
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Chiara Valori | + 2334 word(s) | 2334 | 2021-08-10 08:45:53 | | | |
| 2 | Lindsay Dong | -23 word(s) | 2311 | 2021-08-16 05:34:49 | | |
Video Upload Options
Non-cell-specific therapeutics or therapeutics designed to tackle aberrant pathways within neurons failed to slow down or halt neurodegeneration. It is therefore time to pursue alternative strategies. In line with this, astrocytes emerge as promising therapeutic targets in various neurodegenerative disorders, an hypothesis supported by their importance to maintain the central nervous system homeostasis in health conditions as well as their fundamental and multifaced role in pathological conditions.
1. Introduction
Among glia, astrocytes emerge as a critical and highly heterogeneous population that perform a wide array of functions, spanning from maintaining water and ion homeostasis to modulating neuronal transmission (reviewed in [1][2]). In addition to these fundamental interactions with neurons, they exchange signals also with other glial cell populations and they intimately interact with the blood capillaries to maintain the blood-brain barrier (BBB, [3]) and the glymphatic system [4]. Considering the importance of astrocyte functions for CNS performance, it is reasonable to postulate that astrocyte dysfunction can be a primary event of a pathogenic cascade ultimately leading to neuronal loss. An example that corroborates this view is the case of Alexander’s disease [5], a lethal leukodystrophy caused by mutations in the glial fibrillary acidic protein (GFAP) gene, which encodes an astrocytic intermediate filament protein. In keeping with this, autoantibodies against the astrocytic water channel protein Aquaporin-4 (AQP4) were shown to cause ~80% of occurrences of neuromyelitis optica, an autoimmune condition with a neurodegenerative component [6]. Finally, recent findings suggest that neurotrophic viruses, such as Zika and West Nile viruses, but also SARS-CoV-2, harm the CNS by interfering with astrocyte functions [7]. This amount of evidence provides a first argument challenging the neuron-centric dogma. The second element of confutation of the neuron-centric hypothesis is represented by the evidence that astrocyte dysfunction can also play a key role in the pathogenesis of several chronic neurodegenerative conditions, including Alzheimer’s (AD, [8]), Parkinson’s (PD, [9]), Huntington’s diseases (HD, [10]), and Amyotrophic Lateral Sclerosis (ALS, [11]). A third indication supporting the importance of glial cells in the diseased CNS is based on the growing awareness that “reactive gliosis” cannot be simply considered a stereotyped passive “reaction” to an insult. Rather, it is a highly articulated process where microglia, peripheral immune cells, and astrocytes sense neuronal distress and exchange signals to mount a tailor-made inflammatory milieu in response to distinct forms of injuries. This phenomenon gives rise to a neuroinflammatory reaction that can have either a neuroprotective or a detrimental impact on neurons, depending on the nature and the persistency of the insult (reviewed in [12]).
2. Astrocyte Replacement Therapy
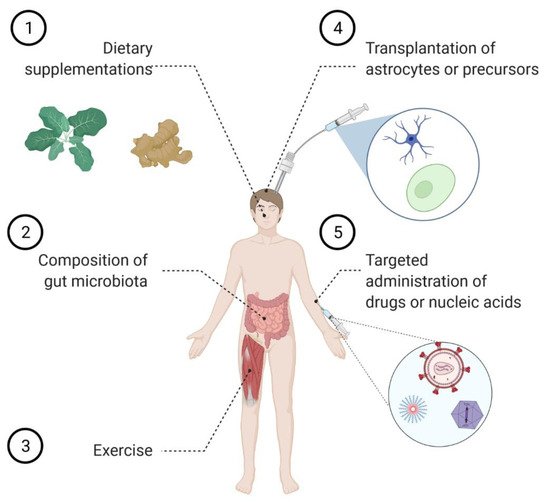
3. Modulating Astrocyte Function by Tuning the Incoming Signaling
By virtue of their strategic position within the CNS, astrocytes are particularly prone to integrate signals arising from different neural cell types as well as from the blood vessels. Because microglia-derived cytokines were reported to drive astrocyte conversion into the neurotoxic A1 phenotype, it was initially hypothesized that blocking this process could be a beneficial therapeutic approach to halt neurodegeneration.
Astrocytes modulate their functions not only by interacting with microglia-derived factors but also in response to signals arising from endothelial cells, which are major constituents of the BBB. For example, endothelial cells were reported to induce Notch signaling in astrocytes, thereby boosting the expression of the astrocyte-specific glutamate transporter EAAT2 (GLT1 in rodents) [22], a protein involved in the maintenance of extracellular glutamate concentrations below excitotoxic levels and a therapeutic target of outstanding relevance for many neurological conditions.
During neuropathology, the BBB can become impaired and lead to soluble factors and inflammatory cell infiltration into the perivascular space. Interestingly, in EAE, the cross-talk between endothelial cells and astrocytes was shown to unleash the expression of tight junction molecules in astrocytes, thus restricting the access to the parenchyma of plasmatic proteins and inflammatory cells, as well as limiting the detrimental phenotype in mice [23][24].
