
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Arrigo Cicero | + 4673 word(s) | 4673 | 2021-05-27 10:38:25 | | | |
| 2 | Vivi Li | Meta information modification | 4673 | 2021-06-09 03:07:43 | | |
Video Upload Options
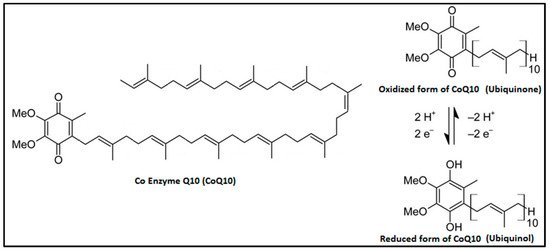
Coenzyme Q10 (CoQ10) is a ubiquitous factor present in cell membranes and mitochondria, both in its reduced (ubiquinol) and oxidized (ubiquinone) forms. Its levels are high in organs with high metabolism such as the heart, kidneys, and liver because it acts as an energy transfer molecule but could be reduced by aging, genetic factors, drugs (e.g., statins), cardiovascular (CV) diseases, degenerative muscle disorders, and neurodegenerative diseases. As CoQ10 is endowed with significant antioxidant and anti-inflammatory features, useful to prevent free radical-induced damage and inflammatory signaling pathway activation, its depletion results in exacerbation of inflammatory processes. Therefore, exogenous CoQ10 supplementation might be useful as an adjuvant in the treatment of cardiovascular diseases such as heart failure, atrial fibrillation, and myocardial infarction and in associated risk factors such as hypertension, insulin resistance, dyslipidemias, and obesity.
1. Introduction
| Organ | Ubiquinone Concentration (µg/g) | Ubiquinol Concentration (µg/g) |
|---|---|---|
| Heart | 132.0 | 61.0 |
| Kidneys | 77.0 | 75.0 |
| Liver | 63.6 | 95.0 |
| Muscle | 39.7 | 65.0 |
| Brain | 13.4 | 23.0 |
| Pancreas | 32.7 | |
| Spleen | 24.6 | |
| Lung | 7.9 | 25.0 |
| Thyroid | 24.7 | |
| Testis | 10.5 | |
| Intestine | 11.5 | 95.0 |
| Colon | 10.7 | |
| Ventricle | 11.8 | |
| Plasma (µmol/mL) | 1.1 | 96.0 |
2. CoQ10 and Cardiovascular Risk Factors
2.1. High Blood Pressure
2.2. Insulin-Resistance and Type 2 Diabetes
2.3. Dyslipidemias
2.4. Systemic Inflammation
3. CoQ10 and Cardiovascular Disease
3.1. CoQ10 and Heart Failure (HF)
3.2. CoQ10 and Myocardial Infarction
3.3. CoQ10 and Atrial Fibrillation
3.4. CoQ10 and Nonischemic Cardiomyopathies
3.5. CoQ10 and Ischemic Stroke
References
- Cicero, A.F.; Colletti, A. Nutraceuticals and Dietary Supplements to Improve Quality of Life and Outcomes in Heart Failure Patients. Curr. Pharm. Des. 2017, 23, 1265–1272.
- Bentinger, M.; Brismar, K.; Dallner, G. The antioxidant role of coenzyme Q. Mitochondrion 2007, 7, S41–S50.
- Saini, R. Coenzyme Q10: The essential nutrient. J. Pharm. Bioallied Sci. 2011, 3, 466–467.
- Aberg, F.; Appelkvist, E.L.; Dallner, G.; Ernster, L. Distribution and redox state of ubiquinones in rat and human tissues. Arch. Biochem. Biophys. 1992, 295, 230–234.
- Miles, M.V.; Horn, P.S.; Morrison, J.A.; Tang, P.H.; DeGrauw, T.; Pesce, A.J. Plasma coenzyme Q10 reference intervals, but not redox status, are affected by gender and race in self-reported healthy adults. Clin. Chim. Acta 2003, 332, 123–132.
- Hernández-Camacho, J.D.; Bernier, M.; López-Lluch, G.; Navas, P. Coenzyme Q10 Supplementation in Aging and Disease. Front. Physiol. 2018, 9, 44.
