
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Wen-Chi Su | + 2338 word(s) | 2338 | 2021-07-09 08:38:57 | | | |
| 2 | Amina Yu | Meta information modification | 2338 | 2021-07-20 03:42:53 | | | | |
| 3 | Amina Yu | Meta information modification | 2338 | 2021-07-21 11:55:25 | | |
Video Upload Options
Amino acids have been implicated with virus infection and replication. Here, we demonstrate the effects of two basic amino acids, arginine and lysine, and their ester derivatives on infection of two enveloped viruses, SARS-CoV-2, and influenza A virus. We found that lysine and its ester derivative can efficiently block infection of both viruses in vitro. Furthermore, the arginine ester derivative caused a significant boost in virus infection. Studies on their mechanism of action revealed that the compounds potentially disturb virus uncoating rather than virus attachment and endosomal acidification. Our findings suggest that lysine supplementation and the reduction of arginine-rich food intake can be considered as prophylactic and therapeutic regimens against these viruses while also providing a paradigm for the development of broad-spectrum antivirals.
1. Introduction
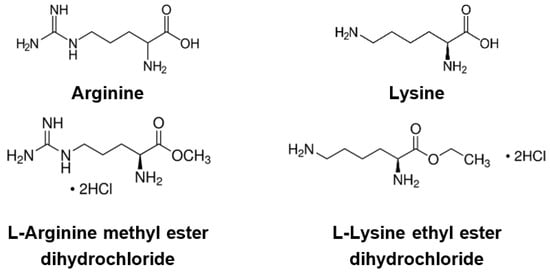
2. Effects of Basic Amino Acids and Their Derivatives on SARS-CoV-2 and Influenza-A Virus Infection
2.1. Lysine and Its Derivative Attenuate SARS-CoV-2 Vpp Infection
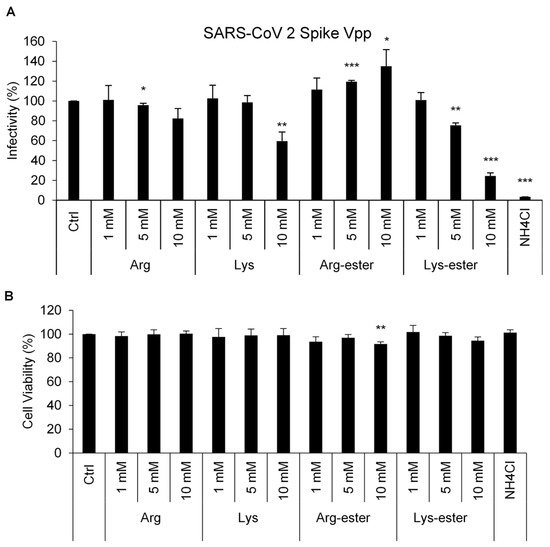
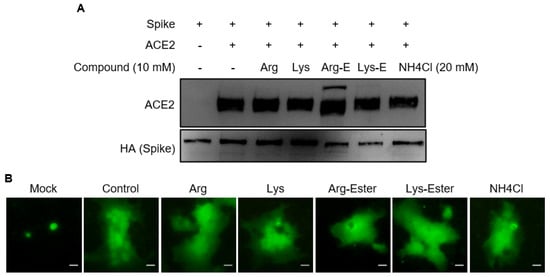
2.2. The Compounds Do Not Interfere the Interaction between Spike and ACE2
2.3. Lysine and Lys-Ester Inhibit IAV Replication
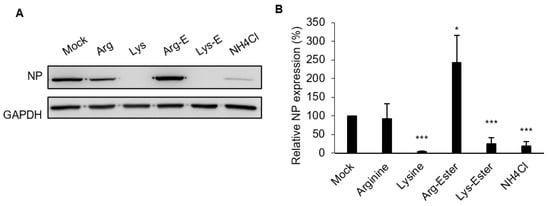
2.4. The Compounds Do Not Affect IAV Binding and Internalization
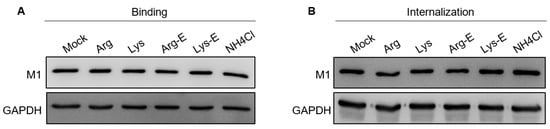
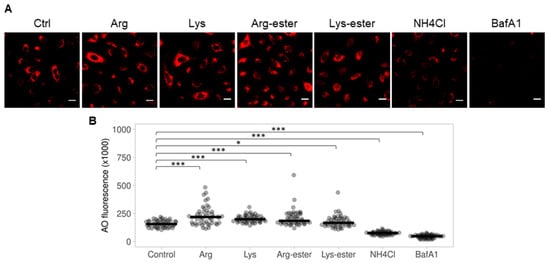
2.5. The Compounds Do Not Reduce Endosomal Acidification
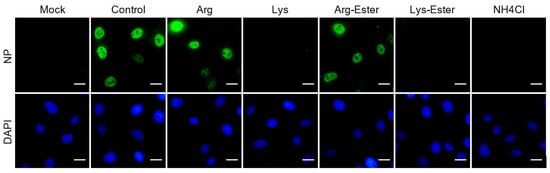
2.6. Lysine and Lys-Ester Reduce Nuclear Distribution of IAV
3. Conclusions
References
- Liu, Y.; Gayle, A.A.; Wilder-Smith, A.; Rocklöv, J. The reproductive number of COVID-19 is higher compared to SARS coronavirus. J. Travel Med. 2020, 27, taaa021.
