
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | M. Carmen Galan | + 1827 word(s) | 1827 | 2021-07-28 13:50:55 | | | |
| 2 | Frida Xu | Meta information modification | 1827 | 2021-07-29 04:56:37 | | |
Video Upload Options
Carbon dots (CDs) have been identified as a promising class of photosensitiser nanomaterials for the specific detection and inactivation of different bacterial species. CDs possess exceptional and tuneable chemical and photoelectric properties that make them excellent candidates for antibacterial theranostic applications, such as great chemical stability, high water solubility, low toxicity and excellent biocompatibility.
1. Introduction
Antimicrobial resistance (AMR) has become a major threat that affects public health. One of the main causes for this problem is the extensive and disproportionate use of antimicrobial agents, which has led to the selection of drug-resistant pathogens that have developed new resistance mechanisms. The fast evolution of new AMR machineries, combined to the slow development and low approval rate of new drugs, has resulted in a major global health crisis [1][2]. Multidrug resistant bacteria isolated in hospitals represent an increasing risk factor especially for surgery and intensive care unit patients [3], making it harder, if not impossible, to treat infections with the consequent increase of medical complications and sanitation costs [4][5].
The development of reliable, cheap, and fast strategies for determining the presence or absence of bacteria or identification of specific species/strains in patient samples could reduce inappropriate prescribing of antibiotics in primary and secondary care [6]. Similarly, the ability to target antimicrobials to specific pathogens could reduce the inappropriate use of broad-spectrum antibiotics, which, as mentioned earlier, drives the emergence of both antibiotic resistance and healthcare-associated infections [7]. Lastly, the targeted delivery of antibiotics directly to the surface of specific cells may enhance their antibacterial activity through increasing local concentration or stimulating intracellular uptake [8]. The issue of antibiotic targeting is of particular relevance when considering Gram-negative bacteria, as the outer membrane of these species presents a formidable barrier to ingress for many antibiotic classes, which reduces treatment options for these organisms and complicates the development of new antibiotics [9]. This is exemplified by the fact that few new agents effective against Gram-negative bacteria are currently in clinical development. For example, oxazolidinones represent the first new chemical class of antibiotic to reach the clinic in over 30 years. These molecules are inhibitors of bacterial protein biosynthesis and represent an important class of drugs that are effective against a range of Gram-positive bacteria including multiresistant pathogens such as methicillin-resistant Staphylococcus aureus (MRSA). However, linezolid, the first such agent to reach the clinic, is becoming compromised by the emergence of resistance [10]. Thus, the prevention of infections through the early detection of pathogens and the development of new antibacterial agents able to circumvent bacterial multidrug resistance (MDR) represent a crucial objective of current biomedical research.
One of the most promising strategies in the fight against MDR pathogens entails the photodynamic inactivation of bacteria promoted by photosensitisers. This strategy relies on the light-promoted generation of highly reactive oxygen species (ROS) able to inactivate bacterial cells in different ways, including membrane destruction and/or irreversible protein and DNA damage [11][12][13]. The most important feature of photodynamic therapies consists of the generation of a closely localized physicochemical environment that is harsh to bacterial cells in ways such as ROS production or temperature increases (known as photothermal (PTT) effects), against which it is difficult to generate a resistance [14][15]. Among the different kind of photosensitisers, carbon dots (CDs) have emerged as a promising class of nanomaterials for the specific detection and inactivation of different bacterial species [16][17]. CDs are a class of quasispherical carbon-based fluorescent nanomaterials with a typical size of 10 nm or below. These materials possess great chemical stability, high water solubility, and outstanding photoelectric properties. In addition, they exhibit low toxicity and excellent biocompatibility [18]. These features, together with their ease of preparation and reduce material costs, makes CDs ideal candidates for antibacterial theranostic applications. Indeed, since their serendipitous discovery in 2004 by Xu et al. [19], CDs have found further applications across many scientific disciplines including semiconductors [20][21], biomedicine [22][23] catalysis [24], sensing and functional materials [25][26], and in the agricultural field [27][28].