- Garrido-Maraver, J.; Oropesa-Ávila, M.; Cordero, M.D.; Vega, A.F.; de la Mata, M.; Delgado, A.; de Miguel, M.; Calero, C.P.; Villanueva Paz, M.; Cotán, D.; et al. Coenzyme Q10 Therapy. Mol. Syndromol. 2014, 5, 187–197.
- De Barcelos, I.P.; Haas, R.H. CoQ10 and Aging. Biology 2019, 8, 28.
- Littarru, G.P.; Tiano, L. Bioenergetic and antioxidant properties of coenzyme Q10: Recent developments. Mol. Biotechnol. 2007, 37, 31–37.
- Malekmohammad, K.; Sewell, R.D.E.; Rafieian-Kopaei, M. Antioxidants and Atherosclerosis: Mechanistic Aspects. Biomolecules 2019, 9, 301.
- Stefely, J.A.; Pagliarini, D.J. Biochemistry of Mitochondrial Coenzyme Q Biosynthesis. Trends Biochem. Sci. 2017, 42, 824–843.
- Szkopińska, A. Ubiquinone. Biosynthesis of quinone ring and its isoprenoid side chain. Intracellular localization. Acta Biochim. Pol. 2000, 47, 469–480.
- Turunen, M.; Olsson, J.; Dallner, G. Metabolism and function of coenzyme Q. Biochim. Biophys. Acta 2004, 1660, 171–199.
- Zhang, Y.; Aberg, F.; Appelkvist, E.L.; Dallner, G.; Ernster, L. Uptake of dietary coenzyme Q supplement is limited in rats. J. Nutr. 1995, 125, 446–453.
- Gutierrez-Mariscal, F.M.; Yubero-Serrano, E.M.; Villalba, J.M.; Lopez-Miranda, J. Coenzyme Q10: From bench to clinic in aging diseases, a translational review. Crit. Rev. Food Sci. Nutr. 2018, 16, 1–18.
- Boroujeni, M.B.; Khayat, Z.K.; Anbari, K.; Niapour, A.; Gholami, M.; Gharravi, A.M. Coenzyme Q10 Protects Skeletal Muscle From Ischemia-Reperfusion Through the NF-kappa B Pathway. Perfusion 2017, 32, 372–377.
- Zhai, J.; Bo, Y.; Lu, Y.; Liu, C.; Zhang, L. Effects of Coenzyme Q10 on Markers of Inflammation: A Systematic Review and Meta-Analysis. PLoS ONE 2017, 12, e0170172.
- Mantle, D.; Hargreaves, I. Coenzyme Q10 and Degenerative Disorders Affecting Longevity: An Overview. Antioxidants 2019, 8, 44.
- Galasko, D.R.; Peskind, E.; Clark, C.M.; Quinn, J.F.; Ringman, J.M.; Jicha, G.A.; Cotman, C.; Cottrell, B.; Montine, T.J.; Thomas, R.G.; et al. Alzheimer’s Disease Cooperative Study Antioxidants for Alzheimer disease: A randomized clinical trial with cerebrospinal fluid biomarker measures. Arch. Neurol. 2012, 69, 836–841.
- Müller, T.; Büttner, T.; Gholipour, A.F.; Kuhn, W. Coenzyme Q10 supplementation provides mild symptomatic benefit in patients with Parkinson’s disease. Neurosci. Lett. 2003, 341, 201–204.
- Parkinson Study Group QE3 Investigators. A randomized clinical trial of high-dosage coenzyme Q10 in early Parkinson disease: No evidence of benefit. JAMA Neurol. 2014, 71, 543–552.
- NCD Risk Factor Collaboration. Worldwide trends in blood pressure from 1975 to 2015: A pooled analysis of 1479 population-based measurement studies with 19.1 million participants. Lancet 2017, 389, 37–55.
- Forouzanfar, M.H.; Liu, P.; Roth, G.A.; Ng, M.; Biryukov, S.; Marczak, L.; Alexander, L.; Estep, K.; Abate, K.A.; Akinyemiju, T.F.; et al. Global Burden of Hypertension and Systolic Blood Pressure of at Least 110 to 115 mm Hg, 1990–2015. JAMA 2017, 317, 165–182.
- Hednerm, T.; Kjeldsen, S.E.; Narkiewicz, K. State of global health-hypertension burden and control. Blood Press 2012, 21, 12.
