Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | mariaenrica tine | + 2506 word(s) | 2506 | 2021-06-23 05:19:00 | | | |
| 2 | Rita Xu | Meta information modification | 2506 | 2021-06-24 04:14:51 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Tine, M. Evolution of Extracellular Vesicles’ Knowledge. Encyclopedia. Available online: https://encyclopedia.pub/entry/11199 (accessed on 21 May 2026).
Tine M. Evolution of Extracellular Vesicles’ Knowledge. Encyclopedia. Available at: https://encyclopedia.pub/entry/11199. Accessed May 21, 2026.
Tine, Mariaenrica. "Evolution of Extracellular Vesicles’ Knowledge" Encyclopedia, https://encyclopedia.pub/entry/11199 (accessed May 21, 2026).
Tine, M. (2021, June 23). Evolution of Extracellular Vesicles’ Knowledge. In Encyclopedia. https://encyclopedia.pub/entry/11199
Tine, Mariaenrica. "Evolution of Extracellular Vesicles’ Knowledge." Encyclopedia. Web. 23 June, 2021.
Copy Citation
Extracellular vesicles (EVs) are a family of particles/vesicles present in blood and body fluids, composed of phospholipid bilayers that carry a variety of molecules that can mediate cell communication, modulating crucial cell processes such as homeostasis, induction/dampening of inflammation, and promotion of repair. Since their discovery in 1946, the scientific interest on EVs has increased exponentially promoting promising studies on their possible roles in health and disease.
exosomes
microvesicles
multivesicular bodies
liposomes
1. Introduction
Extracellular vesicles (EVs) are a family of particles released from the cell that are delimited by a lipid bilayer and cannot replicate, i.e., do not contain a functional nucleus, and can be detected in culture supernatants of different bodily fluids [1][2][3][4][5]. Once considered nothing else than sticky cell debris, they are now recognized as alternative mediators of cell communication, being able to convey a broad range of molecules such as cytokines, inflammatory mediators, and miRNA [1][2][3][4][5][6], crucial in the maintenance of homeostasis, induction/dampening of inflammation, and promotion of repair [1][2][3][4][5][6]. Recognized today as “extracellular vesicles” (EVs), they were initially stratified and nominated based on their size from <100 nm, traditionally named exosomes; 100 to 1000 nm, also recognized as microvesicles, microparticles or ectosomes; and apoptotic bodies when >1000 nm [7], a nomenclature that was eventually modified after the MISEV 18 guidelines.
As shown by the number of publications (Figure 1), there has been an exponential increase in the interest in EVs and their potential applications in understanding the underlying mechanisms of various diseases, such as cancer, cardiovascular, metabolic, neurological, and infectious diseases, among others, which have revealed a role for EVs as promising biomarker candidates for diagnosis, prognosis, and even therapeutic tools, in lung and other diseases [8][9][10][11]. Furthermore, investigations performed with cell cultures of peripheral and epithelial cells from various systems highlight the peculiar release of specific cargoes of these particles when exposed to different triggers, which points to the possibility that EVs might be bioengineered as shuttles of therapeutic drugs, offering an alternative way of drug delivery for disease treatment [9][11].
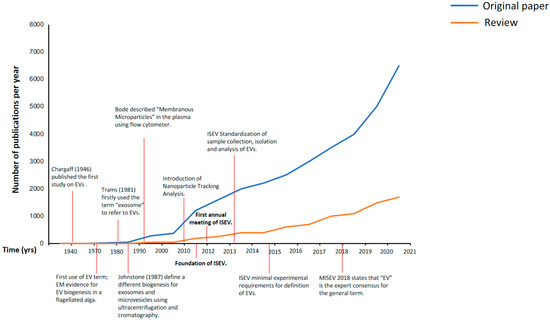
Figure 1. Number of publications per year in PubMed from 1946 to 2021. After the first annual meeting of the International Society of Extracellular Vesicles (ISEV) in 2012, the number of original papers and reviews including any extracellular-vesicle-related terms (exosomes, microvesicles, microparticles, and extracellular vesicles) increased exponentially. EVs: extracellular vesicles; EM: electron microscopy; ISEV: International Society of Extracellular Vesicles; MISEV: Minimal Information for Studies of Extracellular Vesicles.