Ground-breaking studies published in the last few years demonstrated that astrocytes are sensitive to factors of various nature, originating also outside the CNS. In particular, circulating levels of the insulin-like growth factor-1 (IGF-1) were reported to regulate the astrocyte expression of several glutamatergic receptors and the production of eicosanoids, which ensure the optimal neurovascular coupling necessary for intense neuronal activity [25].
Finally, astrocytes have been reported to respond to pesticides and toxins. For example, the herbicide Linuron was shown to target the unfolded protein response in the astrocytes, thereby boosting their neurotoxic role in a mouse model of multiple sclerosis [26].
4. Shared Pathways of Astrocyte Dysfunction in Neurodegenerative Diseases
Several molecular mechanisms shape the response of the astrocytes to distinct neurodegenerative disorders, such as for example AD [27], ALS [11], HD [28], and PD [29]. The identification of common pathogenetic signaling cascades is full of implications, as it suggests that therapeutic agents designed to target astrocytes might have a broad spectrum of applications and be an effective add-on therapy. One of the recurring features in multiple pathologies is the loss of the astrocyte-specific glutamate transporter EAAT2, an event that might lead to neurotoxic accumulation of extracellular glutamate (reviewed in [30]). Surprisingly, the antibiotic ceftriaxone was shown to enhance EAAT2 expression [31] and to successfully ameliorate the phenotype of rodent models of different neurodegenerative conditions [32][33][34][35][36][37]. These promising findings led to the prompt activation of clinical trials to evaluate its therapeutic value in ALS (NCT00349622; NCT00718393) and PD dementia (NCT03413384). Regrettably, a Phase III clinical trial failed at demonstrating the efficacy of ceftriaxone in both slowing down disease progression and prolonging survival of ALS patients [38], while the outcome of the PD dementia trial remains to be determined because patients are still being recruited. Another small molecule apt at enhancing EAAT2 expression is LDN/OSU0212320 [39]. Interestingly, the administration of this compound successfully ameliorated the phenotype of rodent models of ALS [39], AD [40], pain [41], migraine [42], and, very recently, stroke, where the beneficial effect was observed in male, but not in female animals [43]. This last observation remarks that not only the development of neurological conditions, but also the response to treatment can be gender-dependent and calls for the design of preclinical and clinical studies taking this option into consideration.
Notably, selective Nrf2 activation in the astrocytes ameliorated the pathological phenotype in animal models of both ALS [44] and PD [45][46], while its genetic ablation worsened the development of EAE in mice [47]. Furthermore, the neuroprotective effect of transplanted astrocytes in a mouse model of PD was reported to rely on their ability to mount an Nrf2-mediated anti-oxidative and pro-survival environment [16]. At variance with this, ALS mice crossed with animals overexpressing Nrf2 in neurons [48] or subjected to neuronal-targeted viral-mediated gene therapy [49] did not exhibit an extended lifespan. Finally, systemic administration of many promising compounds, according to the in vitro studies, had moderate beneficial effects on animal models of ALS [50][51][52], PD [53][54], and AD [55]. Taken together, these discoveries support the view that Nrf2-activating compounds are indeed promising candidates to control several neurodegenerative conditions as long as the therapy is precisely targeted to the astrocytes.
NF-kB is a ubiquitously expressed transcription factor complex that plays a crucial role in the expression of various inflammatory genes. It is widely acknowledged that NF-kB is activated in the astrocytes in the context of several neurodegenerative disorders. However, a point that remains little understood is whether its activation in the astrocytes triggers a neuroprotective or a neurotoxic response. In particular, astrocyte-specific ablation of NF-kB activity was shown to be beneficial in a mouse model of PD [56], as well as in Drosophila models of Spinocerebellar ataxia 3 and AD [57]. Conversely, this approach led to conflicting results in the context of ALS. In murine models of ALS, it was initially noted that preventing NF-kB activation in astrocytes did not ameliorate the pathological phenotype [58][59] or it was even detrimental [60]. In line with these observations, we demonstrated that activating NF-kB in astrocytes can lead to the production of growth factors that are known to support neuronal survival, namely brain-derived neurotrophic factor (BDNF) and GDNF [61]. However, pharmacological treatment with the NF-kB inhibitor Withaferin A ameliorated the phenotype of several models of ALS [62][63][64].
5. Strategies to Target Astrocytes
A particularly flexible formulation to achieve delivery to the CNS is the synthesis and the assembly of nanoparticles (NPs) apt at delivering nucleic acids, proteins, and small molecules [65].
Innovative strategies are necessary to obtain delivery of chemicals specifically to astrocytes in the context of neuroinflammatory and neurodegenerative disorders, ideally upon systemic administration. To achieve these goals, research is expanding in parallel in two directions. On the one hand, NPs can be decorated with different types of peptidic moieties to achieve both the transport across the BBB and the uptake by the astrocytes. On the other hand, different types of chemistry allowed obtaining NPs apt at selectively targeting astrocytes and delivering nucleic acids [66][67][68] or small molecules [69].
5.1. Peptide-Based Delivery
5.2. Viral Delivery
Viruses have co-evolved to selectively invade host cells and take control of their machinery and metabolism to replicate, thus implying that they are natural gene delivery vectors.