- Digiesi, V.; Cantini, F.; Oradei, A.; Bisi, G.; Guarino, G.C.; Brocchi, A.; Bellandi, F.; Mancini, M.; Littarru, G.P. Coenzyme Q10 in essential hypertension. Mol. Aspects Med. 1994, 15, s257–s263.
- Ignarro, L.J. Biological actions and properties of endothelium-derived nitric oxide formed and released from artery and vein. Circ. Res. 1989, 65, 1–21.
- Fabre, L.F.; Banks, R.C., Jr.; McIsaac, W.M.; Farrell, G. Effects of ubiquinone and related substances on secretion of aldosterone and cortisol. Am. J. Physiol. 1965, 208, 1275–1280.
- Langsjoen, P.; Willis, R.; Folkers, K. Treatment of essential hypertension with coenzyme Q10. Mol. Aspects Med. 1994, 15, S265–S272.
- Zhang, P.; Yang, C.; Guo, H.; Wang, J.; Lin, S.; Li, H.; Yang, Y.; Ling, W. Treatment of coenzyme Q10 for 24 weeks improves lipid and glycemic profile in dyslipidemic individuals. J. Clin. Lipidol. 2018, 12, 417–427.
- Ho, M.J.; Bellusci, A.; Wright, J.M. Blood pressure lowering efficacy of coenzyme Q10 for primary hypertension. Cochrane Database Syst. Rev. 2009, CD007435.
- Rosenfeldt, F.L.; Haas, S.J.; Krum, H.; Hadj, A.; Leong, J.Y.; Watts, G.F. Coenzyme Q10 in the treatment of hypertension: A meta-analysis of the clinical trials. J. Hum. Hypertens. 2007, 21, 297–306.
- Tabrizi, R.; Akbari, M.; Sharifi, N.; Lankarani, K.B.; Kolahdooz, F.; Taghizadeh, M.; Asemi, Z. The Effects of Coenzyme Q10 Supplementation on Blood Pressures Among Patients with Metabolic Diseases: A Systematic Review and Meta-analysis of Randomized Controlled Trials. High Blood Press. Cardiovasc. Prev. 2018, 25, 41–50.
- Dai, Y.L.; Luk, T.H.; Yiu, K.H.; Wang, M.; Yip, P.M.C.; Lee, S.W.L.; Li, S.W.; Tam, S.; Fong, B.; Lau, C.P.; et al. Reversal of mitochondrial dysfunction by coenzyme Q10 supplement improves endothelial function in patients with ischaemic left ventricular systolic dysfunction: A randomized controlled trial. Atherosclerosis 2011, 216, 395–401.
- Lim, S.C.; Lekshminarayanan, R.; Goh, S.K.; Ong, Y.Y.; Subramaniam, T.; Sum, C.F.; Ong, C.N.; Lee, B.L. The effect of coenzyme Q10 on microcirculatory endothelial function of subjects with type 2 diabetes mellitus. Atherosclerosis 2008, 196, 966–969.
- Hamilton, S.J.; Chew, G.T.; Watts, G.F. Coenzyme Q10 improves endothelial dysfunction in statin-treated type 2 diabetic patients. Diabetes Care 2009, 32, 810–812.
- Borghi, C.; Cicero, A.F.G. Nutraceuticals with a clinically detectable blood pressure-lowering effect: A review of available randomized clinical trials and their meta-analyses. Br. J. Clin. Pharmacol. 2017, 83, 163–171.
- Cicero, A.F.G.; Fogacci, F.; Colletti, A. Commentary to: “The Effects of Coenzyme Q10 Supplementation on Blood Pressures Among Patients with Metabolic Diseases: A Systematic Review and Meta-analysis of Randomized Controlled Trials”. Hig. Blood Press. Cardiovasc. Prev. 2018, 25, 51–52.
- Paglialunga, S.; Ludzki, A.; Root-McCaig, J.; Holloway, G.P. In adipose tissue, increased mitochondrial emission of reactive oxygen species is important for short-term high-fat diet-induced insulin resistance in mice. Diabetologia 2015, 58, 1071–1080.
