
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Isabella Bonilha | + 2441 word(s) | 2441 | 2021-05-26 05:39:16 | | | |
| 2 | Catherine Yang | Meta information modification | 2441 | 2021-07-05 06:14:14 | | |
Video Upload Options
High density lipoproteins (HDLs) are commonly known for their anti‑atherogenic properties that include functions such as the promotion of cholesterol efflux and reverse cholesterol transport, as well as antioxidant and anti‑inflammatory activities. However, because of some chronic inflammatory diseases, such as type 2 diabetes mellitus (T2DM), significant changes occur in HDLs in terms of both structure and composition. These alterations lead to the loss of HDLs’ physiological functions, to transformation into dysfunctional lipoproteins, and to increased risk of cardiovascular disease (CVD).
1. Background
Diabetes mellitus (DM) is a chronic metabolic disorder characterized by defective insulin secretion and reduced tissue sensitivity to insulin, leading to hyperglycemia. Mortality and morbidity resulting from DM are consequences of long-term micro and macrovascular complications. The increasing prevalence of DM worldwide represents an important public health problem. The latest edition of the International Diabetes Atlas showed that 463 million adults live with DM and that more than 4 million people aged between 20 and 79 years died from causes related to DM in 2019. In addition, estimates show that there will be 578 million individuals affected by 2030 and 700 million in 2045 [1]. Type 2 diabetes mellitus (T2DM), characterized by an increase in blood glucose as a result of progressive increase in insulin resistance and pancreatic incompetence to meet the progressive demand for insulin production, corresponds to about 90% of all cases of DM and its prevalence is increasing worldwide. T2DM and insulin resistance are also well known for significantly increasing cardiovascular (CV) risk. Notably, in individuals with T2DM, an increase of 2 to 4% in mortality from coronary artery disease is estimated [2]. The dysfunction of pancreatic beta cells, a critical component for the pathogenesis of T2DM, has been attributed to glucotoxicity and high levels of free fatty acids with a high inflammatory response. An additional possible pathogenic mechanism is that hyperglycemia accelerates atherogenesis by increasing the oxidation of lipoproteins.
2. Alterations of HDL Plasma Level and Composition in T2DM
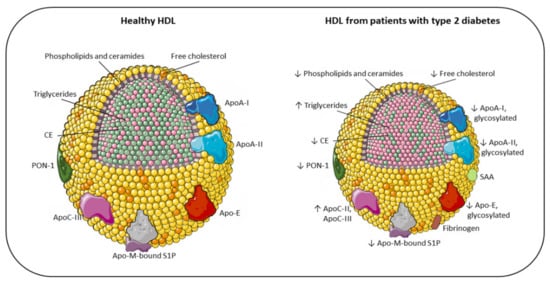
Figure 1. Main compositional differences between the HDL of healthy individuals and the HDL of patients with type 2 diabetes. Apo: apolipoprotein; CE: cholesteryl ester; PON-1: paraoxonase-1; S1P: sphingosine 1-phosphate; SAA: serum amyloid A; TG: triglyceride; ↓ decreased; ↑ increased.
2.1. Modifications of HDL-C Plasma Levels
2.2. Modifications of HDL Size
2.3. Modifications of the Lipid Content
2.4. Modifications of the Protein Component
2.5. Modifications of HDL Due to Glycation and Oxidation
References
- Williams, R.; Karuranga, S.; Malanda, B.; Saeedi, P.; Basit, A.; Besançon, S.; Bommer, C.; Esteghamati, A.; Ogurtsova, K.; Zhang, P.; et al. Global and regional estimates and projections of diabetes-related health expenditure: Results from the International Diabetes Federation Diabetes Atlas, 9th edition. Diabetes Res. Clin. Pract. 2020, 162, 108072.
- Wong, N.K.P.; Nicholls, S.J.; Tan, J.T.M.; Bursill, C.A. The Role of High-Density Lipoproteins in Diabetes and Its Vascular Complications. Int. J. Mol. Sci. 2018, 19, 1680.