This major pitfall was overcome in 2009 when Foust et al. discovered that the adeno-associated virus serotype 9 (AAV9) mainly transduces neurons when administered to neonate mice, while it infects also astrocytes in adult animals [75] and in non-human primates [76]. This promising result prompted additional investigations aiming at increasing the specificity of gene transfer to the astrocytes by either modifying the genome vector and/or by developing engineered capsids to increase the tropism towards astrocytes. In particular, cell-specific gene expression was achieved using astrocyte-specific promoters [77][78][79][80][81][82][83][84][85][86][87].
6. Astrocyte Role in Adult Neurogenesis and as Neuronal Precursors
7. Conclusions
References
- Rossi, D. Astrocyte physiopathology: At the crossroads of intercellular networking, inflammation and cell death. Prog. Neurobiol. 2015, 130, 86–120.
- Verkhratsky, A.; Parpura, V.; Vardjan, N.; Zorec, R. Physiology of Astroglia. Adv. Exp. Med. Biol. 2019, 1175, 45–91.
- Langen, U.H.; Ayloo, S.; Gu, C. Development and Cell Biology of the Blood-Brain Barrier. Annu. Rev. Cell. Dev. Biol. 2019, 35, 591–613.
- Mestre, H.; Mori, Y.; Nedergaard, M. The Brain’s Glymphatic System: Current Controversies. Trends Neurosci. 2020, 43, 458–466.
- Namekawa, M.; Takiyama, Y.; Aoki, Y.; Takayashiki, N.; Sakoe, K.; Shimazaki, H.; Taguchi, T.; Tanaka, Y.; Nishizawa, M.; Saito, K.; et al. Identification of GFAP gene mutation in hereditary adult-onset Alexander’s disease. Ann. Neurol. 2002, 52, 779–785.
- Jarius, S.; Paul, F.; Weinshenker, B.G.; Levy, M.; Kim, H.J.; Wildemann, B. Neuromyelitis optica. Nat. Rev. Dis. Prim. 2020, 6, 85.
- Tavcar, P.; Potokar, M.; Kolenc, M.; Korva, M.; Avsic-Zupanc, T.; Zorec, R.; Jorgacevski, J. Neurotropic Viruses, Astrocytes, and COVID-19. Front. Cell. Neurosci. 2021, 15, 662578.
- Han, X.; Zhang, T.; Liu, H.; Mi, Y.; Gou, X. Astrocyte Senescence and Alzheimer’s Disease: A Review. Front. Aging Neurosci. 2020, 12, 148.
- Miyazaki, I.; Asanuma, M. Neuron-Astrocyte Interactions in Parkinson’s Disease. Cells 2020, 9, 2623.
- Wilton, D.K.; Stevens, B. The contribution of glial cells to Huntington’s disease pathogenesis. Neurobiol. Dis. 2020, 143, 104963.
- Valori, C.F.; Guidotti, G.; Brambilla, L.; Rossi, D. Astrocytes in Motor Neuron Diseases. Adv. Exp. Med. Biol. 2019, 1175, 227–272.
- Sofroniew, M.V. Astrocyte Reactivity: Subtypes, States, and Functions in CNS Innate Immunity. Trends Immunol. 2020, 41, 758–770.
- Pihlaja, R.; Koistinaho, J.; Malm, T.; Sikkila, H.; Vainio, S.; Koistinaho, M. Transplanted astrocytes internalize deposited beta-amyloid peptides in a transgenic mouse model of Alzheimer’s disease. Glia 2008, 56, 154–163.
- Proschel, C.; Stripay, J.L.; Shih, C.H.; Munger, J.C.; Noble, M.D. Delayed transplantation of precursor cell-derived astrocytes provides multiple benefits in a rat model of Parkinsons. EMBO Mol. Med. 2014, 6, 504–518.
- Sun, Y.; Lu, X.J.; Fu, X.; Zhang, Y.; Zhan, Y.; Liu, J.; Zhao, L.; Xia, C.L. Engrafted primary type-2 astrocytes improve the recovery of the nigrostriatal pathway in a rat model of Parkinson’s disease. Mol. Cell. Biochem. 2021, 476, 619–631.
- Serapide, M.F.; L’Episcopo, F.; Tirolo, C.; Testa, N.; Caniglia, S.; Giachino, C.; Marchetti, B. Boosting Antioxidant Self-defenses by Grafting Astrocytes Rejuvenates the Aged Microenvironment and Mitigates Nigrostriatal Toxicity in Parkinsonian Brain via an Nrf2-Driven Wnt/beta-Catenin Prosurvival Axis. Front. Aging Neurosci. 2020, 12, 24.
- Lepore, A.C.; Rauck, B.; Dejea, C.; Pardo, A.C.; Rao, M.S.; Rothstein, J.D.; Maragakis, N.J. Focal transplantation-based astrocyte replacement is neuroprotective in a model of motor neuron disease. Nat. Neurosci. 2008, 11, 1294–1301.
- Kondo, T.; Funayama, M.; Tsukita, K.; Hotta, A.; Yasuda, A.; Nori, S.; Kaneko, S.; Nakamura, M.; Takahashi, R.; Okano, H.; et al. Focal transplantation of human iPSC-derived glial-rich neural progenitors improves lifespan of ALS mice. Stem Cell Rep. 2014, 3, 242–249.
