
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | MEHMET ALI RECAI ONAL | + 1535 word(s) | 1535 | 2021-07-01 09:53:33 | | | |
| 2 | Beatrix Zheng | + 7 word(s) | 1542 | 2021-07-08 03:45:09 | | | | |
| 3 | Beatrix Zheng | + 7 word(s) | 1542 | 2021-07-08 03:57:46 | | |
Video Upload Options
The ferromanganese (FeMn) alloy is produced through the smelting-reduction of manganese ores in submerged arc furnaces. This process generates large amounts of furnace dust that is environmentally problematic for storage. Due to its fineness and high volatile content, this furnace dust cannot be recirculated through the process, either. Conventional MnO2 production requires the pre-reduction of low-grade ores at around 900 °C to convert the manganese oxides present in the ore into their respective acid-soluble forms; however, the furnace dust is a partly reduced by-product. A hydrometallurgical route is proposed to valorize the waste dust for the production of battery-grade MnO2. By using dextrin, a cheap organic reductant, the direct and complete dissolution of the manganese in the furnace dust is possible without any need for high-temperature pre-reduction. The leachate is then purified through pH adjustment followed by direct electrowinning for electrolytic manganese dioxide (EMD) production. An overall manganese recovery rate of >90% is achieved.
1. Introduction
2. Results and Discussions
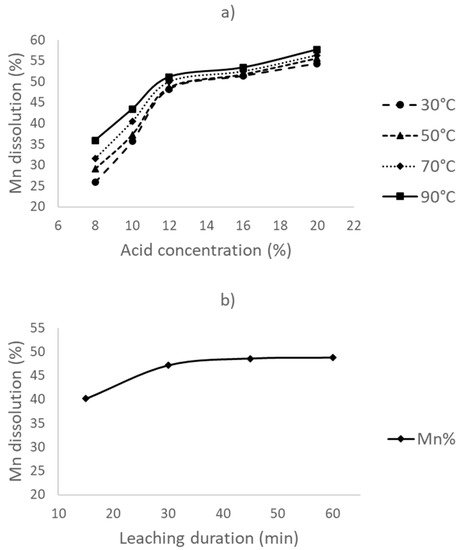
2.1. Leaching Experiments Without Reductant Addition (Direct Acid Leaching)
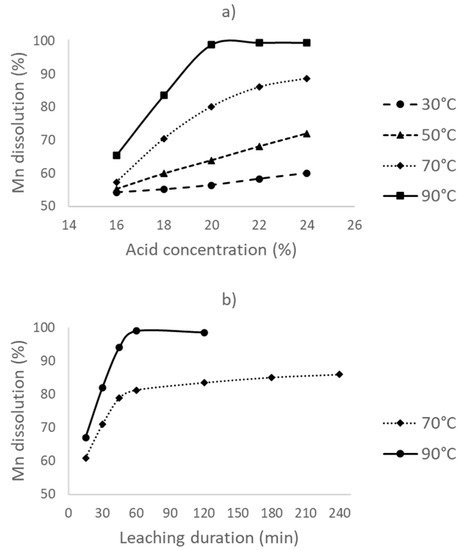
2.2. Leaching with Reductant Addition (Direct Reductive Leaching)
Through direct acid leaching experiments, it was found that maximum recovery of the manganese is less than 60% without any addition of a reductant. This is most likely due to the involvement of MnO2, which can only be effectively dissolved in sulfuric acid in a reducing atmosphere. In the second part of the leaching experiments, dextrin was added as the reductant.
3. Electrolytic MnO2 (EMD) Production

4. Conclusions
-
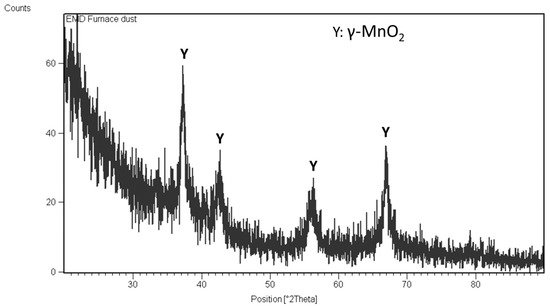
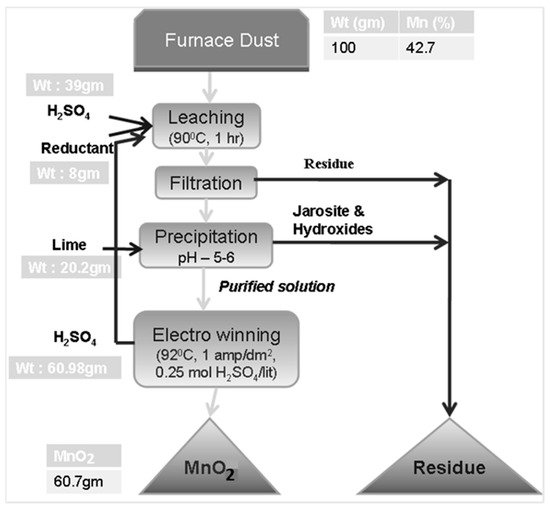
The particle size of the dust (D80) was 32 microns. The XRD result of the furnace dust revealed that Mn3O4, MnO, and silica phases were present in the sample. Mn content was 42.7%.