- Anderson, E.J.; Lustig, M.E.; Boyle, K.E.; Woodlief, T.L.; Kane, D.A.; Lin, C.T.; Price, J.W.; Kang, L.; Rabinovitch, P.S.; Szeto, H.H.; et al. Mitochondrial H2O2 emission and cellular redox state link excess fat intake to insulin resistance in both rodents and humans. J. Clin. Investig. 2009, 119, 573–581.
- Fazakerley, D.J.; Chaudhuri, R.; Yang, P.; Maghzal, G.J.; Thomas, K.C.; Krycer, J.R.; Humphrey, S.J.; Parker, B.L.; Fisher-Wellman, K.H.; Meoli, C.C.; et al. Mitochondrial CoQ deficiency is a common driver of mitochondrial oxidants and insulin resistance. ELife 2018, 7, e32111.
- Raygan, F.; Rezavandi, Z.; Dadkhah Tehrani, S.; Farrokhian, A.; Asemi, Z. The effects of coenzyme Q10 administration on glucose homeostasis parameters, lipid profiles, biomarkers of inflammation and oxidative stress in patients with metabolic syndrome. Eur. J. Nutr. 2016, 55, 2357–2364.
- Saboori, S.; Rad, E.Y.; Mardani, M.; Khosroshahi, M.Z.; Nouri, Y.; Falahi, E. Effect of Q10 supplementation on body weight and body mass index: A systematic review and meta-analysis of randomized controlled clinical trials. Diabetes Metab. Syndr. 2019, 13, 1179–1185.
- Araújo, A.R.; Rosso, N.; Bedogni, G.; Tiribelli, C.; Bellentani, S. Global epidemiology of non-alcoholic fatty liver disease/non-alcoholic steatohepatitis: What we need in the future. Liver Int. 2018, 38, 47–51.
- Buzzetti, E.; Pinzani, M.; Tsochatzis, E.A. The multiple-hit pathogenesis of non-alcoholic fatty liver disease (NAFLD). Metabolism 2016, 65, 1038–1048.
- Chen, Z.; Tian, R.; She, Z.; Cai, J.; Li, H. Role of oxidative stress in the pathogenesis of nonalcoholic fatty liver disease. Free Radic. Biol. Med. 2020, 315–321.
- Hernández-Alvarez, M.I.; Sebastian, D.; Vives, S.; Ivanova, S.; Bartoccioni, P.; Kakimoto, P.; Plana, N.; Veiga, S.R.; Hernández, V.; Vasconcelos, N.; et al. Deficient Endoplasmic Reticulum-Mitochondrial Phosphatidylserine Transfer Causes Liver Disease. Cell 2019, 177, 881–895.
- Mourier, A.; Motori, E.; Brandt, T.; Lagouge, M.; Atanassov, I.; Galiner, A.; Rappl, G.; Brodesser, S.; Hultenby, K.; Dieterich, C.; et al. Mitofusin 2 Is Required to Maintain Mitochondrial Coenzyme Q Levels. J. Cell Biol. 2015, 208, 429–442.
- Cicero, A.F.G.; Colletti, A.; Bellentani, S. Nutraceutical Approach to Non-Alcoholic Fatty Liver Disease (NAFLD): The Available Clinical Evidence. Nutrients 2018, 10, 1153.
- Chen, K.; Chen, X.; Xue, H.; Zhang, P.; Fang, W.; Chen, X.; Ling, W. Coenzyme Q10 attenuates high-fat diet-induced non-alcoholic fatty liver disease through activation of the AMPK pathway. Food Funct. 2019, 10, 814–823.
- Moazen, M.; Mazloom, Z.; Dabbaghmanesh, M.H.; Ahmadi, A. Effect of CoQ10 supplementation on blood pressure, inflammation, and lipid profile in type 2 diabetics. Iran. J. Nutr. Sci. Food Technol. 2013, 8, 145–153.
- Pala, R.; Orhan, C.; Tuzcu, M.; Sahin, N.; Ali, S.; Cinar, V.; Atalay, M.; Sahin, K. Coenzyme Q10 Supplementation Modulates NFκB and Nrf2 Pathways in Exercise Training. J. Sports Sci. Med. 2016, 15, 196–203.