- Izrael, M.; Slutsky, S.G.; Admoni, T.; Cohen, L.; Granit, A.; Hasson, A.; Itskovitz-Eldor, J.; Krush Paker, L.; Kuperstein, G.; Lavon, N.; et al. Safety and efficacy of human embryonic stem cell-derived astrocytes following intrathecal transplantation in SOD1(G93A) and NSG animal models. Stem Cell Res. Ther. 2018, 9, 152.
- Goldberg, N.R.S.; Marsh, S.E.; Ochaba, J.; Shelley, B.C.; Davtyan, H.; Thompson, L.M.; Steffan, J.S.; Svendsen, C.N.; Blurton-Jones, M. Human Neural Progenitor Transplantation Rescues Behavior and Reduces alpha-Synuclein in a Transgenic Model of Dementia with Lewy Bodies. Stem Cells Transl. Med. 2017, 6, 1477–1490.
- Llorente, I.L.; Xie, Y.; Mazzitelli, J.A.; Hatanaka, E.A.; Cinkornpumin, J.; Miller, D.R.; Lin, Y.; Lowry, W.E.; Carmichael, S.T. Patient-derived glial enriched progenitors repair functional deficits due to white matter stroke and vascular dementia in rodents. Sci. Transl. Med. 2021, 13, eaaz6747.
- Lee, M.L.; Martinez-Lozada, Z.; Krizman, E.N.; Robinson, M.B. Brain endothelial cells induce astrocytic expression of the glutamate transporter GLT-1 by a Notch-dependent mechanism. J. Neurochem. 2017, 143, 489–506.
- Horng, S.; Therattil, A.; Moyon, S.; Gordon, A.; Kim, K.; Argaw, A.T.; Hara, Y.; Mariani, J.N.; Sawai, S.; Flodby, P.; et al. Astrocytic tight junctions control inflammatory CNS lesion pathogenesis. J. Clin. Investig. 2017, 127, 3136–3151.
- Mora, P.; Hollier, P.L.; Guimbal, S.; Abelanet, A.; Diop, A.; Cornuault, L.; Couffinhal, T.; Horng, S.; Gadeau, A.P.; Renault, M.A.; et al. Blood-brain barrier genetic disruption leads to protective barrier formation at the Glia Limitans. PLoS Biol. 2020, 18, e3000946.
- Toth, P.; Tarantini, S.; Ashpole, N.M.; Tucsek, Z.; Milne, G.L.; Valcarcel-Ares, N.M.; Menyhart, A.; Farkas, E.; Sonntag, W.E.; Csiszar, A.; et al. IGF-1 deficiency impairs neurovascular coupling in mice: Implications for cerebromicrovascular aging. Aging Cell 2015, 14, 1034–1044.
- Wheeler, M.A.; Jaronen, M.; Covacu, R.; Zandee, S.E.J.; Scalisi, G.; Rothhammer, V.; Tjon, E.C.; Chao, C.C.; Kenison, J.E.; Blain, M.; et al. Environmental Control of Astrocyte Pathogenic Activities in CNS Inflammation. Cell 2019, 176, 581–596.e18.
- Preman, P.; Alfonso-Triguero, M.; Alberdi, E.; Verkhratsky, A.; Arranz, A.M. Astrocytes in Alzheimer’s Disease: Pathological Significance and Molecular Pathways. Cells 2021, 10, 540.
- Gray, M. Astrocytes in Huntington’s Disease. Adv. Exp. Med. Biol. 2019, 1175, 355–381.
- Udovin, L.; Quarracino, C.; Herrera, M.I.; Capani, F.; Otero-Losada, M.; Perez-Lloret, S. Role of Astrocytic Dysfunction in the Pathogenesis of Parkinson’s Disease Animal Models from a Molecular Signaling Perspective. Neural Plast. 2020, 2020, 1859431.
- Todd, A.C.; Hardingham, G.E. The Regulation of Astrocytic Glutamate Transporters in Health and Neurodegenerative Diseases. Int. J. Mol. Sci. 2020, 21, 9607.
- Rothstein, J.D.; Patel, S.; Regan, M.R.; Haenggeli, C.; Huang, Y.H.; Bergles, D.E.; Jin, L.; Dykes Hoberg, M.; Vidensky, S.; Chung, D.S.; et al. Beta-lactam antibiotics offer neuroprotection by increasing glutamate transporter expression. Nature 2005, 433, 73–77.
- Zumkehr, J.; Rodriguez-Ortiz, C.J.; Cheng, D.; Kieu, Z.; Wai, T.; Hawkins, C.; Kilian, J.; Lim, S.L.; Medeiros, R.; Kitazawa, M. Ceftriaxone ameliorates tau pathology and cognitive decline via restoration of glial glutamate transporter in a mouse model of Alzheimer’s disease. Neurobiol. Aging 2015, 36, 2260–2271.