2. Evolution of the EVs Nomenclature: From 1946 to Today
As shown in Figure 1, recent decades have seen a sharp increase in the number of scientific publications describing the physiological and pathological functions of “extracellular vesicles” (EVs), a collective term covering various subtypes of cell-released membranous structures referred to as exosomes, microvesicles, microparticles, ectosomes, oncosomes, apoptotic bodies, and many other names. Unfortunately, these terms, even today, mean different things to different investigators, which creates vexing problems related to their nomenclature. Furthermore, specific issues arise when working with these entities, whose size and amount often make them difficult to obtain as relatively pure preparations and to characterize properly.
A brief description of the development of the knowledge and nomenclature of EVs might help to understand the evolution of the field, possibly clarify the nomenclature, and expose the issues that need to be reviewed in order to progress in the understanding and clinical use of EVs [1][12].
The existence and possible function of EVs were initially suspected in 1946 by Chargaff and West while studying thromboplastin and platelets [13]. They hypothesized that, in addition to the thromboplastic agent, a “variety of minute breakdown products of the blood corpuscles” contributed to the clotting properties [13]. In 1967, Wolf [14] investigated and provided evidence showing that the responsible plasma coagulant material, hypothesized by Chargaff as “minute breakdown products of the blood corpuscles”, was indeed minute particulate material rich in lipid content originating from platelets, as clearly shown by electron microscopy (EM) and referred to as “platelet-dust”. It should be thus considered that it was Wolf who provided the first description of not only EVs but also of their function in the specific conditions investigated.
The term “intracellular and extracellular vesicles” was first used in 1971 by Aaronson et al. [15], who, by using EM, gave the first ultrastructural evidence for the production of a wide variety of intra- and extracellular membranous structures in O. danica, a flagellated alga. Importantly, Aaronson showed that vesicles and other membranous structures arose from several cell organelles, which clearly recognized EV biogenesis as a biological process and not a fixation artefact [15]. They also described how intact vesicles could be recovered by centrifugation. However, despite these first observations, throughout the 1970s, the lack of precise information on the biology of EVs raised important questions about the origin of these membranes, specifically whether they come from viral shedding out of the cell or from random debris of dead cells during ultracentrifugation or are actually released from living cells in a more or less specific process.
Studies in the late 1970s using EM and chemical lysis (nitrogen cavitation) identified in fetal bovine serum numerous elongate and spherical microvesicles with a trilaminar membranous envelope ranging from 30 to 60 nm. These structures were shown to be either normal cell components or the breakdown products of normal cell components, not to be confused with viruses. They were classified as “plasma membranes”, “extracellular membranes”, and “microvesicles” according to the compartment of origin [16].
Later studies further confirmed that they were not virus-like particles nor cell debris but represented a biological entity derived from intra- and extracellular compartments; however, no further information on the biological origin, content, and function of EVs was given [17].
The term exosome (“exo” = outside and “soma” = body) was already introduced in the 1970s [18][19][20], referring to DNA fragments transferred between cells. However, the lack of association of the DNA fragments with lipid bilayers probably prevents these particles from being considered an early description of exosomes or extracellular vesicles. Not until 1981 was the term “exosome” proposed to refer to the exfoliation of “microvesicles” from the plasma membrane by Trams et al. [21]. These authors have contributed important advances to the knowledge and understanding of EV genesis and function. In particular, the fact that exfoliation of membranous vesicles might occur in many different normal and neoplastic cells and that the exfoliative process is selective strongly suggested, based on their lipid composition, that microvesicles consist of specific domains of the plasma membrane [21]. Interestingly, with the use of EM, they described two populations of vesicles, one of which consisted of irregularly shaped vesicles approximately 500 to 1000 nm in diameter which contained another population of smaller, spherical vesicles with an average size of about 40 nm. This was very likely the first description of what later was recognized as a multivesicular body (MVB), the source of exosomes. They also found that constituents of the microvesicles could be transferred to recipient cells and suggested the possibility that the shedding of microvesicles and their interaction with a target cell or target organ represented a physiological phenomenon that takes place in vivo. Conceivably, the vesicle could be incorporated into the recipient cell, thereby producing a modification of the host cell. Of interest, Trams proposed, in 1981, the visionary suggestion of using the liposomes, which contain the “exosome” cargo, to target therapeutic use, a vision that has become a reality for the COVID-19 vaccines [22].
