
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Zongli Xie | + 3030 word(s) | 3030 | 2021-05-13 08:43:12 |
Video Upload Options
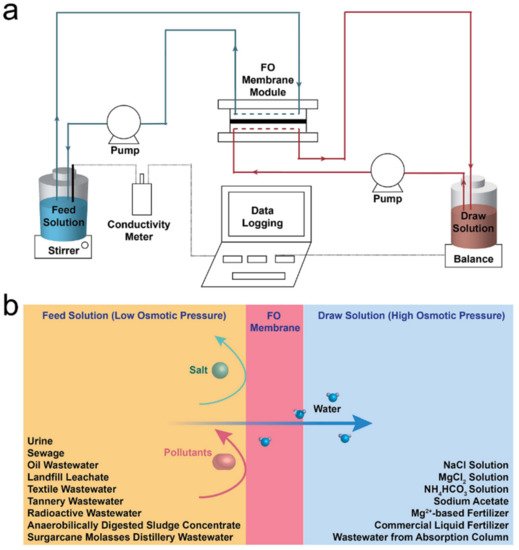
Forward osmosis (FO), driven by the osmotic pressure difference between solutions divided by a semi-permeable membrane, has been recognised as a potential energy-efficient filtration process with a low tendency for fouling and a strong ability to filtrate highly polluted wastewater.
1. Introduction
2. Integration of FO with Other Membrane Technologies
Current studies have revealed the feasibility of the FO filtration process in wastewater treatment. However, drawbacks such as the salt accumulation, requirement of draw solution recovery process, and further water mining from draw solution are still challenges that limit the wide application of stand‐alone FO processes[31]. As a result, integrating the FO process with other membrane filtration processes such as ultrafiltration (UF), nanofiltration (NF), reverse osmosis (RO) and membrane distillation (MD) is proposed (Figure 2a). Some of the combinations are win‐win strategies to apply the advantages of varied technologies and avoid their shortcomings.
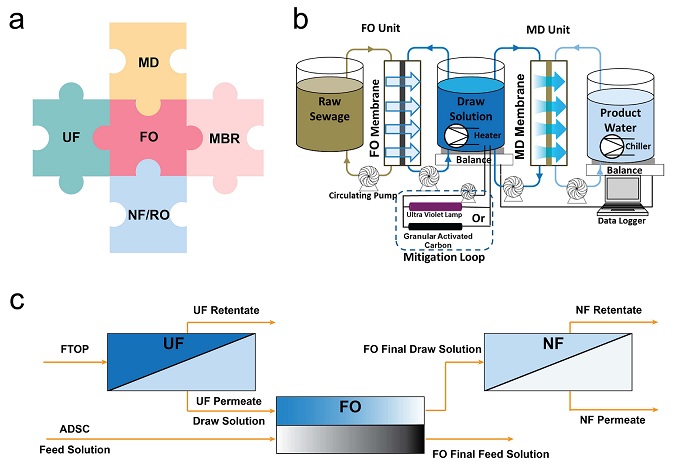 Figure 2. (a) Illustration of the integration of FO with other membrane‐based technologies, (b) schematic diagram of the FO‐MD hybrid system with the mitigation loop for the reduction of the concentration of feed contaminants in the draw solution, reprinted with permission from[32], and (c) schematic diagram of the UF‐FO‐NF integrated system, adapted from[33].
Figure 2. (a) Illustration of the integration of FO with other membrane‐based technologies, (b) schematic diagram of the FO‐MD hybrid system with the mitigation loop for the reduction of the concentration of feed contaminants in the draw solution, reprinted with permission from[32], and (c) schematic diagram of the UF‐FO‐NF integrated system, adapted from[33].
