
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Javier Martin-Broto | + 3548 word(s) | 3548 | 2021-06-15 08:32:49 | | | |
| 2 | Peter Tang | Meta information modification | 3548 | 2021-06-22 09:03:49 | | |
Video Upload Options
Solitary fibrous tumor (SFT) is a malignant condition that exhibits different clinical behaviors ranging from low to high aggressive SFT. Even when surgery alone provides curation rates above 60%, recurrences do occur in a fraction of patients where surgery is unable to provide disease control. Among the systemic therapeutic options, antiangiogenic compounds have shown higher efficacy than chemotherapy by indirect comparisons.
1. Introduction
2. Clinical Aspects
3. Pathogenesis and Pathology
3.1. Morphological Features
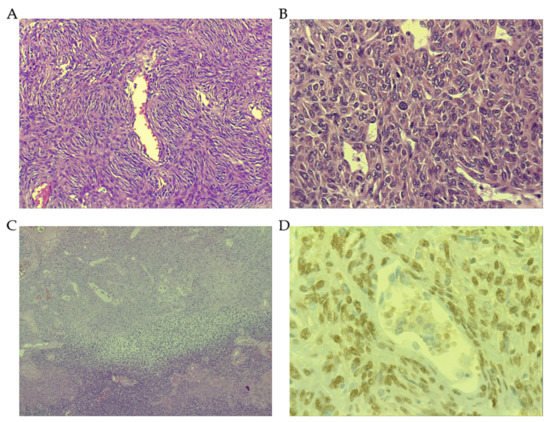
3.2. The Role of Signal Transducer and Activator of Transcription 6 (STAT6)
|
Marker |
Normal Tissues/Precursors |
Mesenchymal Benign Entities |
Mesenchymal Malignant Tumors |
|---|---|---|---|
|
CD 34 |
-Early hematopoietic stem cells |
- Giant cell fibroblastoma - Lipoma |
-SFT * |
|
-Mesenchymal stem cells |
-Kaposi sarcoma |
||
|
-Small-vessels endothelial cells |
-Low-grade myofibroblastic sarcoma |
||
|
-Embryonic fibroblasts |
-Inflammatory myofibroblastic tumors |
||
|
-Endoneurial cells, dermal dendritic interstitial fibroblastic cells |
-DFSP |
||
|
-Adipocitic cells |
-GIST |
||
|
bcl 2 |
-Stem cells -Endocrine tissue -Long-lived cells |
-Schwannoma -Spindle cell lipoma -Dendritic fibromyxolipoma -Neurofibromas (focal) |
-SFT * -Fibrosarcoma -Low-grade myxofibrosarcoma -Dedifferentiated liposarcoma -Synovial sarcoma -DFSP -GIST -Kaposi sarcoma -MPNST |
|
CD99 |
-Bone marrow cells -Leukocytes |
-Fibroma -Giant cell angiofibroma |
-SFT * -Ewing sarcoma -Synovial sarcoma -Rhabdomyosarcoma -Osteosarcoma |
|
STAT 6 |
-SFT * -Unclassified sarcomas of spindle cell or epithelioid morphology (12%) -Desmoid tumors (8%) -Neurofibromas (5%) -Clear cell sarcoma (5%) -Well-diff./dedifferentiated liposarcoma |
* Expression can be lost in dedifferentiated solitary fibrous tumors (SFT); DFSP: Dermatofibrosarcoma protuberans; GIST: Gastrointestinal stromal tumors; MPSNT: Malignant peripheral stealth nerve tumors.
3.3. Other Immunohistochemical Markers
4. Dedifferentiated SFT (DD-SFT)
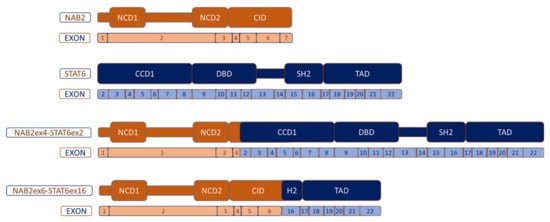
5. Molecular Biology
References
- Kinslow, C.J.; Wang, T.J.C. Incidence of extrameningeal solitary fibrous tumors. Cancer 2020, 126, 4067.