- Gowri, M.S.; Van der Westhuyzen, D.R.; Bridges, S.R.; Anderson, J.W. Decreased protection by HDL from poorly controlled type 2 diabetic subjects against LDL oxidation may Be due to the abnormal composition of HDL. Arterioscler. Thromb. Vasc. Biol. 1999, 19, 2226–2233.
- Milder, T.Y.; Stocker, S.L.; Abdel Shaheed, C.; McGrath-Cadell, L.; Samocha-Bonet, D.; Greenfield, J.R.; Day, R.O. Combination Therapy with an SGLT2 Inhibitor as Initial Treatment for Type 2 Diabetes: A Systematic Review and Meta-Analysis. J. Clin. Med. 2019, 8, 45.
- Barter, P.J. The causes and consequences of low levels of high density lipoproteins in patients with diabetes. Diabetes Metab. J. 2011, 35, 101–106.
- Hedrick, C.C.; Thorpe, S.R.; Fu, M.X.; Harper, C.M.; Yoo, J.; Kim, S.M.; Wong, H.; Peters, A.L. Glycation impairs high-density lipoprotein function. Diabetologia 2000, 43, 312–320.
- Attia, N.; Nakbi, A.; Smaoui, M.; Chaaba, R.; Moulin, P.; Hammami, S.; Hamda, K.B.; Chanussot, F.; Hammami, M. Increased phospholipid transfer protein activity associated with the impaired cellular cholesterol efflux in type 2 diabetic subjects with coronary artery disease. Tohoku J. Exp. Med. 2007, 213, 129–137.
- Gomes Kjerulf, D.; Wang, S.; Omer, M.; Pathak, A.; Subramanian, S.; Han, C.Y.; Tang, C.; den Hartigh, L.J.; Shao, B.; Chait, A. Glycation of HDL blunts its anti-inflammatory and cholesterol efflux capacities in vitro, but has no effect in poorly controlled type 1 diabetes subjects. J. Diabetes Complicat. 2020, 34, 107693.
- Morgantini, C.; Natali, A.; Boldrini, B.; Imaizumi, S.; Navab, M.; Fogelman, A.M.; Ferrannini, E.; Reddy, S.T. Anti-inflammatory and antioxidant properties of HDLs are impaired in type 2 diabetes. Diabetes 2011, 60, 2617–2623.
- Sorrentino, S.A.; Besler, C.; Rohrer, L.; Meyer, M.; Heinrich, K.; Bahlmann, F.H.; Mueller, M.; Horváth, T.; Doerries, C.; Heinemann, M.; et al. Endothelial-vasoprotective effects of high-density lipoprotein are impaired in patients with type 2 diabetes mellitus but are improved after extended-release niacin therapy. Circulation 2010, 121, 110–122.
- Low Wang, C.C.; Hess, C.N.; Hiatt, W.R.; Goldfine, A.B. Clinical Update: Cardiovascular Disease in Diabetes Mellitus: Atherosclerotic Cardiovascular Disease and Heart Failure in Type 2 Diabetes Mellitus—Mechanisms, Management, and Clinical Considerations. Circulation 2016, 133, 2459–2502.
- Drew, B.G.; Rye, K.A.; Duffy, S.J.; Barter, P.; Kingwell, B.A. The emerging role of HDL in glucose metabolism. Nat. Rev. Endocrinol. 2012, 8, 237–245.
- Stancu, C.S.; Toma, L.; Sima, A.V. Dual role of lipoproteins in endothelial cell dysfunction in atherosclerosis. Cell Tissue Res. 2012, 349, 433–446.
- Haase, C.L.; Tybjærg-Hansen, A.; Nordestgaard, B.G.; Frikke-Schmidt, R. HDL Cholesterol and Risk of Type 2 Diabetes: A Mendelian Randomization Study. Diabetes 2015, 64, 3328–3333.
- Vaisar, T.; Couzens, E.; Hwang, A.; Russell, M.; Barlow, C.E.; DeFina, L.F.; Hoofnagle, A.N.; Kim, F. Type 2 diabetes is associated with loss of HDL endothelium protective functions. PLoS ONE 2018, 13, e0192616.