Zhang et al. [34] reported a study about the application of a forward osmosis membrane distillation (FO‐MD) system for sustainable water recovery and acetic acid reuse from oily wastewater. Results indicated that the FO process showed a large water flux, a high oil removal ratio, and a moderate acetic acid permeation rate. After that, the MD process further rejected the NaCl and oil in diluted draw solution, while completed the regeneration of draw solution. The FO‐MD system is also studied for treating wastewater from other sources. Industrial wastewater contains highly toxic heavy metals such as Hg, Cd, and Pb, and its discharge is strictly regulated[35]. Wu et al.[31] investigated the FO‐MD system for the treatment of wastewater containing heavy metals. The result indicated that the FO system could effectively reject more than 97% of Hg, Cd, and Pb, and the MD system achieved around a 100% rejection rate of these heavy metals. Human urine consists of 95% water, 3.5% organics, and 1.5% inorganic salt[36], and is a potential water and nutrients source. Therefore, various technologies have been applied in water and nutrient recovery from urine, including electrodialysis, reverse osmosis, freeze/thaw concentration[37], microbial fuel cell, and ion exchange membrane[38][39][40]. Recently, Liu et al.[38] investigated the feasibility of a FO‐MD hybrid system for treating urine. The FO membrane rejected most of the TOC, TN, and NH4+‐N, but there were still some contaminants accumulated in the draw solution. Then, the MD process further rejected these contaminants, and the hybrid FO‐MD system revealed nearly 100% rejection of TOC, TN, and NH4+‐N.
3. Integration of FO with Other Wastewater Treatment Technologies
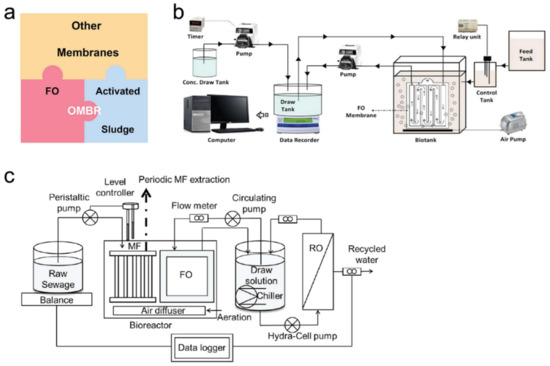
3.1. Integration of FO with Biological Process
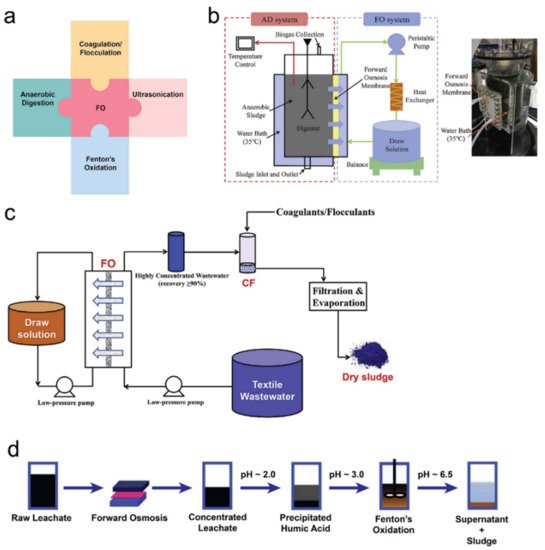
3.2. Integrations of FO with Other Water Treatment Processes
References
- Klaysom, C.; Cath, T.Y.; Depuydt, T.; Vankelecom, I.F. Forward and pressure retarded osmosis: Potential solutions for global challenges in energy and water supply. Chem. Soc. Rev. 2013, 42, 6959–6989.
- Yasukawa, M.; Mishima, S.; Tanaka, Y.; Takahashi, T.; Matsuyama, H. Thin-film composite forward osmosis membrane with high water flux and high pressure resistance using a thicker void-free polyketone porous support. Desalination 2017, 402, 1–9.
- Lutchmiah, K.; Cornelissen, E.R.; Harmsen, D.J.; Post, J.W.; Lampi, K.; Ramaekers, H.; Rietveld, L.C.; Roest, K. Water recovery from sewage using forward osmosis. Water Sci. Technol. 2011, 64, 1443–1449.
- Wang, Z.; Zheng, J.; Tang, J.; Wang, X.; Wu, Z. A pilot-scale forward osmosis membrane system for concentrating low-strength municipal wastewater: Performance and implications. Sci. Rep. 2016, 6, 1–11.
- Wong, S.; Ngadi, N.; Inuwa, I.M.; Hassan, O. Recent advances in applications of activated carbon from biowaste for wastewater treatment: A short review. J. Clean. Prod. 2018, 175, 361–375.
- Paul, R.; Kenway, S.; Mukheibir, P. How scale and technology influence the energy intensity of water recycling systems-an analytical review. J. Clean. Prod. 2019, 215, 1457–1480.
