
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Mariana Boroni | + 4295 word(s) | 4295 | 2021-06-02 07:57:42 | | | |
| 2 | Bruce Ren | -21 word(s) | 4274 | 2021-06-10 03:20:11 | | |
Video Upload Options
The skin is our largest organ and the outermost protective barrier. Its aging reflects both intrinsic and extrinsic processes resulting from the constant insults it is exposed to. Aging in the skin is accompanied by specific epigenetic modifications, accumulation of senescent cells, reduced cellular proliferation/tissue renewal, altered extracellular matrix, and a proinflammatory environment favoring undesirable conditions, including disease onset. Macrophages (Mφ) are the most abundant immune cell type in the skin and comprise a group of heterogeneous and plastic cells that are key for skin homeostasis and host defense. However, they have also been implicated in orchestrating chronic inflammation during aging. Since Mφ are related to innate and adaptive immunity, it is possible that age-modified skin Mφ promote adaptive immunity exacerbation and exhaustion, favoring the emergence of proinflammatory pathologies, such as skin cancer.
1. Introduction
2. Hallmarks of Aging and Macrophages
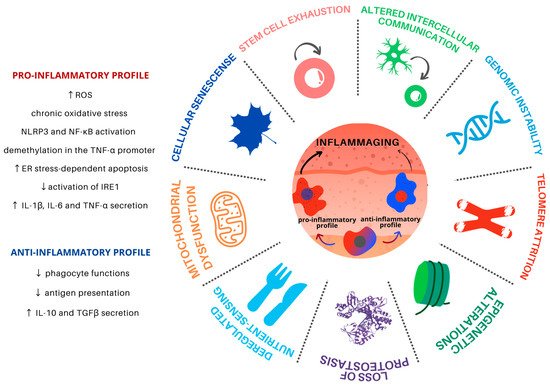
2.1. Genomic Instability
2.2. Telomere Attrition
2.3. Epigenetic Alterations
2.4. Loss of Proteostasis
2.5. Deregulated Nutrient Sensing
2.6. Mitochondrial Dysfunction
2.7. Cellular Senescence
2.8. Stem Cell Exhaustion
2.9. Altered Intercellular Communication
References
- Chung, H.Y.; Kim, D.H.; Lee, E.K.; Chung, K.W.; Chung, S.; Lee, B.; Seo, A.Y.; Chung, J.H.; Jung, Y.S.; Im, E.; et al. Redefining Chronic Inflammation in Aging and Age-Related Diseases: Proposal of the Senoinflammation Concept. Aging Dis. 2019, 10, 367–382.
- López-Otín, C.; Blasco, M.A.; Partridge, L.; Serrano, M.; Kroemer, G. The Hallmarks of Aging. Cell 2013, 153, 1194–1217.
- Mc Auley, M.T.; Guimera, A.M.; Hodgson, D.; Mcdonald, N.; Mooney, K.M.; Morgan, A.E.; Proctor, C.J. Modelling the Molecular Mechanisms of Aging. Biosci. Rep. 2017, 37.
- Tobin, D.J. Introduction to Skin Aging. J. Tissue Viability 2017, 26, 37–46.
- Rodrigues, L.P.; Teixeira, V.R.; Alencar-Silva, T.; Simonassi-Paiva, B.; Pereira, R.W.; Pogue, R.; Carvalho, J.L. Hallmarks of Aging and Immunosenescence: Connecting the Dots. Cytokine Growth Factor Rev. 2021, 59, 9–21.
- Solé-Boldo, L.; Raddatz, G.; Schütz, S.; Mallm, J.-P.; Rippe, K.; Lonsdorf, A.S.; Rodríguez-Paredes, M.; Lyko, F. Single-Cell Transcriptomes of the Human Skin Reveal Age-Related Loss of Fibroblast Priming. Commun. Biol. 2020, 3, 188.
- Robert, L.; Labat-Robert, J.; Robert, A.-M. Physiology of Skin Aging. Pathol. Biol. 2009, 57, 336–341.
- Franceschi, C.; Bonafè, M.; Valensin, S.; Olivieri, F.; De Luca, M.; Ottaviani, E.; De Benedictis, G. Inflamm-Aging. An Evolutionary Perspective on Immunosenescence. Ann. N. Y. Acad. Sci. 2000, 908, 244–254.
- Yanez, D.A.; Lacher, R.K.; Vidyarthi, A.; Colegio, O.R. The Role of Macrophages in Skin Homeostasis. Pflügers Arch. Eur. J. Physiol. 2017, 469, 455–463.
- Bian, Z.; Gong, Y.; Huang, T.; Lee, C.Z.W.; Bian, L.; Bai, Z.; Shi, H.; Zeng, Y.; Liu, C.; He, J.; et al. Deciphering Human Macrophage Development at Single-Cell Resolution. Nature 2020, 582, 571–576.
- Doebel, T.; Voisin, B.; Nagao, K. Langerhans Cells—The Macrophage in Dendritic Cell Clothing. Trends Immunol. 2017, 38, 817–828.
- Merad, M.; Ginhoux, F.; Collin, M. Origin, Homeostasis and Function of Langerhans Cells and Other Langerin-Expressing Dendritic Cells. Nat. Rev. Immunol. 2008, 8, 935–947.
