
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Hanna Jöst | + 1293 word(s) | 1293 | 2021-05-17 11:20:14 | | | |
| 2 | Nora Tang | + 982 word(s) | 2275 | 2021-06-11 11:00:07 | | |
Video Upload Options
A cumulative number of MAVs are being discovered in hematophagous arthropods all over the world. There is a potential impact on the fitness and competence of the vector. Therefore, more research is needed to expand the knowledge about their prevalence, distribution, and diversity. The trend of MAVs reported in the literature enlightens how researches about MAVs are on the increase. The past decades have shown a dramatic surge in the literature recounting novel MSVs and classical arboviruses. The increase in research interest has provided new understanding about viral diversity and evolution. Finally, the new viruses discovered have created curiosity in their use as potential biological control agents.
1. Flaviridae
Flaviviruses are enveloped, spherical, and about 50 nm in diameter. The surface proteins of the viral capsid are arranged in an icosahedral-like symmetry. They have a monopartite, linear, ssRNA(+) genome of about 9.7–12 kb [1]. These viruses can be divided into three main groups: those that are maintained in an arthropod–vertebrate cycle and strictly depend on each other are called dual-host flaviviruses [2]; those that are limited to vertebrates with an unknown arthropod relationship are known as vertebrate-specific flavivirus [3]; and those that replicate only in insect cell lines are termed insect-specific flaviviruses [2][4][5]. Flaviviruses and flavivirus-specific antibodies detected in West Africa are shown in Figure 1.
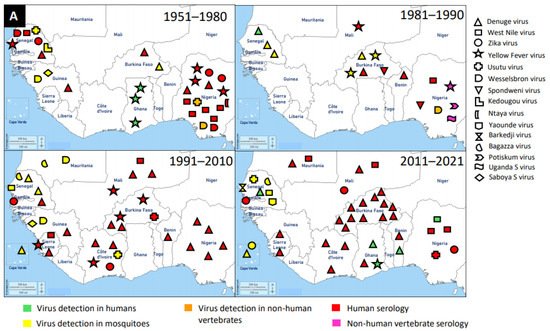
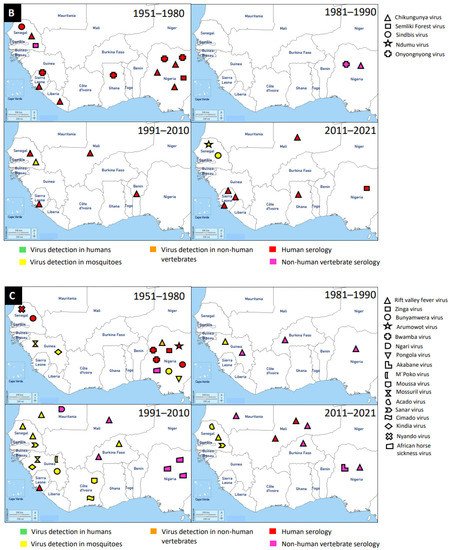
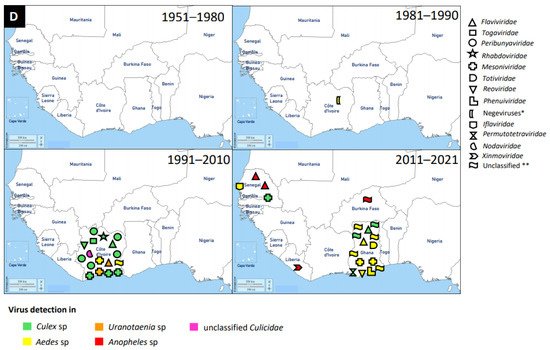
Figure 1. Distribution of MAVs in West Africa. A–C Mosquito-borne viruses (A) Flaviviridae; (B) Togaviridae; (C) Peribunyaviridae, Rhabdoviridae and Reoviridae; (D) Mosquito-specific viruses. Location of the signs in the country was chosen randomly and does not refer to the location where the study was performed; * = Proposed taxon; ** = Unclassified viruses.
The application of next generation sequencing (NGS) techniques in virus discovery has led to the identification and isolation of numerous MSVs [6]. West Africa is not left out in this discovery, and MAVs known to cause outbreaks and those which are not associated with human infections are outlined in Table 1. The first mosquito-specific flavivirus isolated was cell fusing agent virus (CFAV) in 1975 [7], but this virus was never identified in West Africa until 2016 in Ghana [8]. CFAV was isolated from mosquitoes collected via a mosquito surveillance field work from 2015 to 2016 in Ghana. Examples of other MSVs identified in West Africa include: Nounane virus and Nienokoue virus (Côte d’Ivoire); Anopheles flavivirus-like virus and Barkedji virus (Senegal); and Culex flavivirus (Ghana) [9][10][8][11][12]. Nounane and Nienokoue viruses were identified in Tai National Park, a tropical rainforest zone, from the Culex and Uranotaenia mosquitoes respectively [11][12].
Table 1. Mosquito-associated viruses (MAVs) discovered in West Africa from 1951–2021.
| Family | Name of Virus |
Year of Virus Detection |
Country | Source of Virus Detection | Reference |
|---|---|---|---|---|---|
| Flaviviridae | Mosquito-borne viruses | ||||
| Dengue | 2009 | Cape Verde | Aedes aegypti | [13] | |
| 2014/15 | Cape Verde | Ae. aegypti | [14] | ||
| 2016/17 | Ghana | Serology/Human | [15] | ||
| 2016/17 | Ghana | Human | [16] | ||
| 2014/2016 | Ghana | Serology/Human | [17] | ||
| 1974 | Senegal | Ae. luteocephalus, Serology/Human | [18] | ||
| 1983 | Senegal | Human | [19] | ||
| 1990 | Senegal | Aedes sp | [20] | ||
| 2015/19 | Senegal | Human | [21] | ||
| 1999 | Senegal | Aedes sp | [22] | ||
| 2009/10 | Senegal | Ae. aegypti, Serology/Human | [23] | ||
| 1964 | Nigeria | Serology/Human | [24] | ||
| 1975 | Nigeria | Serology/Human | [25] | ||
| 2001 | Nigeria | Serology/Human | [26] | ||
| 2010 * | Nigeria | Serology/Human | [27] | ||
| 2011 | Nigeria | Serology/Human | [28] | ||
| 2013 * | Nigeria | Serology/Human | [29] | ||
| 2014 * | Nigeria | Serology/Human | [30] | ||
| 1987/93 | Benin | Serology/Human | [31] | ||
| 2010 | Benin | Serology/Human | [32] | ||
| 2019 | Benin | Serology/Human; Human | [33] | ||
| 2015 | Mauritania | Serology/Human | [34] | ||
| 2017 | Burkina Faso | Serology/Human | [35] | ||
| 2016 | Burkina Faso | Serology/Human | [36] | ||
| 2019 | Burkina Faso | Serology/Human | [37] | ||
| 1980 | Burkina Faso | Aedes sp | [38] | ||
| 1983/86 | Burkina Faso | Aedes sp | [39] | ||
| 2013/14 | Burkina Faso | Serology/Human | [40] | ||
| 2015/17 | Burkina Faso | Serology/Human | [41] | ||
| 2014 | Burkina Faso | Serology/Human | [42] | ||
| 2016/17 | Burkina Faso | Serology/Human | [43] | ||
| 2016 | Burkina Faso | Serology/Human | [44] | ||
| 1982 | Burkina Faso | Serology/Human | [45] | ||
| 2004 | Burkina Faso | Serology/Human | [46] | ||
| 2016 | Burkina Faso | Serology/Human | [47] | ||
| 2013/14 | Burkina Faso | Serology/Human | [48] | ||
| 2017 | Burkina Faso | Serology/Human | [35] | ||
| 2003/4 | Burkina Faso | Serology/Human | [49] | ||
| 2016 | Burkina Faso | Serology/Human | [50] | ||
| 2009/13 | Mali | Serology/Human | [51] | ||
| 2006 | Mali | Serology/Human | [52] | ||
| 1999 | Côte d’Ivoire | Mosquitoes, Serology/Human | [53] | ||
| 2010 | Côte d’Ivoire | Serology/Human, Human | [54] | ||
| 2011/12 | Côte d’Ivoire | Serology/Human, Human | [55] | ||
| 2017 | Côte d’Ivoire | Serology/Human, Human | [56] | ||
| 2019 | Côte d’Ivoire | Serology/Human | [57] | ||
| Dengue | 1978/91 | Guinea | Mosquitoes | [58] | |
| 2006/8 | Sierra Leone | Serology/Human | [59] | ||
| 2016 * | Sierra Leone | Serology/Human | [60] | ||
| 2012/13 | Sierra Leone | Serology/Human | [61] | ||
| West Nile | 2012/13 | Senegal | Culex sp, Aedes sp | [62] | |
| 1972/75 | Senegal | Serology/Human | [63] | ||