- Iskander, S.M.; Zou, S.; Brazil, B.; Novak, J.T.; He, Z. Energy consumption by forward osmosis treatment of landfill leachate for water recovery. Waste Manag. 2017, 63, 284–291.
- Di Palma, L.; Ferrantelli, P.; Merli, C.; Petrucci, E. Treatment of industrial landfill leachate by means of evaporation and reverse osmosis. Waste Manage. 2002, 22, 951–955.
- Renou, S.; Givaudan, J.; Poulain, S.; Dirassouyan, F.; Moulin, P. Landfill leachate treatment: Review and opportunity. J. Hazard. Mater. 2008, 150, 468–493.
- Lin, J.; Ye, W.; Zeng, H.; Yang, H.; Shen, J.; Darvishmanesh, S.; Luis, P.; Sotto, A.; Van der Bruggen, B. Fractionation of direct dyes and salts in aqueous solution using loose nanofiltration membranes. J. Membr. Sci. 2015, 477, 183–193.
- Lin, J.; Tang, C.Y.; Ye, W.; Sun, S.-P.; Hamdan, S.H.; Volodin, A.; Van Haesendonck, C.; Sotto, A.; Luis, P.; Van der Bruggen, B. Unraveling flux behavior of superhydrophilic loose nanofiltration membranes during textile wastewater treatment. J. Membr. Sci. 2015, 493, 690–702.
- Marcucci, M.; Ciardelli, G.; Matteucci, A.; Ranieri, L.; Russo, M. Experimental campaigns on textile wastewater for reuse by means of different membrane processes. Desalination 2002, 149, 137–143.
- Verliefde, A.R.; Cornelissen, E.; Heijman, S.; Verberk, J.; Amy, G.; Van der Bruggen, B.; Van Dijk, J. The role of electrostatic interactions on the rejection of organic solutes in aqueous solutions with nanofiltration. J. Membr. Sci. 2008, 322, 52–66.
- Van der Bruggen, B.; Cornelis, G.; Vandecasteele, C.; Devreese, I. Fouling of nanofiltration and ultrafiltration membranes applied for wastewater regeneration in the textile industry. Desalination 2005, 175, 111–119.
- Pearce, G. Uf/mf pre-treatment to ro in seawater and wastewater reuse applications: A comparison of energy costs. Desalination 2008, 222, 66–73.
- Wu, X.; Fang, F.; Zhang, K. Graphene oxide modified forward osmosis membranes with improved hydrophilicity and desalination performance. Desalin. Water Treat. 2017, 85, 73–83.
- Wu, X.; Ding, M.; Xu, H.; Yang, W.; Zhang, K.; Tian, H.; Wang, H.; Xie, Z. Scalable ti3c2t x mxene interlayered forward osmosis membranes for enhanced water purification and organic solvent recovery. ACS Nano 2020, 14, 9125–9135.
- Loeb, S. Production of energy from concentrated brines by pressure-retarded osmosis: I. Preliminary technical and economic correlations. J. Membr. Sci. 1976, 1, 49–63.
- Nayak, C.A.; Rastogi, N.K. Forward osmosis for the concentration of anthocyanin from garcinia indica choisy. Sep. Purif. Technol. 2010, 71, 144–151.
- Chung, T.-S.; Li, X.; Ong, R.C.; Ge, Q.; Wang, H.; Han, G. Emerging forward osmosis (fo) technologies and challenges ahead for clean water and clean energy applications. Curr. Opin. Chem. Eng. 2012, 1, 246–257.
- Han, G.; Zhao, B.; Fu, F.; Chung, T.-S.; Weber, M.; Staudt, C.; Maletzko, C. High performance thin-film composite membranes with mesh-reinforced hydrophilic sulfonated polyphenylenesulfone (sppsu) substrates for osmotically driven processes. J. Membr. Sci. 2016, 502, 84–93.
- Achilli, A.; Cath, T.Y.; Marchand, E.A.; Childress, A.E. The forward osmosis membrane bioreactor: A low fouling alternative to mbr processes. Desalination 2009, 239, 10–21.
- Liu, X.; Wu, J.; Hou, L.-a.; Wang, J. Removal of co, sr and cs ions from simulated radioactive wastewater by forward osmosis. Chemosphere 2019, 232, 87–95.
