
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Morgan Hamon | + 2184 word(s) | 2184 | 2021-08-09 13:16:56 | | | |
| 2 | Vicky Zhou | Meta information modification | 2184 | 2021-08-11 09:33:33 | | | | |
| 3 | Vicky Zhou | -2 word(s) | 2182 | 2021-08-12 02:11:51 | | |
Video Upload Options
The kidneys are vital organs performing several essential functions. Their primary function is the filtration of blood and the removal of metabolic waste products as well as fluid homeostasis. Renal filtration is the main pathway for drug removal, highlighting the importance of this organ to the growing field of nanomedicine. The kidneys (i) have a key role in the transport and clearance of nanoparticles (NPs), (ii) are exposed to potential NPs’ toxicity, and (iii) are the targets of diseases that nanomedicine can study, detect, and treat.
1. Introduction
One of the main factors influencing the pharmacokinetics of drugs and therapeutic materials is the clearance rate from the body. After their absorption, nanoparticles enter the systemic circulation, diffuse and interact with the body, and are eventually cleared by the reticuloendothelial system (RES) or the kidney. Herein, we will focus on the kidney’s influence, as renal excretion is one of the most critical factors involved in the excretion of nanomedicines. For more information regarding the optimization of nanodrug pharmacokinetics, we recommend the review by Moss and Siccardi [1].
The kidneys receive 1 to 1.2 L of blood per minute, that is 20–25% of the cardiac output. Each kidney contains an average of one million nephrons, the basic functional unit of the kidney. Each nephron consists of the renal corpuscle, or glomerulus, the proximal, and distal tubules. Blood is transported via the renal vasculature to the glomeruli, where the primary filtrate is passed through fenestrated capillaries into the Bowman’s capsule, which encloses the glomerulus and collects the filtrate. The filtrate is then transported to the proximal and distal renal tubules where nutrients, water, and ions are reabsorbed and waste products secreted.
From the lumen of the capillary to the inside of the Bowman’s capsule, the glomerulus contains four structures that act as filters: the glycocalyx, the endothelium, the glomerular basement membrane (GBM), and the podocytes. The glycocalyx is a negatively charged layer of glycoproteins and glycolipids that protects the underlying endothelial cells and prevents large protein leakage. The endothelial layer is a fenestrated endothelium with large pores of 70–90 nm in diameter. The endothelial cells repose on the negatively charged GBM with pores of 2–8 nm. Its role is to prevent the filtration of large proteins and cellular components from the blood. The last filtration barrier is composed of the podocytes that sit on the urine side of the GBM. Organized as a monolayer, their interdigitating foot processes form the filtration slit sized 4 to 11 nm. Overall, the four layers create a charge- and size-selective filtration barrier with a glomerular functional barrier ’s (GFB) pore size of 4.5–5 nm [2].
Size, shape, and charge of nanoparticles must be considered when designing nanomedicine, as they will decide between renal clearance and accumulation. For example, a positively charged nanomedicine may cross more efficiently the filtration through a negatively charged GBM and podocytes than neutral or negatively charged compounds. An overview of the potential interaction of NP with the kidney is shown in Figure 1 .