- Miller, B.R.; Dorner, J.L.; Shou, M.; Sari, Y.; Barton, S.J.; Sengelaub, D.R.; Kennedy, R.T.; Rebec, G.V. Up-regulation of GLT1 expression increases glutamate uptake and attenuates the Huntington’s disease phenotype in the R6/2 mouse. Neuroscience 2008, 153, 329–337.
- Chotibut, T.; Davis, R.W.; Arnold, J.C.; Frenchek, Z.; Gurwara, S.; Bondada, V.; Geddes, J.W.; Salvatore, M.F. Ceftriaxone increases glutamate uptake and reduces striatal tyrosine hydroxylase loss in 6-OHDA Parkinson’s model. Mol. Neurobiol. 2014, 49, 1282–1292.
- Ho, S.C.; Hsu, C.C.; Pawlak, C.R.; Tikhonova, M.A.; Lai, T.J.; Amstislavskaya, T.G.; Ho, Y.J. Effects of ceftriaxone on the behavioral and neuronal changes in an MPTP-induced Parkinson’s disease rat model. Behav. Brain. Res. 2014, 268, 177–184.
- Bisht, R.; Kaur, B.; Gupta, H.; Prakash, A. Ceftriaxone mediated rescue of nigral oxidative damage and motor deficits in MPTP model of Parkinson’s disease in rats. Neurotoxicology 2014, 44, 71–79.
- Weng, J.C.; Tikhonova, M.A.; Chen, J.H.; Shen, M.S.; Meng, W.Y.; Chang, Y.T.; Chen, K.H.; Liang, K.C.; Hung, C.S.; Amstislavskaya, T.G.; et al. Ceftriaxone prevents the neurodegeneration and decreased neurogenesis seen in a Parkinson’s disease rat model: An immunohistochemical and MRI study. Behav. Brain Res. 2016, 305, 126–139.
- Cudkowicz, M.E.; Titus, S.; Kearney, M.; Yu, H.; Sherman, A.; Schoenfeld, D.; Hayden, D.; Shui, A.; Brooks, B.; Conwit, R.; et al. Safety and efficacy of ceftriaxone for amyotrophic lateral sclerosis: A multi-stage, randomised, double-blind, placebo-controlled trial. Lancet Neurol. 2014, 13, 1083–1091.
- Kong, Q.; Chang, L.C.; Takahashi, K.; Liu, Q.; Schulte, D.A.; Lai, L.; Ibabao, B.; Lin, Y.; Stouffer, N.; Das Mukhopadhyay, C.; et al. Small-molecule activator of glutamate transporter EAAT2 translation provides neuroprotection. J. Clin. Investig. 2014, 124, 1255–1267.
- Takahashi, K.; Kong, Q.; Lin, Y.; Stouffer, N.; Schulte, D.A.; Lai, L.; Liu, Q.; Chang, L.C.; Dominguez, S.; Xing, X.; et al. Restored glial glutamate transporter EAAT2 function as a potential therapeutic approach for Alzheimer’s disease. J. Exp. Med. 2015, 212, 319–332.
- Alotaibi, G.; Rahman, S. Effects of glial glutamate transporter activator in formalin-induced pain behaviour in mice. Eur. J. Pain 2019, 23, 765–783.
- Zhou, X.; Liang, J.; Wang, J.; Fei, Z.; Qin, G.; Zhang, D.; Zhou, J.; Chen, L. Up-regulation of astrocyte excitatory amino acid transporter 2 alleviates central sensitization in a rat model of chronic migraine. J. Neurochem. 2020, 155, 370–389.
- Tejeda-Bayron, F.A.; Rivera-Aponte, D.E.; Malpica-Nieves, C.J.; Maldonado-Martinez, G.; Maldonado, H.M.; Skatchkov, S.N.; Eaton, M.J. Activation of Glutamate Transporter-1 (GLT-1) Confers Sex-Dependent Neuroprotection in Brain Ischemia. Brain Sci. 2021, 11, 76.
- Vargas, M.R.; Johnson, D.A.; Sirkis, D.W.; Messing, A.; Johnson, J.A. Nrf2 activation in astrocytes protects against neurodegeneration in mouse models of familial amyotrophic lateral sclerosis. J. Neurosci. 2008, 28, 13574–13581.
- Chen, P.C.; Vargas, M.R.; Pani, A.K.; Smeyne, R.J.; Johnson, D.A.; Kan, Y.W.; Johnson, J.A. Nrf2-mediated neuroprotection in the MPTP mouse model of Parkinson’s disease: Critical role for the astrocyte. Proc. Natl. Acad. Sci. USA 2009, 106, 2933–2938.
- Wei, Y.; Lu, M.; Mei, M.; Wang, H.; Han, Z.; Chen, M.; Yao, H.; Song, N.; Ding, X.; Ding, J.; et al. Pyridoxine induces glutathione synthesis via PKM2-mediated Nrf2 transactivation and confers neuroprotection. Nat. Commun. 2020, 11, 941.
- Wheeler, M.A.; Clark, I.C.; Tjon, E.C.; Li, Z.; Zandee, S.E.J.; Couturier, C.P.; Watson, B.R.; Scalisi, G.; Alkwai, S.; Rothhammer, V.; et al. MAFG-driven astrocytes promote CNS inflammation. Nature 2020, 578, 593–599.