- Kaplan, D.H. Langerhans Cells: Not Your Average Dendritic Cell. Trends Immunol. 2010, 31, 437.
- Gomez Perdiguero, E.; Klapproth, K.; Schulz, C.; Busch, K.; Azzoni, E.; Crozet, L.; Garner, H.; Trouillet, C.; de Bruijn, M.F.; Geissmann, F.; et al. Tissue-Resident Macrophages Originate from Yolk-Sac-Derived Erythro-Myeloid Progenitors. Nature 2015, 518, 547–551.
- Malissen, B.; Tamoutounour, S.; Henri, S. The Origins and Functions of Dendritic Cells and Macrophages in the Skin. Nat. Rev. Immunol. 2014, 14, 417–428.
- Shapouri-Moghaddam, A.; Mohammadian, S.; Vazini, H.; Taghadosi, M.; Esmaeili, S.-A.; Mardani, F.; Seifi, B.; Mohammadi, A.; Afshari, J.T.; Sahebkar, A. Macrophage Plasticity, Polarization, and Function in Health and Disease. J. Cell. Physiol. 2018, 233, 6425–6440.
- Qian, J.; Olbrecht, S.; Boeckx, B.; Vos, H.; Laoui, D.; Etlioglu, E.; Wauters, E.; Pomella, V.; Verbandt, S.; Busschaert, P.; et al. A Pan-Cancer Blueprint of the Heterogeneous Tumor Microenvironment Revealed by Single-Cell Profiling. Cell Res. 2020, 30, 745–762.
- Zilionis, R.; Engblom, C.; Pfirschke, C.; Savova, V.; Zemmour, D.; Saatcioglu, H.D.; Krishnan, I.; Maroni, G.; Meyerovitz, C.V.; Kerwin, C.M.; et al. Single-Cell Transcriptomics of Human and Mouse Lung Cancers Reveals Conserved Myeloid Populations across Individuals and Species. Immunity 2019, 50, 1317–1334.e10.
- Van Beek, A.A.; Van den Bossche, J.; Mastroberardino, P.G.; de Winther, M.P.J.; Leenen, P.J.M. Metabolic Alterations in Aging Macrophages: Ingredients for Inflammaging? Trends Immunol. 2019, 40, 113–127.
- Tabula Muris Consortium A Single-Cell Transcriptomic Atlas Characterizes Ageing Tissues in the Mouse. Nature 2020, 583, 590–595.
- Vida, C.; de Toda, I.M.; Cruces, J.; Garrido, A.; Gonzalez-Sanchez, M.; De la Fuente, M. Role of Macrophages in Age-Related Oxidative Stress and Lipofuscin Accumulation in Mice. Redox Biol. 2017, 12, 423–437.
- Chambers, E.S.; Vukmanovic-Stejic, M. Skin Barrier Immunity and Ageing. Immunology 2020, 160, 116–125.
- Xu, Y.-P.; Qi, R.-Q.; Chen, W.; Shi, Y.; Cui, Z.-Z.; Gao, X.-H.; Chen, H.-D.; Zhou, L.; Mi, Q.-S. Aging Affects Epidermal Langerhans Cell Development and Function and Alters Their miRNA Gene Expression Profile. Aging 2012, 4, 742–754.
- Foley, K.G.; Pritchard, M.T.; Duncan, F.E. Macrophage-Derived Multinucleated Giant Cells: Hallmarks of the Aging Ovary. Reproduction 2021, 161, V5–V9.
- Canan, C.H.; Gokhale, N.S.; Carruthers, B.; Lafuse, W.P.; Schlesinger, L.S.; Torrelles, J.B.; Turner, J. Characterization of Lung Inflammation and Its Impact on Macrophage Function in Aging. J. Leukoc. Biol. 2014, 96, 473–480.
- Deng, G.; Chen, W.; Wang, P.; Zhan, T.; Zheng, W.; Gu, Z.; Wang, X.; Ji, X.; Sun, Y. Inhibition of NLRP3 Inflammasome-Mediated Pyroptosis in Macrophage by Cycloastragenol Contributes to Amelioration of Imiquimod-Induced Psoriasis-like Skin Inflammation in Mice. Int. Immunopharmacol. 2019, 74, 105682.
- Lu, C.-H.; Lai, C.-Y.; Yeh, D.-W.; Liu, Y.-L.; Su, Y.-W.; Hsu, L.-C.; Chang, C.-H.; Catherine Jin, S.-L.; Chuang, T.-H. Involvement of M1 Macrophage Polarization in Endosomal Toll-Like Receptors Activated Psoriatic Inflammation. Mediat. Inflamm. 2018, 2018, 3523642.
- Liu, T.; Deng, Z.; Xie, H.; Chen, M.; Xu, S.; Peng, Q.; Sha, K.; Xiao, W.; Zhao, Z.; Li, J. ADAMDEC1 Promotes Skin Inflammation in Rosacea via Modulating the Polarization of M1 Macrophages. Biochem. Biophys. Res. Commun. 2020, 521, 64–71.