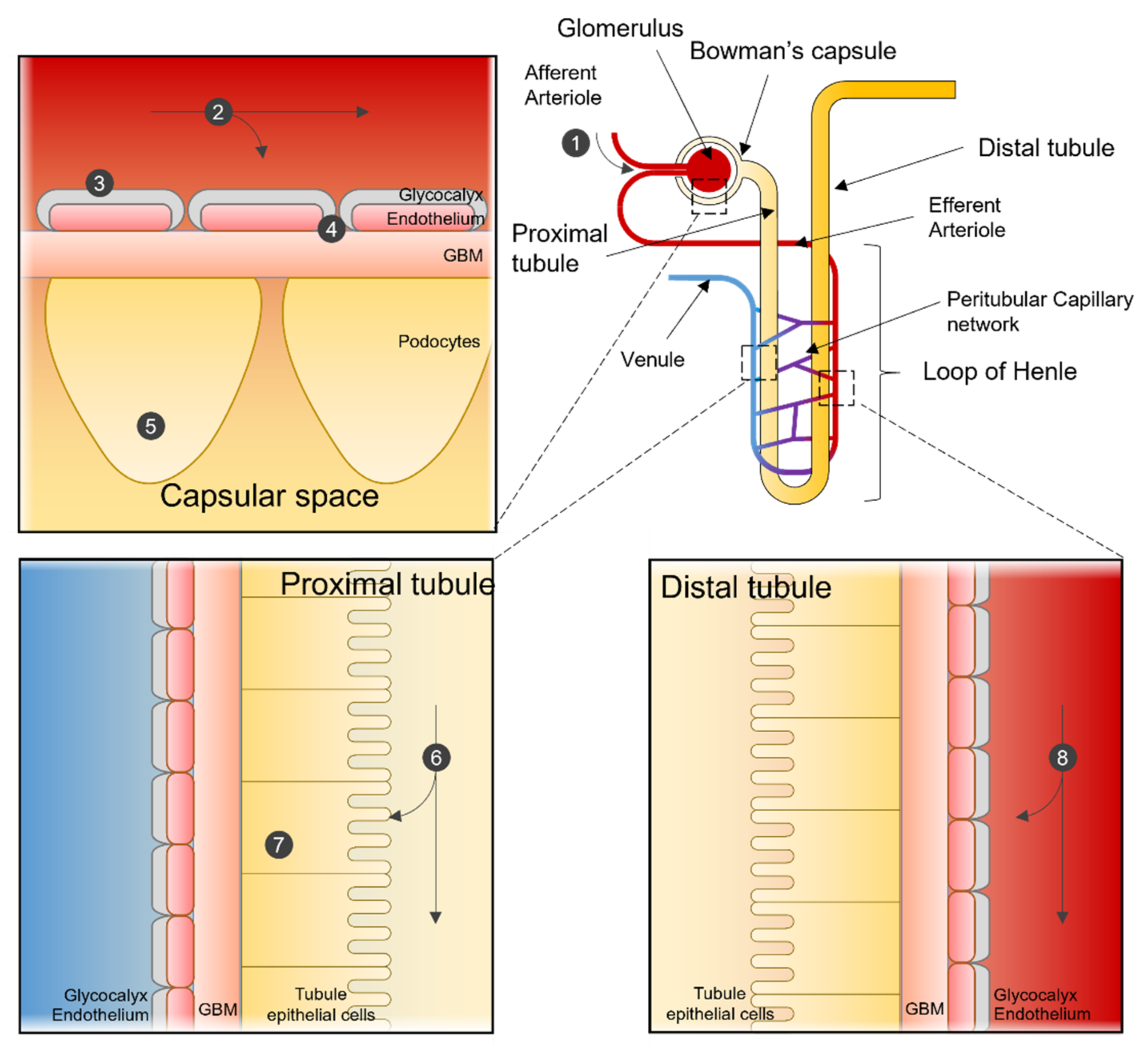 Figure 1. Nanoparticle interactions in the Nephron. Sites of interaction and potential uptake of NPs that are subject to extraction or used to target the kidney. NPs enter the kidney via the renal artery and are transported to the afferent arteriole (1). Depending on particle characteristics, NPs remain in the blood stream or are subjected to renal filtration from the blood in the glomerular capillaries (2). NPs can be designed so that renal structures, such as the glycocalyx (3), endothelial cells, or the glomerular basement membrane (4), can function in selecting NPs for filtration. After filtration, NPs can interact with podocytes in the Bowman’s lumen (5). NPs in the filtrate are then transported to the proximal tubule, where they interact with proximal epithelial cells (6) and are potentially reabsorbed. Pro-drug NPs can be activated in the lysosome of proximal tubular cells (7). NPs not selected for renal filtration can interact with the renal tubular comparted after being transported from the efferent arteriole to the peritubular network (8).