| 1998/99 | Senegal | Masonia uniformis | [64] | ||
| 1951/55 | Nigeria | Serology/Human | [65] | ||
| 1968/69 | Nigeria | Serology/human | [66] | ||
| 1963 | Nigeria | Serology/Human | [67] | ||
| 1975 | Nigeria | Serology/Human | [68] | ||
| 1987 | Nigeria | Serology/Horses | [69] | ||
| 2011/12 | Nigeria | Serology/Horses | [70] | ||
| 2018 | Nigeria | Human | [71] | ||
| 2015 | Mauritania | Serology/Human | [34] | ||
| 2009/13 | Mali | Serology/Human | [51] | ||
| 2006/8 | Sierra Leone | Serology/Human | [59] | ||
| Zika | 2016 | Cape Verde | Ae. aegytpi | [72] | |
| 2016 | Mali | Serology/Human | [73] | ||
| 1972/75 | Senegal | Serology/Human | [63] | ||
| 1999 | Côte d’Ivoire | Mosquitoes, Serology/Human | [53] | ||
| 2010/14 | Gambia | Serology | [74] | ||
| 1975 * | Nigeria | Serology | [25] | ||
| 1951/55 | Nigeria | Serology/Human | [65] | ||
| 2018 | Nigeria | Serology | [71] | ||
| Yellow Fever | 1989 | Nigeria | Serology/horses | [69] | |
| 1951/55 | Nigeria | Serology/Human | [65] | ||
| 1968/69 | Nigeria | Serology/Human | [66] | ||
| 1975 | Nigeria | Serology/Human | [68] | ||
| 1972/75 | Senegal | Serology/Human | [63] | ||
| 1976 | Senegal | Aedes sp | [75] | ||
| 2003/8 | Burkina Faso | Serology/Human | [76] | ||
| 1983/86 | Burkina Faso | Aedes sp | [39] | ||
| 1983 | Burkina Faso | Aedes sp | [77] | ||
| 1999 | Burkina Faso | Serology/Human | [78] | ||
| 1987 | Mali | Serology/Human | [79] | ||
| 2006 | Mali | Serology/Human | [52] | ||
| 1999 | Côte d’Ivoire | Mosquitoes, Serology/Human | [53] | ||
| 2006/09 | Sierra Leone | Serology/Human | [59] | ||
| 1977/80 | Ghana | Human | [80] | ||
| 1963 | Ghana | Human | [80] | ||
| 1969/70 | Ghana | Human | [80] | ||
| 2011 | Ghana | Human | [81] | ||
| Usutu | 2012/13 | Senegal | Culex sp | [62] | |
| 1972/1977 | Senegal | Aedes sp | [82] | ||
| 1972 | Nigeria | Turdus libonyanus | [83] | ||
| 2018 | Nigeria | Serology/Human | [71] | ||
| 2004 | Burkina Faso | Serology/Human | [83] | ||
| 2004 | Côte d’Ivoire | Culex quinquefasciatus | [83] | ||
| Wesselsbron | 1972/75 | Senegal | Serology/Human | [63] | |
| 1971 * | Nigeria | Serology/Horse | [84] | ||
| 1975 | Nigeria | Serology/Human | [68] | ||
| 1989 * | Nigeria | Serology/Horse | [69] | ||
| 1998 | Mauritania | Aedes vexans | [64] | ||
| 1978/91 | Guinea | Mosquitoes | [58] | ||
| Spondweni | 1982 * | Burkina Faso | Serology/Human | [85] | |
| Kedougou | 1978 * | Senegal | Aedes sp | [86] | |
| Ntaya | 1977 | Nigeria | Serology/Human | [87] | |
| Yaounde | 2012/13 | Senegal | Culex sp | [62] | |
| Bagazza | 2012/13 | Senegal | Culex sp | [62] | |
| 1998/99 | Senegal | Aedes fowleri | [64] | ||
| 1998/99 | Mauritania | Culex neavei | [64] | ||
| Barkedji | 2012/13 | Senegal | Culex sp, Aedes sp | [62] | |
| Potiskum | 1989 * | Nigeria | Serology/Horses | [69] | |
| Uganda S | 1989 * | Nigeria | Serology/Horses | [69] | |
| Saboya | 1978/91 | Guinea | Mosquitoes | [58] | |
| Togaviridae | |||||
| Chikungunya | 1963 | Nigeria | Aedes sp | [88] | |
| 1969 | Nigeria | Serology/Human | [89] | ||
| 1968/69 | Nigeria | Serology/Human | [66] | ||
| 1989 * | Nigeria | Serology/Horse | [69] | ||
| 1974 | Nigeria | Serology/Human | [90] | ||
| 2009/10 | Senegal | Serology, Aedes sp | [91] | ||
| 1972/75 | Senegal | Serology/Human | [63] | ||
| 2016/17 | Ghana | Serology/Human | [15] | ||
| 2006 | Benin | Serology/Human | [92] | ||
| 2009/13 | Mali | Serology/Human | [51] | ||
| Togaviridae | |||||
| Chikungunya | 2012/13 | Sierra Leone | Serology/Human | [93] | |
| 2016/17 | Sierra Leone | Serology/Human | [94] | ||
| 2006/8 | Sierra Leone | Serology/Human | [59] | ||
| 2012/13 | Sierra Leone | Serology/Human | [61] | ||
| 1975/77 | Sierra Leone | Serology/Human | [87] | ||
| 1977 | Liberia | Serology/Human | [87] | ||
| Semliki Forest | 1971 | Senegal | Serology/Horses | [95] | |
| 1951/55 | Nigeria | Serology/Human | [65] | ||
| 2014 * | Nigeria | Serology/Human | [96] | ||
| Sindbis | 2012/13 | Senegal | Culex sp | [62] | |
| 1972/75 | Senegal | Serology | [63] | ||
| Ndumu | 2012/13 | Senegal | Several mosquitoes | [63] | |
| Onyongnyong | 1989 * | Nigeria | Serology/Horses | [69] | |
| 1974 | Nigeria | Serology/Human | [87] | ||
| 1975 | Nigeria | Serology/Human | [87] | ||
| 1954 | Ghana | Serology/Human | [87] | ||
| 1975/77 | Sierra Leone | Serology/Human | [87] | ||
| Phenuiviridae | |||||
| Rift Valley fever | 1993/96 | Burkina Faso | Aedes sp | [39] | |
| 1987 | Burkina Faso | Serology/Sheep | [97] | ||
| 2005/7 | Burkina Faso | Serology/livestock | [98] | ||
| 1985/87 | Burkina Faso | Serology/livestock | [99] | ||
| 2006/8 | Sierra Leone | Serology/Human | [59] | ||
| 1987 * | Guinea | Serology/Bats | [100] | ||
| 1978/91 | Guinea | Mosquitoes | [58] | ||
| 2016 | Mali | Serology/Human | [101] | ||
| 2015 | Mali | Serology/Human | [102] | ||
| 2005/14 | Mali | Serology/Bovine | [103] | ||
| 2016 | Niger | Serology/Human/ Livestock |
[104] | ||
| 1959 | Nigeria | Serology/Sheep | [105] | ||
| 2016 | Nigeria | Serology/Camels | [106] | ||
| 1989 | Nigeria | Seroloy/Horses | [69] | ||
| 1998 | Senegal | Culex sp, Serology | [107] | ||
| 2012/13 | Senegal | Aedes ochraceus | [62] | ||
| 1998/99 | Senegal | Culex poicilipes | [64] | ||
| 1998 | Mauritania | Culex poicilipes | [64] | ||
| 2015 | Mauritania | Serology/Human | [34] | ||
| Peribunyaviridae | |||||
| Zinga | 1975/1977 | Nigeria | Serology/Human | [87] | |
| Bunyamwera | 1978 * | Senegal | Serology/Human | [63] | |
| 1951/55 | Nigeria | Serology/Human | [65] | ||
| 1963 | Nigeria | M. africana | [88] | ||
| 1978/91 | Guinea | Mosquitoes | [58] | ||
| Arumowot | 1968/69 | Nigeria | Serology/Human | [66] | |
| Bwamba | 1969/72 | Nigeria | Serology/Human | [108] | |
| 1951/55 | Nigeria | Serology/Human | [65] | ||
| Ngari | 2010 | Mauritania | Serology/Goat | [109] | |
| Nyando | 1972 * | Senegal | Serology/Human | [110] | |
| Pongola | 1963 | Nigeria | M. africana | [88] | |