- Tiefenbach, J.; Magomedova, L.; Liu, J.; Reunov, A.A.; Tsai, R.; Eappen, N.S.; Jockusch, R.A.; Nislow, C.; Cummins, C.L.; Krause, H.M. Idebenone and coenzyme Q10 are novel PPARα/γ ligands, with potential for treatment of fatty liver diseases. Dis. Model. Mech. 2018, 11.
- Farsi, F.; Mohammadshahi, M.; Alavinejad, P.; Rezazadeh, A.; Zarei, M.; Engali, K.A. Functions of Coenzyme Q10 Supplementation on Liver Enzymes, Markers of Systemic Inflammation, and Adipokines in Patients Affected by Nonalcoholic Fatty Liver Disease: A Double-Blind, Placebo-Controlled, Randomized Clinical Trial. J. Am. Coll. Nutr. 2016, 35, 346–353.
- Lee, B.J.; Tseng, Y.F.; Yen, C.H.; Lin, P.T. Effects of coenzyme Q10 supplementation (300 mg/day) on antioxidation and anti-inflammation in coronary artery disease patients during statins therapy: A randomized, placebo-controlled trial. Nutr. J. 2013, 12, 1–9.
- Gokbel, H.; Gergerlioglu, H.S.; Okudan, N.; Belviranli, M. Effects of coenzyme Q10 supplementation on plasma adiponectin, interleukin-6, and tumor necrosis factor-alpha levels in men. J. Med. Food 2010, 13, 216–218.
- Farhangi, M.A.; Alipour, B.; Jafarvand, E.; Khoshbaten, M. Oral coenzyme Q10 supplementation in patients with nonalcoholic fatty liver disease: Effects on serum vaspin, chemerin, pentraxin 3, insulin resistance and oxidative stress. Arch. Med. Res. 2014, 45, 589–595.
- Samimi, M.; Zarezade Mehrizi, M.; Foroozanfard, F.; Akbari, H.; Jamilian, M.; Asemi, Z. The effects of coenzyme Q10 supplementation on glucose metabolism and lipid profiles in women with polycystic ovary syndrome: A randomized, double-blind, placebo-controlled trial. Clin. Endocrinol. 2017, 86, 560–566.
- Rahmani, E.; Jamilian, M.; Samimi, M.; Zarezade Mehrizi, M.; Aghadavod, E.; Akbari, E.; Tamtaji, O.R.; Asemi, Z. The effects of coenzyme Q10 supplementation on gene expression related to insulin, lipid and inflammation in patients with polycystic ovary syndrome. Gynecol. Endocrinol. 2018, 34, 217–222.
- Izadi, A.; Shirazi, S.; Taghizadeh, S.; Gargari, B.P. Independent and Additive Effects of Coenzyme Q10 and Vitamin E on Cardiometabolic Outcomes and Visceral Adiposity in Women with Polycystic Ovary Syndrome. Arch. Med. Res. 2019, 50, 1–10.
- Ates, O.; Bilen, H.; Keles, S.; Hakan Alp, H.; Keleş, M.S.; Yıldırım, K.; Ondaş, O.; Pınar, L.C.; Civelekler, C.; Baykal, O. Plasma coenzyme Q10 levels in type 2 diabetic patients with retinopathy. Int. J. Ophthalmol. 2013, 6, 675–679.
- El-ghoroury, E.A.; Raslan, H.M.; Badawy, E.A.; El-Saaid, G.S.; Agybi, M.H.; Siam, I.; Salem, S.I. Malondialdehyde and coenzyme Q10 in platelets and serum in type 2 diabetes mellitus: Correlation with glycemic control. Blood Coagul. Fibrinolysis 2009, 20, 248–251.
- Yamashita, S.; Yamamoto, Y. Simultaneous detection of ubiquinol and ubiquinone in human plasma as a marker of oxidative stress. Anal. Biochem. 1997, 250, 66–73.
- Hasegawa, G.; Yamamoto, Y.; Zhi, J.G.; Yamasaki, M.; Yano, M.; Nakajima, T.; Fukui, M.; Yoshikawa, T.; Nakamura, N. Daily profile of plasma %CoQ10 level, a biomarker of oxidative stress, in patients with diabetes manifesting postprandial hyperglycaemia. Acta Diabetol. 2005, 42, 179–181.