2. General Antibacterial Mechanisms of Action of CDs
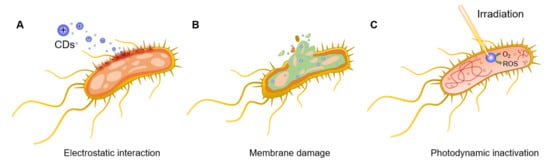
3. CDs as Bacteria Targeting and Antibacterial Agents
3.1. Applications of CDs as Labelling and Bactericidal Agents (Theranostics)
3.2. Applications of CDs for Biofilm Eradication and Inhibition
4. Conclusions
References
- Brown, E.D.; Wright, G.D. Antibacterial drug discovery in the resistance era. Nature 2016, 529, 336–343.
- Roope, L.S.J.; Smith, R.D.; Pouwels, K.B.; Buchanan, J.; Abel, L.; Eibich, P.; Butler, C.C.; Tan, P.S.; Walker, A.S.; Robotham, J.V.; et al. The challenge of antimicrobial resistance: What economics can contribute. Science 2019, 364.
- Cassini, A.; Högberg, L.D.; Plachouras, D.; Quattrocchi, A.; Hoxha, A.; Simonsen, G.S.; Colomb-Cotinat, M.; Kretzschmar, M.E.; Devleesschauwer, B.; Cecchini, M.; et al. Attributable deaths and disability-adjusted life-years caused by infections with antibiotic-resistant bacteria in the EU and the European Economic Area in 2015: A population-level modelling analysis. Lancet Infect. Dis. 2019, 19, 56–66.
- Van Duin, D.; Paterson, D.L. Multidrug-Resistant Bacteria in the Community: Trends and Lessons Learned. Infect. Dis. Clin. North. Am. 2016, 30, 377–390.
- Hofer, U. The cost of antimicrobial resistance. Nat. Rev. Microbiol. 2019, 17, 3.
- O’Neill, J. Tackling Drug-Resistant Infections Globally: Final Report and Recommendations; HM Government: London, UK, 2016.
- Karam, G.; Chastre, J.; Wilcox, M.H.; Vincent, J.-L. Antibiotic strategies in the era of multidrug resistance. Crit. Care 2016, 20, 136.
- Zhang, L.; Pornpattananangkul, D.; Hu, C.M.J.; Huang, C.M. Development of Nanoparticles for Antimicrobial Drug Delivery. Curr. Med. Chem. 2010, 17, 585–594.
- Zgurskaya, H.I.; López, C.A.; Gnanakaran, S. Permeability Barrier of Gram-Negative Cell Envelopes and Approaches to Bypass It. ACS Infect. Dis. 2015, 1, 512–522.
- Mendes, R.E.; Deshpande, L.M.; Jones, R.N. Linezolid update: Stable in vitro activity following more than a decade of clinical use and summary of associated resistance mechanisms. Drug Resist. Updat. 2014, 17, 1–12.
- Tim, M. Strategies to optimize photosensitizers for photodynamic inactivation of bacteria. J. Photochem. Photobiol. B 2015, 150, 2–10.
- Yougbaré, S.; Mutalik, C.; Krisnawati, D.I.; Kristanto, H.; Jazidie, A.; Nuh, M.; Cheng, T.-M.; Kuo, T.-R. Nanomaterials for the Photothermal Killing of Bacteria. Nanomaterials 2020, 10, 1123.
- Knoblauch, R.; Geddes, C.D. Carbon nanodots in photodynamic antimicrobial therapy: A review. Materials 2020, 13, 4004.
- Gao, S.; Yan, X.; Xie, G.; Zhu, M.; Ju, X.; Stang, P.J.; Tian, Y.; Niu, Z. Membrane intercalation-enhanced photodynamic inactivation of bacteria by a metallacycle and TAT-decorated virus coat protein. Proc. Natl. Acad. Sci. USA 2019, 116, 23437–23443.
- Chen, Y.; Gao, Y.; Chen, Y.; Liu, L.; Mo, A.; Peng, Q. Nanomaterials-based photothermal therapy and its potentials in antibacterial treatment. J. Control. Release 2020, 328, 251–262.
- Dong, X.; Ge, L.; Abu Rabe, D.I.; Mohammed, O.O.; Wang, P.; Tang, Y.; Kathariou, S.; Yang, L.; Sun, Y.P. Photoexcited state properties and antibacterial activities of carbon dots relevant to mechanistic features and implications. Carbon 2020, 170, 137–145.
