
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Jitendra Kumar Sinha | + 2572 word(s) | 2572 | 2021-07-01 05:25:59 | | | |
| 2 | Lindsay Dong | Meta information modification | 2572 | 2021-07-06 03:14:17 | | |
Video Upload Options
Epilepsy is a non-communicable disease of the brain that affects people of all ages. It is characterized by episodes of spontaneous and abnormal electrical activity in the brain. It is often accompanied by depression, anxiety, and substantially increased morbidity and mortality. A large number of third-generation antiepileptic drugs are available, but they have multiple side effects causing a decline in the quality of life. The inheritance and etiology of epilepsy are complex with multiple underlying genetic and epigenetic mechanisms. Different neurotransmitters play intricate functions to maintain the normal physiology of various neurons.
1. Introduction
2. Role of Genes, Genetics and Inheritance
The newly emerging genetic technology has played a significant role in the discovery of a variety of genes that are associated with epilepsy. The advancement of genomic techniques and gene sequencing has substantially enhanced the knowledge about the genetic variations taking place in the human genome. Studies estimate that there is an underlying genetic cause in about half of all cases of epilepsy [8]. Currently, with the emerging research of epigenetic biomarkers, MicroRNAs (miRNAs) have been assumed to play a significant role. MiRNA molecules 19-25 nucleotides long regulate gene expression as post-transcriptional modifiers [9]. Differential expression of more than 100 miRNAs has been reported in epilepsy. Among them, because of their predominant role in biological processes related to epilepsy, such as neurodegeneration, neuronal growth, and neuroinflammation, miR-132, miR-155, and miR-146a have been highlighted primarily [9]. Some cases of genetic mutations result in the core symptom of epilepsy, while changes in a few of the genes are responsible for malformations in the gross development of the brain that cause seizures. There are around 84 genes classified as epilepsy genes based on the OMIM database results. Mutation in these classified genes leads to epilepsy as a core symptom. There are approximately 73 genes that are categorized as neurodevelopment-associated epilepsy genes [8]. According to the OMIM database (https://www.ncbi.nlm.nih.gov/omim, accessed on 24 April 2021), there are about 536 genes responsible for causing associated diseases of epilepsy [8][10].
3. Epigenetics Involved in Epilepsy
Recent studies show epigenetics playing essential roles in temporal lobe epilepsy [11]. Therefore, studying the role of epigenetic changes in the development of the disease has become an emerging topic in the area of research. The knowledge of epigenetic mechanisms helps in providing the putative conceptual framework in the development of therapies that can help in the prevention of the disease. Epileptogenesis should be considered as a target point for developing therapy when there is an increment in the severity and frequency of impulsive recurrent seizures. Various processes that take place along with epileptogenesis are mossy fiber sprouting, dysfunction of adenosine together with gliosis, aberrant connectivity, neuronal cell loss, and neuroinflammation [12][13]. TCF4, MECP2, UBE3A, and CHD2 are some of the regulatory genes associated with epilepsy. Among these, the CHD2 gene is responsible for encoding a protein that remodels chromatin, and deregulation of CHD2 might have a downstream effect on other genes [14].
Epigenetic modifications are also responsible for many of the pathological changes that take place during epileptogenesis. Many changes have been shown to occur in the central nervous system cells that alter the gene expression due to DNA methylation and histone acetylation and methylation [15][16].
Various processes like histone modifications that involve either adding up or eliminating the acetyl or methyl groups are suggested to be associated with epileptogenesis. According to the hypothesis of DNA methylation implicated in epileptogenesis, seizures can induce epigenetic modifications and can exaggerate the process of epileptogenesis. DNA hypermethylation, along with the amplified activity of DNA methylating enzymes has been implicated in the development of experimental and human epilepsy [17].
Histone modification is one of the epigenetic mechanisms that have the considerable potential to alter the neuronal expression of genes by exerting their additive effects in a correlated manner [11]. The principal role of histone proteins is to support the tertiary and quaternary structure of the DNA. Disruption in these vital epigenetic machinery leads to various disorders such as epilepsy, autism, Rett syndrome, etc.
DNA methylation is dependent on several biochemical enzymatic reactions. One such reaction pathway is the S-adenosylmethionine-dependent transmethylation pathway, which is controlled by glycine and adenosine under the regulation of adenosine kinase (ADK). In chronic epilepsy, it is observed that there is an increase in the ADK and a resulting decline in adenosine, which leads to elevated DNA methylation in the brain. Thus, interference with methylation of DNA gives the new conceptual prospect to control and prevent epilepsy. Glycine modifying therapies can also be regarded as an alternative opportunity that affects the process of DNA methylation and, ultimately, the process of epileptogenesis. Therefore, understanding the epigenetics of epileptogenesis might help in the discovery and development of therapeutic interventions [12][18].
