| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Elena Codruta Cozma | + 1744 word(s) | 1744 | 2021-07-05 11:06:14 | | | |
| 2 | Vivi Li | Meta information modification | 1744 | 2021-07-20 03:58:31 | | | | |
| 3 | Conner Chen | Meta information modification | 1744 | 2021-09-22 04:39:24 | | |
Video Upload Options
Melanomas of the skin are poorly circumscribed lesions, very frequently asymptomatic but unfortunately with a continuous growing incidence. In this landscape, one can distinguish melanomas originating in the mucous membranes and located in areas not exposed to the sun, namely the vulvo-vaginal melanomas. By contrast with cutaneous melanomas, the incidence of these types of melanomas is constant, being diagnosed in females in their late sixties. While hairy skin and glabrous skin melanomas of the vulva account for 5% of all cancers located in the vulva, melanomas of the vagina and urethra are particularly rare conditions. The location in areas less accessible to periodic inspection determines their diagnosis in advanced stages, often metastatic.
1. Introduction
2. Risk Factors
3. Clinical Manifestations
| Number of Patients |
Tumor Localisation | Main Signs and Symptoms |
Others Signs and Symptoms |
References |
|---|---|---|---|---|
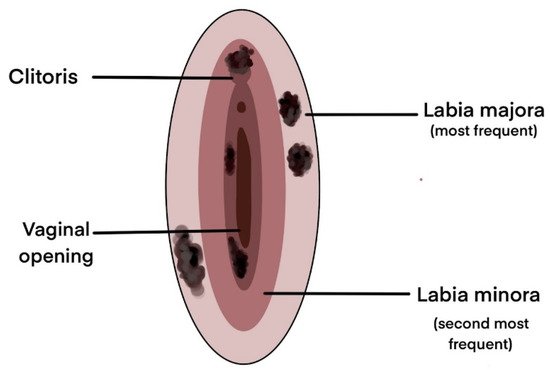
| 51 | Labia minora | Pain, Palpable mass, Genital bleeding, Pruritus | Dysuria, Ulceration | [53] |
| 20 | Labia majora | Genital bleeding, Pruritus, Palpable mass | Pain, Dysuria, Unhealing sore, Urinary difficulties | [32] |
| 10 | Labia majora | Pruritus | - | [43] |
| 11 | Not specified | Pruritus, Pain, Genital bleeding | - | [56] |
| 14 | Labia minora | Pruritus | - | [54] |
| 31 | Vagina | Genital bleeding, Pain, Palpable mass | Abnormal vaginal secretion, Urinary difficulties | [55] |
| 33 | Not specified | Palpable mass, Genital bleeding, Pain, Pruritus | Abnormal vaginal secretion, Dysuria, Dyspareunia, Ulceration | [57] |
| 198 | Unilateral, Clitoris | Genital bleeding, Pain, Pruritus | - | [15] |
References
- Liu, Y.; Sheikh, M.S. Melanoma: Molecular Pathogenesis and Therapeutic Management. Mol. Cell. Pharmacol. 2014, 6, 228.
- Mort, R.L.; Jackson, I.J.; Patton, E.E. The Melanocyte Lineage in Development and Disease. Development 2015, 142, 620–632.
- Ali, S.A.; Naaz, I. Current Challenges in Understanding the Story of Skin Pigmentation—Bridging the Morpho-Anatomical and Functional Aspects of Mammalian Melanocytes. In Muscle Cell and Tissue; Sakuma, K., Ed.; InTech: London, UK, 2015.
- Skin Cancer Statistics. Available online: (accessed on 25 April 2021).
- Melanoma Skin Cancer Statistics. Available online: (accessed on 25 April 2021).
- Weir, H.K.; Marrett, L.D.; Cokkinides, V.; Barnholtz-Sloan, J.; Patel, P.; Tai, E.; Jemal, A.; Li, J.; Kim, J.; Ekwueme, D.U. Melanoma in Adolescents and Young Adults (Ages 15–39 Years): United States, 1999–2006. J. Am. Acad. Dermatol. 2011, 65, S38–S49.
- Olsen, C.M.; Thompson, J.F.; Pandeya, N.; Whiteman, D.C. Evaluation of Sex-Specific Incidence of Melanoma. JAMA Dermatol. 2020, 156, 553–560.