- Korenak, J.; Hélix-Nielsen, C.; Bukšek, H.; Petrinić, I. Efficiency and economic feasibility of forward osmosis in textile wastewater treatment. J. Clean. Prod. 2019, 210, 1483–1495.
- Gao, Y.; Fang, Z.; Liang, P.; Huang, X. Direct concentration of municipal sewage by forward osmosis and membrane fouling behavior. Bioresour. Technol. 2018, 247, 730–735.
- Chekli, L.; Kim, Y.; Phuntsho, S.; Li, S.; Ghaffour, N.; Leiknes, T.; Shon, H.K. Evaluation of fertilizer-drawn forward osmosis for sustainable agriculture and water reuse in arid regions. J. Environ. Manag. 2017, 187, 137–145.
- Ming Xie; Mingxin Zheng; Paul Cooper; William E. Price; Long D. Nghiem; Menachem Elimelech; Osmotic dilution for sustainable greenwall irrigation by liquid fertilizer: Performance and implications. Journal of Membrane Science 2015, 494, 32-38, 10.1016/j.memsci.2015.07.026.
- Mona Gulied; Fares Al Momani; MajedA Khraisheh; Rahul Bhosale; Ahmed AlNouss; Influence of draw solution type and properties on the performance of forward osmosis process: Energy consumption and sustainable water reuse. Chemosphere 2019, 233, 234-244, 10.1016/j.chemosphere.2019.05.241.
- M.J. Luján-Facundo; J.L. Soler-Cabezas; José-Antonio Mendoza-Roca; M.C. Vincent-Vela; A. Bes-Piá; S. Doñate-Hernández; A study of the osmotic membrane bioreactor process using a sodium chloride solution and an industrial effluent as draw solutions. Chemical Engineering Journal 2017, 322, 603-610, 10.1016/j.cej.2017.04.062.
- Wu, X.; Tanner, J.; Ng, D.; Acharya, D.; Xie, Z. Sewage concentration via a graphene oxide modified thin-film nanocomposite forward osmosis membrane: Enhanced performance and mitigated fouling. Chem. Eng. J. 2020, 127718.
- Chia-Yu Wu; Shiao-Shing Chen; Dai-Zhou Zhang; Jun Kobayashi; Hg removal and the effects of coexisting metals in forward osmosis and membrane distillation. Water Science and Technology 2017, 75, 2622-2630, 10.2166/wst.2017.143.
- Ming Xie; Long D. Nghiem; William E. Price; Menachem Elimelech; A Forward Osmosis–Membrane Distillation Hybrid Process for Direct Sewer Mining: System Performance and Limitations. Environmental Science & Technology 2013, 47, 13486-13493, 10.1021/es404056e.
- Carlos Carbonell-Alcaina; Jose Luis Soler-Cabezas; Amparo Bes-Piá; María Cinta Vincent-Vela; Jose Antonio Mendoza-Roca; Laura Pastor-Alcañiz; Silvia Álvarez-Blanco; Integrated Membrane Process for the Treatment and Reuse of Residual Table Olive Fermentation Brine and Anaerobically Digested Sludge Centrate. Membranes 2020, 10, 253, 10.3390/membranes10100253.
- Sui Zhang; Peng Wang; Xiuzhu Fu; Tai-Shung Chung; Sustainable water recovery from oily wastewater via forward osmosis-membrane distillation (FO-MD). Water Research 2014, 52, 112-121, 10.1016/j.watres.2013.12.044.
- Meltem Urgun-Demirtas; Paul L. Benda; Patricia S. Gillenwater; M. Cristina Negri; Hui Xiong; Seth Snyder; Achieving very low mercury levels in refinery wastewater by membrane filtration. Journal of Hazardous Materials 2012, 215-216, 98-107, 10.1016/j.jhazmat.2012.02.040.
- Bo-Bertil Lind; Zsófia Ban; Stefan Bydén; Volume reduction and concentration of nutrients in human urine. Ecological Engineering 2001, 16, 561-566, 10.1016/s0925-8574(00)00107-5.
- M. Maurer; W. Pronk; Tove A. Larsen; Treatment processes for source-separated urine. Water Research 2006, 40, 3151-3166, 10.1016/j.watres.2006.07.012.