4. Neurotransmitter Release Machinery in Epilepsy
4.1. Glutamate Receptors
Glutamate is an excitatory neurotransmitter responsible for stimulating an increase in calcium and sodium conduction through ligand-gated ion channels (Figure 1). A wide spectrum of anti-convulsant properties is displayed by AMPA antagonists and NMDA antagonists in animal models with acute and chronic epilepsy [19]. Once seizures begin, the activity-dependent plasticity of the glutamate receptors becomes a vital feature of the epileptic brain.
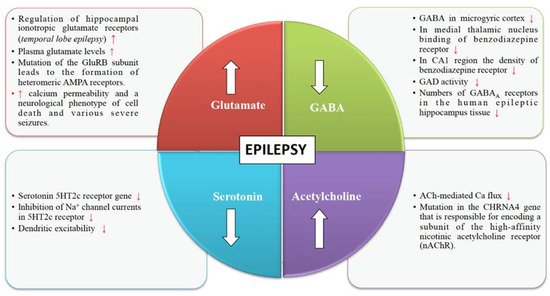
4.2. GABA Receptors
Various pieces of evidence from several clinical and experimental data underline the role of GABA in the mechanism and management of epilepsy (Figure 1). The synaptic inhibition by GABA plays an essential role in regulating neuronal excitability, which has been linked to epilepsy. GABAergic neurotransmission that is controlled by Cl– permeable GABAA receptors can exhibit both seizure-repressing and -stimulating activity [20].
4.3. Cholinergic Receptors
The structural and functional diversity of the neuronal nicotinic acetylcholine receptors (nAChRs) perform modulatory functions throughout the mammalian brain (Figure 1). Nicotinic receptors are involved in various developmental mechanisms such as memory, attention, and learning. Disruptions in cholinergic mechanisms can lead to several disorders such as epilepsy, Parkinson’s disease, dementia, schizophrenia, autism, Alzheimer’s disease, etc. [21].
4.4. Serotonin Receptors
Serotonergic neurotransmission has been shown to have a potential role in epilepsy [22]. Serotonin receptors (5-HTRs) have been therefore considered as promising candidate targets for the development of new AEDs (Figure 1). Studies show 5-HT regulates a wide variety of focal and generalized seizures. Agents like 5-hydroxytryptophan and 5-HT reuptake blockers are known to increase the extracellular serotonin levels and hence inhibit focal as well as generalized seizures.
5. Drug Resistant Epilepsy
Drug-resistant epilepsy (DRE) is also known as refractory epilepsy or pharmaco-resistant epilepsy. It can be defined as a failure of two or more sufficient trials of tolerated, chosen, and appropriately used AEDs regimens, which can be administered as monotherapies or in combination to get relief from seizures. Around one out of four patients with seizures develop DRE [23]. DRE patients have increased risks of injuries, psychosocial problems, and premature death [24][25][26].
5.1. Alterations in the Drug Targets
5.2. The Inability of the Drugs to Reach Their Targets
5.3. Real Targets Missed by the Drugs
6. Non-Conventional Therapeutic Strategies
6.1. Ruling Out Pseudo-Resistance
6.2. Combination Therapy
6.3. Non-Drug Treatment
7. Modern Approaches for Treatment
7.1. MTOR Pathway
7.2. Inflammatory Pathways
7.3. Breakdown of Blood-Brain Barrier
| S. No. | Treatment Approaches | Interventions Used | Action Mechanism | Main Uses | References |
|---|---|---|---|---|---|
| A. | PHARMACEUTICAL APPROACHES | Gabapentin | Ca2+ blockage | Used for generalised and focal seizures. | [40][41] |
| (Anti-epileptic drugs) | Carbamazepine | Na+ channel blockage | Decrease nerve impulses that are responsible for causing seizures. | [40] | |
| Lamotrigine | Na+ channel blockage | Used as a first- line drug for generalized and focal seizures. | [42][41] | ||
| Tiagabine | GABA potentiation | Used for partial seizures in adjunctive therapy. | [40] | ||
| Zonisamide | Na+ channel blockage | Used for generalized and focal seizures. | [40][41] | ||
| Vigabatrin | GABA potentiation | Used for infantile spasms and for focal onset of seizures. | [40] | ||