- Paulson, K.G.; Gupta, D.; Kim, T.S.; Veatch, J.R.; Byrd, D.R.; Bhatia, S.; Wojcik, K.; Chapuis, A.G.; Thompson, J.A.; Madeleine, M.M.; et al. Age-Specific Incidence of Melanoma in the United States. JAMA Dermatol. 2020, 156, 57–64.
- Davis, L.E.; Shalin, S.C.; Tackett, A.J. Current State of Melanoma Diagnosis and Treatment. Cancer Biol. Ther. 2019, 20, 1366–1379.
- Matthews, N.H.; Li, W.-Q.; Qureshi, A.A.; Weinstock, M.A.; Cho, E. Cutaneous Melanoma: Etiology and Therapy. Codon Publ. 2017.
- Corona, R.; Mele, A.; Amini, M.; De Rosa, G.; Coppola, G.; Piccardi, P.; Fucci, M.; Pasquini, P.; Faraggiana, T. Interobserver Variability on the Histopathologic Diagnosis of Cutaneous Melanoma and Other Pigmented Skin Lesions. J. Clin. Oncol. 1996, 14, 1218–1223.
- Troxel, D.B. Pitfalls in the Diagnosis of Malignant Melanoma: Findings of a Risk Management Panel Study. Am. J. Surg. Pathol. 2003, 27, 1278–1283.
- Trimble, E.L. Melanomas of the Vulva and Vagina. Oncol. Williston Park 1996, 10, 1017–1023.
- Qurrat ul Ain, Q.; Rao, B. A Rare Case Report: Malignant Vulvar Melanoma. Indian J. Gynecol. Oncol. 2020, 18.
- Boer, F.L.; Ten Eikelder, M.L.G.; Kapiteijn, E.H.; Creutzberg, C.L.; Galaal, K.; van Poelgeest, M.I.E. Vulvar Malignant Melanoma: Pathogenesis, Clinical Behaviour and Management: Review of the Literature. Cancer Treat. Rev. 2019, 73, 91–103.
- Ragnarsson-Olding, B.K. Primary Malignant Melanoma of the Vulva—An Aggressive Tumor for Modeling the Genesis of Non-UV Light-Associated Melanomas. Acta Oncol. 2004, 43, 421–435.
- Heinzelmann-Schwarz, V.A.; Nixdorf, S.; Valadan, M.; Diczbalis, M.; Olivier, J.; Otton, G.; Fedier, A.; Hacker, N.F.; Scurry, J.P. A Clinicopathological Review of 33 Patients with Vulvar Melanoma Identifies C-KIT as a Prognostic Marker. Int. J. Mol. Med. 2014, 33, 784–794.
- Wohlmuth, C.; Wohlmuth-Wieser, I. Vulvar Malignancies: An Interdisciplinary Perspective. J. Dtsch. Dermatol. Ges. 2019, 17, 1257–1276.
- Jamaer, E.; Liang, Z.; Stagg, B. Primary Malignant Melanoma of the Vagina. BMJ Case Rep. 2020, 13, e232200.
- Kalampokas, E.; Kalampokas, T.; Damaskos, C. Primary Vaginal Melanoma, A Rare and Aggressive Entity. A Case Report and Review of the Literature. In Vivo 2017, 31, 133–139.
- Wohlmuth, C.; Wohlmuth-Wieser, I.; May, T.; Vicus, D.; Gien, L.T.; Laframboise, S. Malignant Melanoma of the Vulva and Vagina: A US Population-Based Study of 1863 Patients. Am. J. Clin. Dermatol. 2020, 217, 285–295.
- Hu, D.-N.; Yu, G.-P.; McCormick, S.A. Population-Based Incidence of Vulvar and Vaginal Melanoma in Various Races and Ethnic Groups with Comparisons to Other Site-Specific Melanomas. Melanoma Res. 2010, 20, 153–158.
- You, J.S.; Jones, P.A. Cancer Genetics and Epigenetics: Two Sides of the Same Coin? Cancer Cell 2012, 22, 9–20.
- Rastrelli, M.; Tropea, S.; Rossi, C.R.; Alaibac, M. Melanoma: Epidemiology, Risk Factors, Pathogenesis, Diagnosis and Classification. In Vivo 2014, 28, 1005–1011.
- Gandini, S.; Sera, F.; Cattaruzza, M.S.; Pasquini, P.; Picconi, O.; Boyle, P.; Melchi, C.F. Meta-Analysis of Risk Factors for Cutaneous Melanoma: II. Sun Exposure. Eur. J. Cancer 2005, 41, 45–60.