- Liu, Z.; Zhang, J.; Jiang, P.; Yin, Z.; Liu, Y.; Liu, Y.; Wang, X.; Hu, L.; Xu, Y.; Liu, W. Paeoniflorin Inhibits the Macrophage-Related Rosacea-like Inflammatory Reaction through the Suppressor of Cytokine Signaling 3-Apoptosis Signal-Regulating Kinase 1-p38 Pathway. Medicine 2021, 100, e23986.
- Abdellatif, A.A.; Zaki, A.M.; Abdo, H.M.; Aly, D.G.; Emara, T.A.; El-Toukhy, S.; Emam, H.M.; Abdelwahab, M.S. Assessment of Serum Levels of Granulocyte-Macrophage Colony-Stimulating Factor (GM-CSF) among Non-Segmental Vitiligo Patients: A Pilot Study. Acta Derm. Alp. Pannonica Adriat 2015, 24, 43–45.
- Farag, A.G.A.; Hammam, M.A.; Habib, M.S.; Elnaidany, N.F.; Kamh, M.E. Macrophage Migration Inhibitory Factor as an Incriminating Agent in Vitiligo. An. Bras. Dermatol. 2018, 93, 191–196.
- Ishida, Y.; Kuninaka, Y.; Yamamoto, Y.; Nosaka, M.; Kimura, A.; Furukawa, F.; Mukaida, N.; Kondo, T. Pivotal Involvement of the CX3CL1-CX3CR1 Axis for the Recruitment of M2 Tumor-Associated Macrophages in Skin Carcinogenesis. J. Investig. Dermatol. 2020, 140, 1951–1961.e6.
- Weber, C.; Telerman, S.B.; Reimer, A.S.; Sequeira, I.; Liakath-Ali, K.; Arwert, E.N.; Watt, F.M. Macrophage Infiltration and Alternative Activation during Wound Healing Promote MEK1-Induced Skin Carcinogenesis. Cancer Res. 2016, 76, 805–817.
- Castro-Garza, J.; Luévano-Martínez, M.L.; Villarreal-Treviño, L.; Gosálvez, J.; Fernández, J.L.; Dávila-Rodríguez, M.I.; García-Vielma, C.; González-Hernández, S.; Cortés-Gutiérrez, E.I. Mycobacterium Tuberculosis Promotes Genomic Instability in Macrophages. Mem. Inst. Oswaldo Cruz 2018, 113, 161–166.
- Herrtwich, L.; Nanda, I.; Evangelou, K.; Nikolova, T.; Horn, V.; Sagar; Erny, D.; Stefanowski, J.; Rogell, L.; Klein, C.; et al. DNA Damage Signaling Instructs Polyploid Macrophage Fate in Granulomas. Cell 2018, 174, 1325–1326.
- Horn, V.; Triantafyllopoulou, A. DNA Damage Signaling and Polyploid Macrophages in Chronic Inflammation. Curr. Opin. Immunol. 2018, 50, 55–63.
- Sidler, C. Chapter 29—Genomic Instability and Aging: Causes and Consequences. In Genome Stability; Kovalchuk, I., Kovalchuk, O., Eds.; Academic Press: Boston, MA, USA, 2016; pp. 511–525. ISBN 9780128033098.
- Janssens, S.; Tschopp, J. Signals from within: The DNA-Damage-Induced NF-κB Response. Cell Death Differ. 2006, 13, 773–784.
- Lloberas, J.; Tur, J.; Vico, T.; Celada, A. Molecular and Cellular Aspects of Macrophage Aging. In Handbook of Immunosenescence; Springer: Cham, Switzerland, 2018; pp. 1–32.
- Yue, Z.; Nie, L.; Zhang, P.; Chen, Q.; Lv, Q.; Wang, Q. Tissue-Resident Macrophage Inflammaging Aggravates Homeostasis Dysregulation in Age-Related Diseases. Cell. Immunol. 2021, 361, 104278.
- Stanley, S.E.; Armanios, M. The Short and Long Telomere Syndromes: Paired Paradigms for Molecular Medicine. Curr. Opin. Genet. Dev. 2015, 33, 1–9.
- De Maeyer, R.P.H.; Chambers, E.S. The Impact of Ageing on Monocytes and Macrophages. Immunol. Lett. 2021, 230, 1–10.
- Kang, Y.; Zhang, H.; Zhao, Y.; Wang, Y.; Wang, W.; He, Y.; Zhang, W.; Zhang, W.; Zhu, X.; Zhou, Y.; et al. Telomere Dysfunction Disturbs Macrophage Mitochondrial Metabolism and the NLRP3 Inflammasome through the PGC-1α/TNFAIP3 Axis. Cell Rep. 2018, 22, 3493–3506.
- Jurk, D.; Wilson, C.; Passos, J.F.; Oakley, F.; Correia-Melo, C.; Greaves, L.; Saretzki, G.; Fox, C.; Lawless, C.; Anderson, R.; et al. Chronic Inflammation Induces Telomere Dysfunction and Accelerates Ageing in Mice. Nat. Commun. 2014, 2, 4172.
- Jose, S.S.; Bendickova, K.; Kepak, T.; Krenova, Z.; Fric, J. Chronic Inflammation in Immune Aging: Role of Pattern Recognition Receptor Crosstalk with the Telomere Complex? Front. Immunol. 2017, 8, 1078.