Figure 1. Nanoparticle interactions in the Nephron. Sites of interaction and potential uptake of NPs that are subject to extraction or used to target the kidney. NPs enter the kidney via the renal artery and are transported to the afferent arteriole (1). Depending on particle characteristics, NPs remain in the blood stream or are subjected to renal filtration from the blood in the glomerular capillaries (2). NPs can be designed so that renal structures, such as the glycocalyx (3), endothelial cells, or the glomerular basement membrane (4), can function in selecting NPs for filtration. After filtration, NPs can interact with podocytes in the Bowman’s lumen (5). NPs in the filtrate are then transported to the proximal tubule, where they interact with proximal epithelial cells (6) and are potentially reabsorbed. Pro-drug NPs can be activated in the lysosome of proximal tubular cells (7). NPs not selected for renal filtration can interact with the renal tubular comparted after being transported from the efferent arteriole to the peritubular network (8).
2. Nanotechnology for Renal Therapy
2.1. Drug Delivery
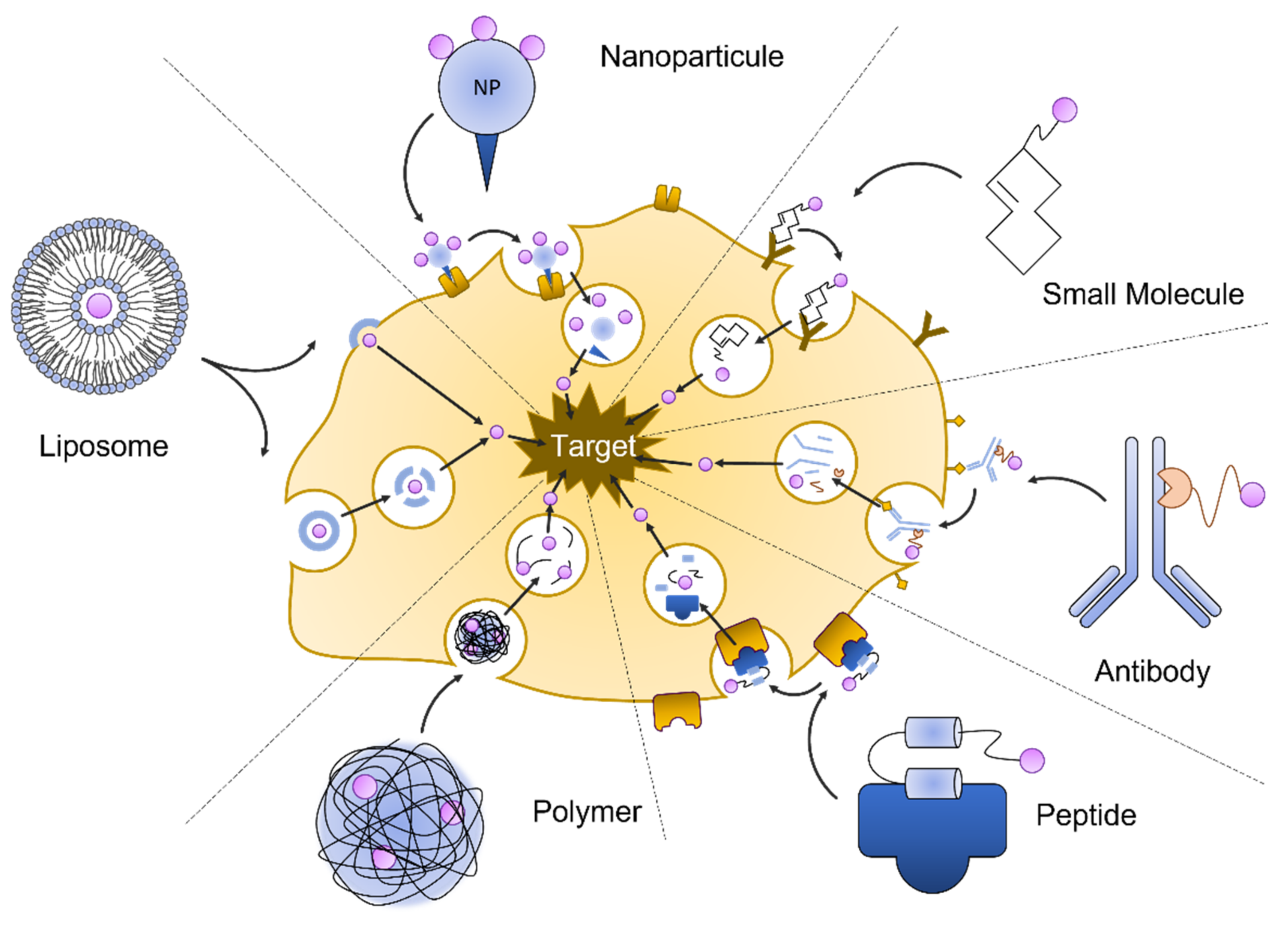
2.1.1. Liposomes
2.1.2. Antibodies
2.1.3. Protein/Peptide-Based Carrier
2.1.4. Small Molecule Pro-Drugs
2.1.5. Nanoparticles
2.1.6. Hydropolymeric Carriers
2.2. Tissue Engineering
2.2.1. Renal Replacement Technology
2.2.2. Kidney Regeneration
References
- Moss, D.M.; Siccardi, M. Optimizing Nanomedicine Pharmacokinetics Using Physiologically Based Pharmacokinetics Modelling. Br. J. Pharm. 2014, 171, 3963–3979.