- Huang, H.; Chi, H.; Liao, D.; Zou, Y. Effects of coenzyme Q10 on cardiovascular and metabolic biomarkers in overweight and obese patients with type 2 diabetes mellitus: A pooled analysis. Diabetes Metab. Syndr. Obes. 2018, 11, 875–886.
- Maheshwari, R.A.; Balaraman, R.; Sen, A.K.; Seth, A.K. Effect of coenzyme Q10 alone and its combination with metformin on streptozotocin-nicotinamide-induced diabetic nephropathy in rats. Indian J. Pharmacol. 2014, 46, 627–632.
- Maheshwari, R.; Balaraman, R.; Sen, A.K.; Shukla, D.; Seth, A. Effect of concomitant administration of coenzyme Q10 with sitagliptin on experimentally induced diabetic nephropathy in rats. Ren. Fail. 2017, 39, 130–139.
- Lee, S.K.; Lee, J.O.; Kim, J.H.; Kim, N.; You, G.; Moon, J.W.; Sha, J.; Kim, S.J.; Lee, Y.W.; Kang, H.J.; et al. Coenzyme Q10 increases the fatty acid oxidation through AMPK-mediated PPARalpha induction in 3T3-L1 preadipocytes. Cell Signal. 2012, 24, 2329–2336.
- Feige, J.N.; Gelman, L.; Michalik, L.; Desvergne, B.; Wahli, W. From molecular action to physiological outputs: Peroxisome proliferator activated receptors are nuclear receptors at the crossroads of key cellular functions. Prog. Lipid Res. 2006, 45, 120–159.
- Tsai, K.L.; Chen, L.H.; Chiou, S.H.; Chiou, G.Y.; Chen, Y.C.; Chou, H.Y.; Chen, L.K.; Chen, H.Y.; Chiu, T.H.; Tsai, C.S.; et al. Coenzyme Q10 suppresses oxLDL-induced endothelial oxidative injuries by the modulation of LOX-1-mediated ROS generation via the AMPK/PKC/NADPH oxidase signaling pathway. Mol. Nutr. Food Res. 2011, 55, S227–S240.
- Kaikkonen, J.; Nyyssönen, K.; Porkkala-Sarataho, E.; Poulsen, H.E.; Metsä-Ketelä, T.; Hayn, M.; Salonen, R.; Salonen, J.T. Effect of oral coenzyme Q10 supplementation on the oxidation resistance of human VLDL+LDL fraction: Absorption and antioxidative properties of oil and granule-based preparations. Free Radic. Biol. Med. 1997, 22, 1195–1202.
- Sarmiento, A.; Diaz-Castro, J.; Pulido-Moran, M.; Moreno-Fernandez, J.; Kajarabille, N.; Chirosa, I.; Guisado, I.M.; Javier Chirosa, L.; Guisado, R.; Ochoa, J.J. Short-term ubiquinol supplementation reduces oxidative stress associated with strenuous exercise in healthy adults: A randomized trial. Biofactors 2016, 42, 612–622.
- Sharifi, N.; Tabrizi, R.; Moosazadeh, M.; Mirhosseini, N.; Lankarani, K.B.; Akbari, M.; Chamani, M.; Kolahdooz, F.; Asemi, Z. The effects of coenzyme Q10 supplementation on lipid profiles among patients with metabolic diseases: A systematic review and meta-analysis of randomized controlled trials. Curr. Pharm. Des. 2018, 24, 2729–2742.
- Sahebkar, A.; Simental-Mendıa, L.E.; Stefanutti, C.; Pirro, M. Supplementation with coenzyme Q10 reduces plasma lipoprotein(a) concentrations but not other lipid indices: A systematic review and meta-analysis. Pharmacol. Res. 2016, 105, 198–209.
- Geovanini, G.R.; Libby, P. Atherosclerosis and inflammation: Overview and updates. Clin. Sci. 2018, 132, 1243–1252.
- Farsi, F.; Heshmati, J.; Keshtkar, A.; Irandoost, P.; Meri, A.; Akbari, A.; Jannani, L.; Morshedzadeh, N.; Vafa, M. Can Coenzyme Q10 Supplementation Effectively Reduce Human Tumor Necrosis Factor-α and interleukin-6 Levels in Chronic Inflammatory Diseases? A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Pharmacol. Res. 2019, 148, 104290.