| Akabane | 2015 | Nigeria | Serology/Livestock | [111] | |
| M’Poko | 1978/91 | Guinea | Mosquitoes | [58] | |
| Rhabdoviridae | |||||
| Mossuril | 1978/91 | Guinea | Mosquitoes | [58] | |
| Reoviridae | |||||
| Acado | 2012/13 | Senegal | Culex sp | [62] | |
| Sanar | 2012/13 | Senegal | Culex neavei, M. uniformis | [62] | |
| 1998/99 | Senegal | Culex poicilipes | [64] | ||
| Kindia | 1978/91 | Guinea | Mosquitoes | [58] | |
| African horse sickeness | 1971 * | Nigeria | Serology/Horse | [84] | |
| 1993 * | Nigeria | Serology/Camels/Donkey/ Dogs/Horses |
[112] | ||
| 1993 * | Nigeria | Serology/Horse | [113] | ||
| 1995 * | Nigeria | Serology/Horse | [114] | ||
| Flaviviridae | Mosquito-specific viruses | ||||
| Culex flavivirus | 2016 | Ghana | Culex sp | [115] | |
| Cell fusing agent virus | 2016 | Ghana | Aedes aegypti | [115] | |
| Anopheles flavivirus-like 2 | 2012 | Senegal | Anopheles sp | [116] | |
| Anopheles flavivirus-like 1 | 2012 | Senegal | Anopheles sp | [116] | |
| Nienokoue | 2004 | Côte d’Ivoire | Culex species mosquitoes | [117] | |
| Nounane | 2004 | Côte d’Ivoire | Uranotaenia mashonaensis | [118] | |
| Togaviridae | |||||
| Taï Forest alphavirus | 2004 | Côte d’Ivoire | Culex decens | [119] | |
| Peribunyaviridae | |||||
| Ferak | 2004 | Côte d’Ivoire | Culex decens | [120] | |
| Jonchet | 2004 | Côte d’Ivoire | Culex sp | [120] | |
| Herbert | 2004 | Côte d’Ivoire | Culex nebulosus | [121] | |
| Tai | 2004 | Côte d’Ivoire | Culex sp | [121] | |
| Rhabdoviridae | |||||
| Moussa | 2004 | Côte d’Ivoire | Culex decens | [122] | |
| Mesoniviridae | |||||
| Odorna | 2016 | Ghana | Aedes aegypti | [115] | |
| Dianke | 2013 | Senegal | Culex poicilipes | [123] | |
| Cavally | 2016 | Ghana | Aedes aegypti | [115] | |
| Cavally | 2004 | Côte d’Ivoire | Aedes harrisoni | [124] | |
| Nse | 2004 | Côte d’Ivoire | Culex nebulosus | [125] | |
| Meno | 2004 | Côte d’Ivoire | Uranotaenia chorleyi | [125] | |
| Hana | 2004 | Côte d’Ivoire | Culex sp | [125] | |
| Moumo | 2004 | Côte d’Ivoire | Culex sp | [125] | |
| Totiviridae | |||||
| Aedes aegypti totivirus | 2016 | Ghana | Aedes aegypti | [115] | |
| Reoviridae | |||||
| Aedes pseudoscutellaris reovirus | 2015 | Ghana | Aedes aegypti | [115] | |
| Cimodo | 2008 | Côte d’Ivoire | Culex nebulosus | [126] | |
| Phenuiviridae | |||||
| Phasi Charoen-like phasivirus | 2016 | Ghana | Aedes aegypti | [115] | |
| Gouleako | 2004 | Côte d’Ivoire | Culex sp | [127] | |
| Negeviruses ** | |||||
| Dezidougou | 1984 | Côte d’Ivoire | Aedes aegypti | [128] | |
| Iflaviridae | |||||
| Aedes vexans iflavirus | 2017 | Senegal | Aedes vexans | [129] | |
| Permutotetraviridae | |||||
| Culex permutotetra-like virus | 2016 | Ghana | Culex sp | [115] | |
| Nodaviridae | |||||
| Mosinovirus | 2004 | Côte d’Ivoire | Culicidae | [130] | |
| Xinmoviridae | |||||
| Bolahun virus variant 2 | 2012/15 | Liberia | Anopheles gambiae | [116] | |
| Unclassified *** | |||||
| Unclassified Riboviria | Bolahun virus variant 1 | 2012/15 | Burkina Faso | Anopheles gambiae | [116] |
| Unclassified Riboviria | Aedes aegypti virga-like virus | 2016 | Ghana | Aedes aegypti | [115] |
| Unclassified Riboviria | West Accra | 2015 | Ghana | Aedes aegypti | [115] |
| Unclassified Riboviria | Mole Culex | 2016 | Ghana | Culex sp | [115] |
| Unclassified Riboviria | Goutanap | 2004 | Côte d’Ivoire | Culex nebulosus | [131] |
| Unclassified Riboviria | Goutanap | 2016 | Ghana | Culex sp | [115] |
| Unclassified Riboviria | Tesano Aedes | 2016 | Ghana | Aedes aegypti | [115] |
| Unclassified Riboviria | Korle-bu Aedes | 2016 | Ghana | Aedes aegypti | [115] |
* = Year of research publication used; ** = Proposed taxon [128]; *** = Unclassified viruses; Year/Year = Study period.
The presence of flaviviruses in West Africa is not limited to MSVs, but also medically important MBVs, such as DENV, WNV, ZIKV, YFV, and Usutu virus (USUV). McNamara et al. seem to provide the first report on mosquito-borne flaviviruses, including YFV, WNV, ZIKV, and Uganda S virus in the 1950s [116]. West Africa has experienced several outbreaks of DENV and it is the main MBV circulating in the human population and posed a major public health threat. It is likely that the first detection of DENV in West Africa was in Ibadan, Nigeria in 1964 from febrile patients [132]. However, the most recent outbreak of DENV was reported in Ouagadougou, Burkina Faso in 2017 [133]. In addition to DENV outbreaks in Burkina Faso, there were reports in other countries, such as Nigeria, Côte d’Ivoire, Senegal, Sierra Leone, Benin, Mali, Cape Verde, Ghana, Mauritania, and Guinea in a decreasing order of incidence. Therefore, Burkina Faso had several DENV reports unlike other West African countries. The main vectors for DENV are Aedes mosquitoes (mainly Aedes aegypti) through which DENV is transmitted to humans (amplifying host). DENV was mainly detected via serological investigations in humans, although Ae. aegypti mosquitoes were also documented as a source of detection.
2. Togaviridae
Members of this family are enveloped, spherical, icosahedral, and 65–70 nm in diameter. They have a capsid with icosahedral symmetry made of 240 monomers. Additionally, they have a monopartite, linear, ssRNA(+) genome of 9.7–11.8 kb [134]. Alphaviruses are mostly transmitted by mosquito vectors and they have amplifying vertebrate hosts. Acute infections in vertebrates are characterised by a high titer of the virus making it possible for mosquitoes to be infected in the course of blood feeding [135].
Tai Forest alphavirus (TFAV), Eilat virus (EILV), Agua Salud alphavirus (ASALV), and Mwinilunga alphavirus (MWAV) are the main mosquito-specific members of this family [136][137]. However, only TFAV was identified and isolated in West Africa. TFAV was isolated from Culex decens mosquitoes collected in 2004 in Côte d’Ivoire [136]. TFAV has a relationship with western equine encephalitis virus complex through phylogenetic analyses [136].