| Perampanel | Glutamate (AMPA) antagonist | Used for partial seizures with focal onset. | [40][41] | ||
| B. | THERAPEUTIC APPROACHES | Progressive muscle relaxation | Tense a group of muscles while breathing in and relaxes them while breathing out. | Improves sleep and overall well-being. Enhances control over epilepsy by the patients. | [43] |
| Yoga | Release tension in key joints through combination of body postures. | Decrease in automatic dysfunction during onset of seizures. | [43][44] | ||
| Cognitive behavioural therapy | Restructuring of maladaptive thought patterns. | Improvement in anxiety and depression and enhanced psychosocial functioning. | [43][45] | ||
| Vagus nerve stimulation | Used to generate impulse through electric current in vagus nerve. | Used as an adjunctive therapy for partial onset of seizures. | [46] | ||
| C. | NATURAL APPROACHES | Ketogenic diet | Neurotransmitter modulation in brain by ketone bodies. | Successful in reducing seizures and enhancing motor function. | [47][48][49] |
| Vitamin D3 | Increase Ca2+ uptake and decrease neuronal excitability. | Produces anti-convulsant effect and prevent seizures. | [50][51] | ||
| Herbal treatments | Found to be involved in potentiation of GABAergic activity in brain. | Herbal medications control epileptic seizures and reduce side effects and increase cognitive effects of AEDs. | [52][53] |
8. Conclusions
References
- Scharfman, H.E. The neurobiology of epilepsy. Curr. Neurol. Neurosci. Rep. 2007, 7, 348–354.
- Kobylarek, D.; Iwanowski, P.; Lewandowska, Z.; Limphaibool, N.; Szafranek, S.; Labrzycka, A.; Kozubski, W. Advances in the Potential Biomarkers of Epilepsy. Front. Neurol. 2019, 10, 685.
- Fisher, R.S.; Acevedo, C.; Arzimanoglou, A.; Bogacz, A.; Cross, J.H.; Elger, C.E.; Engel, J.; Forsgren, L.; French, J.A.; Glynn, M.; et al. ILAE Official Report: A practical clinical definition of epilepsy. Epilepsia 2014, 55, 475–482.
- Löscher, W.; Klitgaard, H.; Twyman, R.E.; Schmidt, D. New avenues for anti-epileptic drug discovery and development. Nat. Rev. Drug Discov. 2013, 12, 757–776.
- Schmidt, D. Is antiepileptogenesis a realistic goal in clinical trials? Concerns and new horizons. Epileptic Disord 2012, 14, 105–113.
- Brodie, M.J.; Barry, S.J.E.; Bamagous, G.A.; Norrie, J.D.; Kwan, P. Patterns of treatment response in newly diagnosed epilepsy. Neurology 2012, 78, 1548–1554.
- Mishra, P.; Mittal, A.K.; Rajput, S.K.; Sinha, J.K. Cognition and memory impairment attenuation via reduction of oxidative stress in acute and chronic mice models of epilepsy using antiepileptogenic Nux vomica. J. Ethnopharmacol. 2021, 267, 113509.
- Wang, J.; Lin, Z.-J.; Liu, L.; Xu, H.-Q.; Shi, Y.-W.; Yi, Y.-H.; He, N.; Liao, W.-P. Epilepsy-associated genes. Seizure 2017, 44, 11–20.
- Martins-Ferreira, R.; Chaves, J.; Carvalho, C.; Bettencourt, A.; Chorão, R.; Freitas, J.; Samões, R.; Boleixa, D.; Lopes, J.; Ramalheira, J.; et al. Circulating microRNAs as potential biomarkers for genetic generalized epilepsies: A three microRNA panel. Eur. J. Neurol. 2020, 27, 660–666.
- Grønskov, K.; Brøndum-Nielsen, K.; Dedic, A.; Hjalgrim, H. A nonsense mutation in FMR1 causing fragile X syndrome. Eur. J. Hum. Genet. 2011, 19, 489–491.
- Toth, M. Epigenetic Neuropharmacology: Drugs Affecting the Epigenome in the Brain. Annu. Rev. Pharmacol. Toxicol. 2021, 61, 181–201.
- Eboison, D. The Biochemistry and Epigenetics of Epilepsy: Focus on Adenosine and Glycine. Front. Mol. Neurosci. 2016, 9, 26.
- Kobow, K.; Blümcke, I. The emerging role of DNA methylation in epileptogenesis. Epilepsia 2012, 53, 11–20.