-
The leaching of the furnace dust without a reductant resulted in <60% Mn dissolution, with a 12% acid concentration, a 30 °C leaching temperature, and a 30 min leaching duration.
-
Dextrin, a green, cheap, and water-soluble organic reductant, was found suitable as a reductant.
-
98.9% Mn dissolution is obtained by the optimum leaching conditions of a 90 °C leaching temperature, a 1 h leaching duration, a 20% acid concentration, and an 8% dextrin addition.
-
Optimized conditions for electrolytic manganese dioxide (EMD) production were: a current density of 1 amp/dm2 and a sulfuric acid concentration of 0.25 M. The product obtained through the electrolysis process is a pure gamma MnO2 phase.
-
A total manganese recovery of 90.3% from the furnace dust is possible with the proposed flow sheet.
References
- Kronenberg, M.L.; Blomgren, G.E. Primary Batteries—Lithium Batteries. In Comprehensive Treatise of Electrochemistry; Bockris, J.O., Conway, B.E., Yeager, E., White, R.E., Eds.; Springer: Boston, MA, USA, 1981; pp. 247–278. ISBN 9781461566892.
- Sattar, R.; Ilyas, S.; Kousar, S.; Khalid, A.; Sajid, M.; Bukhari, S.I. Study on the reduction roasting of spent LiNixCoyMnzO2 lithium-ion battery cathode materials. Environ. Eng. Res. 2020, 25, 88–95.
- Munir, H.; Srivastava, R.R.; Kim, H.; Ilyas, S.; Khosa, M.K.; Yameen, B. Leaching of exhausted LNCM cathode batteries in ascorbic acid lixiviant: A green recycling approach, reaction kinetics and process mechanism. J. Chem. Technol. Biotechnol. 2020, 95, 2286–2294.
- Biswal, A.; Tripathy, B.C.; Sanjay, K.; Subbaiah, T.; Minakshi, M. Electrolytic manganese dioxide (EMD): A perspective on worldwide production, reserves and its role in electrochemistry. RSC Adv. 2015, 5, 58255–58283.
- de Araujo, J.A.M.; de Castro, M.M.R.; Lins, V.F.C. Reuse of furnace fines of ferro alloy in the electrolytic manganese production. Hydrometallurgy 2006, 84, 204–210.
- Matricardi, L.R. Disposal of ferromanganese furnace fume. In Proceedings of the Electric Furnace Conference, Houston, TX, USA, 9–12 December 1975; pp. 73–75.
- Lee, Y.E.; Kozak, D.S. The role of zinc in the eruption of high carbon FeMn smelting furnace. In Proceedings of the Electric Furnace Conference, Washington, DC, USA, 7–10 November 1993; pp. 145–150.
- Nkosi, S.; Steenkamp, J.D.; Groot, D.R.; Gous, J.P. Hydrometallurgical process for the recovery of manganese from pelletized silicomanganese submerged arc furnace dust. In Proceedings of the Fray International Symposium, Cancun, Mexico, 4–7 December 2011; pp. 309–320.
- Hamano, T.; Zhang, G.; Brown, P.; Ostrovski, O. Manganese furnace dust: Drying and reduction of zinc oxide by tar. ISIJ Int. 2008, 48, 906–911.
- Shen, R.; Zhang, G.; Dell’Amico, M.; Brown, P.; Ostrovski, O. A feasibility study of recycling of manganese furnace dust. In Proceedings of the XI International Conference on Innovations in the Ferro Alloy Industry, Infacon XI, Maharashtra, India, 18–21 February 2007; pp. 507–519.