- Bevona, C.; Goggins, W.; Quinn, T.; Fullerton, J.; Tsao, H. Cutaneous Melanomas Associated with Nevi. Arch. Dermatol. 2003, 139, 1620–1624.
- Stern, R.S.; PUVA Follow up Study. The Risk of Melanoma in Association with Long-Term Exposure to PUVA. J. Am. Acad. Dermatol. 2001, 44, 755–761.
- Mihajlovic, M.; Vlajkovic, S.; Jovanovic, P.; Stefanovic, V. Primary Mucosal Melanomas: A Comprehensive Review. Int. J. Clin. Exp. Pathol. 2012, 5, 739–753.
- Rapi, V.; Dogan, A.; Schultheis, B.; Hartmann, F.; Rezniczek, G.A.; Tempfer, C.B. Melanoma of the Vagina: Case Report and Systematic Review of the Literature. Anticancer Res. 2017, 37, 6911–6920.
- Stang, A.; Streller, B.; Eisinger, B.; Jöckel, K.H. Population-Based Incidence Rates of Malignant Melanoma of the Vulva in Germany. Gynecol. Oncol. 2005, 96, 216–221.
- Tasseron, E.W.; van der Esch, E.P.; Hart, A.A.; Brutel de la Rivière, G.; Aartsen, E.J. A Clinicopathological Study of 30 Melanomas of the Vulva. Gynecol. Oncol. 1992, 46, 170–175.
- Wechter, M.E.; Gruber, S.B.; Haefner, H.K.; Lowe, L.; Schwartz, J.L.; Reynolds, K.R.; Johnston, C.M.; Johnson, T.M. Vulvar Melanoma: A Report of 20 Cases and Review of the Literature. J. Am. Acad. Dermatol. 2004, 50, 554–562.
- Mert, I.; Semaan, A.; Winer, I.; Morris, R.T.; Ali-Fehmi, R. Vulvar/Vaginal Melanoma: An Updated Surveillance Epidemiology and End Results Database Review, Comparison with Cutaneous Melanoma and Significance of Racial Disparities. Int. J. Gynecol. Cancer 2013, 23, 1118–1126.
- Hassanein, A.M.; Mrstik, M.E.; Hardt, N.S.; Morgan, L.A.; Wilkinson, E.J. Malignant Melanoma Associated with Lichen Sclerosus in the Vulva of a 10-Year-Old. Pediatr. Dermatol. 2004, 21, 473–476.
- Hieta, N.; Kurki, S.; Rintala, M.; Söderlund, J.; Hietanen, S.; Orte, K. Association of Vulvar Melanoma with Lichen Sclerosus. Acta Derm. Venereol. 2019, 99, 339–340.
- Zekan, J.; Sirotkovic-Skerlev, M.; Skerlev, M. Oncogenic Aspects of HPV Infections of the Female Genital Tract. In DNA Replication-Current Advances; Seligmann, H., Ed.; InTech: London, UK, 2011.
- Münger, K.; Baldwin, A.; Edwards, K.M.; Hayakawa, H.; Nguyen, C.L.; Owens, M.; Grace, M.; Huh, K. Mechanisms of Human Papillomavirus-Induced Oncogenesis. J. Virol. 2004, 78, 11451–11460.
- Crum, C.P.; McLachlin, C.M.; Tate, J.E.; Mutter, G.L. Pathobiology of Vulvar Squamous Neoplasia. Curr. Opin. Obstet. Gynecol. 1997, 9, 63–69.
- Swetter, S.M.; Tsao, H.; Bichakjian, C.K.; Curiel-Lewandrowski, C.; Elder, D.E.; Gershenwald, J.E.; Guild, V.; Grant-Kels, J.M.; Halpern, A.C.; Johnson, T.M.; et al. Guidelines of Care for the Management of Primary Cutaneous Melanoma. J. Am. Acad. Dermatol. 2019, 80, 208–250.
- Edwards, L. Pigmented Vulvar Lesions: Pigmented Vulvar Lesions. Dermatol. Ther. 2010, 23, 449–457.
- Resende, F.S.; Conforti, C.; Giuffrida, R.; de Barros, M.H.; Zalaudek, I. Raised Vulvar Lesions: Be Aware! Dermatol. Pract. Concept. 2018, 8, 158–161.
- Sand, F.L.; Thomsen, S.F. Clinician’s Update on the Benign, Premalignant, and Malignant Skin Tumours of the Vulva: The Dermatologist’s View. Int. Sch. Res. Not. 2017, 2017, 2414569.