Medically important mosquito-borne alphaviruses that are emerging public health threats include Venezuelan (VEEV) and eastern (EEEV) equine encephalitis viruses, and CHIKV. These viruses can cause outbreaks of severe meningoencephalitis with frequent lethal consequences, or polyarthritis with agonizing and chronic joint pain [135]. Likely, the first epidemic of CHIKV in West Africa occurred in 1969 in Ibadan, Nigeria [138]. It is probable that the virus was first isolated from Aedes mosquitoes (Ae. aegypti, Ae. grahami, and Ae. taylori) collected in 1963 in Lagos, Nigeria, and serological survey revealed antibodies in the sera of study participants [139]. The recent detection of CHIKV in West Africa occurred between 2016 and 2017 in Sierra Leone and this was via Ion Torrent semiconductor sequencing to profile pathogen spectrum in archived human yellow fever virus-negative sera [140]. A broader spectrum of pathogens was suggested by the distribution of sequencing reads and must be considered in clinical diagnostics and epidemiological surveillance [140]. Until now, a greater number of cases of CHIKV were reported in Sierra Leone, Nigeria, and Senegal, compared to the very few cases in Ghana, Benin, Mali, and Liberia. In general, CHIKV was mainly detected by serology in humans, although there were a few Aedes mosquito detections.
3. Peribunyaviridae
These viruses are enveloped, spherical, or pleomorphic virions 80–120 nm in diameter. They have a segmented, linear ssRNA(-) genome, with L segment between 6.8 and 12 kb, M segment between 3.2 and 4.9, and S segment between 1 and 3 kb [141]. This is a newly established family which includes four genera for globally distributed viruses: Orthobunyavirus, Herbevirus, Pacuvirus, and Shangavirus. Mostly, peribunyaviruses are maintained in a vertebrate–arthropod transmission cycle [142].
The mosquito-specific members of the family include Ferak virus, Jonchet virus, Herbert virus, and Tai virus. Interestingly, all these MSVs were identified in Côte d’Ivoire [143][144][145]. These viruses were identified via a mosquito surveillance study, where mosquitoes were trapped from February to June 2004 in Taï National Park, Côte d’Ivoire [146]. The mosquito samples were further analysed to identify and isolate these viruses [143][144][145]. However, the viruses were mainly isolated from Culex mosquitoes.
The mosquito-borne members of the Peribunyaviridae include Zinga virus, Bunyamwera virus, Arumowot virus, Bwamba virus, Ngari virus, Nyando virus, Pangola virus, Akabane virus, and M’poko virus [147][148]. Bunyamwera virus is the prominent MBV of this family. Bunyamwera virus was first detected in 1963 in West Africa, Nigeria, from Mansonia africana mosquitoes [149]. The virus was also reported to be detected in humans and mosquitoes in Senegal and Guinea respectively [150][116].
4. Other Virus Families
An increasing number of MAVs, especially the mosquito-specific ones, are distributed globally. MAVs were also classified under the following virus families: Rhabdoviridae, Mesoniviridae, Totiviridae, Reoviridae, Phenuiviridae, Permutotetraviridae, Iflaviridae, Xinmoviridae, Nodaviridae, and Negeviruses (proposed taxon). The latest mosquito-specific mesonivirus is Dianke virus, which was isolated from mosquitoes in Eastern Senegal [151]. Interestingly, Dianke virus was identified in different species of mosquitoes, including Aedes sp, Culex sp, Mansonia sp, and Uranotaenia sp. This explains why mesoniviruses are said to have a broad host range and geographical distribution. Another recent and novel mosquito-specific iflavirus isolated from Aedes vexans mosquitoes in Senegal is Aedes vexans iflavirus [3]. Ae. vexans mosquito is a competent vector of numerous MBVs, such as RVFV and ZIKV. Therefore, it is possible that Aedes vexans iflavirus could interfere with the replication and transmission potentials of RVFV and ZIKV. This review therefore suggests studies involving the interaction of MSVs with MBVs to answer the search for a probable biological vector control tool against arboviruses.
The medically important mosquito-borne member of the family Phenuiviridae, with several reports, is the RVFV. RVFV was reported in Burkina Faso, Sierra Leone, Guinea, Mali, Nigeria, Senegal, and Mauritania. It is likely that the first report of RVFV was in 1959 in Nigeria [13]. However, the latest RVFV case was detected in 2016 in Mali, Niger, and Nigeria [14][15][16], and this is the first occurrence of RVFV in Niger. RVFV was detected in culicine mosquitoes and serological investigations in humans, bats, livestock, horses, and camels.
References
- WHO. Dengue Fever in Cape Verde—Update 1; World Health Organization: Geneva, Switzerland, 2009.
- Guedes, D.R.D.; Gomes, E.T.B.; Paiva, M.H.S.; de Melo-Santos, M.A.V.; Alves, J.; Gómez, L.F.; Ayres, C.F.J. Circulation of DENV2 and DENV4 in Aedes aegypti (Diptera: Culicidae) Mosquitoes from Praia, Santiago Island, Cabo Verde. J. Insect Sci. 2017, 17, 86.
- Manu, S.K.; Bonney, J.H.K.; Pratt, D.; Abdulai, F.N.; Agbosu, E.E.; Frimpong, P.O.; Adiku, T.K. Arbovirus Circulation among Febrile Patients at the Greater Accra Regional Hospital, Ghana. BMC Res. Notes 2019, 12, 332.
- Amoako, N.; Duodu, S.; Dennis, F.E.; Bonney, J.H.K.; Asante, K.P.; Ameh, J.; Mosi, L.; Hayashi, T.; Agbosu, E.E.; Pratt, D.; et al. Detection of Dengue Virus among Children with Suspected Malaria, Accra, Ghana. Emerg. Infect. Dis. 2018, 24, 1544–1547.
- Bonney, J.H.K.; Hayashi, T.; Dadzie, S.; Agbosu, E.; Pratt, D.; Nyarko, S.; Asiedu-Bekoe, F.; Ido, E.; Sarkodie, B.; Ohta, N.; et al. Molecular Detection of Dengue Virus in Patients Suspected of Ebola Virus Disease in Ghana. PLoS ONE 2018, 13, e0208907.
- Robin, Y.; Cornet, M.; Heme, G.; Le Gonidec, G. Isolement Du Virus de La Dengue Au Sénégal [Dengue Virus Isolation in Senegal]. Ann. l’Institut Pasteur Virol. 1980, 131, 149–154.
- Saluzzo, J.F.; Cornet, M.; Castagnet, P.; Rey, C.; Digoutte, J.P. Isolation of Dengue 2 and Dengue 4 Viruses from Patients in Senegal. Trans. R. Soc. Trop. Med. Hyg. 1986, 80, 5.
- Traore-Lamizana, M.; Zeller, H.; Monlun, E.; Mondo, M.; Hervy, J.-P.; Adam, F.; Digoutte, J.-P. Dengue 2 Outbreak in Southeastern Senegal During 1990: Virus Isolations from Mosquitoes (Diptera: Culicidae). J. Med. Entomol. 1994, 31, 623–627.
- Dieng, I.; dos Passos Cunha, M.; Diagne, M.M.; Sembène, P.M.; de Andrade Zanotto, P.M.; Faye, O.; Faye, O.; Sall, A.A. Origin and Spread of the Dengue Virus Type 1, Genotype V in Senegal, 2015–2019. Viruses 2021, 13, 57.
- Diallo, M.; Ba, Y.; Sall, A.A.; Diop, O.M.; Ndione, J.A.; Mondo, M.; Girault, L.; Mathiot, C. Amplification of the Sylvatic Cycle of Dengue Virus Type 2, Senegal, 1999–2000: Entomologic Findings and Epidemiologic Considerations. Emerg. Infect. Dis. 2003, 9, 362–367.
- Faye, O.; Ba, Y.; Faye, O.; Talla, C.; Diallo, D.; Chen, R.; Mondo, M.; Ba, R.; Macondo, E.; Siby, T.; et al. Urban Epidemic of Dengue Virus Serotype 3 Infection, Senegal, 2009. Emerg. Infect. Dis. 2014, 20, 456–459.
- Carey, D.; Causey, O.; Reddy, S.; Cooke, A. Dengue Viruses from Febrile Patients in Nigeria, 1964–1968. Lancet 1971, 297, 105–106.
- Fagbami, A.H.; Fabiyi, A. Arbovirus Studies in Two Towns in Western State of Nigeria. Trop. Geogr. Med. 1975, 27, 59–62.
- Baba, M.M.; Talle, M. The Effect of Climate on Dengue Virus Infections in Nigeria. N. Y. Sci. J. 2011, 4, 28–33.