- Qianliang Liu; Caihong Liu; Lei Zhao; Weichao Ma; Huiling Liu; Jun Ma; Integrated forward osmosis-membrane distillation process for human urine treatment. Water Research 2016, 91, 45-54, 10.1016/j.watres.2015.12.045.
- Ryan C. Tice; Younggy Kim; Energy efficient reconcentration of diluted human urine using ion exchange membranes in bioelectrochemical systems. Water Research 2014, 64, 61-72, 10.1016/j.watres.2014.06.037.
- Jeremy A. O'Neal; Treavor H. Boyer; Phosphate recovery using hybrid anion exchange: Applications to source-separated urine and combined wastewater streams. Water Research 2013, 47, 5003-5017, 10.1016/j.watres.2013.05.037.
- Aftab, B.; Khan, S.J.; Maqbool, T.; Hankins, N.P. Heavy metals removal by osmotic membrane bioreactor (ombr) and their effect on sludge properties. Desalination 2017, 403, 117–127.
- Luo, W.; Hai, F.I.; Price, W.E.; Guo, W.; Ngo, H.H.; Yamamoto, K.; Nghiem, L.D. Phosphorus and water recovery by a novel osmotic membrane bioreactor–reverse osmosis system. Bioresour. Technol. 2016, 200, 297–304.
- Zhao, J.; Li, Y.; Pan, S.; Tu, Q.; Zhu, H. Performance of a forward osmotic membrane bioreactor for anaerobic digestion of waste sludge with increasing solid concentration. J. Environ. Manag. 2019, 246, 239–246.
- Han, G.; Liang, C.-Z.; Chung, T.-S.; Weber, M.; Staudt, C.; Maletzko, C. Combination of forward osmosis (fo) process with coagulation/flocculation (cf) for potential treatment of textile wastewater. Water Res. 2016, 91, 361–370.
- Iskander, S.M.; Novak, J.T.; He, Z. Reduction of reagent requirements and sludge generation in fenton’s oxidation of landfill leachate by synergistically incorporating forward osmosis and humic acid recovery. Water Res. 2019, 151, 310–317.
- Yao, M.; Duan, L.; Wei, J.; Qian, F.; Hermanowicz, S.W. Carbamazepine removal from wastewater and the degradation mechanism in a submerged forward osmotic membrane bioreactor. Bioresour. Technol. 2020, 314, 123732.
- Huang, L.; Lee, D.-J. Membrane bioreactor: A mini review on recent r&d works. Bioresour. Technol. 2015, 194, 383–388.
- Praveen, P.; Loh, K.-C. Osmotic membrane bioreactor for phenol biodegradation under continuous operation. J. Hazard. Mater. 2016, 305, 115–122.
- Qiu, G.; Ting, Y.-P. Direct phosphorus recovery from municipal wastewater via osmotic membrane bioreactor (ombr) for wastewater treatment. Bioresour. Technol. 2014, 170, 221–229.
- Xiao, D.; Tang, C.Y.; Zhang, J.; Lay, W.C.; Wang, R.; Fane, A.G. Modeling salt accumulation in osmotic membrane bioreactors: Implications for fo membrane selection and system operation. J. Membr. Sci. 2011, 366, 314–324.
- Qiu, G.; Ting, Y.-P. Osmotic membrane bioreactor for wastewater treatment and the effect of salt accumulation on system performance and microbial community dynamics. Bioresour. Technol. 2013, 150, 287–297.
- Kim, S. Scale-up of osmotic membrane bioreactors by modeling salt accumulation and draw solution dilution using hollow-fiber membrane characteristics and operation conditions. Bioresour. Technol. 2014, 165, 88–95.
- Ab Hamid, N.H.; Wang, D.K.; Smart, S.; Ye, L. Achieving stable operation and shortcut nitrogen removal in a long-term operated aerobic forward osmosis membrane bioreactor (fombr) for treating municipal wastewater. Chemosphere 2020, 260, 127581.
- Chen, L.; Gu, Y.; Cao, C.; Zhang, J.; Ng, J.-W.; Tang, C. Performance of a submerged anaerobic membrane bioreactor with forward osmosis membrane for low-strength wastewater treatment. Water Res. 2014, 50, 114–123.