- De Simone, P.; Silipo, V.; Buccini, P.; Mariani, G.; Marenda, S.; Eibenschutz, L.; Ferrari, A.; Catricalà, C. Vulvar Melanoma: A Report of 10 Cases and Review of the Literature. Melanoma Res. 2008, 18, 127–133.
- Genton, C.Y.; Kunz, J.; Schreiner, W.E. Primary Malignant Melanoma of the Vagina and Cervix Uteri: Report of a Case with Utrastructural Study. Virchows Arch. A Path. Anat. Histol. 1981, 393, 245–250.
- Hewitt, P. Sequel to a Case of Recurrent Melanosis of Both Groins and Back: The Disease Reappearing in the Brain, Heart, Pancreas, Liver and Other Organs. Lancet 1861, 77, 263.
- Panizzon, R.G. Vulvar Melanoma. Semin. Dermatol. 1996, 15, 67–70.
- Pirlamarla, A.K.; Tang, J.; Amin, B.; Kabarriti, R. Vulvar Melanoma with Isolated Metastasis to the Extraocular Muscles: Case Report and Brief Literature Review. Anticancer Res. 2018, 38, 3763–3766.
- Campaner, A.B.; Fernandes, G.L.; de Cardoso, F.A.; Veasey, J.V. Vulvar Melanoma: Relevant Aspects in Therapeutic Management. An. Bras. Dermatol. 2017, 92, 398–400.
- Baderca, F.; Cojocaru, S.; Lazăr, E.; Lăzureanu, C.; Lighezan, R.; Alexa, A.; Raica, M.; Nicola, T. Amelanotic Vulvar Melanoma: Case Report and Review of the Literature. Rom. J. Morphol. Embryol. 2008, 49, 219–228.
- Filippetti, R.; Pitocco, R. Amelanotic Vulvar Melanoma: A Case Report. Am. J. Dermatopathol. 2015, 37, e75–e77.
- Mukeya, G.K.; Kakoka, I.M.; Mwansa, J.C.; Kalau, W.A. Mélanome malin vulvaire: À propos d’un cas observé à l’Hôpital du Cinquantenaire de Lubumbashi. Pan Afr. Med. J. 2020, 36, 124.
- Sinasac, S.E.; Petrella, T.M.; Rouzbahman, M.; Sade, S.; Ghazarian, D.; Vicus, D. Melanoma of the Vulva and Vagina: Surgical Management and Outcomes Based on a Clinicopathologic Reviewof 68 Cases. J. Obstet. Gynaecol. Can. 2019, 41, 762–771.
- Verschraegen, C.F.; Benjapibal, M.; Supakarapongkul, W.; Levy, L.B.; Ross, M.; Atkinson, E.N.; Bodurka-Bevers, D.; Kavanagh, J.J.; Kudelka, A.P.; Legha, S.S. Vulvar Melanoma at the M. D. Anderson Cancer Center: 25 Years Later. Int. J. Gynecol. Cancer 2001, 11, 359–364.
- Vaccari, S.; Barisani, A.; Salvini, C.; Pirola, S.; Preti, E.P.; Pennacchioli, E.; Iacobone, A.D.; Patrizi, A.; Tosti, G. Thin Vulvar Melanoma: A Challenging Diagnosis. Dermoscopic Features of a Case Series. Clin. Exp. Dermatol. 2020, 45, 187–193.
- Pandey, G.; Dave, P.; Patel, S.; Patel, B.; Arora, R.; Parekh, C.; Begum, D. Female Genital Tract Melanoma: Analysis from a Regional Cancer Institute. J. Turk. Soc. Obstet. Gynecol. 2020, 17, 46–51.
- Baiocchi, G.; Duprat, J.P.; Neves, R.I.; Fukazawa, E.M.; Landman, G.; Guimarães, G.C.; Valadares, L.J. Vulvar Melanoma: Report on Eleven Cases and Review of the Literature. Sao Paulo Med. J. 2010, 128, 38–41.
- Joste, M.; Dion, L.; Brousse, S.; Nyangoh Timoh, K.; Rousseau, C.; Reilhac, A.; Laviolle, B.; Lesimple, T.; Lavoue, V.; Leveque, J. Vulvar and Vaginal Melanomas: A Retrospective Study Spanning 19 Years from a Tertiary Center. J. Gynecol. Obstet. Hum. Reprod. 2021, 50, 102091.