Although Trams’ publication was often cited during the 1980s, the term “exosome” was not used again to refer to EVs until 1986 [23] and again in 1987 by Rose Johnstone and colleagues [24] after they had described in reticulocytes the formation of multivesicular structures, which, after their fusion with membranes, released the vesicles they contained into the medium. By this mechanism, the reticulocyte could rid itself of proteins and structures that were no longer required in order to become a mature red cell [25]. These authors were the first to report about exosomes as intraluminal vesicles of multivesicular endosomes that were secreted upon fusion with the plasma membrane [26], defining for the first time different biogenesis pathways for exosomes and microvesicles [27].
Initially described as a means to extrude obsolete components by a very specific cell type, the reticulocyte, exosomes remained minimally investigated for the following 10 years. In addition, most cell biologists remained skeptical about the actual existence of this “weird” secretion pathway and were convinced that exosomes were merely membrane fragments artificially released upon in vitro cell handling. Subsequent to their discovery in reticulocytes, the MVB structures and exosomes were eventually shown to be present in many cell types, including B lymphocytes in 1996 [28], dendritic cells in 1998 [29], platelet [30] and epithelial cells in 1991 [31], and neurons in 2005 [32]. Owing to the original findings that the MVB/exosome complex could have an important role in the immune activation and stimulation of adaptive immune responses [28][29], a renewed interest in exosomes arose in the immunology field, which provided essential information about their generation and biology [33]. It is now recognized that the generation of exosomes, with budding, fission, and segregation occurring within the MVB lumen, does not occur by default but is governed by specific processes. Furthermore, the discharge of exosomes has been shown to take place not only by constitutive but also by regulated exocytosis of MVBs activated in response to specific intracellular signals [34]. Nowadays, exosomes are described in mammals and invertebrates and appear to be involved in many different processes [35][36][37][38].
2.1. Exosomes or Microvesicles or Extracellular Vesicles (EVs)
The technological outgrowth started in the 1990s, which introduced new methods facilitating the study of EVs beyond the EM (nanoparticle-tracking analysis, dynamic light scattering, high-resolution flow cytometry), contributing to a progressive expansion of the scientific knowledge and publications on the field (Figure 1 and Figure 2). However, researchers continued to refer to EVs indiscriminately as either exosomes or microvesicles but also as ectosomes, membrane particles, exosome-like vesicles, and apoptotic vesicles [33]. In the period from the late 1990s to the early 2010s, the growth of the curves of the scientific production referred to as “exosomes” or “microvesicles” were completely overlapped (Figure 2), as much as the two terms were in researchers’ minds.
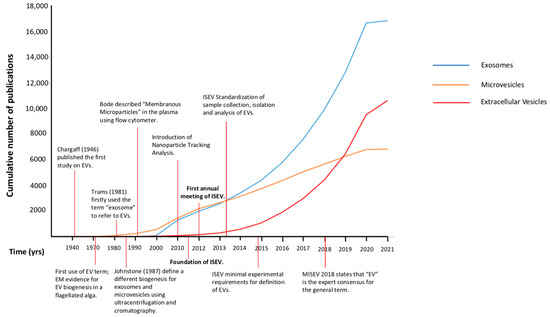
Figure 2. Cumulative number of publications. Comparative evolution of the use of different terms for EVs (exosomes, microvesicles or extracellular vesicles) in the literature from 1946 to 2021. EVs: extracellular vesicles; EM: electron microscopy; ISEV: International Society of Extracellular Vesicles; MISEV: minimal information for studies of extracellular vesicles.