- Makabenta, J.M.V.; Nabawy, A.; Li, C.-H.; Schmidt-Malan, S.; Patel, R.; Rotello, V.M. Nanomaterial-based therapeutics for antibiotic-resistant bacterial infections. Nat. Rev. Microbiol. 2021, 19, 23–36.
- Lim, S.Y.; Shen, W.; Gao, Z. Carbon quantum dots and their applications. Chem. Soc. Rev. 2014, 44, 362–381.
- Xu, X.; Ray, R.; Gu, Y.; Ploehn, H.J.; Gearheart, L.; Raker, K.; Scrivens, W.A. Electrophoretic Analysis and Purification of Fluorescent Single-Walled Carbon Nanotube Fragments. J. Am. Chem. Soc. 2004, 126, 12736–12737.
- Wang, X.; Feng, Y.; Dong, P.; Huang, J. A Mini Review on Carbon Quantum Dots: Preparation, Properties, and Electrocatalytic Application. Front. Chem. 2019, 7, 671.
- Yu, H.; Shi, R.; Zhao, Y.; Waterhouse, G.I.N.; Wu, L.-Z.; Tung, C.-H.; Zhang, T. Smart Utilization of Carbon Dots in Semiconductor Photocatalysis. Adv. Mater. 2016, 28, 9454–9477.
- Mohammadinejad, R.; Dadashzadeh, A.; Moghassemi, S.; Ashrafizadeh, M.; Dehshahri, A.; Pardakhty, A.; Sassan, H.; Sohrevardi, S.-M.; Mandegary, A. Shedding light on gene therapy: Carbon dots for the minimally invasive image-guided delivery of plasmids and noncoding RNAs—A review. J. Adv. Res. 2019, 18, 81–93.
- Du, J.; Xu, N.; Fan, J.; Sun, W.; Peng, X. Carbon Dots for In Vivo Bioimaging and Theranostics. Small 2019, 15, 1805087.
- Li, W.; Wei, Z.; Wang, B.; Liu, Y.; Song, H.; Tang, Z.; Yang, B.; Lu, S. Carbon quantum dots enhanced the activity for the hydrogen evolution reaction in ruthenium-based electrocatalysts. Mater. Chem. Front. 2019, 4, 277–284.
- Kang, Z.; Lee, S.-T. Carbon dots: Advances in nanocarbon applications. Nanoscale 2019, 11, 19214–19224.
- Li, M.; Chen, T.; Gooding, J.J.; Liu, J. Review of Carbon and Graphene Quantum Dots for Sensing. ACS Sens. 2019, 4, 1732–1748.
- Li, Y.; Xu, X.; Wu, Y.; Zhuang, J.; Zhang, X.; Zhang, H.; Lei, B.; Hu, C.; Liu, Y. A review on the effects of carbon dots in plant systems. Mater. Chem. Front. 2020, 4, 437–448.
- Swift, T.A.; Oliver, T.A.A.; Galan, M.C.; Whitney, H.M. Functional nanomaterials to augment photosynthesis: Evidence and considerations for their responsible use in agricultural applications. Interface Focus 2019, 9, 20180048.
- Bing, W.; Sun, H.; Yan, Z.; Ren, J.; Qu, X. Programmed Bacteria Death Induced by Carbon Dots with Different Surface Charge. Small 2016, 12, 4713–4718.
- Saravanan, A.; Maruthapandi, M.; Das, P.; Ganguly, S.; Margel, S.; Luong, J.H.T.; Gedanken, A. Applications of N-Doped Carbon Dots as Antimicrobial Agents, Antibiotic Carriers, and Selective Fluorescent Probes for Nitro Explosives. ACS Appl. Nano Mater. 2020, 3, 8023–8031.
- Yang, J.; Zhang, X.; Ma, Y.-H.; Gao, G.; Chen, X.; Jia, H.-R.; Li, Y.-H.; Chen, Z.; Wu, F.-G. Carbon Dot-Based Platform for Simultaneous Bacterial Distinguishment and Antibacterial Applications. ACS Appl. Mater. Interfaces 2016, 8, 32170–32181.