- Scott, R.P.; Quaggin, S.E. Review Series: The Cell Biology of Renal Filtration. J. Cell Biol. 2015, 209, 199–210.
- Alavi, M.; Karimi, N.; Safaei, M. Application of Various Types of Liposomes in Drug Delivery Systems. Adv. Pharm. Bull. 2017, 7, 3–9.
- Lai, L.W.; Moeckel, G.W.; Lien, Y.H. Kidney-Targeted Liposome-Mediated Gene Transfer in Mice. Gene Ther. 1997, 4, 426–431.
- Shirai, T.; Kohara, H.; Tabata, Y. Inflammation Imaging by Silica Nanoparticles with Antibodies Orientedly Immobilized. J. Drug Target. 2012, 20, 535–543.
- Durigutto, P.; Sblattero, D.; Biffi, S.; De Maso, L.; Garrovo, C.; Baj, G.; Colombo, F.; Fischetti, F.; Di Naro, A.F.; Tedesco, F.; et al. Targeted Delivery of Neutralizing Anti-C5 Antibody to Renal Endothelium Prevents Complement-Dependent Tissue Damage. Front. Immunol. 2017, 8, 1093.
- Sutthasupha, P.; Lungkaphin, A. The Potential Roles of Chitosan Oligosaccharide in Prevention of Kidney Injury in Obese and Diabetic Conditions. Food Funct. 2020, 11, 7371–7388.
- Fu, Y.; Lin, Q.; Gong, T.; Sun, X.; Zhang, Z.-R. Renal-Targeting Triptolide-Glucosamine Conjugate Exhibits Lower Toxicity and Superior Efficacy in Attenuation of Ischemia/Reperfusion Renal Injury in Rats. Acta Pharm. Sin. 2016, 37, 1467–1480.
- Huang, C.; Zeng, T.; Li, J.; Tan, L.; Deng, X.; Pan, Y.; Chen, Q.; Li, A.; Hu, J. Folate Receptor-Mediated Renal-Targeting Nanoplatform for the Specific Delivery of Triptolide to Treat Renal Ischemia/Reperfusion Injury. ACS Biomater. Sci. Eng. 2019, 5, 2877–2886.