- Ikura, K.; Hanai, K.; Shinjyo, T.; Uchigata, Y. HDL cholesterol as a predictor for the incidence of lower extremity amputation and wound-related death in patients with diabetic foot ulcers. Atherosclerosis 2015, 239, 465–469.
- Russo, G.T.; De Cosmo, S.; Viazzi, F.; Pacilli, A.; Ceriello, A.; Genovese, S.; Guida, P.; Giorda, C.; Cucinotta, D.; Pontremoli, R.; et al. Plasma Triglycerides and HDL-C Levels Predict the Development of Diabetic Kidney Disease in Subjects With Type 2 Diabetes: The AMD Annals Initiative. Diabetes Care 2016, 39, 2278–2287.
- Qi, Q.; Liang, L.; Doria, A.; Hu, F.B.; Qi, L. Genetic predisposition to dyslipidemia and type 2 diabetes risk in two prospective cohorts. Diabetes 2012, 61, 745–752.
- Bakogianni, M.C.; Kalofoutis, C.A.; Skenderi, K.I.; Kalofoutis, A.T. Clinical evaluation of plasma high-density lipoprotein subfractions (HDL2, HDL3) in non-insulin-dependent diabetics with coronary artery disease. J. Diabetes Complicat. 2001, 15, 265–269.
- Russo, G.T.; Giandalia, A.; Romeo, E.L.; Alibrandi, A.; Horvath, K.V.; Asztalos, B.F.; Cucinotta, D. Markers of Systemic Inflammation and Apo-AI Containing HDL Subpopulations in Women with and without Diabetes. Int. J. Endocrinol. 2014, 2014, 607924.
- Garvey, W.T.; Kwon, S.; Zheng, D.; Shaughnessy, S.; Wallace, P.; Hutto, A.; Pugh, K.; Jenkins, A.J.; Klein, R.L.; Liao, Y. Effects of insulin resistance and type 2 diabetes on lipoprotein subclass particle size and concentration determined by nuclear magnetic resonance. Diabetes 2003, 52, 453–462.
- Mora, S.; Otvos, J.D.; Rosenson, R.S.; Pradhan, A.; Buring, J.E.; Ridker, P.M. Lipoprotein particle size and concentration by nuclear magnetic resonance and incident type 2 diabetes in women. Diabetes 2010, 59, 1153–1160.
- Goldberg, I.J. Clinical review 124: Diabetic dyslipidemia: Causes and consequences. J. Clin. Endocrinol. Metab. 2001, 86, 965–971.
- Awadallah, S.; Madkour, M.; Hamidi, R.A.; Alwafa, E.A.; Hattab, M.; Zakkour, B.; Al-Matroushi, A.; Ahmed, E.; Al-Kitbi, M. Plasma levels of Apolipoprotein A1 and Lecithin:Cholesterol Acyltransferase in type 2 diabetes mellitus: Correlations with haptoglobin phenotypes. Diabetes Metab. Syndr. 2017, 11 (Suppl. 2), S543–S546.
- Fournier, N.; Myara, I.; Atger, V.; Moatti, N. Reactivity of lecithin-cholesterol acyl transferase (LCAT) towards glycated high-density lipoproteins (HDL). Clin. Chim. Acta 1995, 234, 47–61.
- Rizzo, M.; Otvos, J.; Nikolic, D.; Montalto, G.; Toth, P.P.; Banach, M. Subfractions and subpopulations of HDL: An update. Curr. Med. Chem. 2014, 21, 2881–2891.
- Kontush, A.; Chapman, M.J. Why is HDL functionally deficient in type 2 diabetes? Curr. Diabetes Rep. 2008, 8, 51–59.
- Cardner, M.; Yalcinkaya, M.; Goetze, S.; Luca, E.; Balaz, M.; Hunjadi, M.; Hartung, J.; Shemet, A.; Kränkel, N.; Radosavljevic, S.; et al. Structure-function relationships of HDL in diabetes and coronary heart disease. JCI Insight 2020, 5.
- Ståhlman, M.; Fagerberg, B.; Adiels, M.; Ekroos, K.; Chapman, J.M.; Kontush, A.; Borén, J. Dyslipidemia, but not hyperglycemia and insulin resistance, is associated with marked alterations in the HDL lipidome in type 2 diabetic subjects in the DIWA cohort: Impact on small HDL particles. Biochim. Biophys. Acta 2013, 1831, 1609–1617.
