibit the growth and metastasis in colorectal cancer. Appl. Mater. Today 2019, 16, 351–366.
- Wathoni, N.; Rusdin, A.; Motoyama, K.; Joni, I.M.; Lesmana, R.; Muchtaridi, M. Nanoparticle drug delivery systems for α-mangostin. Nanotechnol. Sci. Appl. 2020, 13, 23–36.
- Sriyanti, I.; Edikresnha, D.; Rahma, A.; Munir, M.M.; Rachmawati, H.; Khairurrijal, K. Mangosteen pericarp extract embedded in electrospun PVP nanofiber mats: Physicochemical properties and release mechanism of α-mangostin. Int. J. Nanomedicine 2018, 13, 4927–4941.
- Wathoni, N.; Meylina, L.; Rusdin, A.; Fouad, A.; Mohammed, A.; Tirtamie, D.; Herdiana, Y.; Motoyama, K.; Panatarani, C.; Joni, I.M.; et al. The Potential Cytotoxic Activity Enhancement of α -Mangostin in Chitosan-Kappa Carrageenan-Loaded Nanoparticle against MCF-7 Cell Line. Polymer 2021, 13, 1–13.
- Pham, D.T.; Saelim, N.; Tiyaboonchai, W. Alpha mangostin loaded crosslinked silk fibroin-based nanoparticles for cancer chemotherapy. Colloids Surf B Biointerfaces 2019, 181, 705–713.
- Dissanayake, S.; Denny, W.A.; Gamage, S.; Sarojini, V. Recent developments in anticancer drug delivery using cell penetrating and tumor targeting peptides. J. Control. Release 2017, 250, 62–76.
- Bazylinska, U.; Saczko, J. Nanoemulsion-templated polylelectrolyte multifunctional nanocapsules for DNA entrapment and bioimaging. Colloids Surf B Biointerfaces 2016, 137, 191–202.
- Mcmahon, K.M.; Foit, L.; Angeloni, N.L.; Giles, F.J.; Gordon, L.I.; Thaxton, C.S. Nanotechnology-Based Precision Tools for the Detection and Treatment of Cancer. Cancer Treat Res 2015, 166, 129–150.
- Sepand, M.R.; Ranjbar, S.; Kempson, I.M.; Akbariani, M.; Muganda, W.C.A.; Müller, M.; Ghahremani, M.H.; Raoufi, M. Targeting non-apoptotic cell death in cancer treatment by nanomaterials: Recent advances and future outlook. Nanomed. Nanotechnol. Biol. Med. 2020, 29, 102243.
- Herdiana, Y.; Wathoni, N.; Shamsuddin, S.; Joni, I.M.; Muchtaridi, M. Chitosan-Based Nanoparticles of Targeted Drug Delivery System in Breast Cancer Treatmen. Polymers 2021, 13, 1717.
- Peng, F.; Setyawati, M.I.; Tee, J.K.; Ding, X.; Wang, J.; Nga, M.E.; Ho, H.K.; Leong, D.T. Nanoparticles promote in vivo breast cancer cell intravasation and extravasation by inducing endothelial leakiness. Nat. Nanotechnol. 2019, 14, 279–286.
- Verma, R.K.; Yu, W.; Shrivastava, A.; Shankar, S.; Srivastava, R.K. α-Mangostin-encapsulated PLGA nanoparticles inhibit pancreatic carcinogenesis by targeting cancer stem cells in human, and transgenic (KrasG12D, and KrasG12D/tp53R270H) mice. Sci. Rep. 2016, 6, 1–13.