- Vargas, M.R.; Burton, N.C.; Kutzke, J.; Gan, L.; Johnson, D.A.; Schafer, M.; Werner, S.; Johnson, J.A. Absence of Nrf2 or its selective overexpression in neurons and muscle does not affect survival in ALS-linked mutant hSOD1 mouse models. PLoS ONE 2013, 8, e56625.
- Nanou, A.; Higginbottom, A.; Valori, C.F.; Wyles, M.; Ning, K.; Shaw, P.; Azzouz, M. Viral delivery of antioxidant genes as a therapeutic strategy in experimental models of amyotrophic lateral sclerosis. Mol. Ther. 2013, 21, 1486–1496.
- Feng, X.; Peng, Y.; Liu, M.; Cui, L. DL-3-n-butylphthalide extends survival by attenuating glial activation in a mouse model of amyotrophic lateral sclerosis. Neuropharmacology 2012, 62, 1004–1010.
- Mead, R.J.; Higginbottom, A.; Allen, S.P.; Kirby, J.; Bennett, E.; Barber, S.C.; Heath, P.R.; Coluccia, A.; Patel, N.; Gardner, I.; et al. S[+] Apomorphine is a CNS penetrating activator of the Nrf2-ARE pathway with activity in mouse and patient fibroblast models of amyotrophic lateral sclerosis. Free Radic. Biol. Med. 2013, 61, 438–452.
- Neymotin, A.; Calingasan, N.Y.; Wille, E.; Naseri, N.; Petri, S.; Damiano, M.; Liby, K.T.; Risingsong, R.; Sporn, M.; Beal, M.F.; et al. Neuroprotective effect of Nrf2/ARE activators, CDDO ethylamide and CDDO trifluoroethylamide, in a mouse model of amyotrophic lateral sclerosis. Free Radic. Biol. Med. 2011, 51, 88–96.
- Park, J.S.; Leem, Y.H.; Park, J.E.; Kim, D.Y.; Kim, H.S. Neuroprotective Effect of beta-Lapachone in MPTP-Induced Parkinson’s Disease Mouse Model: Involvement of Astroglial p-AMPK/Nrf2/HO-1 Signaling Pathways. Biomol. Ther. (Seoul) 2019, 27, 178–184.
- Inose, Y.; Izumi, Y.; Takada-Takatori, Y.; Akaike, A.; Koyama, Y.; Kaneko, S.; Kume, T. Protective effects of Nrf2-ARE activator on dopaminergic neuronal loss in Parkinson disease model mice: Possible involvement of heme oxygenase-1. Neurosci. Lett. 2020, 736, 135268.
- Ikram, M.; Muhammad, T.; Rehman, S.U.; Khan, A.; Jo, M.G.; Ali, T.; Kim, M.O. Hesperetin Confers Neuroprotection by Regulating Nrf2/TLR4/NF-kappaB Signaling in an Abeta Mouse Model. Mol. Neurobiol. 2019, 56, 6293–6309.
- Hammond, S.L.; Bantle, C.M.; Popichak, K.A.; Wright, K.A.; Thompson, D.; Forero, C.; Kirkley, K.S.; Damale, P.U.; Chong, E.K.P.; Tjalkens, R.B. NF-kappaB Signaling in Astrocytes Modulates Brain Inflammation and Neuronal Injury Following Sequential Exposure to Manganese and MPTP During Development and Aging. Toxicol. Sci. 2020, 177, 506–520.
- Li, Y.X.; Sibon, O.C.M.; Dijkers, P.F. Inhibition of NF-kappaB in astrocytes is sufficient to delay neurodegeneration induced by proteotoxicity in neurons. J. Neuroinflamm. 2018, 15, 261.
- Crosio, C.; Valle, C.; Casciati, A.; Iaccarino, C.; Carri, M.T. Astroglial inhibition of NF-kappaB does not ameliorate disease onset and progression in a mouse model for amyotrophic lateral sclerosis (ALS). PLoS ONE 2011, 6, e17187.
- Frakes, A.E.; Ferraiuolo, L.; Haidet-Phillips, A.M.; Schmelzer, L.; Braun, L.; Miranda, C.J.; Ladner, K.J.; Bevan, A.K.; Foust, K.D.; Godbout, J.P.; et al. Microglia induce motor neuron death via the classical NF-kappaB pathway in amyotrophic lateral sclerosis. Neuron 2014, 81, 1009–1023.
- Lee, J.Y.; Lee, J.D.; Phipps, S.; Noakes, P.G.; Woodruff, T.M. Absence of toll-like receptor 4 (TLR4) extends survival in the hSOD1 G93A mouse model of amyotrophic lateral sclerosis. J. Neuroinflamm. 2015, 12, 90.
- Brambilla, L.; Martorana, F.; Guidotti, G.; Rossi, D. Dysregulation of Astrocytic HMGB1 Signaling in Amyotrophic Lateral Sclerosis. Front. Neurosci. 2018, 12, 622.
- Patel, P.; Julien, J.P.; Kriz, J. Early-stage treatment with Withaferin A reduces levels of misfolded superoxide dismutase 1 and extends lifespan in a mouse model of amyotrophic lateral sclerosis. Neurotherapeutics 2015, 12, 217–233.