- Kinslow, C.J.; Bruce, S.S.; Rae, A.I.; Sheth, S.A.; McKhann, G.M.; Sisti, M.B.; Bruce, J.N.; Sonabend, A.M.; Wang, T.J.C. Solitary-fibrous tumor/hemangiopericytoma of the central nervous system: A population-based study. J. Neuro-Oncol. 2018, 138, 173–182.
- Fletcher, C.D. The evolving classification of soft tissue tumours—An update based on the new 2013 WHO classification. Histopathology 2014, 64, 2–11.
- Kallen, M.E.; Hornick, J.L. The 2020 WHO Classification: What’s New in Soft Tissue Tumor Pathology? Am. J. Surg. Pathol. 2021, 45, e1–e23.
- O’Neill, A.C.; Tirumani, S.H.; Do, W.S.; Keraliya, A.R.; Hornick, J.L.; Shinagare, A.B.; Ramaiya, N.H. Metastatic Patterns of Solitary Fibrous Tumors: A Single-Institution Experience. AJR. Am. J. Roentgenol. 2017, 208, 2–9.
- Demicco, E.G.; Park, M.S.; Araujo, D.M.; Fox, P.S.; Bassett, R.L.; Pollock, R.E.; Lazar, A.J.; Wang, W.L. Solitary fibrous tumor: A clinicopathological study of 110 cases and proposed risk assessment model. Mod. Pathol. 2012, 25, 1298–1306.
- Baldi, G.G.; Stacchiotti, S.; Mauro, V.; Dei Tos, A.P.; Gronchi, A.; Pastorino, U.; Duranti, L.; Provenzano, S.; Marrari, A.; Libertini, M.; et al. Solitary fibrous tumor of all sites: Outcome of late recurrences in 14 patients. Clin. Sarcoma Res. 2013, 3, 4.
- Fritchie, K.; Jensch, K.; Moskalev, E.A.; Caron, A.; Jenkins, S.; Link, M.; Brown, P.D.; Rodriguez, F.J.; Guajardo, A.; Brat, D.; et al. The impact of histopathology and NAB2-STAT6 fusion subtype in classification and grading of meningeal solitary fibrous tumor/hemangiopericytoma. Acta Neuropathol. 2019, 137, 307–319.
- Salas, S.; Resseguier, N.; Blay, J.Y.; Le Cesne, A.; Italiano, A.; Chevreau, C.; Rosset, P.; Isambert, N.; Soulie, P.; Cupissol, D.; et al. Prediction of local and metastatic recurrence in solitary fibrous tumor: Construction of a risk calculator in a multicenter cohort from the French Sarcoma Group (FSG) database. Ann. Oncol. 2017, 28, 1979–1987.
- Lee, J.C.; Fletcher, C.D. Malignant fat-forming solitary fibrous tumor (so-called “lipomatous hemangiopericytoma”): Clinicopathologic analysis of 14 cases. Am. J. Surg. Pathol. 2011, 35, 1177–1185.
- Dong, S.S.; Wang, N.; Yang, C.P.; Zhang, G.C.; Liang, W.H.; Zhao, J.; Qi, Y. Giant Cell-Rich Solitary Fibrous Tumor in the Nasopharynx: Case Report and Literature Review. OncoTargets Ther. 2020, 13, 6819–6826.
- Thway, K.; Hayes, A.; Ieremia, E.; Fisher, C. Heterologous osteosarcomatous and rhabdomyosarcomatous elements in dedifferentiated solitary fibrous tumor: Further support for the concept of dedifferentiation in solitary fibrous tumor. Ann. Diagn. Pathol. 2013, 17, 457–463.
- Doyle, L.A.; Vivero, M.; Fletcher, C.D.; Mertens, F.; Hornick, J.L. Nuclear expression of STAT6 distinguishes solitary fibrous tumor from histologic mimics. Mod. Pathol. 2014, 27, 390–395.