It was Gyorgy et al. [7] in 2011 who, aiming to clarify the confusion created by the heterogeneity of vesicle terminology, suggested that the large number of mobile membrane-limited vesicles contained in the extracellular environment should be termed “extracellular vesicles” and not microparticles, as particle suggest a solid, particulate structure rather than a vesicular one. EVs would include exosomes, activation- or apoptosis-induced microvesicles/microparticles, and apoptotic bodies [7]. They also proposed, to make the terminology unambiguous, to classify EVs into three broad classes based on their biogenesis, secretory components, and size: exosomes, microvesicles, and apoptotic bodies. Exosomes are 50–100 nm EVs of endosomal origin containing certain surface markers including tetraspanins. Microvesicles are larger than exosomes (100–1000 nm) and are derived from the plasma membrane of cells through direct outward budding. They contain membrane components, as do their parent cells. Apoptotic bodies are released from cells that undergo apoptosis and can be 50–5000 nm in diameter. They may contain DNA fragments, noncoding RNAs, and cell organelles [7].
By 2013, Gould and Raposo [39] highlighted how the expanding interest in EVs had also introduced some vexing problems related to their nomenclature and how the proposed generic terms meant different things to different investigators. The term exosome has been used, and still is nowadays, in three different ways: some investigators following the original biogenetic definition (i.e., vesicles that bud into endosomes and are released when the resulting MVBs fuse with the plasma membrane [24][28]); others preferring the original, broad definition (i.e., secreted vesicles that ‘‘may serve a physiologic function’’ [21][40]); and others still employing an empirical definition based on differential centrifugation (i.e., vesicles that sediment only after centrifugation at 70,000–100,000 g [24][41]). A similar range of definitions was evident for the term microvesicle, which some define as vesicles that bud from the plasma membrane [33][42], others define as all secreted vesicles [43], and others still define on the basis of differential centrifugation (i.e., vesicles that sediment at 10,000 g [33][41]).
It is of interest how the term exosome gained popularity among investigators, as evidenced by the increasing number of publications over the years referring to exosomes (Figure 2). Probably the fact that the exosome, along with the MVB, were the EVs most studied and more clearly described attracted its use, even when it might not have been appropriated. The description of MVBs in immune cells and how the resulting exosomes had a function in adaptive immunity gave the exosomes a relevance never reached by “microvesicles” being mere pieces of the cell membrane [44].
Soon after the description of MVBs and their 40 to 80 nm exosomes cargo by Johnstone in 1985 [25], differential centrifugation and ultracentrifugation above 100,000 g was used in order to isolate and differentiate the smallest vesicles, the exosomes, from EVs larger than 100 nm, the microvesicles, and apoptotic bodies [26]. This technique was used broadly, alone or along with EM and other techniques, to study the properties, biological content, and identification markers of these smallest EVs, the exosomes. Extensive literature has revealed how exosomes can be identified by the expression of endosome tetraspanins (CD37, CD53, CD63, CD81, and CD82, among others), which are possibly responsible for cell penetration, invasion and fusion events [45][46]. Furthermore, in 2007 Valadi et al. [47] showed that exosomes obtained from human mast cell line cultures, identified by ultracentrifugation and expression of CD63 by FACS analysis, contained RNA from approximately 1000 genes, many of which were not present in the cytoplasm of the donor cell. Further analysis revealed also the presence of miRNA, which was transferable to other human and mouse cells. However, in addition to exosomes, various other membrane-derived vesicles (~50–4000 nm in diameter) in the circulation were also found to contain miRNA [48].
2.2. The International Society for Extracellular Vesicles (ISEV) 2013: First Attempt to Introduce Some Order into EV Science
As the interest and consequent number of publications on EVs, mainly exosomes (the ‘‘buzz term’’ for EV-related science), increased, by 2013, it was becoming evident that most studies were not clearly defining the origin of EVs under study, neither were they defining the relative contributions of exosomes and of other secreted membrane vesicles in the proposed functions. It was by then clearly recognized that a major ongoing challenge was to establish methods that would allow discrimination between exosomes and microvesicles, as differences in properties such as size, morphology, buoyant density, and protein composition seemed insufficient for a clear distinction. Paradoxically, the increasing interest for the content and function had progressively reduced the relevance for a precise nomenclature in classifying EVs, therefore leading once again to a confusing scientific production. In response to this confusion, in 2013, the International Society for Extracellular Vesicles (ISEV), a group of scientists with collective long-term expertise in the field of EV biology, proposed a series of criteria, based on best current practice, that represented the minimal characterization of EVs that should be reported by investigators. The idea was that adoption of these criteria should aid researchers in planning studies and reporting their results. In addition, the ISEV’s recommendations suggested appropriate controls that should be included in EV-related functional studies [2][3].