- Gao, Y.; Fang, Z.; Chen, C.; Zhu, X.; Liang, P.; Qiu, Y.; Zhang, X.; Huang, X. Evaluating the performance of inorganic draw solution concentrations in an anaerobic forward osmosis membrane bioreactor for real municipal sewage treatment. Bioresour. Technol. 2020, 307, 123254.
- Zhang, J.; Loong, W.L.C.; Chou, S.; Tang, C.; Wang, R.; Fane, A.G. Membrane biofouling and scaling in forward osmosis membrane bioreactor. J. Membr. Sci. 2012, 403, 8–14.
- Gu, Y.; Chen, L.; Ng, J.-W.; Lee, C.; Chang, V.W.-C.; Tang, C.Y. Development of anaerobic osmotic membrane bioreactor for low-strength wastewater treatment at mesophilic condition. J. Membr. Sci. 2015, 490, 197–208.
- Lay, W.C.; Liu, Y.; Fane, A.G. Impacts of salinity on the performance of high retention membrane bioreactors for water reclamation: A review. Water Res. 2010, 44, 21–40.
- Ferrari, F.; Balcazar, J.L.; Rodriguez-Roda, I.; Pijuan, M. Anaerobic membrane bioreactor for biogas production from concentrated sewage produced during sewer mining. Sci. Total Environ. 2019, 670, 993–1000.
- Luján-Facundo, M.J.; Soler-Cabezas, J.L.; Mendoza-Roca, J.A.; Vincent-Vela, M.C.; Bes-Piá, A.; Doñate-Hernández, S. A study of the osmotic membrane bioreactor process using a sodium chloride solution and an industrial effluent as draw solutions. Chem. Eng. J. 2017, 322, 603–610.
- Wang, K.Y.; Ong, R.C.; Chung, T.-S. Double-skinned forward osmosis membranes for reducing internal concentration polarization within the porous sublayer. Ind. Eng. Chem. Res. 2010, 49, 4824–4831.
- Holloway, R.W.; Regnery, J.; Nghiem, L.D.; Cath, T.Y. Removal of trace organic chemicals and performance of a novel hybrid ultrafiltration-osmotic membrane bioreactor. Environ. Sci. Technol. 2014, 48, 10859–10868.
- Luján-Facundo, M.J.; Fernández-Navarro, J.; Alonso-Molina, J.L.; Amorós-Muñoz, I.; Moreno, Y.; Mendoza-Roca, J.A.; Pastor-Alcañiz, L. The role of salinity on the changes of the biomass characteristics and on the performance of an ombr treating tannery wastewater. Water Res. 2018, 142, 129–137.
- Park, S.H.; Park, B.; Shon, H.K.; Kim, S. Modeling full-scale osmotic membrane bioreactor systems with high sludge retention and low salt concentration factor for wastewater reclamation. Bioresour. Technol. 2015, 190, 508–515.
- Qiu, G.; Law, Y.-M.; Das, S.; Ting, Y.-P. Direct and complete phosphorus recovery from municipal wastewater using a hybrid microfiltration-forward osmosis membrane bioreactor process with seawater brine as draw solution. Environ. Sci. Technol. 2015, 49, 6156–6163.
- Luo, W.; Hai, F.I.; Kang, J.; Price, W.E.; Nghiem, L.D.; Elimelech, M. The role of forward osmosis and microfiltration in an integrated osmotic-microfiltration membrane bioreactor system. Chemosphere 2015, 136, 125–132.
- Wang, X.; Yuan, B.; Chen, Y.; Li, X.; Ren, Y. Integration of micro-filtration into osmotic membrane bioreactors to prevent salinity build-up. Bioresour. Technol. 2014, 167, 116–123.
- Zou, S.; Yuan, H.; Childress, A.; He, Z. Energy Consumption by Recirculation: A Missing Parameter When Evaluating forward Osmosis. Environ. Sci. Technol. 2016, 50, 6827–6829.
- Cath, T.Y.; Gormly, S.; Beaudry, E.G.; Flynn, M.T.; Adams, V.D.; Childress, A.E. Membrane contactor processes for wastewater reclamation in space: Part i. Direct osmotic concentration as pretreatment for reverse osmosis. J. Membr. Sci. 2005, 257, 85–98.
- McGinnis, R.L.; Elimelech, M. Energy requirements of ammonia–carbon dioxide forward osmosis desalination. Desalination 2007, 207, 370–382.