- Swarup, V.; Phaneuf, D.; Dupre, N.; Petri, S.; Strong, M.; Kriz, J.; Julien, J.P. Deregulation of TDP-43 in amyotrophic lateral sclerosis triggers nuclear factor kappaB-mediated pathogenic pathways. J. Exp. Med. 2011, 208, 2429–2447.
- Kumar, S.; Phaneuf, D.; Julien, J.P. Withaferin-A Treatment Alleviates TAR DNA-Binding Protein-43 Pathology and Improves Cognitive Function in a Mouse Model of FTLD. Neurotherapeutics 2021, 18, 286–296.
- Thangudu, S.; Cheng, F.Y.; Su, C.H. Advancements in the Blood-Brain Barrier Penetrating Nanoplatforms for Brain Related Disease Diagnostics and Therapeutic Applications. Polymers 2020, 12, 3055.
- Joshi, C.R.; Raghavan, V.; Vijayaraghavalu, S.; Gao, Y.; Saraswathy, M.; Labhasetwar, V.; Ghorpade, A. Reaching for the Stars in the Brain: Polymer-Mediated Gene Delivery to Human Astrocytes. Mol. Ther. Nucleic. Acids 2018, 12, 645–657.
- Proulx, J.; Joshi, C.; Vijayaraghavalu, S.; Saraswathy, M.; Labhasetwar, V.; Ghorpade, A.; Borgmann, K. Arginine-Modified Polymers Facilitate Poly (Lactide-Co-Glycolide)-Based Nanoparticle Gene Delivery to Primary Human Astrocytes. Int. J. Nanomed. 2020, 15, 3639–3647.
- Fatima, N.; Gromnicova, R.; Loughlin, J.; Sharrack, B.; Male, D. Gold nanocarriers for transport of oligonucleotides across brain endothelial cells. PLoS ONE 2020, 15, e0236611.
- Papa, S.; Veneruso, V.; Mauri, E.; Cremonesi, G.; Mingaj, X.; Mariani, A.; De Paola, M.; Rossetti, A.; Sacchetti, A.; Rossi, F.; et al. Functionalized nanogel for treating activated astrocytes in spinal cord injury. J. Control. Release 2020, 330, 218–228.
- Guidotti, G.; Brambilla, L.; Rossi, D. Exploring Novel Molecular Targets for the Treatment of High-Grade Astrocytomas Using Peptide Therapeutics: An Overview. Cells 2020, 9, 490.
- Martorana, F.; Brambilla, L.; Valori, C.F.; Bergamaschi, C.; Roncoroni, C.; Aronica, E.; Volterra, A.; Bezzi, P.; Rossi, D. The BH4 domain of Bcl-X(L) rescues astrocyte degeneration in amyotrophic lateral sclerosis by modulating intracellular calcium signals. Hum. Mol. Genet. 2012, 21, 826–840.
- Valiante, S.; Falanga, A.; Cigliano, L.; Iachetta, G.; Busiello, R.A.; La Marca, V.; Galdiero, M.; Lombardi, A.; Galdiero, S. Peptide gH625 enters into neuron and astrocyte cell lines and crosses the blood-brain barrier in rats. Int. J. Nanomed. 2015, 10, 1885–1898.
- Villa-Cedillo, S.A.; Rodriguez-Rocha, H.; Zavala-Flores, L.M.; Montes-de-Oca-Luna, R.; Garcia-Garcia, A.; Loera-Arias, M.J.; Saucedo-Cardenas, O. Asn194Lys mutation in RVG29 peptide increases GFP transgene delivery by endocytosis to neuroblastoma and astrocyte cells. J. Pharm. Pharmacol. 2017, 69, 1352–1363.
- Villa-Cedillo, S.A.; Soto-Dominguez, A.; Rodriguez-Rocha, H.; Garcia-Garcia, A.; de Jesus Loera-Arias, M.; Rivera-Chavez, L.F.; Acosta-Espinoza, E.J.; Valdes, J.; Zavala-Flores, L.M.; Montes-de-Oca-Luna, R.; et al. The mRVG-9R peptide as a potential therapeutic vector to the central nervous system cells. Cell Biol. Int. 2019, 43, 809–819.
- Foust, K.D.; Nurre, E.; Montgomery, C.L.; Hernandez, A.; Chan, C.M.; Kaspar, B.K. Intravascular AAV9 preferentially targets neonatal neurons and adult astrocytes. Nat. Biotechnol. 2009, 27, 59–65.
- Samaranch, L.; Salegio, E.A.; San Sebastian, W.; Kells, A.P.; Foust, K.D.; Bringas, J.R.; Lamarre, C.; Forsayeth, J.; Kaspar, B.K.; Bankiewicz, K.S. Adeno-associated virus serotype 9 transduction in the central nervous system of nonhuman primates. Hum. Gene Ther. 2012, 23, 382–389.
- Ortinski, P.I.; Dong, J.; Mungenast, A.; Yue, C.; Takano, H.; Watson, D.J.; Haydon, P.G.; Coulter, D.A. Selective induction of astrocytic gliosis generates deficits in neuronal inhibition. Nat. Neurosci. 2010, 13, 584–591.