References
- Théry, C.; Witwer, K.W.; Aikawa, E.; Alcaraz, M.J.; Anderson, J.D.; Andriantsitohaina, R.; Antoniou, A.; Arab, T.; Archer, F. Minimal information for studies of extracellular vesicles (MISEV2018): A position statement of the International Society for Extracellular Vesicles and update of the MISEV2014 guidelines. J. Extracell. Vesicles 2018, 7, 1535750.
- Lötvall, J.; Hill, A.F.; Hochberg, F.; Buzás, E.I.; Di Vizio, D.; Gardiner, C.; Gho, Y.S.; Kurochkin, I.V.; Mathivanan, S.; Quesenberry, P.; et al. Minimal experimental requirements for definition of extracellular vesicles and their functions: A position statement from the International Society for Extracellular Vesicles. J. Extracell. Vesicles 2014, 3, 26913.
- Witwer, K.W.; Buzás, E.I.; Bemis, L.T.; Bora, A.; Lässer, C.; Lötvall, J.; Nolte-’t Hoen, E.N.; Piper, M.G.; Sivaraman, S.; Skog, J.; et al. Standardization of sample collection, isolation and analysis methods in extracellular vesicle research. J. Extracell. Vesicles 2013, 2, 20360.
- Fernández-Messina, L.; Gutiérrez-Vázquez, C.; Rivas-García, E.; Sánchez-Madrid, F.; Fuente, H.D. Immunomodulatory role of microRNAs transferred by extracellular vesicles. Biol. Cell 2015, 107, 61–77.
- EV-TRACK Consortium; Van Deun, J.; Mestdagh, P.; Agostinis, P.; Akay, Ö.; Anand, S.; Anckaert, J.; Martinez, Z.A.; Baetens, T.; Beghein, E. EV-TRACK: Transparent reporting and centralizing knowledge in extracellular vesicle research. Nat. Methods 2017, 14, 228–232.
- Słomka, A.; Urban, S.K.; Lukacs-Kornek, V.; Żekanowska, E.; Kornek, M. Large Extracellular Vesicles: Have We Found the Holy Grail of Inflammation? Front. Immunol. 2018, 9, 2723.
- György, B.; Szabó, T.G.; Pásztói, M.; Pál, Z.; Misják, P.; Aradi, B.; László, V.; Pállinger, E.; Pap, E.; Kittel, A.; et al. Membrane vesicles, current state-of-the-art: Emerging role of extracellular vesicles. Cell Mol. Life Sci. 2011, 68, 2667–2688.
- Shah, R.; Patel, T.; Freedman, J.E. Circulating Extracellular Vesicles in Human Disease. N. Engl. J. Med. 2018, 379, 958–966.
- Hartjes, T.A.; Mytnyk, S.; Jenster, G.W.; van Steijn, V.; van Royen, M.E. Extracellular Vesicle Quantification and Characterization: Common Methods and Emerging Approaches. Bioengineering 2019, 6, 7.
- Simeone, P.; Bologna, G.; Lanuti, P.; Pierdomenico, L.; Guagnano, M.T.; Pieragostino, D.; Del Boccio, P.; Vergara, D.; Marchisio, M.; Miscia, S.; et al. Extracellular Vesicles as Signaling Mediators and Disease Biomarkers across Biological Barriers. Int. J. Mol. Sci. 2020, 21, 2514.
- Vader, P.; Mol, E.A.; Pasterkamp, G.; Schiffelers, R.M. Extracellular vesicles for drug delivery. Adv Drug Deliv. Rev. 2016, 106, 148–156.
- Aucamp, J.; Bronkhorst, A.J.; Badenhorst, C.P.S.; Pretorius, P.J. A historical and evolutionary perspective on the biological significance of circulating DNA and extracellular vesicles. Cell Mol. Life Sci. 2016, 73, 4355–4381.
- Chargaff, E.; West, R. The biological significance of the thromboplastic protein of blood. J. Biol. Chem. 1946, 166, 189–197.
- Wolf, P. The nature and significance of platelet products in human plasma. Br. J. Haematol. 1967, 13, 269–288.