- Dagrada, G.P.; Spagnuolo, R.D.; Mauro, V.; Tamborini, E.; Cesana, L.; Gronchi, A.; Stacchiotti, S.; Pierotti, M.A.; Negri, T.; Pilotti, S. Solitary fibrous tumors: Loss of chimeric protein expression and genomic instability mark dedifferentiation. Mod. Pathol. 2015, 28, 1074–1083.
- Barron, L.; Wynn, T.A. Fibrosis is regulated by Th2 and Th17 responses and by dynamic interactions between fibroblasts and macrophages. Am. J. Physiol. Gastrointest. Liver Physiol. 2011, 300, G723–G728.
- Ingram, J.L.; Antao-Menezes, A.; Mangum, J.B.; Lyght, O.; Lee, P.J.; Elias, J.A.; Bonner, J.C. Opposing actions of Stat1 and Stat6 on IL-13-induced up-regulation of early growth response-1 and platelet-derived growth factor ligands in pulmonary fibroblasts. J. Immunol. 2006, 177, 4141–4148.
- Demicco, E.G.; Harms, P.W.; Patel, R.M.; Smith, S.C.; Ingram, D.; Torres, K.; Carskadon, S.L.; Camelo-Piragua, S.; McHugh, J.B.; Siddiqui, J.; et al. Extensive survey of STAT6 expression in a large series of mesenchymal tumors. Am. J. Clin. Pathol. 2015, 143, 672–682.
- Doyle, L.A.; Tao, D.; Marino-Enriquez, A. STAT6 is amplified in a subset of dedifferentiated liposarcoma. Mod. Pathol. 2014, 27, 1231–1237.
- Ouladan, S.; Trautmann, M.; Orouji, E.; Hartmann, W.; Huss, S.; Buttner, R.; Wardelmann, E. Differential diagnosis of solitary fibrous tumors: A study of 454 soft tissue tumors indicating the diagnostic value of nuclear STAT6 relocation and ALDH1 expression combined with in situ proximity ligation assay. Int. J. Oncol. 2015, 46, 2595–2605.
- Yokoi, T.; Tsuzuki, T.; Yatabe, Y.; Suzuki, M.; Kurumaya, H.; Koshikawa, T.; Kuhara, H.; Kuroda, M.; Nakamura, N.; Nakatani, Y.; et al. Solitary fibrous tumour: Significance of p53 and CD34 immunoreactivity in its malignant transformation. Histopathology 1998, 32, 423–432.
- Waller, E.K.; Olweus, J.; Lund-Johansen, F.; Huang, S.; Nguyen, M.; Guo, G.R.; Terstappen, L. The “common stem cell” hypothesis reevaluated: Human fetal bone marrow contains separate populations of hematopoietic and stromal progenitors. Blood 1995, 85, 2422–2435.
- Quirici, N.; Soligo, D.; Bossolasco, P.; Servida, F.; Lumini, C.; Deliliers, G.L. Isolation of bone marrow mesenchymal stem cells by anti-nerve growth factor receptor antibodies. Exp. Hematol. 2002, 30, 783–791.
- De Francesco, F.; Tirino, V.; Desiderio, V.; Ferraro, G.; D’Andrea, F.; Giuliano, M.; Libondi, G.; Pirozzi, G.; De Rosa, A.; Papaccio, G. Human CD34/CD90 ASCs are capable of growing as sphere clusters, producing high levels of VEGF and forming capillaries. PLoS ONE 2009, 4, e6537.
- Hasegawa, T.; Matsuno, Y.; Shimoda, T.; Hirohashi, S.; Hirose, T.; Sano, T. Frequent expression of bcl-2 protein in solitary fibrous tumors. Jpn. J. Clin. Oncol. 1998, 28, 86–91.
- Takizawa, I.; Saito, T.; Kitamura, Y.; Arai, K.; Kawaguchi, M.; Takahashi, K.; Hara, N. Primary solitary fibrous tumor (SFT) in the retroperitoneum. Urol. Oncol. 2008, 26, 254–259.