- Dawurung, J.; Baba, M.; Stephen, G.; Jonas, S.; Bukbuk, D.; Dawurung, C. Serological Evidence of Acute Dengue Virus Infection among Febrile Patients Attending Plateau State Specialist Hospital Jos, Nigeria. Rep. Opin. 2010, 2, 71–76.
- Idris, A.; Baba, M.; Thairu, Y.; Bamidele, O. Sero-Prevalence of Dengue Type-3 Virus among Patients with Febrile Illnesses Attending a Tertiary Hospital in Maiduguri, Nigeria. Int. J. Med. Med. Sci. 2013, 5, 560–563.
- Adedayo, F.; Nioma, I.; Olanrewaju, M.; Adeyinka, A.; Ebele, A. Serological Evidence of Recent Dengue Virus Infection Among Febrile Children in a Semi Arid Zone. Am. J. Infect. Dis. 2013, 9, 7–10.
- Oladipo, E.; Amanetu, C.; Gbadero, T.; Oloke, J. Detectable Anti-Dengue Virus IgM Antibodies among Healthy Individuals in Ogbomoso, Oyo State, Nigeria. Am. J. Infect. Dis. 2014, 10, 64–67.
- Eisenhut, M.; Schwarz, T.F.; Hegenscheid, B. Seroprevalence of Dengue, Chikungunya and Sindbis Virus Infections in German Aid Workers. Infection 1999, 27, 82–85.
- Gautret, P.; Botelho-Nevers, E.; Charrel, R.N.; Parola, P. Dengue Virus Infections in Travellers Returning from Benin to France, July–August 2010. Euro Surveill. 2010, 15, 19657.
- Fourié, T.; Luciani, L.; Amrane, S.; Zandotti, C.; Leparc-Goffart, I.; Ninove, L.; Nougairède, A. Dengue Virus Type 1 Infection in Traveler Returning from Benin to France, 2019. Emerg. Infect. Dis. 2020, 26, 1946–1949.
- Bob, N.S.; Bâ, H.; Fall, G.; Ishagh, E.; Diallo, M.Y.; Sow, A.; Sembene, P.M.; Faye, O.; El Kouri, B.; Sidi, M.L.; et al. Detection of the Northeastern African Rift Valley Fever Virus Lineage During the 2015 Outbreak in Mauritania. Open Forum Infect. Dis. 2017, 4, ofx087.
- Tougma, S.A.; Zoungrana Yaméogo, W.N.; Dahourou, D.L.; Salou Kagoné, I.A.; Compaoré, T.R.; Kaboré, A.; Kagoné, T.; Drabo, M.K.; Meda, N. Dengue Virus Infection and Pregnancy Outcomes during the 2017 Outbreak in Ouagadougou, Burkina Faso: A Retrospective Cohort Study. PLoS ONE 2020, 15, e0238431.
- Tarnagda, Z.; Cissé, A.; Bicaba, B.W.; Diagbouga, S.; Sagna, T.; Ilboudo, A.K.; Tialla, D.; Lingani, M.; Sondo, K.A.; Yougbaré, I.; et al. Dengue Fever in Burkina Faso, 2016. Emerg. Infect. Dis. 2018, 24, 170–172.
- Sondo, K.A.; Ouattara, A.; Diendéré, E.A.; Diallo, I.; Zoungrana, J.; Zémané, G.; Da, L.; Gnamou, A.; Meda, B.; Poda, A.; et al. Dengue Infection during Pregnancy in Burkina Faso: A Cross-Sectional Study. BMC Infect. Dis. 2019, 19, 997.
- Hervy, J.-P.; Legros, F.; Roche, J.-C.; Monteny, N.; Diaco, B. Circulation Du Virus Dengue 2 Dans Plusieurs Milieux Boisés Des Savanes Soudaniennes de La Région de Bobo-Dioulasso (Burkina Faso). Cah. ORSTOM Entomol. Méd. Parasitol. 1984, 22, 135–143.
- Robert, V.; Lhuillier, M.; Meunier, D.; Sarthou, J.L.; Monteny, N.; Digoutte, J.P.; Cornet, M.; Germain, M.; Cordellier, R. Yellow Fever Virus, Dengue 2 and Other Arboviruses Isolated from Mosquitos, in Burkina Faso, from 1983 to 1986. Entomological and Epidemiological Considerations. Bull. Soc. Pathol. Exot. 1993, 86, 90–100.
- Ridde, V.; Agier, I.; Bonnet, E.; Carabali, M.; Dabiré, K.R.; Fournet, F.; Ly, A.; Meda, I.B.; Parra, B. Presence of Three Dengue Serotypes in Ouagadougou (Burkina Faso): Research and Public Health Implications. Infect. Dis. Poverty 2016, 5, 23.
- Lim, J.K.; Carabali, M.; Edwards, T.; Barro, A.; Lee, J.-S.; Dahourou, D.; Lee, K.S.; Nikiema, T.; Shin, M.Y.; Bonnet, E.; et al. Estimating the Force of Infection for Dengue Virus Using Repeated Serosurveys, Ouagadougou, Burkina Faso. Emerg. Infect. Dis. 2021, 27, 130–139.
- Lim, J.K.; Carabali, M.; Lee, J.-S.; Lee, K.-S.; Namkung, S.; Lim, S.-K.; Ridde, V.; Fernandes, J.; Lell, B.; Matendechero, S.H.; et al. Evaluating Dengue Burden in Africa in Passive Fever Surveillance and Seroprevalence Studies: Protocol of Field Studies of the Dengue Vaccine Initiative. BMJ Open 2018, 8, e017673.
- Im, J.; Balasubramanian, R.; Ouedraogo, M.; Wandji Nana, L.R.; Mogeni, O.D.; Jeon, H.J.; van Pomeren, T.; Haselbeck, A.; Lim, J.K.; Prifti, K.; et al. The Epidemiology of Dengue Outbreaks in 2016 and 2017 in Ouagadougou, Burkina Faso. Heliyon 2020, 6, e04389.
- Hashimoto, T.; Kutsuna, S.; Maeki, T.; Tajima, S.; Takaya, S.; Katanami, Y.; Yamamoto, K.; Takeshita, N.; Hayakawa, K.; Kato, Y.; et al. A Case of Dengue Fever Imported from Burkina Faso to Japan in October 2016. Jpn. J. Infect. Dis. 2017, 70, 675–677.
- Gonzalez, J.P.; Du Saussay, C.; Gautun, J.C.; McCormick, J.B.; Mouchet, J. Dengue in Burkina Faso (Ex-Upper Volta): Seasonal Epidemics in the Urban Area of Ouagadougou. Bull. Soc. Pathol. Exot. Fil. 1985, 78, 7–14.
- Fournet, F.; Rican, S.; Vaillant, Z.; Roudot, A.; Meunier-Nikiema, A.; Kassié, D.; Dabiré, R.; Salem, G. The Influence of Urbanization Modes on the Spatial Circulation of Flaviviruses within Ouagadougou (Burkina Faso). Int. J. Environ. Res. Public Health 2016, 13, 1226.
- Eldin, C.; Gautret, P.; Nougairede, A.; Sentis, M.; Ninove, L.; Saidani, N.; Million, M.; Brouqui, P.; Charrel, R.; Parola, P. Identification of Dengue Type 2 Virus in Febrile Travellers Returning from Burkina Faso to France, Related to an Ongoing Outbreak, October to November 2016. Eurosurveillance 2016, 21, 30425.
- Diallo, I.; Sondo, K.A.; Tieno, H.; Tamelokpo, E.Y.; Zoungrana, J.; Sagna, Y.; Savadogo, M.; Poda, A.; Guira, O.; Diendéré, E.A.; et al. À Propos de 98 Cas de Dengue Hospitalisés Dans Une Clinique Privée de Ouagadougou: Aspects Épidémiologiques, Diagnostiques et Évolutifs. Bull. Soc. Pathol. Exot. 2017, 110, 291–296.
- Collenberg, E.; Ouedraogo, T.; Ganamé, J.; Fickenscher, H.; Kynast-Wolf, G.; Becher, H.; Kouyaté, B.; Kräusslich, H.-G.; Sangaré, L.; Tebit, D.M. Seroprevalence of Six Different Viruses among Pregnant Women and Blood Donors in Rural and Urban Burkina Faso: A Comparative Analysis. J. Med. Virol. 2006, 78, 683–692.
- Baronti, C.; Piorkowski, G.; Touret, F.; Charrel, R.; de Lamballerie, X.; Nougairede, A. Complete Coding Sequences of Two Dengue Virus Type 2 Strains Isolated from an Outbreak in Burkina Faso in 2016. Genome Announc. 2017, 5, e00209-17.