- Fan, L.; Feng, Y.; Chen, G.C.; Qin, L.Q.; Fu, C.L.; Chen, L.H. Effects of coenzyme Q10 supplementation on inflammatory markers: A systematic review and meta-analysis of randomized controlled trials. Pharmacol. Res. 2017, 119, 128–136.
- Bozkurt, B. What Is New in Heart Failure Management in 2017? Update on ACC/AHA Heart Failure Guidelines. Curr. Cardiol. Rep. 2018, 20, 39.
- Jessup, M.; Marwick, T.H.; Ponikowski, P.; Voors, A.A.; Yancy, C.W. 2016 ESC and ACC/AHA/HFSA heart failure guideline update—What is new and why is it important? Nat. Rev. Cardiol. 2016, 13, 623–628.
- Liu, L.; Eisen, H.J. Epidemiology of heart failure and scope of the problem. Cardiol. Clin. 2014, 32, 1–8.
- Mozaffarian, D.; Benjamin, E.J.; Go, A.S.; Arnett, D.K.; Blaha, M.J.; Cushman, M.; Das, S.R.; de Ferranti, S.; Després, J.P.; Fullerton, H.J.; et al. American Heart Association Statistics Committee; Stroke Statistics Subcommittee. Heart Disease and Stroke Statistics-2016 Update: A Report from the American Heart Association. Circulation 2016, 133, e38–e360.
- Maggioni, A.P. Epidemiology of Heart Failure in Europe. Heart Fail. Clin. 2015, 11, 625–635.
- Meyer, S.; Brouwers, F.P.; Voors, A.A.; Hillege, H.L.; de Boer, R.A.; Gansevoort, R.T.; van der Harst, P.; Rienstra, M.; van Gelder, I.C.; van Veldhuisen, D.J.; et al. Sex differences in new-onset heart failure. Clin. Res. Cardiol. 2015, 104, 342–350.
- Kannel, W.B. Incidence and epidemiology of heart failure. Heart Fail. Rev. 2000, 5, 167–173.
- Florkowski, C.M.; Molyneux, S.L.; Young, J.M. Coenzyme Q10 and congestive heart failure: An evolving evidence base. Kardiol. Polska. 2015, 73, 73–79.
- Folkers, K.; Vadhanavikit, S.; Mortensen, S.A. Biochemical rationale and myocardial tissue data on the effective therapy of cardiomyopathy with coenzyme Q10. Proc. Natl. Acad. Sci. USA 1985, 82, 901–904.
- Kitamura, N.; Yamaguchi, A.; Otaki, M.; Sawatani, O.; Minoji, T.; Tamura, H.; Atobe, M. Myocardial tissue level of coenzyme Q10 in patients with cardiac failure. Biomed. Clin. Asp. Coenzyme Q. 1984, 4, 221–229.
- Judy, W.V.; Stogsdill, W.W.; Folkers, K. Myocardial preservation by therapy with coenzyme Q10 during heart surgery. Clin. Investig. 1993, 71, 155–161.
- Weber, C.; Bysted, A.; Hłlmer, G. The coenzyme Q10 content of the average Danish diet. Int. J. Vitam. Nutr. Res. 1997, 67, 123–129.
- Onur, S.; Niklowitz, P.; Jacobs, G.; Lieb, W.; Menke, T.; Döring, F. Association between serum level of ubiquinol and NT-proBNP, a marker for chronic heart failure, in healthy elderly subjects. Biofactors 2015, 41, 35–43.
- Mortensen, S.A.; Rosenfeldt, F.; Kumar, A.; Dolliner, P.; Filipiak, K.J.; Pella, D.; Alehagen, U.; Steurer, G.; Littarru, G.P. Q-SYMBIO Study Investigators. The effect of coenzyme Q10 on morbidity and mortality in chronic heart failure: Results from Q-SYMBIO: A randomized double-blind trial. JACC Heart Fail. 2014, 2, 641–649.
- Lei, L.; Liu, Y. Efficacy of coenzyme Q10 in patients with cardiac failure: A meta-analysis of clinical trials. BMC Cardiovasc. Disord. 2017, 17, 196.
- Fotino, A.D.; Thompson-Paul, A.M.; Bazzano, L.A. Effect of coenzyme Q10 supplementation on heart failure: A meta-analysis. Am. J. Clin. Nutr. 2013, 97, 268–275.