- Suster, S.; Fisher, C.; Moran, C.A. Expression of bcl-2 oncoprotein in benign and malignant spindle cell tumors of soft tissue, skin, serosal surfaces, and gastrointestinal tract. Am. J. Surg. Pathol. 1998, 22, 863–872.
- Baer, R. Bcl-2 breathes life into embryogenesis. Am. J. Pathol. 1994, 145, 7–10.
- Aronica, M.A.; Goenka, S.; Boothby, M. IL-4-dependent induction of BCL-2 and BCL-X(L)IN activated T lymphocytes through a STAT6- and pi 3-kinase-independent pathway. Cytokine 2000, 12, 578–587.
- Hirakawa, N.; Naka, T.; Yamamoto, I.; Fukuda, T.; Tsuneyoshi, M. Overexpression of bcl-2 protein in synovial sarcoma: A comparative study of other soft tissue spindle cell sarcomas and an additional analysis by fluorescence in situ hybridization. Hum. Pathol. 1996, 27, 1060–1065.
- Manara, M.C.; Pasello, M.; Scotlandi, K. CD99: A Cell Surface Protein with an Oncojanus Role in Tumors. Genes 2018, 9, 159.
- Maitra, A.; Hansel, D.E.; Argani, P.; Ashfaq, R.; Rahman, A.; Naji, A.; Deng, S.; Geradts, J.; Hawthorne, L.; House, M.G.; et al. Global expression analysis of well-differentiated pancreatic endocrine neoplasms using oligonucleotide microarrays. Clin. Cancer Res. 2003, 9, 5988–5995.
- Manara, M.C.; Bernard, G.; Lollini, P.L.; Nanni, P.; Zuntini, M.; Landuzzi, L.; Benini, S.; Lattanzi, G.; Sciandra, M.; Serra, M.; et al. CD99 acts as an oncosuppressor in osteosarcoma. Mol. Biol. Cell 2006, 17, 1910–1921.
- Scotlandi, K.; Zuntini, M.; Manara, M.C.; Sciandra, M.; Rocchi, A.; Benini, S.; Nicoletti, G.; Bernard, G.; Nanni, P.; Lollini, P.L.; et al. CD99 isoforms dictate opposite functions in tumour malignancy and metastases by activating or repressing c-Src kinase activity. Oncogene 2007, 26, 6604–6618.
- Bremond, A.; Meynet, O.; Mahiddine, K.; Coito, S.; Tichet, M.; Scotlandi, K.; Breittmayer, J.P.; Gounon, P.; Gleeson, P.A.; Bernard, A.; et al. Regulation of HLA class I surface expression requires CD99 and p230/golgin-245 interaction. Blood 2009, 113, 347–357.
- Olson, N.J.; Linos, K. Dedifferentiated Solitary Fibrous Tumor: A Concise Review. Arch. Pathol. Lab. Med. 2018, 142, 761–766.
- Han, Y.; Zhang, Q.; Yu, X.; Han, X.; Wang, H.; Xu, Y.; Qiu, X.; Jin, F. Immunohistochemical detection of STAT6, CD34, CD99 and BCL-2 for diagnosing solitary fibrous tumors/hemangiopericytomas. Int. J. Clin. Exp. Pathol. 2015, 8, 13166–13175.
- Fletcher, C.D.M.; Bridge, J.A.; Lee, J.C. Extrapleural solitary fibrous tumor. In WHO Classification of Tumors of Soft Tissue and Bone; Fletcher, C.D.M., Bridge, J.A., Hogendoom, P.C.W., Mertens, F., Eds.; IARC: Lyon, France, 2013; pp. 80–82.
- Yang, J.W.; Song, D.H.; Jang, I.S.; Ko, G.H. Dedifferentiated solitary fibrous tumor of thoracic cavity. Korean J. Pathol. 2014, 48, 250–253.
- Schneider, N.; Hallin, M.; Thway, K. STAT6 Loss in Dedifferentiated Solitary Fibrous Tumor. Int. J. Surg. Pathol. 2017, 25, 58–60.