- Zhang, J.; Rane, G.; Dai, X.; Shanmugam, M.K.; Arfuso, F.; Samy, R.P.; Lai, M.K.P.; Kappei, D.; Kumar, A.P.; Sethi, G. Ageing and the Telomere Connection: An Intimate Relationship with Inflammation. Ageing Res. Rev. 2016, 25, 55–69.
- Chen, S.; Yang, J.; Wei, Y.; Wei, X. Epigenetic Regulation of Macrophages: From Homeostasis Maintenance to Host Defense. Cell. Mol. Immunol. 2020, 17, 36–49.
- Ivashkiv, L.B. Epigenetic Regulation of Macrophage Polarization and Function. Trends Immunol. 2013, 34, 216–223.
- Daskalaki, M.G.; Tsatsanis, C.; Kampranis, S.C. Histone Methylation and Acetylation in Macrophages as a Mechanism for Regulation of Inflammatory Responses. J. Cell. Physiol. 2018, 233, 6495–6507.
- Horvath, S. DNA Methylation Age of Human Tissues and Cell Types. Genome Biol. 2013, 14, R115.
- Boroni, M.; Zonari, A.; de Oliveira, C.R.; Alkatib, K.; Cruz, E.A.O.; Brace, L.E.; de Carvalho, J.L. Highly Accurate Skin-Specific Methylome Analysis Algorithm as a Platform to Screen and Validate Therapeutics for Healthy Aging. Clin. Epigenet. 2020, 12, 105.
- Dekkers, K.F.; Neele, A.E.; Jukema, J.W.; Heijmans, B.T.; de Winther, M.P.J. Human Monocyte-to-Macrophage Differentiation Involves Highly Localized Gain and Loss of DNA Methylation at Transcription Factor Binding Sites. Epigenet. Chromatin. 2019, 12, 34.
- Gowers, I.R.; Walters, K.; Kiss-Toth, E.; Read, R.C.; Duff, G.W.; Wilson, A.G. Age-Related Loss of CpG Methylation in the Tumour Necrosis Factor Promoter. Cytokine 2011, 56, 792–797.
- Satoh, T.; Takeuchi, O.; Vandenbon, A.; Yasuda, K.; Tanaka, Y.; Kumagai, Y.; Miyake, T.; Matsushita, K.; Okazaki, T.; Saitoh, T.; et al. The Jmjd3-Irf4 Axis Regulates M2 Macrophage Polarization and Host Responses against Helminth Infection. Nat. Immunol. 2010, 11, 936–944.
- Kittan, N.A.; Allen, R.M.; Dhaliwal, A.; Cavassani, K.A.; Schaller, M.; Gallagher, K.A.; Carson, W.F., 4th; Mukherjee, S.; Grembecka, J.; Cierpicki, T.; et al. Cytokine Induced Phenotypic and Epigenetic Signatures Are Key to Establishing Specific Macrophage Phenotypes. PLoS ONE 2013, 8, e78045.
- de Groot, A.E.; Pienta, K.J. Epigenetic Control of Macrophage Polarization: Implications for Targeting Tumor-Associated Macrophages. Oncotarget 2018, 9, 20908–20927.
- Otoupalova, E.; Smith, S.; Cheng, G.; Thannickal, V.J. Oxidative Stress in Pulmonary Fibrosis. Compr. Physiol. 2011, 10, 509–547.
- Williams, R.; Laskovs, M.; Williams, R.I.; Mahadevan, A.; Labbadia, J. A Mitochondrial Stress-Specific Form of HSF1 Protects against Age-Related Proteostasis Collapse. Dev. Cell 2020, 54, 758–772.e5.
- Bonam, S.R.; Ruff, M.; Muller, S. HSPA8/HSC70 in Immune Disorders: A Molecular Rheostat That Adjusts Chaperone-Mediated Autophagy Substrates. Cells 2019, 8, 849.
- Tsukahara, K.; Tamatsu, Y.; Sugawara, Y.; Shimada, K. Morphological Study of the Relationship between Solar Elastosis and the Development of Wrinkles on the Forehead and Lateral Canthus. Arch. Dermatol. 2012, 148, 913–917.
- Rossetti, D.; Kielmanowicz, M.G.; Vigodman, S.; Hu, Y.P.; Chen, N.; Nkengne, A.; Oddos, T.; Fischer, D.; Seiberg, M.; Lin, C.B. A Novel Anti-Ageing Mechanism for Retinol: Induction of Dermal Elastin Synthesis and Elastin Fibre Formation. Int. J. Cosmet. Sci. 2011, 33, 62–69.
- Genovese, L.; Corbo, A.; Sibilla, S. An Insight into the Changes in Skin Texture and Properties Following Dietary Intervention with a Nutricosmeceutical Containing a Blend of Collagen Bioactive Peptides and Antioxidants. Skin Pharmacol. Physiol. 2017, 30, 146–158.
- Jiang, M.; Yan, F.; Avram, M.; Lu, Z. A Prospective Study of the Safety and Efficacy of a Combined Bipolar Radiofrequency, Intense Pulsed Light, and Infrared Diode Laser Treatment for Global Facial Photoaging. Lasers Med. Sci. 2017, 32, 1051–1061.