- Aaronson, S.; Behrens, U.; Orner, R.; Haines, T.H. Ultrastructure of intracellular and extracellular vesicles, membranes, and myelin figures produced by Ochromonas danica. J. Ultrastruct. Res. 1971, 35, 418–430.
- Dalton, A.J. Microvesicles and vesicles of multivesicular bodies versus “virus-like” particles. J. Natl. Cancer Inst. 1975, 54, 1137–1148.
- van Blitterswijk, W.J.; Emmelot, P.; Hilkmann, H.A.; Hilgers, J.; Feltkamp, C.A. Rigid plasma-membrane-derived vesicles, enriched in tumour-associated surface antigens (MLr), occurring in the ascites fluid of a murine leukaemia (GRSL). Int. J. Cancer 1979, 23, 62–70.
- Fox, A.S.; Yoon, S.B. DNA-induced transformation in Drosophila: Locus-specificity and the establishment of transformed stocks. Proc. Natl. Acad. Sci. USA 1970, 67, 1608–1615.
- Fox, A.S.; Yoon, S.B.; Gelbart, W.M. DNA-induced transformation in Drosophila: Genetic analysis of transformed stocks. Proc. Natl. Acad. Sci. USA 1971, 68, 342–346.
- Mishra, N.C.; Tatum, E.L. Non-Mendelian inheritance of DNA-induced inositol independence in Neurospora. Proc. Natl. Acad. Sci. USA 1973, 70, 3875–3879.
- Trams, E.G.; Lauter, C.J.; Salem, N.; Heine, U. Exfoliation of membrane ecto-enzymes in the form of micro-vesicles. Biochim. Biophys. Acta 1981, 645, 63–70.
- Mulligan, M.J.; Lyke, K.E.; Kitchin, N.; Absalon, J.; Gurtman, A.; Lockhart, S.; Neuzil, K.; Raabe, V.; Bailey, R.; Swanson, K.A.; et al. Phase I/II study of COVID-19 RNA vaccine BNT162b1 in adults. Nature 2020, 586, 589–593.
- Kassis, S.; Lauter, C.J.; Stojanov, M.; Salem, N., Jr. Exfoliation of the beta-adrenergic receptor and the regulatory components of adenylate cyclase by cultured rat glioma C6 cells. Biochim. Biophys. Acta 1986, 886, 474–482.
- Johnstone, R.M.; Adam, M.; Hammond, J.R.; Orr, L.; Turbide, C. Vesicle formation during reticulocyte maturation. Association of plasma membrane activities with released vesicles (exosomes). J. Biol. Chem. 1987, 262, 9412–9420.
- Pan, B.T.; Teng, K.; Wu, C.; Adam, M.; Johnstone, R.M. Electron Microscopic Evidence for Externalization of the Transferrin Receptor in Vesicular Form in Sheep Reticulocytes. J. Cell Biol. 1985, 101, 942–948.
- Johnstone, R.M.; Bianchini, A.; Teng, K. Reticulocyte maturation and exosome release: Transferrin receptor containing exosomes shows multiple plasma membrane functions. Blood 1989, 74, 1844–1851.
- van Niel, G.; D’Angelo, G.; Raposo, G. Shedding light on the cell biology of extracellular vesicles. Nat. Rev. Mol. Cell Biol. 2018, 19, 213–228.
- Raposo, G.; Nijman, H.W.; Stoorvogel, W.; Liejendekker, R.; Harding, C.V.; Melief, C.J.; Geuze, H.J. B lymphocytes secrete antigen-presenting vesicles. J. Exp. Med. 1996, 183, 1161–1172.
- Zitvogel, L.; Regnault, A.; Lozier, A.; Wolfers, J.; Flament, C.; Tenza, D.; Ricciardi-Castagnoli, P.; Raposo, G.; Amigorena, S. Eradication of established murine tumors using a novel cell-free vaccine: Dendritic cell-derived exosomes. Nat. Med. 1998, 4, 594–600.
- Heijnen, H.F.; Schiel, A.E.; Fijnheer, R.; Geuze, H.J.; Sixma, J.J. Activated platelets release two types of membrane vesicles: Microvesicles by surface shedding and exosomes derived from exocytosis of multivesicular bodies and alpha-granules. Blood 1999, 94, 3791–3799.
