
Video Upload Options
Forest connectivity is critical for the long-term persistence and range shifts of forest-dependent species. In the face of contemporary climate change, the climate connectivity of forested areas is one of the most important factors that determine species’ ability to track suitable climate niches. Climate connectivity is defined as the degree to which a landscape allows species to shift their ranges to suitable habitats that are the same or cooler than their current habitats as the climate warms. Improving climate connectivity is both a necessary action and a viable solution to reduce the likelihood of local extinctions and biodiversity loss. This is particularly urgent considering the rapid rate of climate change, which is expected to cause large shifts in the distributions of species in European forests.
1. Introduction
2. Identify Forest Patches
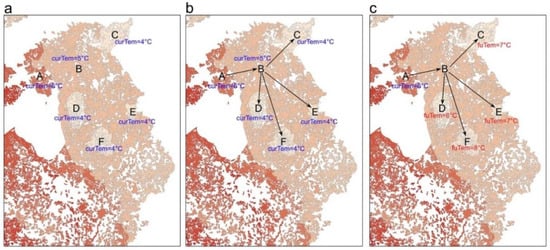
3. Evaluate Climate Connectivity
References
- Haddad, N.M.; Brudvig, L.A.; Clobert, J.; Davies, K.F.; Gonzalez, A.; Holt, R.D.; Lovejoy, T.E.; Sexton, J.O.; Austin, M.P.; Collins, C.D.; et al. Habitat fragmentation and its lasting impact on Earth’s ecosystems. Sci. Adv. 2015, 1, e1500052.
- Watson, J.E.M.; Evans, T.; Venter, O.; Williams, B.; Tulloch, A.; Stewart, C.; Thompson, I.; Ray, J.C.; Murray, K.; Salazar, A.; et al. The exceptional value of intact forest ecosystems. Nat. Ecol. Evol. 2018, 2, 599–610.
- Betts, M.G.; Wolf, C.; Ripple, W.J.; Phalan, B.; Millers, K.A.; Duarte, A.; Butchart, S.H.M.; Levi, T. Global forest loss disproportionately erodes biodiversity in intact landscapes. Nature 2017, 547, 441–444.
- Littlefield, C.E.; McRae, B.H.; Michalak, J.L.; Lawler, J.J.; Carroll, C. Connecting today’s climates to future climate analogs to facilitate movement of species under climate change. Conserv. Biol. 2017, 31, 1397–1408.
- Guo, F.; Lenoir, J.; Bonebrake, T.C. Land-use change interacts with climate to determine elevational species redistribution. Nat. Commun. 2018, 9, 1315.
- Honnay, O.; Verheyen, K.; Butaye, J.; Jacquemyn, H.; Bossuyt, B.; Hermy, M. Possible effects of habitat fragmentation and climate change on the range of forest plant species. Ecol. Lett. 2002, 5, 525–530.
- Walther, G.R.; Post, E.; Convey, P.; Menzel, A.; Parmesan, C.; Beebee, T.J.; Fromentin, J.M.; Hoegh-Guldberg, O.; Bairlein, F. Ecological responses to recent climate change. Nature 2002, 416, 389–395.
- Pecl, G.T.; Araújo, M.B.; Bell, J.D.; Blanchard, J.; Bonebrake, T.C.; Chen, I.-C.; Clark, T.D.; Colwell, R.K.; Danielsen, F.; Evengård, B.; et al. Biodiversity redistribution under climate change: Impacts on ecosystems and human well-being. Science 2017, 355, eaai9214.
- Lenoir, J.; Svenning, J.-C. Climate-related range shifts—A global multidimensional synthesis and new research directions. Ecography 2015, 38, 15–28.
- Dullinger, S.; Dendoncker, N.; Gattringer, A.; Leitner, M.; Mang, T.; Moser, D.; Mucher, C.A.; Plutzar, C.; Rounsevell, M.; Willner, W.; et al. Modelling the effect of habitat fragmentation on climate-driven migration of European forest understorey plants. Divers. Distrib. 2015, 21, 1375–1387.
- McGuire, J.L.; Lawler, J.J.; McRae, B.H.; Nunez, T.A.; Theobald, D.M. Achieving climate connectivity in a fragmented landscape. Proc. Natl. Acad. Sci. USA 2016, 113, 7195–7200.