- Charles-de-Sá, L.; Gontijo-de-Amorim, N.F.; Rigotti, G.; Sbarbati, A.; Bernardi, P.; Benati, D.; Bizon Vieira Carias, R.; Maeda Takiya, C.; Borojevic, R. Photoaged Skin Therapy with Adipose-Derived Stem Cells. Plast. Reconstr. Surg. 2020, 145, 1037e–1049e.
- Pain, S.; Berthélémy, N.; Naudin, C.; Degrave, V.; André-Frei, V. Understanding Solar Skin Elastosis-Cause and Treatment. J. Cosmet. Sci. 2018, 69, 175–185.
- Song, Y.; Shen, H.; Du, W.; Goldstein, D.R. Inhibition of X-Box Binding Protein 1 Reduces Tunicamycin-Induced Apoptosis in Aged Murine Macrophages. Aging Cell 2013, 12, 794–801.
- Martinon, F.; Chen, X.; Lee, A.-H.; Glimcher, L.H. TLR Activation of the Transcription Factor XBP1 Regulates Innate Immune Responses in Macrophages. Nat. Immunol. 2010, 11, 411–418.
- Shenderov, K.; Riteau, N.; Yip, R.; Mayer-Barber, K.D.; Oland, S.; Hieny, S.; Fitzgerald, P.; Oberst, A.; Dillon, C.P.; Green, D.R.; et al. Cutting Edge: Endoplasmic Reticulum Stress Licenses Macrophages To Produce Mature IL-1β in Response to TLR4 Stimulation through a Caspase-8– and TRIF-Dependent Pathway. J. Immunol. 2014, 192, 2029–2033.
- Fattah, E.A.; Bhattacharya, A.; Herron, A.; Safdar, Z.; Tony Eissa, N. Critical Role for IL-18 in Spontaneous Lung Inflammation Caused by Autophagy Deficiency. J. Immunol. 2015, 194, 5407–5416.
- Shin, J.N.; Fattah, E.A.; Bhattacharya, A.; Ko, S.; Eissa, N.T. Inflammasome Activation by Altered Proteostasis. J. Biol. Chem. 2013, 288, 35886–35895.
- Sun, W.; Pang, Y.; Liu, Z.; Sun, L.; Liu, B.; Xu, M.; Dong, Y.; Feng, J.; Jiang, C.; Kong, W.; et al. Macrophage Inflammasome Mediates Hyperhomocysteinemia-Aggravated Abdominal Aortic Aneurysm. J. Mol. Cell. Cardiol. 2015, 81, 96–106.
- Xiu, F.; Catapano, M.; Diao, L.; Stanojcic, M.; Jeschke, M.G. Prolonged Endoplasmic Reticulum–Stressed Hepatocytes Drive an Alternative Macrophage Polarization. Shock 2015, 44, 44–51.
- Mahadevan, N.R.; Rodvold, J.; Sepulveda, H.; Rossi, S.; Drew, A.F.; Zanetti, M. Transmission of Endoplasmic Reticulum Stress and pro-Inflammation from Tumor Cells to Myeloid Cells. Proc. Natl. Acad. Sci. USA 2011, 108, 6561–6566.
- Aunan, J.R.; Watson, M.M.; Hagland, H.R.; Søreide, K. Molecular and Biological Hallmarks of Ageing. Br. J. Surg. 2016, 103, e29–e46.
- Barzilai, N.; Ferrucci, L. Insulin Resistance and Aging: A Cause or a Protective Response? J. Gerontol. A Biol. Sci. Med. Sci. 2012, 67, 1329–1331.
- Sanchez-Garrido, J.; Shenoy, A.R. Regulation and Repurposing of Nutrient Sensing and Autophagy in Innate Immunity. Autophagy 2020, 1–21.
- Tabibzadeh, S. Signaling Pathways and Effectors of Aging. Front. Biosci. 2021, 26, 50–96.
- Qin, K.; Han, C.; Zhang, H.; Li, T.; Li, N.; Cao, X. NAD+ Dependent Deacetylase Sirtuin 5 Rescues the Innate Inflammatory Response of Endotoxin Tolerant Macrophages by Promoting Acetylation of p65. J. Autoimmun. 2017, 81, 120–129.
- Viola, A.; Munari, F.; Sánchez-Rodríguez, R.; Scolaro, T.; Castegna, A. The Metabolic Signature of Macrophage Responses. Front. Immunol. 2019, 10, 1462.
- Ip, W.K.E.; Hoshi, N.; Shouval, D.S.; Snapper, S.; Medzhitov, R. Anti-Inflammatory Effect of IL-10 Mediated by Metabolic Reprogramming of Macrophages. Science 2017, 356, 513–519.
- Wang, F.; Zhang, S.; Vuckovic, I.; Jeon, R.; Lerman, A.; Folmes, C.D.; Dzeja, P.P.; Herrmann, J. Glycolytic Stimulation Is Not a Requirement for M2 Macrophage Differentiation. Cell Metab. 2018, 28, 463–475.e4.
- Binger, K.J.; Gebhardt, M.; Heinig, M.; Rintisch, C.; Schroeder, A.; Neuhofer, W.; Hilgers, K.; Manzel, A.; Schwartz, C.; Kleinewietfeld, M.; et al. High Salt Reduces the Activation of IL-4- and IL-13-Stimulated Macrophages. J. Clin. Investig. 2015, 125, 4223–4238.