- Symonds, J.D. CHD2 epilepsy: Epigenetics and the quest for precision medicine. Dev. Med. Child Neurol. 2019, 62, 549–550.
- Caramaschi, D.; Hatcher, C.; Mulder, R.H.; Felix, J.F.; Cecil, C.A.M.; Relton, C.L.; Walton, E. Epigenome-wide association study of seizures in childhood and adolescence. Clin. Epigenet. 2020, 12, 1–13.
- Feng, J.; Zhou, Y.; Campbell, S.L.; Le, T.; Li, E.; Sweatt, J.D.; Silva, A.J.; Fan, G. Dnmt1 and Dnmt3a maintain DNA methylation and regulate synaptic function in adult forebrain neurons. Nat. Neurosci. 2010, 13, 423–430.
- Henshall, D.C.; Kobow, K. Epigenetics and Epilepsy. Cold Spring Harb. Perspect. Med. 2015, 5, a022731.
- Boison, D. Adenosine Kinase: Exploitation for Therapeutic Gain. Pharmacol. Rev. 2013, 65, 906–943.
- Chapman, A.G. Glutamate and Epilepsy. J. Nutr. 2000, 130, 1043S–1045S.
- Wang, Y.; Wang, Y.; Chen, Z. Double-edged GABAergic synaptic transmission in seizures: The importance of chloride plasticity. Brain Res. 2018, 1701, 126–136.
- Dani, J.A.; Bertrand, D. Nicotinic Acetylcholine Receptors and Nicotinic Cholinergic Mechanisms of the Central Nervous System. Annu. Rev. Pharmacol. Toxicol. 2007, 47, 699–729.
- Bourin, M. Mechanisms of Action of Anxiolytics. In Psychiatry and Neuroscience Update; Springer Science and Business Media LLC: New York, NY, USA, 2021; pp. 195–211.
- Kwan, P.; Schachter, S.C.; Brodie, M.J. Drug-Resistant Epilepsy. N. Engl. J. Med. 2011, 365, 919–926.
- Mohanraj, R.; Norrie, J.; Stephen, L.J.; Kelly, K.; Hitiris, N.; Brodie, M.J. Mortality in adults with newly diagnosed and chronic epilepsy: A retrospective comparative study. Lancet Neurol. 2006, 5, 481–487.
- Lawn, N.D.; Bamlet, W.R.; Radhakrishnan, K.; O’Brien, P.C.; So, E.L. Injuries due to seizures in persons with epilepsy: A population-based study. Neurology 2004, 63, 1565–1570.
- McCagh, J.; Fisk, J.E.; Baker, G.A. Epilepsy, psychosocial and cognitive functioning. Epilepsy Res. 2009, 86, 1–14.
- Loup, F.; Picard, F.; Yonekawa, Y.; Wieser, H.-G.; Fritschy, J.-M. Selective changes in GABAA receptor subtypes in white matter neurons of patients with focal epilepsy. Brain 2009, 132, 2449–2463.
- Remy, S.; Beck, H. Molecular and cellular mechanisms of pharmacoresistance in epilepsy. Brain 2005, 129, 18–35.
- Tishler, D.M.; Weinberg, K.I.; Hinton, D.R.; Barbaro, N.; Annett, G.M.; Raffel, C. MDR1 Gene Expression in Brain of Patients with Medically Intractable Epilepsy. Epilepsia 1995, 36, 1–6.
- Vincent, A.; Irani, S.R.; Lang, B. The growing recognition of immunotherapy-responsive seizure disorders with autoantibodies to specific neuronal proteins. Curr. Opin. Neurol. 2010, 23, 144–150.
- McKnight, K.; Jiang, Y.; Hart, Y.; Cavey, A.; Wroe, S.; Blank, M.; Shoenfeld, Y.; Vincent, A.; Palace, J.; Lang, B. Serum antibodies in epilepsy and seizure-associated disorders. Neurology 2005, 65, 1730–1736.
- Smith, D.; DeFalla, B.; Chadwick, D. The misdiagnosis of epilepsy and the management of refractory epilepsy in a specialist clinic. Qjm: Int. J. Med. 1999, 92, 15–23.
- Mishra, P.; Sinha, J.K.; Rajput, S.K. Efficacy of Cicuta virosa medicinal preparations against pentylenetetrazole-induced seizures. Epilepsy Behav. 2021, 115, 107653.
- Spencer, S.; Huh, L. Outcomes of epilepsy surgery in adults and children. Lancet Neurol. 2008, 7, 525–537.
- Vezzani, A. Before Epilepsy Unfolds: Finding the epileptogenesis switch. Nat. Med. 2012, 18, 1626–1627.