- Zhang, J.; Lu, X.; Tang, D.; Wu, S.; Hou, X.; Liu, J.; Wu, P. Phosphorescent Carbon Dots for Highly Efficient Oxygen Photosensitization and as Photo-oxidative Nanozymes. ACS Appl. Mater. Interfaces 2018, 10, 40808–40814.
- Xu, N.; Du, J.; Yao, Q.; Ge, H.; Li, H.; Xu, F.; Gao, F.; Xian, L.; Fan, J.; Peng, X. Precise photodynamic therapy: Penetrating the nuclear envelope with photosensitive carbon dots. Carbon 2020, 159, 74–82.
- Cui, F.; Ye, Y.; Ping, J.; Sun, X. Carbon dots: Current advances in pathogenic bacteria monitoring and prospect applications. Biosens. Bioelectron. 2020, 156.
- Verma, A.; Arshad, F.; Ahmad, K.; Goswami, U.; Samanta, S.K.; Sahoo, A.K.; Sk, M.P. Role of surface charge in enhancing antibacterial activity of fluorescent carbon dots. Nanotechnology 2020, 31.
- Sun, R.; Chen, H.; Sutrisno, L.; Kawazoe, N.; Chen, G. Nanomaterials and their composite scaffolds for photothermal therapy and tissue engineering applications. Sci. Technol. Adv. Mater. 2021, 22, 404–428.
- Li, H.; Huang, J.; Song, Y.; Zhang, M.; Wang, H.; Lu, F.; Huang, H.; Liu, Y.; Dai, X.; Gu, Z.; et al. Degradable Carbon Dots with Broad-Spectrum Antibacterial Activity. ACS Appl. Mater. Interfaces 2018, 10, 26936–26946.
- Hall-Stoodley, L.; Costerton, J.W.; Stoodley, P. Bacterial biofilms: From the Natural environment to infectious diseases. Nat. Rev. Microbiol. 2004, 2, 95–108.
- Vestby, L.K.; Grønseth, T.; Simm, R.; Nesse, L.L. Bacterial Biofilm and its Role in the Pathogenesis of Disease. Antibiotics 2020, 9, 59.
- Lu, F.; Ma, Y.; Wang, H.; Zhang, M.; Wang, B.; Zhang, Y.; Huang, H.; Liao, F.; Liu, Y.; Kang, Z. Water-solvable carbon dots derived from curcumin and citric acid with enhanced broad-spectrum antibacterial and antibiofilm activity. Mater. Today Commun. 2021, 26.
- Ran, H.H.; Cheng, X.; Bao, Y.W.; Hua, X.W.; Gao, G.; Zhang, X.; Jiang, Y.W.; Zhu, Y.X.; Wu, F.G. Multifunctional quaternized carbon dots with enhanced biofilm penetration and eradication efficiencies. J. Mater. Chem. B 2019, 7, 5104–5114.
- Li, P.; Liu, S.; Cao, W.; Zhang, G.; Yang, X.; Gong, X.; Xing, X. Low-toxicity carbon quantum dots derived from gentamicin sulfate to combat antibiotic resistance and eradicate mature biofilms. Chem. Commun. 2020, 56, 2316–2319.
- Liang, G.; Shi, H.; Qi, Y.; Li, J.; Jing, A.; Liu, Q.; Feng, W.; Li, G.; Gao, S. Specific anti-biofilm activity of carbon quantum dots by destroying p. Gingivalis biofilm related genes. Int. J. Nanomed. 2020, 15, 5473–5489.
- Otis, G.; Bhattacharya, S.; Malka, O.; Kolusheva, S.; Bolel, P.; Porgador, A.; Jelinek, R. Selective Labeling and Growth Inhibition of Pseudomonas aeruginosa by Aminoguanidine Carbon Dots. ACS Infect. Dis. 2019, 5, 292–302.
- Wang, H.; Zhang, M.; Ma, Y.; Wang, B.; Shao, M.; Huang, H.; Liu, Y.; Kang, Z. Selective inactivation of Gram-negative bacteria by carbon dots derived from natural biomass: Artemisia argyi leaves. J. Mater. Chem. B 2020, 8, 2666–2672.