- Huang, S.C.-C.; Smith, A.M.; Everts, B.; Colonna, M.; Pearce, E.L.; Schilling, J.D.; Pearce, E.J. Metabolic Reprogramming Mediated by the mTORC2-IRF4 Signaling Axis Is Essential for Macrophage Alternative Activation. Immunity 2016, 45, 817–830.
- Knipper, J.A.; Willenborg, S.; Brinckmann, J.; Bloch, W.; Maaß, T.; Wagener, R.; Krieg, T.; Sutherland, T.; Munitz, A.; Rothenberg, M.E.; et al. Interleukin-4 Receptor α Signaling in Myeloid Cells Controls Collagen Fibril Assembly in Skin Repair. Immunity 2015, 43, 803–816.
- Minutti, C.M.; Jackson-Jones, L.H.; García-Fojeda, B.; Knipper, J.A.; Sutherland, T.E.; Logan, N.; Ringqvist, E.; Guillamat-Prats, R.; Ferenbach, D.A.; Artigas, A.; et al. Local Amplifiers of IL-4Rα–mediated Macrophage Activation Promote Repair in Lung and Liver. Science 2017, 356, 1076–1080.
- Weng, S.-Y.; Wang, X.; Vijayan, S.; Tang, Y.; Kim, Y.O.; Padberg, K.; Regen, T.; Molokanova, O.; Chen, T.; Bopp, T.; et al. IL-4 Receptor Alpha Signaling through Macrophages Differentially Regulates Liver Fibrosis Progression and Reversal. EBioMedicine 2018, 29, 92–103.
- Gan, L.; Zheng, W.; Chabot, J.-G.; Unterman, T.G.; Quirion, R. Nuclear/cytoplasmic Shuttling of the Transcription Factor FoxO1 Is Regulated by Neurotrophic Factors. J. Neurochem. 2005, 93, 1209–1219.
- Betz, C.; Hall, M.N. Where Is mTOR and What Is It Doing There? J. Cell Biol. 2013, 203, 563–574.
- Knuever, J.; Willenborg, S.; Ding, X.; Akyüz, M.D.; Partridge, L.; Niessen, C.M.; Brüning, J.C.; Eming, S.A. Myeloid Cell-Restricted Insulin/IGF-1 Receptor Deficiency Protects against Skin Inflammation. J. Immunol. 2015, 195, 5296–5308.
- Prattichizzo, F.; De Nigris, V.; Mancuso, E.; Spiga, R.; Giuliani, A.; Matacchione, G.; Lazzarini, R.; Marcheselli, F.; Recchioni, R.; Testa, R.; et al. Short-Term Sustained Hyperglycaemia Fosters an Archetypal Senescence-Associated Secretory Phenotype in Endothelial Cells and Macrophages. Redox Biol. 2018, 15, 170–1811.
- Chung, S.; Ranjan, R.; Lee, Y.G.; Park, G.Y.; Karpurapu, M.; Deng, J.; Xiao, L.; Kim, J.Y.; Unterman, T.G.; Christman, J.W. Distinct Role of FoxO1 in M-CSF- and GM-CSF-Differentiated Macrophages Contributes LPS-Mediated IL-10: Implication in Hyperglycemia. J. Leukoc. Biol. 2015, 97, 327–339.
- Spadaro, O.; Goldberg, E.L.; Camell, C.D.; Youm, Y.-H.; Kopchick, J.J.; Nguyen, K.Y.; Bartke, A.; Sun, L.Y.; Dixit, V.D. Growth Hormone Receptor Deficiency Protects against Age-Related NLRP3 Inflammasome Activation and Immune Senescence. Cell Rep. 2016, 14, 1571–1580.
- Qing, L.; Fu, J.; Wu, P.; Zhou, Z.; Yu, F.; Tang, J. Metformin Induces the M2 Macrophage Polarization to Accelerate the Wound Healing via Regulating AMPK/mTOR/NLRP3 Inflammasome Singling Pathway. Am. J. Transl. Res. 2019, 11, 655–668.
- Koo, S.-J.; Garg, N.J. Metabolic Programming of Macrophage Functions and Pathogens Control. Redox Biol. 2019, 24, 101198.
- Shekhova, E. Mitochondrial Reactive Oxygen Species as Major Effectors of Antimicrobial Immunity. PLoS Pathog. 2020, 16, e1008470.
- Salminen, A.; Ojala, J.; Kaarniranta, K.; Kauppinen, A. Mitochondrial Dysfunction and Oxidative Stress Activate Inflammasomes: Impact on the Aging Process and Age-Related Diseases. Cell. Mol. Life Sci. 2012, 69, 2999–3013.
- Yarbro, J.R.; Emmons, R.S.; Pence, B.D. Macrophage Immunometabolism and Inflammaging: Roles of Mitochondrial Dysfunction, Cellular Senescence, CD38, and NAD. Immunometabolism 2020, 2, e200026.
- Plataki, M.; Cho, S.J.; Harris, R.M.; Huang, H.-R.; Yun, H.S.; Schiffer, K.T.; Stout-Delgado, H.W. Mitochondrial Dysfunction in Aged Macrophages and Lung during Primary Streptococcus Pneumoniae Infection Is Improved with Pirfenidone. Sci. Rep. 2019, 9, 971.
