Solitary fibrous tumor (SFT) is a malignant condition that exhibits different clinical behaviors ranging from low to high aggressive SFT. Even when surgery alone provides curation rates above 60%, recurrences do occur in a fraction of patients where surgery is unable to provide disease control. Among the systemic therapeutic options, antiangiogenic compounds have shown higher efficacy than chemotherapy by indirect comparisons.
- solitary fibrous tumor
- anti-angiogenics
- tumor biology
1. Introduction
2. Clinical Aspects
3. Pathogenesis and Pathology
3.1. Morphological Features
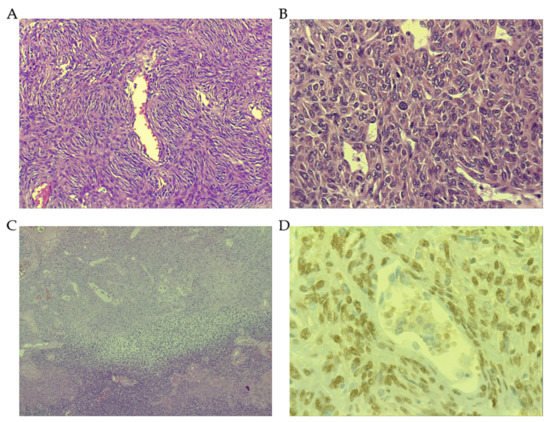
3.2. The Role of Signal Transducer and Activator of Transcription 6 (STAT6)
|
Marker |
Normal Tissues/Precursors |
Mesenchymal Benign Entities |
Mesenchymal Malignant Tumors |
|---|---|---|---|
|
CD 34 |
-Early hematopoietic stem cells |
- Giant cell fibroblastoma - Lipoma |
-SFT * |
|
-Mesenchymal stem cells |
-Kaposi sarcoma |
||
|
-Small-vessels endothelial cells |
-Low-grade myofibroblastic sarcoma |
||
|
-Embryonic fibroblasts |
-Inflammatory myofibroblastic tumors |
||
|
-Endoneurial cells, dermal dendritic interstitial fibroblastic cells |
-DFSP |
||
|
-Adipocitic cells |
-GIST |
||
|
bcl 2 |
-Stem cells -Endocrine tissue -Long-lived cells |
-Schwannoma -Spindle cell lipoma -Dendritic fibromyxolipoma -Neurofibromas (focal) |
-SFT * -Fibrosarcoma -Low-grade myxofibrosarcoma -Dedifferentiated liposarcoma -Synovial sarcoma -DFSP -GIST -Kaposi sarcoma -MPNST |
|
CD99 |
-Bone marrow cells -Leukocytes |
-Fibroma -Giant cell angiofibroma |
-SFT * -Ewing sarcoma -Synovial sarcoma -Rhabdomyosarcoma -Osteosarcoma |
|
STAT 6 |
-SFT * -Unclassified sarcomas of spindle cell or epithelioid morphology (12%) -Desmoid tumors (8%) -Neurofibromas (5%) -Clear cell sarcoma (5%) -Well-diff./dedifferentiated liposarcoma |
* Expression can be lost in dedifferentiated solitary fibrous tumors (SFT); DFSP: Dermatofibrosarcoma protuberans; GIST: Gastrointestinal stromal tumors; MPSNT: Malignant peripheral stealth nerve tumors.
3.3. Other Immunohistochemical Markers
4. Dedifferentiated SFT (DD-SFT)
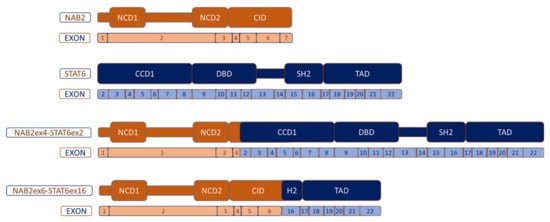
5. Molecular Biology
This entry is adapted from the peer-reviewed paper 10.3390/cancers13122913