- Su, J.; Yin, H.; Kong, F. Ecological networks in response to climate change and the human footprint in the Yangtze River Delta urban agglomeration, China. Landsc. Ecol. 2020, 36, 2095–2112.
- Keeley, A.T.H.; Ackerly, D.D.; Cameron, D.R.; Heller, N.E.; Huber, P.R.; Schloss, C.A.; Thorne, J.H.; Merenlender, A.M. New concepts, models, and assessments of climate-wise connectivity. Environ. Res. Lett. 2018, 13, 073002.
- Parks, S.A.; Carroll, C.; Dobrowski, S.Z.; Allred, B.W. Human land uses reduce climate connectivity across North America. Glob. Chang. Biol. 2020, 26, 2944–2955.
- Steinacker, C.; Beierkuhnlein, C.; Jaeschke, A. Assessing the exposure of forest habitat types to projected climate change-Implications for Bavarian protected areas. Ecol. Evol. 2019, 9, 14417–14429.
- Kullman, L. 20th Century Climate Warming and Tree-limit Rise in the Southern Scandes of Sweden. Ambio 2001, 30, 72–80.
- Devictor, V.; van Swaay, C.; Brereton, T.; Brotons, L.; Chamberlain, D.; Heliölä, J.; Herrando, S.; Julliard, R.; Kuussaari, M.; Lindström, Å.; et al. Differences in the climatic debts of birds and butterflies at a continental scale. Nat. Clim. Chang. 2012, 2, 121–124.
- Penteriani, V.; Zarzo-Arias, A.; Novo-Fernandez, A.; Bombieri, G.; Lopez-Sanchez, C.A. Responses of an endangered brown bear population to climate change based on predictable food resource and shelter alterations. Glob. Chang. Biol. 2019, 25, 1133–1151.
- Araujo, M.B.; Alagador, D.; Cabeza, M.; Nogues-Bravo, D.; Thuiller, W. Climate change threatens European conservation areas. Ecol. Lett. 2011, 14, 484–492.
- Wessely, J.; Hülber, K.; Gattringer, A.; Kuttner, M.; Moser, D.; Rabitsch, W.; Schindler, S.; Dullinger, S.; Essl, F. Habitat-based conservation strategies cannot compensate for climate-change-induced range loss. Nat. Clim. Chang. 2017, 7, 823–827.
- Virkkala, R.; Heikkinen, R.K.; Fronzek, S.; Kujala, H.; Leikola, N. Does the protected area network preserve bird species of conservation concern in a rapidly changing climate? Biodivers. Conserv. 2012, 22, 459–482.
- Rüter, S.; Vos, C.C.; van Eupen, M.; Rühmkorf, H. Transboundary ecological networks as an adaptation strategy to climate change: The example of the Dutch—German border. Basic Appl. Ecol. 2014, 15, 639–650.
- Hamann, A.; Roberts, D.R.; Barber, Q.E.; Carroll, C.; Nielsen, S.E. Velocity of climate change algorithms for guiding conservation and management. Glob. Chang. Biol. 2015, 21, 997–1004.
- Brito-Morales, I.; Garcia Molinos, J.; Schoeman, D.S.; Burrows, M.T.; Poloczanska, E.S.; Brown, C.J.; Ferrier, S.; Harwood, T.D.; Klein, C.J.; McDonald-Madden, E.; et al. Climate Velocity Can Inform Conservation in a Warming World. Trends Ecol. Evol. 2018, 33, 441–457.
- Dobrowski, S.Z.; Parks, S.A. Climate change velocity underestimates climate change exposure in mountainous regions. Nat. Commun. 2016, 7, 12349.
- Carroll, C.; Parks, S.A.; Dobrowski, S.Z.; Roberts, D.R. Climatic, topographic, and anthropogenic factors determine connectivity between current and future climate analogs in North America. Glob. Chang. Biol. 2018, 24, 5318–5331.
- Senior, R.A.; Hill, J.K.; Edwards, D.P. Global loss of climate connectivity in tropical forests. Nat. Clim. Chang. 2019, 9, 623–626.
- Fick, S.E.; Hijmans, R.J. WorldClim 2: New 1-km spatial resolution climate surfaces for global land areas. Int. J. Climatol. 2017, 37, 4302–4315.
- Chen, I.C.; Hill, J.K.; Ohlemuller, R.; Roy, D.B.; Thomas, C.D. Rapid range shifts of species associated with high levels of climate warming. Science 2011, 333, 1024–1026.