- Ryther, R.C.C.; Wong, M. Mammalian Target of Rapamycin (mTOR) Inhibition: Potential for Antiseizure, Antiepileptogenic, and Epileptostatic Therapy. Curr. Neurol. Neurosci. Rep. 2012, 12, 410–418.
- Vezzani, A.; Friedman, A.; Dingledine, R.J. The role of inflammation in epileptogenesis. Neuropharmacol. 2013, 69, 16–24.
- Frigerio, F.; Frasca, A.; Weissberg, I.; Parrella, S.; Friedman, A.; Vezzani, A.; Noe’, F.M. Long-lasting pro-ictogenic effects induced in vivo by rat brain exposure to serum albumin in the absence of concomitant pathology. Epilepsia 2012, 53, 1887–1897.
- Cacheaux, L.P.; Ivens, S.; David, Y.; Lakhter, A.J.; Bar-Klein, G.; Shapira, M.Y.; Heinemann, U.; Friedman, A.; Kaufer, D. Transcriptome Profiling Reveals TGF- Signaling Involvement in Epileptogenesis. J. Neurosci. 2009, 29, 8927–8935.
- Ventola, C.L. Epilepsy Management: Newer Agents, Unmet Needs, and Future Treatment Strategies. P T Peer-Rev. J. Formul. Manag. 2014, 39, 776–792.
- Schmidt, D.; Schachter, S.C. Drug treatment of epilepsy in adults. BMJ 2014, 348, g254.
- Brodie, M.; Yuen, A. Lamotrigine substitution study: Evidence for synergism with sodium valproate? Epilepsy Res. 1997, 26, 423–432.
- Haut, S.R.; Gursky, J.M.; Privitera, M. Behavioral interventions in epilepsy. Curr. Opin. Neurol. 2019, 32, 227–236.
- Panebianco, M.; Sridharan, K.; Ramaratnam, S. Yoga for epilepsy. Cochrane Database Syst. Rev. 2017, 2017, CD001524.
- Ramaratnam, S.; A Baker, G.; Goldstein, L.H. Psychological treatments for epilepsy. Cochrane Database Syst. Rev. 2016, 2, CD002029.
- Milby, A.H.; Halpern, C.H.; Baltuch, G.H. Vagus nerve stimulation in the treatment of refractory epilepsy. J. Neurosurg. 2009, 6, 228–237.
- Longo, R.; Peri, C.; Cricrì, D.; Coppi, L.; Caruso, D.; Mitro, N.; De Fabiani, E.; Crestani, M. Ketogenic Diet: A New Light Shining on Old but Gold Biochemistry. Nutrients 2019, 11, 2497.
- Neal, E.G.; Chaffe, H.; Schwartz, R.H.; Lawson, M.S.; Edwards, N.; Fitzsimmons, G.; Whitney, A.; Cross, J.H. The ketogenic diet for the treatment of childhood epilepsy: A randomised controlled trial. Lancet Neurol. 2008, 7, 500–506.
- Çubukçu, D.; Güzel, O.; Arslan, N. Effect of Ketogenic Diet on Motor Functions and Daily Living Activities of Children With Multidrug-Resistant Epilepsy: A Prospective Study. J. Child Neurol. 2018, 33, 718–723.
- Pendo, K.; DeGiorgio, C.M. Vitamin D3 for the Treatment of Epilepsy: Basic Mechanisms, Animal Models, and Clinical Trials. Front. Neurol. 2016, 7, 218.
- Holló, A.; Clemens, Z.; Kamondi, A.; Lakatos, P.; Szűcs, A. Correction of vitamin D deficiency improves seizure control in epilepsy: A pilot study. Epilepsy Behav. 2012, 24, 131–133.
- Liu, W.; Ge, T.; Pan, Z.; Leng, Y.; Lv, J.; Li, B. The effects of herbal medicine on epilepsy. Oncotarget 2017, 8, 48385–48397.
- Sucher, N.J.; Carles, M.C. A pharmacological basis of herbal medicines for epilepsy. Epilepsy Behav. 2015, 52, 308–318.
- Grone, B.P.; Baraban, S.C. Animal models in epilepsy research: Legacies and new directions. Nat. Neurosci. 2015, 18, 339–343.
- Ghosh, S.; Sinha, J.K.; Raghunath, M. Epigenomic maintenance through dietary intervention can facilitate DNA repair process to slow down the progress of premature aging. IUBMB Life 2016, 68, 717–721.