- Safronetz, D.; Sacko, M.; Sogoba, N.; Rosenke, K.; Martellaro, C.; Traoré, S.; Cissé, I.; Maiga, O.; Boisen, M.; Nelson, D.; et al. Vectorborne Infections, Mali. Emerg. Infect. Dis. 2016, 22, 340–342.
- Phoutrides, E.K.; Coulibaly, M.B.; George, C.M.; Sacko, A.; Traore, S.; Bessoff, K.; Wiley, M.R.; Kolivras, K.N.; Adelman, Z.; Traore, M.; et al. Dengue Virus Seroprevalence Among Febrile Patients in Bamako, Mali: Results of a 2006 Surveillance Study. Vector Borne Zoonotic Dis. 2011, 11, 1479–1485.
- Akoua-Koffi, C.; Diarrassouba, S.; Benie, V.; Ngbichi, J.; Bozoua, T.; Bosson, A.; Akran, V.; Carnevale, P.; Ehouman, A. Investigation Autour d’un Cas Mortel de Fièvre Jaune En Côte d’Ivoire En 1999. Bull. Soc. Pathol. Exot. 2001, 94, 227–230.
- Aoussi, E.B.F.; Ehui, E.; Kassi, N.A.; Kouakou, G.; Nouhou, Y.; Adjogoua, E.V.; Eholié, S.; Bissagnéné, E. Seven Native Cases of Dengue in Abidjan, Ivory Coast. Méd. Mal. Infect. 2014, 44, 433–436.
- L’Azou, M.; Jean-Marie, J.; Bessaud, M.; Cabié, A.; Césaire, R.; de Lamballerie, X.; Courbil, R.; Richard, P. Dengue Seroprevalence in the French West Indies: A Prospective Study in Adult Blood Donors. Am. J. Trop. Med. Hyg. 2015, 92, 1137–1140.
- WHO. Dengue Fever—Côte d’Ivoire—Disease Outbreak News 4 August 2017; WHO: Geneva, Switzerland, 2017.
- WHO. Weekly Bulletin on Outbreaks and Other Emergencies—Week 42 2019, 14–20 October 2019; WHO: Geneva, Switzerland, 2019.
- Butenko, A.M. Arbovirus Circulation in the Republic of Guinea. Med. Parazitol. 1996, 2, 40–45.
- Schoepp, R.J.; Rossi, C.A.; Khan, S.H.; Goba, A.; Fair, J.N. Undiagnosed Acute Viral Febrile Illnesses, Sierra Leone. Emerg. Infect. Dis. 2014, 20, 1176–1182.
- de Araújo Lobo, J.M.; Mores, C.N.; Bausch, D.G.; Christofferson, R.C. Short Report: Serological Evidence of Under-Reported Dengue Circulation in Sierra Leone. PLoS Negl. Trop. Dis. 2016, 10, e0004613.
- Dariano, D.F.; Taitt, C.R.; Jacobsen, K.H.; Bangura, U.; Bockarie, A.S.; Bockarie, M.J.; Lahai, J.; Lamin, J.M.; Leski, T.A.; Yasuda, C.; et al. Surveillance of Vector-Borne Infections (Chikungunya, Dengue, and Malaria) in Bo, Sierra Leone, 2012–2013. Am. J. Trop. Med. Hyg. 2017, 97, 1151–1154.
- Ndiaye, E.H.; Diallo, D.; Fall, G.; Ba, Y.; Faye, O.; Dia, I.; Diallo, M. Arboviruses Isolated from the Barkedji Mosquito-Based Surveillance System, 2012–2013. BMC Infect. Dis. 2018, 18, 642.
- Renaudet, J.; Jan, C.; Ridet, J.; Adam, C.; Robin, Y. A Serological Survey of Arboviruses in the Human Population of Senegal. Bull. Soc. Pathol. Exot. Fil. 1978, 71, 131–140.
- Diallo, M.; Nabeth, P.; Ba, K.; Sall, A.A.; Ba, Y.; Mondo, M.; Girault, L.; Abdalahi, M.O.; Mathiot, C. Mosquito Vectors of the 1998-1999 Outbreak of Rift Valley Fever and Other Arboviruses (Bagaza, Sanar, Wesselsbron and West Nile) in Mauritania and Senegal. Med. Vet. Entomol. 2005, 19, 119–126.
- Macnamara, F.N.; Horn, D.W.; Porterfield, J.S. Yellow Fever and Other Arthropod-Borne Viruses; a Consideration of Two Serological Surveys Made in South Western Nigeria. Trans. R. Soc. Trop. Med. Hyg. 1959, 53, 202–212.
- Guyer, B. Serological Survey for Arboviruses in Igbo-Ora, Western Nigeria. Ann. Trop. Med. Parasitol. 1972, 66, 243–250.
- Tomori, O.; Fagbami, A.; Fabiyi, A. Isolations of West Nile Virus from Man in Nigeria. Trans. R. Soc. Trop. Med. Hyg. 1978, 72, 103–104.
- Fagbami, A.H. Zika Virus Infections in Nigeria: Virological and Seroepidemiological Investigations in Oyo State. J. Hyg. 1979, 83, 213–219.
- Olaleye, O.D.; Oladosu, L.A.; Omilabu, S.A.; Baba, S.S.; Fagbami, A.H. Complement Fixing Antibodies against Arboviruses in Horses at Lagos, Nigeria. Rev. Elev. Med. Vet. Pays Trop. 1989, 42, 321–325.
- Sule, W.F.; Oluwayelu, D.O.; Adedokun, R.A.M.; Rufai, N.; McCracken, F.; Mansfield, K.L.; Johnson, N. High Seroprevelance of West Nile Virus Antibodies Observed in Horses from Southwestern Nigeria. Vector Borne Zoonotic Dis. 2015, 15, 218–220.
- Oderinde, B.S.; Mora-Cárdenas, E.; Carletti, T.; Baba, M.M.; Marcello, A. Prevalence of Locally Undetected Acute Infections of Flaviviruses in North-Eastern Nigeria. Virus Res. 2020, 286, 198060.
- Campos, M.; Ward, D.; Morales, R.F.; Gomes, A.R.; Silva, K.; Sepúlveda, N.; Gomez, L.F.; Clark, T.G.; Campino, S. Surveillance of Aedes aegypti Populations in the City of Praia, Cape Verde: Zika Virus Infection, Insecticide Resistance and Genetic Diversity. Parasites Vectors 2020, 13, 481.
- Diarra, I.; Nurtop, E.; Sangaré, A.K.; Sagara, I.; Pastorino, B.; Sacko, S.; Zeguimé, A.; Coulibaly, D.; Fofana, B.; Gallian, P.; et al. Zika Virus Circulation in Mali. Emerg. Infect. Dis. 2020, 26, 945–952.
- Buechler, C.R.; Bailey, A.L.; Weiler, A.M.; Barry, G.L.; Breitbach, M.E.; Stewart, L.M.; Jasinska, A.J.; Freimer, N.B.; Apetrei, C.; Phillips-Conroy, J.E.; et al. Seroprevalence of Zika Virus in Wild African Green Monkeys and Baboons. mSphere 2017, 2, e00393-16.
- Salaun, J.J.; Germain, M.; Robert, V.; Robin, Y.; Monath, T.P.; Camicas, J.L.; Digoutte, J.P. Yellow Fever in Senegal from 1976 to 1980 (Author’s Transl). Med. Trop. 1981, 41, 45–51.
- Yaro, S.; Zango, A.; Rouamba, J.; Diabaté, A.; Dabiré, R.; Kambiré, C.; Tiendrebeogo, S.M.R.; Yonli, T.; Ouango, J.G.; Diagbouga, S.P. Situation Épidémiologique de La Fièvre Jaune Au Burkina Faso de 2003 à 2008. Bull. Soc. Pathol. Exot. 2010, 103, 44–47.
- Baudon, D.; Robert, V.; Roux, J.; Lhuillier, M.; Saluzzo, J.F.; Sarthou, J.L.; Cornet, M.; Stanghellini, A.; Gazin, P.; Molez, J.F. The 1983 Yellow Fever Epidemic in Burkina Faso. Bull. World Health Organ. 1986, 64, 873–882.