- Wilson, J.M.; Whitney, J.A.; Neutra, M.R. Biogenesis of the apical endosome-lysosome complex during differentiation of absorptive epithelial cells in rat ileum. J. Cell Sci. 1991, 100, 133–143.
- Marzesco, A.M.; Janich, P.; Wilsch-Brauninger, M.; Dubreuil, V.; Langenfeld, K.; Corbeil, D.; Huttner, W.B. Release of extracellular membrane particles carrying the stem cell marker prominin-1 (CD133) from neural progenitors and other epithelial cells. J. Cell Sci. 2005, 118, 2849–2858.
- Théry, C.; Ostrowski, M.; Segura, E. Membrane vesicles as conveyors of immune responses. Nat. Rev. Immunol. 2009, 9, 581–593.
- Cocucci, E.; Racchetti, G.; Meldolesi, J. Shedding microvesicles: Artefacts no more. Trends Cell Biol. 2009, 19, 43–51.
- Bobrie, A.; Colombo, M.; Raposo, G.; Théry, C. Exosome secretion: Molecular mechanisms and roles in immune responses. Traffic 2011, 12, 1659–1668.
- Saeedi, S.; Israel, S.; Nagy, C.; Turecki, G. The emerging role of exosomes in mental disorders. Transl. Psychiatry 2019, 9, 122.
- Larabi, A.; Barnich, N.; Nguyen, H.T.T. Emerging Role of Exosomes in Diagnosis and Treatment of Infectious and Inflammatory Bowel Diseases. Cells 2020, 9, 1111.
- Kalluri, R.; LeBleu, V.S. The biology, function, and biomedical applications of exosomes. Science 2020, 367, eaau6977.
- Gould, S.J.; Raposo, G. As we wait: Coping with an imperfect nomenclature for extracellular vesicles. J. Extracell. Vesicles 2013, 2, 20389.
- Booth, A.M.; Fang, Y.; Fallon, J.K.; Yang, J.M.; Hildreth, J.E.; Gould, S.J. Exosomes and HIV Gag bud from endosome-like domains of the T cell plasma membrane. J. Cell Biol. 2006, 172, 923–935.
- Thery, C.; Amigorena, S.; Raposo, G.; Clayton, A. Isolation and characterization of exosomes from cell culture supernatants and biological fluids. Curr. Protoc. Cell Biol. 2006, 30, 1–29.
- Simons, M.; Raposo, G. Exosomes–vesicular carriers for intercellular communication. Curr. Opin. Cell Biol. 2009, 21, 575–581.
- Bess, J.W.; Gorelick, R.J.; Bosche, W.J.; Henderson, L.E.; Arthur, L.O. Microvesicles are a source of contaminating cellular proteins found in purified HIV-1 preparations. Virology 1997, 230, 134–144.
- Pulecio, J.; Petrovic, J.; Prete, F.; Chiaruttini, G.; Lennon-Dumenil, A.M.; Desdouets, C.; Gasman, S.; Burrone, O.R.; Benvenuti, F. Cdc42-mediated MTOC polarization in dendritic cells controls targeted delivery of cytokines at the immune synapse. J. Exp. Med. 2010, 207, 2719–2732.
- Andreu, Z.; Yáñez-Mó, M. Tetraspanins in extracellular vesicle formation and function. Front. Immunol. 2014, 5, 1–12.
- Gurunathan, S.; Kang, M.H.; Kim, J.H. A Comprehensive Review on Factors Influences Biogenesis, Functions, Therapeutic and Clinical Implications of Exosomes. Int. J. Nanomed. 2021, 16, 1281–1312.
- Valadi, H.; Ekström, K.; Bossios, A.; Sjöstrand, M.; Lee, J.J.; Lötvall, J.O. Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nat. Cell Biol. 2007, 9, 654–659.
- Vickers, K.; Remaley, A. Lipid-based carriers of microRNAs and intercellular communication. Curr. Opin. Lipidol. 2012, 23, 91–97.
More
Information
Subjects:
Cardiac & Cardiovascular Systems
Contributor
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
1.2K
Revisions:
2 times
(View History)
Update Date:
24 Jun 2021
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No