- Xiang, X.; Zou, S.; He, Z. Energy consumption of water recovery from wastewater in a submerged forward osmosis system using commercial liquid fertilizer as a draw solute. Sep. Purif. Technol. 2017, 174, 432–438.
- Vinardell, S.; Astals, S.; Mata-Alvarez, J.; Dosta, J. Techno-economic analysis of combining forward osmosis-reverse osmosis and anaerobic membrane bioreactor technologies for municipal wastewater treatment and water production. Bioresour. Technol. 2020, 297, 122395.
- Hu, Y.-y.; Wu, J.; Li, H.-z.; Poncin, S.; Wang, K.-j.; Zuo, J.-e. Novel insight into high solid anaerobic digestion of swine manure after thermal treatment: Kinetics and microbial community properties. J. Environ. Manag. 2019, 235, 169–177.
- Castelló, E.; Braga, L.; Fuentes, L.; Etchebehere, C. Possible causes for the instability in the h2 production from cheese whey in a cstr. Int. J. Hydrog. Energy 2018, 43, 2654–2665.
- Gao, Y.; Fang, Z.; Liang, P.; Zhang, X.; Qiu, Y.; Kimura, K.; Huang, X. Anaerobic digestion performance of concentrated municipal sewage by forward osmosis membrane: Focus on the impact of salt and ammonia nitrogen. Bioresour. Technol. 2019, 276, 204–210.
- Lau, W.-J.; Ismail, A. Polymeric nanofiltration membranes for textile dye wastewater treatment: Preparation, performance evaluation, transport modelling, and fouling control—A review. Desalination 2009, 245, 321–348.
- Verma, A.K.; Dash, R.R.; Bhunia, P. A review on chemical coagulation/flocculation technologies for removal of colour from textile wastewaters. J. Environ. Manag. 2012, 93, 154–168.
- Chen, T.; Gao, B.; Yue, Q. Effect of dosing method and ph on color removal performance and floc aggregation of polyferric chloride–polyamine dual-coagulant in synthetic dyeing wastewater treatment. Colloids Surf. Physicochem. Eng. Aspects 2010, 355, 121–129.
- Liang, C.-Z.; Sun, S.-P.; Li, F.-Y.; Ong, Y.-K.; Chung, T.-S. Treatment of highly concentrated wastewater containing multiple synthetic dyes by a combined process of coagulation/flocculation and nanofiltration. J. Membr. Sci. 2014, 469, 306–315.
- Deng, Y.; Englehardt, J.D. Treatment of landfill leachate by the fenton process. Water Res. 2006, 40, 3683–3694.
- Zhao, X.; Wei, X.; Xia, P.; Liu, H.; Qu, J. Removal and transformation characterization of refractory components from biologically treated landfill leachate by fe2+/naclo and fenton oxidation. Sep. Purif. Technol. 2013, 116, 107–113.
- Hermosilla, D.; Cortijo, M.; Huang, C.P. Optimizing the treatment of landfill leachate by conventional fenton and photo-fenton processes. Sci. Total Environ. 2009, 407, 3473–3481.
- Li, H.; Jin, Y.; Nie, Y. Application of alkaline treatment for sludge decrement and humic acid recovery. Bioresour. Technol. 2009, 100, 6278–6283.
- Morozesk, M.; Bonomo, M.M.; da Costa Souza, I.; Rocha, L.D.; Duarte, I.D.; Martins, I.O.; Dobbss, L.B.; Carneiro, M.T.W.D.; Fernandes, M.N.; Matsumoto, S.T. Effects of humic acids from landfill leachate on plants: An integrated approach using chemical, biochemical and cytogenetic analysis. Chemosphere 2017, 184, 309–317.
- Nguyen, N.C.; Nguyen, H.T.; Chen, S.-S.; Nguyen, N.T.; Li, C.-W. Application of forward osmosis (fo) under ultrasonication on sludge thickening of waste activated sludge. Water Sci. Technol. 2015, 72, 1301–1307.
- Jeremy A. O'Neal; Treavor H. Boyer; Phosphate recovery using hybrid anion exchange: Applications to source-separated urine and combined wastewater streams. Water Research 2013, 47, 5003-5017, 10.1016/j.watres.2013.05.037.