- Schirosi, L.; Lantuejoul, S.; Cavazza, A.; Murer, B.; Yves Brichon, P.; Migaldi, M.; Sartori, G.; Sgambato, A.; Rossi, G. Pleuro-pulmonary solitary fibrous tumors: A clinicopathologic, immunohistochemical, and molecular study of 88 cases confirming the prognostic value of de Perrot staging system and p53 expression, and evaluating the role of c-kit, BRAF, PDGFRs (alpha/beta), c-met, and EGFR. Am. J. Surg. Pathol. 2008, 32, 1627–1642.
- Kurisaki-Arakawa, A.; Akaike, K.; Hara, K.; Arakawa, A.; Takahashi, M.; Mitani, K.; Yao, T.; Saito, T. A case of dedifferentiated solitary fibrous tumor in the pelvis with TP53 mutation. Virchows Arch. 2014, 465, 615–621.
- Akaike, K.; Kurisaki-Arakawa, A.; Hara, K.; Suehara, Y.; Takagi, T.; Mitani, K.; Kaneko, K.; Yao, T.; Saito, T. Distinct clinicopathological features of NAB2-STAT6 fusion gene variants in solitary fibrous tumor with emphasis on the acquisition of highly malignant potential. Hum. Pathol. 2015, 46, 347–356.
- Hu, F.; Xue, M.; Li, Y.; Jia, Y.J.; Zheng, Z.J.; Yang, Y.L.; Guan, M.P.; Sun, L.; Xue, Y.M. Early Growth Response 1 (Egr1) Is a Transcriptional Activator of NOX4 in Oxidative Stress of Diabetic Kidney Disease. J. Diabetes Res. 2018, 2018, 3405695.
- Arakawa, Y.; Miyake, H.; Horiguchi, H.; Inokuchi, T.; Hino, N.; Ogasawara, T.; Kuroda, T.; Yamasaki, S. Overexpression of IGF2 and IGF2 receptor in malignant solitary fibrous tumor with hypoglycemia: A case report. Surg. Case Rep. 2018, 4, 106.
- Yamada, Y.; Kohashi, K.; Kinoshita, I.; Yamamoto, H.; Iwasaki, T.; Yoshimoto, M.; Ishihara, S.; Toda, Y.; Itou, Y.; Koga, Y.; et al. Clinicopathological review of solitary fibrous tumors: Dedifferentiation is a major cause of patient death. Virchows Arch. 2019, 475, 467–477.
- England, D.M.; Hochholzer, L.; McCarthy, M.J. Localized benign and malignant fibrous tumors of the pleura. A clinicopathologic review of 223 cases. Am. J. Surg. Pathol. 1989, 13, 640–658.
- Martin-Broto, J.; Stacchiotti, S.; Lopez-Pousa, A.; Redondo, A.; Bernabeu, D.; de Alava, E.; Casali, P.G.; Italiano, A.; Gutierrez, A.; Moura, D.S.; et al. Pazopanib for treatment of advanced malignant and dedifferentiated solitary fibrous tumour: A multicentre, single-arm, phase 2 trial. Lancet Oncol. 2019, 20, 134–144.
- Chen, R.H.; Du, Y.; Han, P.; Wang, H.B.; Liang, F.Y.; Feng, G.K.; Zhou, A.J.; Cai, M.Y.; Zhong, Q.; Zeng, M.S.; et al. ISG15 predicts poor prognosis and promotes cancer stem cell phenotype in nasopharyngeal carcinoma. Oncotarget 2016, 7, 16910–16922.
- Bahrami, A.; Lee, S.; Schaefer, I.M.; Boland, J.M.; Patton, K.T.; Pounds, S.; Fletcher, C.D. TERT promoter mutations and prognosis in solitary fibrous tumor. Mod. Pathol. 2016, 29, 1511–1522.
- Demicco, E.G.; Wani, K.; Ingram, D.; Wagner, M.; Maki, R.G.; Rizzo, A.; Meeker, A.; Lazar, A.J.; Wang, W.L. TERT promoter mutations in solitary fibrous tumour. Histopathology 2018, 73, 843–851.