- Hu, T.; Liu, Y.; Zhao, M.; Zhuang, Q.; Xu, L.; He, Q. A comparison of COVID-19, SARS and MERS. PeerJ 2020, 8, e9725.
- Kaur, N.; Singh, R.; Dar, Z.; Bijarnia, R.K.; Dhingra, N.; Kaur, T. Genetic comparison among various coronavirus strains for the identification of potential vaccine targets of SARS-CoV2. Infect. Genet. Evol. 2021, 89, 104490.
- Ngai, J.C.; Ko, F.W.; Ng, S.S.; To, K.-W.; Tong, M.; Hui, D.S. The long-term impact of severe acute respiratory syndrome on pulmonary function, exercise capacity and health status. Respirology 2010, 15, 543–550.
- Rubin, R. As their numbers grow, COVID-19 “Long Haulers” stump experts. JAMA 2020, 324, 1381–1383.
- Arbeitskreis Blut, U.B.B.K. Influenza virus. Transfus. Meds. Hemother. 2009, 36, 32–39.
- Hashemi, S.A.; Safamanesh, S.; Ghasemzadeh-moghaddam, H.; Ghafouri, M.; Azimian, A. High prevalence of SARS-CoV-2 and influenza A virus (H1N1) coinfection in dead patients in Northeastern Iran. J. Med. Virol. 2021, 93, 1008–1012.
- Bai, L.; Zhao, Y.; Dong, J.; Liang, S.; Guo, M.; Liu, X.; Wang, X.; Huang, Z.; Sun, X.; Zhang, Z.; et al. Co-infection of influenza A virus enhances SARS-CoV-2 infectivity. bioRxiv 2020.
- Zhang, A.J.; Lee, A.C.; Chan, J.F.; Liu, F.; Li, C.; Chen, Y.; Chu, H.; Lau, S.Y.; Wang, P.; Chan, C.C.; et al. Co-infection by severe acute respiratory syndrome coronavirus 2 and influenza A(H1N1)pdm09 virus enhances the severity of pneumonia in golden Syrian hamsters. Clin. Infect. Dis. 2020, 72, e978–e992.
- Lakadamyali, M.; Rust, M.J.; Zhuang, X. Endocytosis of influenza viruses. Microbes Infect. 2004, 6, 929–936.
- Li, S.; Sieben, C.; Ludwig, K.; Höfer, C.T.; Chiantia, S.; Herrmann, A.; Eghiaian, F.; Schaap, I.A.T. pH-Controlled two-step uncoating of influenza virus. Biophys. J. 2014, 106, 1447–1456.
- Ou, X.; Liu, Y.; Lei, X.; Li, P.; Mi, D.; Ren, L.; Guo, L.; Guo, R.; Chen, T.; Hu, J.; et al. Characterization of spike glycoprotein of SARS-CoV-2 on virus entry and its immune cross-reactivity with SARS-CoV. Nat. Commun. 2020, 11, 1620.
- Helenius, A. Virus entry: What has pH got to do with it? Nat. Cell Biol 2013, 15, 125.
- V’kovski, P.; Kratzel, A.; Steiner, S.; Stalder, H.; Thiel, V. Coronavirus biology and replication: Implications for SARS-CoV-2. Nat. Rev. Microbiol. 2021, 19, 155–170.
- Repnik, U.; Česen, M.H.; Turk, B. The endolysosomal system in cell death and survival. Cold Spring Harb. Perspect. Biol. 2013, 5, a008755.
- Hoffmann, M.; Kleine-Weber, H.; Schroeder, S.; Krüger, N.; Herrler, T.; Erichsen, S.; Schiergens, T.S.; Herrler, G.; Wu, N.-H.; Nitsche, A.; et al. SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell 2020, 181, 271–280.e278.