- Brinck, J.W.; Thomas, A.; Lauer, E.; Jornayvaz, F.R.; Brulhart-Meynet, M.C.; Prost, J.C.; Pataky, Z.; Lofgren, P.; Hoffstedt, J.; Eriksson, M.; et al. Diabetes Mellitus Is Associated With Reduced High-Density Lipoprotein Sphingosine-1-Phosphate Content and Impaired High-Density Lipoprotein Cardiac Cell Protection. Arterioscler. Thromb. Vasc. Biol. 2016, 36, 817–824.
- Feingold, K.R. Dyslipidemia in Diabetes. In Endotext; Feingold, K.R., Anawalt, B., Boyce, A., Chrousos, G., de Herder, W.W., Dungan, K., Grossman, A., Hershman, J.M., Hofland, H.J., Kaltsas, G., et al., Eds.; Endotext: South Dartmouth, MA, USA, 2000.
- Zhang, P.; Gao, J.; Pu, C.; Zhang, Y. Apolipoprotein status in type 2 diabetes mellitus and its complications (Review). Mol. Med. Rep. 2017, 16, 9279–9286.
- Caron, S.; Verrijken, A.; Mertens, I.; Samanez, C.H.; Mautino, G.; Haas, J.T.; Duran-Sandoval, D.; Prawitt, J.; Francque, S.; Vallez, E.; et al. Transcriptional activation of apolipoprotein CIII expression by glucose may contribute to diabetic dyslipidemia. Arterioscler. Thromb. Vasc. Biol. 2011, 31, 513–519.
- Aroner, S.A.; Furtado, J.D.; Sacks, F.M.; Tsai, M.Y.; Mukamal, K.J.; McClelland, R.L.; Jensen, M.K. Apolipoprotein C-III and its defined lipoprotein subspecies in relation to incident diabetes: The Multi-Ethnic Study of Atherosclerosis. Diabetologia 2019, 62, 981–992.
- Gordon, S.M.; Davidson, W.S.; Urbina, E.M.; Dolan, L.M.; Heink, A.; Zang, H.; Lu, L.J.; Shah, A.S. Erratum. The Effects of Type 2 Diabetes on Lipoprotein Composition and Arterial Stiffness in Male Youth. Diabetes 2013;62:2958-2967. Diabetes 2016, 65, 2100.
- Cohen, M.P. Diabetes and Protein Glycosylation, 1st ed.; York, S.-V.N., Ed.; Springer: New York, NY, USA, 1986; Volume 1, p. XII.
- Pan, B.; Ren, H.; Ma, Y.; Liu, D.; Yu, B.; Ji, L.; Pan, L.; Li, J.; Yang, L.; Lv, X.; et al. High-density lipoprotein of patients with type 2 diabetes mellitus elevates the capability of promoting migration and invasion of breast cancer cells. Int. J. Cancer 2012, 131, 70–82.
- Hermo, R.; Mier, C.; Mazzotta, M.; Tsuji, M.; Kimura, S.; Gugliucci, A. Circulating levels of nitrated apolipoprotein A-I are increased in type 2 diabetic patients. Clin. Chem. Lab. Med. 2005, 43, 601–606.
- Curtiss, L.K.; Witztum, J.L. Plasma apolipoproteins AI, AII, B, CI, and E are glucosylated in hyperglycemic diabetic subjects. Diabetes 1985, 34, 452–461.
- Calvo, C.; Talussot, C.; Ponsin, G.; Berthezene, F. Non enzymatic glycation of apolipoprotein A-I. Effects on its self-association and lipid binding properties. Biochem. Biophys. Res. Commun. 1988, 153, 1060–1067.
- Lapolla, A.; Brioschi, M.; Banfi, C.; Tremoli, E.; Bonfante, L.; Cristoni, S.; Seraglia, R.; Traldi, P. On the search for glycated lipoprotein ApoA-I in the plasma of diabetic and nephropathic patients. J. Mass Spectrom. 2008, 43, 74–81.