- Giuliani, A.; Prattichizzo, F.; Micolucci, L.; Ceriello, A.; Procopio, A.D.; Rippo, M.R. Mitochondrial (Dys) Function in Inflammaging: Do MitomiRs Influence the Energetic, Oxidative, and Inflammatory Status of Senescent Cells? Mediat. Inflamm. 2017, 2017, 2309034.
- Minhas, P.S.; Liu, L.; Moon, P.K.; Joshi, A.U.; Dove, C.; Mhatre, S.; Contrepois, K.; Wang, Q.; Lee, B.A.; Coronado, M.; et al. Macrophage de Novo NAD+ Synthesis Specifies Immune Function in Aging and Inflammation. Nat. Immunol. 2019, 20, 50–63.
- Franceschi, C.; Garagnani, P.; Vitale, G.; Capri, M.; Salvioli, S. Inflammaging and “Garb-Aging”. Trends Endocrinol. Metab. 2017, 28, 199–212.
- Sreedhar, A.; Aguilera-Aguirre, L.; Singh, K.K. Mitochondria in Skin Health, Aging, and Disease. Cell Death Dis. 2020, 11, 444.
- Tanaka, H.; Okada, T.; Konishi, H.; Tsuji, T. The Effect of Reactive Oxygen Species on the Biosynthesis of Collagen and Glycosaminoglycans in Cultured Human Dermal Fibroblasts. Arch. Dermatol. Res. 1993, 285, 352–355.
- Rinnerthaler, M.; Bischof, J.; Streubel, M.K.; Trost, A.; Richter, K. Oxidative Stress in Aging Human Skin. Biomolecules 2015, 5, 545–589.
- Ives, A.; Nomura, J.; Martinon, F.; Roger, T.; LeRoy, D.; Miner, J.N.; Simon, G.; Busso, N.; So, A. Xanthine Oxidoreductase Regulates Macrophage IL1β Secretion upon NLRP3 Inflammasome Activation. Nat. Commun. 2015, 6, 6555.
- Fuente, M.; Miquel, J. An Update of the Oxidation-Inflammation Theory of Aging: The Involvement of the Immune System in Oxi-Inflamm-Aging. Curr. Pharm. Des. 2009, 15, 3003–3026.
- Herranz, N.; Gil, J. Mechanisms and Functions of Cellular Senescence. J. Clin. Investig. 2018, 128, 1238–1246.
- Gruber, F.; Kremslehner, C.; Eckhart, L.; Tschachler, E. Cell Aging and Cellular Senescence in Skin Aging—Recent Advances in Fibroblast and Keratinocyte Biology. Exp. Gerontol. 2020, 130, 110780.
- Coppé, J.-P.; Desprez, P.-Y.; Krtolica, A.; Campisi, J. The Senescence-Associated Secretory Phenotype: The Dark Side of Tumor Suppression. Annu. Rev. Pathol. 2010, 5, 99–118.
- Kale, A.; Sharma, A.; Stolzing, A.; Desprez, P.-Y.; Campisi, J. Role of Immune Cells in the Removal of Deleterious Senescent Cells. Immun. Ageing 2020, 17, 16.
- Ogata, Y.; Yamada, T.; Hasegawa, S.; Sanada, A.; Iwata, Y.; Arima, M.; Nakata, S.; Sugiura, K.; Akamatsu, H. SASP-Induced Macrophage Dysfunction May Contribute to Accelerated Senescent Fibroblast Accumulation in the Dermis. Exp. Dermatol. 2021, 30, 84–91.
- Gomez, C.R.; Karavitis, J.; Palmer, J.L.; Faunce, D.E.; Ramirez, L.; Nomellini, V.; Kovacs, E.J. Interleukin-6 Contributes to Age-Related Alteration of Cytokine Production by Macrophages. Mediators Inflamm. 2010, 2010, 475139.
- Stout, R.D.; Suttles, J. Immunosenescence and Macrophage Functional Plasticity: Dysregulation of Macrophage Function by Age-Associated Microenvironmental Changes. Immunol. Rev. 2005, 205, 60–71.
- Cao Dinh, H.; Njemini, R.; Onyema, O.O.; Beyer, I.; Liberman, K.; De Dobbeleer, L.; Renmans, W.; Vander Meeren, S.; Jochmans, K.; Delaere, A.; et al. Strength Endurance Training but Not Intensive Strength Training Reduces Senescence-Prone T Cells in Peripheral Blood in Community-Dwelling Elderly Women. J. Gerontol. A Biol. Sci. Med. Sci. 2019, 74, 1870–1878.
- Díaz-Del Cerro, E.; Vida, C.; Martínez de Toda, I.; Félix, J.; De la Fuente, M. The Use of a Bed with an Insulating System of Electromagnetic Fields Improves Immune Function, Redox and Inflammatory States, and Decrease the Rate of Aging. Environ. Health 2020, 19, 118.
- Sotiropoulou, P.A.; Blanpain, C. Development and Homeostasis of the Skin Epidermis. Cold Spring Harb. Perspect. Biol. 2012, 4, a008383.