- Vogels, R.; Macagno, N.; Griewank, K.; Groenen, P.; Verdijk, M.; Fonville, J.; Kusters, B.; Figarella-Branger, D.; Wesseling, P.; Bouvier, C.; et al. Prognostic significance of NAB2-STAT6 fusion variants and TERT promotor mutations in solitary fibrous tumors/hemangiopericytomas of the CNS: Not (yet) clear. Acta Neuropathol. 2019, 137, 679–682.
- Chmielecki, J.; Crago, A.M.; Rosenberg, M.; O’Connor, R.; Walker, S.R.; Ambrogio, L.; Auclair, D.; McKenna, A.; Heinrich, M.C.; Frank, D.A.; et al. Whole-exome sequencing identifies a recurrent NAB2-STAT6 fusion in solitary fibrous tumors. Nat. Genet. 2013, 45, 131–132.
- Robinson, D.R.; Wu, Y.M.; Kalyana-Sundaram, S.; Cao, X.; Lonigro, R.J.; Sung, Y.S.; Chen, C.L.; Zhang, L.; Wang, R.; Su, F.; et al. Identification of recurrent NAB2-STAT6 gene fusions in solitary fibrous tumor by integrative sequencing. Nat. Genet. 2013, 45, 180–185.
- Mohajeri, A.; Tayebwa, J.; Collin, A.; Nilsson, J.; Magnusson, L.; von Steyern, F.V.; Brosjo, O.; Domanski, H.A.; Larsson, O.; Sciot, R.; et al. Comprehensive genetic analysis identifies a pathognomonic NAB2/STAT6 fusion gene, nonrandom secondary genomic imbalances, and a characteristic gene expression profile in solitary fibrous tumor. Genes Chromosomes Cancer 2013, 52, 873–886.
- Lai, A.Y.; Wade, P.A. Cancer biology and NuRD: A multifaceted chromatin remodelling complex. Nat. Rev. Cancer 2011, 11, 588–596.
- Reynolds, N.; Latos, P.; Hynes-Allen, A.; Loos, R.; Leaford, D.; O’Shaughnessy, A.; Mosaku, O.; Signolet, J.; Brennecke, P.; Kalkan, T.; et al. NuRD suppresses pluripotency gene expression to promote transcriptional heterogeneity and lineage commitment. Cell Stem Cell 2012, 10, 583–594.
- Li, J.; Rodriguez, J.P.; Niu, F.; Pu, M.; Wang, J.; Hung, L.W.; Shao, Q.; Zhu, Y.; Ding, W.; Liu, Y.; et al. Structural basis for DNA recognition by STAT6. Proc. Nat. Acad. Sci. USA 2016, 113, 13015–13020.
- Barthelmess, S.; Geddert, H.; Boltze, C.; Moskalev, E.A.; Bieg, M.; Sirbu, H.; Brors, B.; Wiemann, S.; Hartmann, A.; Agaimy, A.; et al. Solitary fibrous tumors/hemangiopericytomas with different variants of the NAB2-STAT6 gene fusion are characterized by specific histomorphology and distinct clinicopathological features. Am. J. Pathol. 2014, 184, 1209–1218.
- Tai, H.C.; Chuang, I.C.; Chen, T.C.; Li, C.F.; Huang, S.C.; Kao, Y.C.; Lin, P.C.; Tsai, J.W.; Lan, J.; Yu, S.C.; et al. NAB2-STAT6 fusion types account for clinicopathological variations in solitary fibrous tumors. Mod. Pathol. 2015, 28, 1324–1335.
- Bieg, M.; Moskalev, E.A.; Will, R.; Hebele, S.; Schwarzbach, M.; Schmeck, S.; Hohenberger, P.; Jakob, J.; Kasper, B.; Gaiser, T.; et al. Gene Expression in Solitary Fibrous Tumors (SFTs) Correlates with Anatomic Localization and NAB2-STAT6 Gene Fusion Variants. Am. J. Pathol. 2021.