- von Jonquieres, G.; Mersmann, N.; Klugmann, C.B.; Harasta, A.E.; Lutz, B.; Teahan, O.; Housley, G.D.; Frohlich, D.; Kramer-Albers, E.M.; Klugmann, M. Glial promoter selectivity following AAV-delivery to the immature brain. PLoS ONE 2013, 8, e65646.
- Meng, X.; Yang, F.; Ouyang, T.; Liu, B.; Wu, C.; Jiang, W. Specific gene expression in mouse cortical astrocytes is mediated by a 1740bp-GFAP promoter-driven combined adeno-associated virus 2/5/7/8/9. Neurosci. Lett. 2015, 593, 45–50.
- Vagner, T.; Dvorzhak, A.; Wojtowicz, A.M.; Harms, C.; Grantyn, R. Systemic application of AAV vectors targeting GFAP-expressing astrocytes in Z-Q175-KI Huntington’s disease mice. Mol. Cell. Neurosci. 2016, 77, 76–86.
- Taschenberger, G.; Tereshchenko, J.; Kugler, S. A MicroRNA124 Target Sequence Restores Astrocyte Specificity of gfaABC1D-Driven Transgene Expression in AAV-Mediated Gene Transfer. Mol. Ther. Nucleic. Acids 2017, 8, 13–25.
- Griffin, J.M.; Fackelmeier, B.; Fong, D.M.; Mouravlev, A.; Young, D.; O’Carroll, S.J. Astrocyte-selective AAV gene therapy through the endogenous GFAP promoter results in robust transduction in the rat spinal cord following injury. Gene. Ther. 2019, 26, 198–210.
- Pajarillo, E.; Johnson, J., Jr.; Rizor, A.; Nyarko-Danquah, I.; Adinew, G.; Bornhorst, J.; Stiboller, M.; Schwerdtle, T.; Son, D.S.; Aschner, M.; et al. Astrocyte-specific deletion of the transcription factor Yin Yang 1 in murine substantia nigra mitigates manganese-induced dopaminergic neurotoxicity. J. Biol. Chem. 2020, 295, 15662–15676.
- Koh, W.; Park, Y.M.; Lee, S.E.; Lee, C.J. AAV-Mediated Astrocyte-Specific Gene Expression under Human ALDH1L1 Promoter in Mouse Thalamus. Exp. Neurobiol. 2017, 26, 350–361.
- Mudannayake, J.M.; Mouravlev, A.; Fong, D.M.; Young, D. Transcriptional activity of novel ALDH1L1 promoters in the rat brain following AAV vector-mediated gene transfer. Mol. Ther. Methods Clin. Dev. 2016, 3, 16075.
- Birolini, G.; Verlengia, G.; Talpo, F.; Maniezzi, C.; Zentilin, L.; Giacca, M.; Conforti, P.; Cordiglieri, C.; Caccia, C.; Leoni, V.; et al. SREBP2 gene therapy targeting striatal astrocytes ameliorates Huntington’s disease phenotypes. Brain 2021, awab186.
- Guo, L.; Gao, T.; Gao, C.; Jia, X.; Ni, J.; Han, C.; Wang, Y. Stimulation of astrocytic sigma-1 receptor is sufficient to ameliorate inflammation- induced depression. Behav. Brain Res. 2021, 410, 113344.
- Vicidomini, C.; Guo, N.; Sahay, A. Communication, Cross Talk, and Signal Integration in the Adult Hippocampal Neurogenic Niche. Neuron 2020, 105, 220–235.
- Araki, T.; Ikegaya, Y.; Koyama, R. The effects of microglia-and astrocyte-derived factors on neurogenesis in health and disease. Eur. J. Neurosci. 2020.
- Marchetti, B.; Tirolo, C.; L’Episcopo, F.; Caniglia, S.; Testa, N.; Smith, J.A.; Pluchino, S.; Serapide, M.F. Parkinson’s disease, aging and adult neurogenesis: Wnt/beta-catenin signalling as the key to unlock the mystery of endogenous brain repair. Aging Cell 2020, 19, e13101.
- Casse, F.; Richetin, K.; Toni, N. Astrocytes’ Contribution to Adult Neurogenesis in Physiology and Alzheimer’s Disease. Front. Cell. Neurosci. 2018, 12, 432.
- Richetin, K.; Steullet, P.; Pachoud, M.; Perbet, R.; Parietti, E.; Maheswaran, M.; Eddarkaoui, S.; Begard, S.; Pythoud, C.; Rey, M.; et al. Tau accumulation in astrocytes of the dentate gyrus induces neuronal dysfunction and memory deficits in Alzheimer’s disease. Nat. Neurosci. 2020, 23, 1567–1579.
- Myszczynska, M.A.; Ojamies, P.N.; Lacoste, A.M.B.; Neil, D.; Saffari, A.; Mead, R.; Hautbergue, G.M.; Holbrook, J.D.; Ferraiuolo, L. Applications of machine learning to diagnosis and treatment of neurodegenerative diseases. Nat. Rev. Neurol. 2020, 16, 440–456.