- Dunnwald, M.; Chinnathambi, S.; Alexandrunas, D.; Bickenbach, J.R. Mouse Epidermal Stem Cells Proceed through the Cell Cycle. J. Cell. Physiol. 2003, 195, 194–201.
- Li, L.; Ngo, H.T.T.; Hwang, E.; Wei, X.; Liu, Y.; Liu, J.; Yi, T.-H. Conditioned Medium from Human Adipose-Derived Mesenchymal Stem Cell Culture Prevents UVB-Induced Skin Aging in Human Keratinocytes and Dermal Fibroblasts. Int. J. Mol. Sci. 2019, 21, 49.
- Ma, T.; Fu, B.; Yang, X.; Xiao, Y.; Pan, M. Adipose Mesenchymal Stem Cell-Derived Exosomes Promote Cell Proliferation, Migration, and Inhibit Cell Apoptosis via Wnt/β-Catenin Signaling in Cutaneous Wound Healing. J. Cell. Biochem. 2019, 120, 10847–10854.
- Castilho, R.M.; Squarize, C.H.; Chodosh, L.A.; Williams, B.O.; Gutkind, J.S. mTOR Mediates Wnt-Induced Epidermal Stem Cell Exhaustion and Aging. Cell Stem Cell 2009, 5, 279–289.
- Ide, S.; Yahara, Y.; Kobayashi, Y.; Strausser, S.A.; Ide, K.; Watwe, A.; Xu-Vanpala, S.; Privratsky, J.R.; Crowley, S.D.; Shinohara, M.L.; et al. Yolk-Sac-Derived Macrophages Progressively Expand in the Mouse Kidney with Age. eLife 2020, 9, e51756.
- Ferrer, I.R.; West, H.C.; Henderson, S.; Ushakov, D.S.; e Sousa, P.S.; Strid, J.; Chakraverty, R.; Yates, A.J.; Bennett, C.L. A Wave of Monocytes Is Recruited to Replenish the Long-Term Langerhans Cell Network after Immune Injury. Sci. Immunol. 2019, 4, eaax8704.
- Lucas, T.; Waisman, A.; Ranjan, R.; Roes, J.; Krieg, T.; Müller, W.; Roers, A.; Eming, S.A. Differential Roles of Macrophages in Diverse Phases of Skin Repair. J. Immunol. 2010, 184, 3964–3977.
- Rodrigues, M.; Kosaric, N.; Bonham, C.A.; Gurtner, G.C. Wound Healing: A Cellular Perspective. Physiol. Rev. 2019, 99, 665–706.
- Fisher, G.; Rittié, L. Restoration of the Basement Membrane after Wounding: A Hallmark of Young Human Skin Altered with Aging. J. Cell Commun. Signal. 2018, 12, 401–411.
- Lee, S.; Szilagyi, E.; Chen, L.; Premanand, K.; DiPietro, L.A.; Ennis, W.; Bartholomew, A.M. Activated Mesenchymal Stem Cells Increase Wound Tensile Strength in Aged Mouse Model via Macrophages. J. Surg. Res. 2013, 181, 20–24.
- Morgun, E.I.; Vorotelyak, E.A. Epidermal Stem Cells in Hair Follicle Cycling and Skin Regeneration: A View From the Perspective of Inflammation. Front. Cell Dev. Biol. 2020, 8, 581697.
- Kolter, J.; Feuerstein, R.; Zeis, P.; Hagemeyer, N.; Paterson, N.; d’Errico, P.; Baasch, S.; Amann, L.; Masuda, T.; Lösslein, A.; et al. A Subset of Skin Macrophages Contributes to the Surveillance and Regeneration of Local Nerves. Immunity 2019, 50, 1482–1497.e7.
- Shook, B.A.; Wasko, R.R.; Rivera-Gonzalez, G.C.; Salazar-Gatzimas, E.; López-Giráldez, F.; Dash, B.C.; Muñoz-Rojas, A.R.; Aultman, K.D.; Zwick, R.K.; Lei, V.; et al. Myofibroblast Proliferation and Heterogeneity Are Supported by Macrophages during Skin Repair. Science 2018, 362, eaar2971.
- Zhuang, Y.; Lyga, J. Inflammaging in Skin and Other Tissues—the Roles of Complement System and Macrophage. Inflamm. Allergy Drug Targets 2014, 13, 153–161.
- Doles, J.; Storer, M.; Cozzuto, L.; Roma, G.; Keyes, W.M. Age-Associated Inflammation Inhibits Epidermal Stem Cell Function. Genes Dev. 2012, 26, 2144–2153.
- Rando, T.A.; Chang, H.Y. Aging, Rejuvenation, and Epigenetic Reprogramming: Resetting the Aging Clock. Cell 2012, 148, 46–57.
- Pilkington, S.M.; Barron, M.J.; Watson, R.E.B.; Griffiths, C.E.M.; Bulfone-Paus, S. Aged Human Skin Accumulates Mast Cells with Altered Functionality That Localize to Macrophages and Vasoactive Intestinal Peptide-Positive Nerve Fibres. Br. J. Dermatol. 2019, 180, 849–858.
- Fulop, T.; Witkowski, J.M.; Olivieri, F.; Larbi, A. The Integration of Inflammaging in Age-Related Diseases. Semin. Immunol. 2018, 40, 17–35.