- He, X.; Yuan, Z.; Wu, X.; Xu, C.; Li, W. Low Molecular Weight Hydroxyethyl Chitosan-Prednisolone Conjugate for Renal Targeting Therapy: Synthesis, Characterization and in Vivo Studies. Theranostics 2012, 2, 1054–1063.
- Yang, B.; Gao, J.; Pei, Q.; Xu, H.; Yu, H. Engineering Prodrug Nanomedicine for Cancer Immunotherapy. Adv. Sci. 2020, 7, 2002365.
- Wischnjow, A.; Sarko, D.; Janzer, M.; Kaufman, C.; Beijer, B.; Brings, S.; Haberkorn, U.; Larbig, G.; Kübelbeck, A.; Mier, W. Renal Targeting: Peptide-Based Drug Delivery to Proximal Tubule Cells. Bioconjug. Chem. 2016, 27, 1050–1057.
- Kamaly, N.; He, J.C.; Ausiello, D.A.; Farokhzad, O.C. Nanomedicines for Renal Disease: Current Status and Future Applications. Nat. Rev. Nephrol. 2016, 12, 738–753.
- Tripathy, N.; Wang, J.; Tung, M.; Conway, C.; Chung, E.J. Transdermal Delivery of Kidney-Targeting Nanoparticles Using Dissolvable Microneedles. Cell Mol. Bioeng. 2020, 13, 475–486.
- Chen, H.-C.; Cheng, C.-Y.; Lin, H.-C.; Chen, H.-H.; Chen, C.-H.; Yang, C.-P.; Yang, K.-H.; Lin, C.-M.; Lin, T.-Y.; Shih, C.-M.; et al. Multifunctions of Excited Gold Nanoparticles Decorated Artificial Kidney with Efficient Hemodialysis and Therapeutic Potential. ACS Appl. Mater. Interfaces 2016, 8, 19691–19700.
- Murugesan, S.; Mousa, S.; Vijayaraghavan, A.; Ajayan, P.M.; Linhardt, R.J. Ionic Liquid-Derived Blood-Compatible Composite Membranes for Kidney Dialysis. J. Biomed. Mater. Res. Part B Appl. Biomater. 2006, 79B, 298–304.
- Reddy, A.R.N.; Reddy, Y.N.; Krishna, D.R.; Himabindu, V. Multi Wall Carbon Nanotubes Induce Oxidative Stress and Cytotoxicity in Human Embryonic Kidney (HEK293) Cells. Toxicology 2010, 272, 11–16.
- Herrera Sanchez, M.B.; Bruno, S.; Grange, C.; Tapparo, M.; Cantaluppi, V.; Tetta, C.; Camussi, G. Human Liver Stem Cells and Derived Extracellular Vesicles Improve Recovery in a Murine Model of Acute Kidney Injury. Stem Cell Res. Ther. 2014, 5, 124.
- Cantaluppi, V.; Medica, D.; Mannari, C.; Stiaccini, G.; Figliolini, F.; Dellepiane, S.; Quercia, A.D.; Migliori, M.; Panichi, V.; Giovannini, L.; et al. Endothelial Progenitor Cell-Derived Extracellular Vesicles Protect from Complement-Mediated Mesangial Injury in Experimental Anti-Thy1.1 Glomerulonephritis. Nephrol. Dial. Transplant. 2015, 30, 410–422.
- Mollet, B.B.; Bogaerts, I.L.J.; van Almen, G.C.; Dankers, P.Y.W. A Bioartificial Environment for Kidney Epithelial Cells Based on a Supramolecular Polymer Basement Membrane Mimic and an Organotypical Culture System. J. Tissue Eng. Regen. Med. 2017, 11, 1820–1834.