- Barennes, H.; Baldet, T.; Cassel, A.-M.; Kabiré, C.; Kambou, C. An Epidemic Risk of Yellow Fever in Burkina Faso despite a Rapid Immunisation Riposte: Role of a Multidisciplinary Investigation Team. Sante 2002, 12, 323–329.
- Cordellier, R. The Epidemiology of Yellow Fever in Western Africa. Bull. World Health Organ. 1991, 69, 73–84.
- Agadzi, V.K.; Boatin, B.A.; Appawu, M.A.; Mingle, J.A.; Addy, P.A. Yellow Fever in Ghana, 1977–1980. Bull. World Health Organ. 1984, 62, 577–583.
- WHO. Yellow Fever in Ghana (Emergencies Preparedness, Response); WHO: Geneva, Switzerland, 2012.
- Cornet, M.; Robin, Y.; Chateau, R.; Heme, G.; Adam, C.; Valade, M.; Gonidec, G.; Jan, C.; Renaudet, J.; Dieng, P.L.; et al. Isolements d’arbovirus Au Sénégal Oriental à Partir de Moustiques (1972–1977) et Notes Sur l’épidémiologie Des Virus Transmis Par Les Aedes, En Particulier Du Virus Amaril. Cah. ORSTOM. Sér. Entomol. Méd. Parasitol. 1979, 17, 149–163.
- Nikolay, B.; Diallo, M.; Boye, C.S.B.; Sall, A.A. Usutu Virus in Africa. Vector Borne Zoonotic Dis. 2011, 11, 1417–1423.
- Kemp, G.; Humburg, J.; Alhaji, I. Isolation and Identification of African Horse-Sickness Virus in Nigeria. Vet. Rec. 1971, 89, 127–128.
- Wolfe, M.; Calisher, C.; Mcguire, K. Spondweni Virus Infection in a Foreign Resident of Upper Volta. Lancet 1982, 320, 1306–1308.
- Robin, Y.; Cornet, M.; Le Gonidec, G.; Chateau, R.; Heme, G. [Kedougou Virus (Ar D14701): A New Arbovirus (“Flavivirus”) Isolated in Senegal (Author’s Transl)]. Ann. Microbiol. 1978, 129, 239–244.
- Woodruff, A.W.; Bowen, E.T.; Platt, G.S. Viral Infections in Travellers from Tropical Africa. BMJ 1978, 1, 956–958.
- Boorman, J.P.T.; Draper, C.C. Isolations of Arboviruses in the Lagos Area of Nigeria, and a Survey of Antibodies to Them in Man and Animals. Trans. R. Soc. Trop. Med. Hyg. 1968, 62, 269–277.
- Moore, D.L.; Reddy, S.; Akinkugbe, F.M.; Lee, V.H.; David-West, T.S.; Causey, O.R.; Carey, D.E. An Epidemic of Chikungunya Fever at Ibadan, Nigeria, 1969. Ann. Trop. Med. Parasitol. 1974, 68, 59–68.
- Tomori, O.; Fagbami, A.; Fabiyi, A. The 1974 Epidemic of Chikungunya Fever in Children in Ibadan. Trop. Geogr. Med. 1975, 27, 413–417.
- Sow, A.; Faye, O.; Diallo, M.; Diallo, D.; Chen, R.; Faye, O.; Diagne, C.T.; Guerbois, M.; Weidmann, M.; Ndiaye, Y.; et al. Chikungunya Outbreak in Kedougou, Southeastern Senegal in 2009–2010. Open Forum Infect. Dis. 2018, 5, ofx259.
- Bacci, A.; Marchi, S.; Massougbodji, A.; Perrin, R.X.; Chippaux, J.-P.; Sambri, V.; Landini, M.P.; Fievet, N.; Varani, S.; Rossini, G. High Seroprevalence of Chikungunya Virus Antibodies Among Pregnant Women Living in an Urban Area in Benin, West Africa. Am. J. Trop. Med. Hyg. 2015, 92, 1133–1136.
- Ansumana, R.; Jacobsen, K.H.; Gbakima, A.A.; Hodges, M.H.; Lamin, J.M.; Leski, T.A.; Malanoski, A.P.; Lin, B.; Bockarie, M.J.; Stenger, D.A. Presumptive Self-Diagnosis of Malaria and Other Febrile Illnesses in Sierra Leone. Pan Afr. Med. J. 2013, 15.
- Zhang, Y.; Ye, F.; Xia, L.X.; Zhu, L.W.; Kamara, I.L.; Huang, K.Q.; Zhang, Y.; Liu, J.; Kargbo, B.; Wang, J.; et al. Next-Generation Sequencing Study of Pathogens in Serum from Patients with Febrile Jaundice in Sierra Leone. Biomed. Environ. Sci. 2019, 32, 363–370.
- Odend’hal, S. Semliki Forest Virus. In The Geographical Distribution of Animal Viral Diseases; Elsevier: Amsterdam, The Netherlands, 1983; pp. 373–375.
- Hubálek, Z.; Rudolf, I.; Nowotny, N. Arboviruses Pathogenic for Domestic and Wild Animals. Adv. Virus. Res. 2014, 89, 201–275.
- Gonzalez, J.P.; Le Guenno, B.; Some, M.J.R.; Akakpo, J.A. Serological Evidence in Sheep Suggesting Phlebovirus Circulation in a Rift Valley Fever Enzootic Area in Burkina Faso. Trans. R. Soc. Trop. Med. Hyg. 1992, 86, 680–682.
- Boussini, H.; Lamien, C.E.; Nacoulma, O.G.; Kabore, A.; Poda, G.; Viljoen, G.J. Prevalence of Rift Valley Fever in Domestic Ruminants in the Central and Northern Regions of Burkina Faso. Rev. Sci. Tech. OIE 2014, 33, 893–901.
- Akakpo, A.J.; Some, M.J.; Bornarel, P.; Jouan, A.; Gonzalez, J.P. Epidemiology of Rift Valley Fever in Western Africa. I. Serologic Survey in Domestic Ruminants of Burkina Faso. Bull. Soc. Pathol. Exot. Fil. 1989, 82, 321–331.
- Boiro, I.; Konstaninov, O.K.; Numerov, A.D. Isolation of Rift Valley Fever Virus from Bats in the Republic of Guinea. Bull. Soc. Pathol. Exot. Fil. 1987, 80, 62–67.
- Tong, C.; Javelle, E.; Grard, G.; Dia, A.; Lacrosse, C.; Fourié, T.; Gravier, P.; Watier-Grillot, S.; Lancelot, R.; Letourneur, F.; et al. Tracking Rift Valley Fever: From Mali to Europe and Other Countries, 2016. Eurosurveillance 2019, 24, 1800213.
- Haneche, F.; Leparc-Goffart, I.; Simon, F.; Hentzien, M.; Martinez-Pourcher, V.; Caumes, E.; Maquart, M. Rift Valley Fever in Kidney Transplant Recipient Returning from Mali with Viral RNA Detected in Semen up to Four Months from Symptom Onset, France, Autumn 2015. Eurosurveillance 2016, 21, 30222.
- Subudhi, S.; Dakouo, M.; Sloan, A.; Stein, D.R.; Grolla, A.; Jones, S.; Dibernardo, A.; Rosenke, K.; Sas, M.; Traore, A.; et al. Seroprevalence of Rift Valley Fever Virus Antibodies in Cattle in Mali, 2005–2014. Am. J. Trop. Med. Hyg. 2018, 98, 872–874.
- Lagare, A.; Fall, G.; Ibrahim, A.; Ousmane, S.; Sadio, B.; Abdoulaye, M.; Alhassane, A.; Mahaman, A.E.; Issaka, B.; Sidikou, F.; et al. First Occurrence of Rift Valley Fever Outbreak in Niger, 2016. Vet. Med. Sci. 2019, 5, 70–78.
- Ferguson, W. Identification of Rift Valley Fever in Nigeria. Bull. Epizoot Dis. Afr. 1959, 7, 317–318.
- Musa, A.A.; Yila, S.A.; Allam, L.; Sackey, A.; Alhaji, N.B.; Garba, B.S.; Mambula-Machunga, S.; Nafarnda, W.D.; Owolodun, O.A.; Dzikwi, A.A. Serological Evidence of Rift Valley Fever Infection and Risk Factors among One-Humped Camels (Camelus Dromedarius) in Northern Nigeria. bioRxiv 2020.