- Shang, J.; Wan, Y.; Luo, C.; Ye, G.; Geng, Q.; Auerbach, A.; Li, F. Cell entry mechanisms of SARS-CoV-2. Proc. Natl. Acad. Sci. USA 2020, 117, 11727–11734.
- Tang, T.; Bidon, M.; Jaimes, J.A.; Whittaker, G.R.; Daniel, S. Coronavirus membrane fusion mechanism offers a potential target for antiviral development. Antivir. Res. 2020, 178, 104792.
- Chitalia, V.C.; Munawar, A.H. A painful lesson from the COVID-19 pandemic: The need for broad-spectrum, host-directed antivirals. J. Transl. Med. 2020, 18, 390.
- Becht, H. Induction of an arginine-rich component during infection with influenza virus. J. Gen. Virol. 1969, 4, 215–220.
- Eaton, M.D.; Scala, A.R.; Low, I.E. Amino acid imbalance and incomplete viral replication. Arch. Für Die Gesamte Virusforsch. 1964, 14, 583–598.
- Griffith, R.S.; DeLong, D.C.; Nelson, J.D. Relation of arginine-lysine antagonism to herpes simplex growth in tissue culture. Chemotherapy 1981, 27, 209–213.
- Rossi, M.; Jacobs, B. Chapter 20—Herpes simplex virus. In Integrative Medicine (Fourth Edition); Rakel, D., Ed.; Elsevier: Amsterdam, The Netherlands, 2018; pp. 191–197.e192.
- Luiking, Y.C.; Deutz, N.E.P. Biomarkers of arginine and lysine excess. J. Nutr. 2007, 137, 1662S–1668S.
- Mailoo, V.J.; Rampes, S. Lysine for herpes simplex prophylaxis: A review of the evidence. Integr. Med. (Encinitas) 2017, 16, 42–46.
- Griffith, R.S.; Norins, A.L.; Kagan, C. A multicentered study of lysine therapy in Herpes simplex infection. Dermatologica 1978, 156, 257–267.
- Griffith, R.S.; Walsh, D.E.; Myrmel, K.H.; Thompson, R.W.; Behforooz, A. Success of L-lysine therapy in frequently recurrent herpes simplex infection. Treatment and prophylaxis. Dermatologica 1987, 175, 183–190.
- Thein, D.J.; Hurt, W.C. Lysine as a prophylactic agent in the treatment of recurrent herpes simplex labialis. Oral Surg. Oral Med. Oral Pathol. 1984, 58, 659–666.
- Al-Bari, M.A.A. Targeting endosomal acidification by chloroquine analogs as a promising strategy for the treatment of emerging viral diseases. Pharm. Res. Perspect 2017, 5, e00293.
- Dabydeen, S.A.; Meneses, P.I. The role of NH4Cl and cysteine proteases in Human Papillomavirus type 16 infection. Virol. J. 2009, 6, 109.
- Matlin, K.S. Ammonium chloride slows transport of the influenza virus hemagglutinin but does not cause mis-sorting in a polarized epithelial cell line. J. Biol. Chem. 1986, 261, 15172–15178.
- Oomens, A.G.P.; Wertz, G.W. The baculovirus GP64 protein mediates highly stable infectivity of a human respiratory syncytial virus lacking its homologous transmembrane glycoproteins. J. Virol. 2004, 78, 124–135.
- Su, W.-C.; Chen, Y.-C.; Tseng, C.-H.; Hsu, P.W.-C.; Tung, K.-F.; Jeng, K.-S.; Lai, M.M.C. Pooled RNAi screen identifies ubiquitin ligase Itch as crucial for influenza A virus release from the endosome during virus entry. Proc. Natl. Acad. Sci. USA 2013, 110, 17516–17521.
- Krolenko, S.A.; Adamyan, S.Y.; Belyaeva, T.N.; Mozhenok, T.P. Acridine orange accumulation in acid organelles of normal and vacuolated frog skeletal muscle fibres. Cell Biol. Int. 2006, 30, 933–939.
- Bayer, N.; Schober, D.; Prchla, E.; Murphy, R.F.; Blaas, D.; Fuchs, R. Effect of bafilomycin A1 and nocodazole on endocytic transport in HeLa cells: Implications for viral uncoating and infection. J. Virol. 1998, 72, 9645–9655.
- Shulla, A.; Heald-Sargent, T.; Subramanya, G.; Zhao, J.; Perlman, S.; Gallagher, T. A transmembrane serine protease is linked to the severe acute respiratory syndrome coronavirus receptor and activates virus entry. J. Virol. 2011, 85, 873–882.