- Diallo, M.; Lochouarn, L.; Sall, A.A.; Mondo, M.; Ba, K.; Girault, L.; Mathiot, C. First Isolation of the Rift Valley Fever Virus from Culex Poicilipes (Diptera: Culicidae) in Nature. Am. J. Trop. Med. Hyg. 2000, 62, 702–704.
- Tomor, O.; Monath, T.P.; Lee, V.; Fagbami, A.; Fabiyi, A. Bwamba Virus Infection: A Sero-Survey of Veterbrates in Five Ecological Zones in Nigeria. Trans. R. Soc. Trop. Med. Hyg. 1974, 68, 461–465.
- Eiden, M.; Vina-Rodriguez, A.; El Mamy, B.O.; Isselmou, K.; Ziegler, U.; Höper, D.; Jäckel, S.; Balkema-Buschmann, A.; Unger, H.; Doumbia, B.; et al. Ngari Virus in Goats during Rift Valley Fever Outbreak, Mauritania, 2010. Emerg. Infect. Dis. 2014, 20, 2174–2176.
- Digoutte, J.P.; Gagnard, V.J.; Bres, P.; Pajot, F.X. Nyando Virus Infection in Man. Bull. Soc. Pathol. Exot. Filiales 1972, 65, 751–758.
- Oluwayelu, D.O.; Aiki-Raji, C.O.; Umeh, E.C.; Mustapha, S.O.; Adebiyi, A.I. Serological Investigation of Akabane Virus Infection in Cattle and Sheep in Nigeria. Adv. Virol. 2016, 2016, 1–4.
- Baba, S.S.; Olaleye, O.D.; Ayanbadejo, O.A. Haemagglutination-Inhibiting Antibodies against African Horse Sickness Virus in Domestic Animals in Nigeria. Vet. Res. 1993, 24, 483–487.
- Oladosu, L.A.; Olayeye, O.D.; Baba, S.S.; Omilabu, S.A. Isolation and Identification of African Horse Sickness Virus during an Outbreak in Lagos, Nigeria. Rev. Sci. Tech. OIE 1993, 12, 873–877.
- Adeyefa, C.A.; Hamblin, C. Continuing Prevalence of African Horse Sickness in Nigeria. Rev. Elev. Med. Vet. Pays Trop. 1995, 48, 31–33.
- Amoa-Bosompem, M.; Kobayashi, D.; Murota, K.; Faizah, A.N.; Itokawa, K.; Fujita, R.; Osei, J.H.N.; Agbosu, E.; Pratt, D.; Kimura, S.; et al. Entomological Assessment of the Status and Risk of Mosquito-Borne Arboviral Transmission in Ghana. Viruses 2020, 12, 147.
- Fauver, J.R.; Grubaugh, N.D.; Krajacich, B.J.; Weger-Lucarelli, J.; Lakin, S.M.; Fakoli, L.S.; Bolay, F.K.; Diclaro, J.W.; Dabiré, K.R.; Foy, B.D.; et al. West African Anopheles Gambiae Mosquitoes Harbor a Taxonomically Diverse Virome Including New Insect-Specific Flaviviruses, Mononegaviruses, and Totiviruses. Virology 2016, 498, 288–299.
- Junglen, S.; Korries, M.; Grasse, W.; Wieseler, J.; Kopp, A.; Hermanns, K.; León-Juárez, M.; Drosten, C.; Kümmerer, B.M. Host Range Restriction of Insect-Specific Flaviviruses Occurs at Several Levels of the Viral Life Cycle. mSphere 2017, 2, e00375-16.
- Junglen, S.; Kopp, A.; Kurth, A.; Pauli, G.; Ellerbrok, H.; Leendertz, F.H. A New Flavivirus and a New Vector: Characterization of a Novel Flavivirus Isolated from Uranotaenia Mosquitoes from a Tropical Rain Forest. J. Virol. 2009, 83, 4462–4468.
- Hermanns, K.; Zirkel, F.; Kopp, A.; Marklewitz, M.; Rwego, I.B.; Estrada, A.; Gillespie, T.R.; Drosten, C.; Junglen, S. Discovery of a Novel Alphavirus Related to Eilat Virus. J. Gen. Virol. 2017, 98, 43–49.
- Marklewitz, M.; Zirkel, F.; Kurth, A.; Drosten, C.; Junglen, S. Evolutionary and Phenotypic Analysis of Live Virus Isolates Suggests Arthropod Origin of a Pathogenic RNA Virus Family. Proc. Natl. Acad. Sci. USA 2015, 112, 7536–7541.
- Marklewitz, M.; Zirkel, F.; Rwego, I.B.; Heidemann, H.; Trippner, P.; Kurth, A.; Kallies, R.; Briese, T.; Lipkin, W.I.; Drosten, C.; et al. Discovery of a Unique Novel Clade of Mosquito-Associated Bunyaviruses. J. Virol. 2013, 87, 12850–12865.
- Quan, P.-L.; Junglen, S.; Tashmukhamedova, A.; Conlan, S.; Hutchison, S.K.; Kurth, A.; Ellerbrok, H.; Egholm, M.; Briese, T.; Leendertz, F.H.; et al. Moussa Virus: A New Member of the Rhabdoviridae Family Isolated from Culex Decens Mosquitoes in Côte d’Ivoire. Virus Res. 2010, 147, 17–24.
- Diagne, M.M.; Gaye, A.; Ndione, M.H.D.; Faye, M.; Fall, G.; Dieng, I.; Widen, S.G.; Wood, T.G.; Popov, V.; Guzman, H.; et al. Dianke Virus: A New Mesonivirus Species Isolated from Mosquitoes in Eastern Senegal. Virus Res. 2020, 275, 197802.
- Zirkel, F.; Kurth, A.; Quan, P.-L.; Briese, T.; Ellerbrok, H.; Pauli, G.; Leendertz, F.H.; Lipkin, W.I.; Ziebuhr, J.; Drosten, C.; et al. An Insect Nidovirus Emerging from a Primary Tropical Rainforest. MBio 2011, 2, e00077-11.
- Zirkel, F.; Roth, H.; Kurth, A.; Drosten, C.; Ziebuhr, J.; Junglen, S. Identification and Characterization of Genetically Divergent Members of the Newly Established Family Mesoniviridae. J. Virol. 2013, 87, 6346–6358.
- Hermanns, K.; Zirkel, F.; Kurth, A.; Drosten, C.; Junglen, S. Cimodo Virus Belongs to a Novel Lineage of Reoviruses Isolated from African Mosquitoes. J. Gen. Virol. 2014, 95, 905–909.
- Marklewitz, M.; Handrick, S.; Grasse, W.; Kurth, A.; Lukashev, A.; Drosten, C.; Ellerbrok, H.; Leendertz, F.H.; Pauli, G.; Junglen, S. Gouleako Virus Isolated from West African Mosquitoes Constitutes a Proposed Novel Genus in the Family Bunyaviridae. J. Virol. 2011, 85, 9227–9234.
- Vasilakis, N.; Forrester, N.L.; Palacios, G.; Nasar, F.; Savji, N.; Rossi, S.L.; Guzman, H.; Wood, T.G.; Popov, V.; Gorchakov, R.; et al. Negevirus: A Proposed New Taxon of Insect-Specific Viruses with Wide Geographic Distribution. J. Virol. 2013, 87, 2475–2488.
- Parry, R.; Naccache, F.; Ndiaye, E.H.; Fall, G.; Castelli, I.; Lühken, R.; Medlock, J.; Cull, B.; Hesson, J.C.; Montarsi, F.; et al. Identification and RNAi Profile of a Novel Iflavirus Infecting Senegalese Aedes vexans arabiensis Mosquitoes. Viruses 2020, 12, 440.
- Schuster, S.; Zirkel, F.; Kurth, A.; van Cleef, K.W.R.; Drosten, C.; van Rij, R.P.; Junglen, S. A Unique Nodavirus with Novel Features: Mosinovirus Expresses Two Subgenomic RNAs, a Capsid Gene of Unknown Origin, and a Suppressor of the Antiviral RNA Interference Pathway. J. Virol. 2014, 88, 13447–13459.
- Kallies, R.; Kopp, A.; Zirkel, F.; Estrada, A.; Gillespie, T.; Drosten, C.; Junglen, S. Genetic Characterization of Goutanap Virus, a Novel Virus Related to Negeviruses, Cileviruses and Higreviruses. Viruses 2014, 6, 4346–4357.