- Sander, S.; Coleman, C.I.; Patel, A.A.; Kluger, J.; White, C.M. The impact of coenzyme Q10 on systolic function in patients with chronic heart failure. J. Card Fail. 2006, 12, 464–472.
- Belardinelli, R.; Mucaj, A.; Lacalaprice, F.; Solenghi, M.; Principi, F.; Tiano, L.; Littarru, G.P. Coenzyme Q10 improves contractility of dysfunctional myocardium in chronic heart failure. Biofactors 2005, 25, 137–145.
- Munkholm, H.; Hansen, H.H.; Rasmussen, K. Coenzyme Q10 treatment in serious heart failure. Biofactors 1999, 9, 285–289.
- Keogh, A.; Fenton, S.; Leslie, C.; Aboyoun, C.; Macdonald, P.; Zhao, Y.C.; Bailey, M.; Rosenfeldt, F. Randomised double-blind, placebo-controlled trial of coenzyme Q, therapy in class II and III systolic heart failure. Heart Lung Circ. 2003, 12, 135–141.
- Langsjoen, P.H. Lack of effect of coenzyme Q on left ventricular function in patients with congestive heart failure. J. Am. Coll. Cardiol. 2000, 35, 816–817.
- Soja, A.M.; Mortensen, S.A. Treatment of congestive heart failure with coenzyme Q10 illuminated by metaanalysis of clinical trials. Mol. Asp. Med. 1997, 18, 159–168.
- Swedberg, K.; Hoffman-Bang, C.; Rehnqvist, N.; Astrom, H. Coenzyme Q10 as adjunctive in treatment of congestive heart failure. J. Card Fail. 1995, 1, 101–107.
- Shi, H.; Noguchi, N.; Niki, E. Dynamics of antioxidant action of ubiquinol: A reappraisal. Biofactors 1999, 9, 141–148.
- Morisco, C.; Trimuco, B.; Condorelh, M. Effect of coenzyme therapy in patients with congestive heart failure: A long term multicentre randomized study. Clin. Investig. 1993, 71, S134–S136.
- Mortensen, S.A.; Leth, A.; Agner, E.; Rohde, M. Coenzyme Q10: Clinical benefits with biochemical correlates suggesting a scientific breakthrough in the management of chronic heart failure. Int. J. Tissue React. 1990, 12, 155–162.
- Beyer, R. An analysis of coenzyme Q in free radical generation and as an antioxidant. Biochem. Cell Biol. 1992, 70, 390–403.
- Niibori, K.; Wroblewski, K.P.; Yokoyama, H.; Juan, A.; Crestanello, J.A.; Whitman, G.J.R. Bioenergetic effect of liposomal coenzyme Q10 on myocardial ischaemia reperfusion injury. Biofactors 1999, 9, 307–313.
- Cohn, J.N.; Ferrari, R.; Sharpe, N. Cardiac remodelling. Concepts and clinical implications: A consensus paper from an international forum on cardiac remodeling. Behalf of an International Forum on Cardiac Remodeling. J. Am. Coll. Cardiol. 2000, 35, 569–582.
- Ulla, A.; Mohamed, M.K.; Sikder, B.; Rahman, A.T.; Sumi, F.A.; Hossain, M.; Mahmud, H.; Rahman, G.M.S.; Alam, M.A. Coenzyme Q10 prevents oxidative stress and fibrosis in isoprenaline induced cardiac remodeling in aged rats. BMC Pharmacol. Toxicol. 2017, 18.
- Singh, R.B.; Niaz, M.A.; Rastogi, S.S.; Sharma, J.P.; Kumar, R.; Bishnoi, I.; Beegom, R. Plasma levels of antioxidant vitamins and oxidative stress in patients with suspected acute myocardial infarction. Acta Cardiol. 1994, 49, 411–452.
- Grech, E.D.; Jackson, M.; Ramsdale, D.R. Reperfusion injury after acute myocardial infarction. Br. Med. J. 1995, 310, 477–478.
- Singh, R.B.; Fedacko, J.; Mojto, V.; Pella, D. Coenzyme Q10 Modulates Remodeling Possibly by Decreasing Angiotensin-Converting Enzyme in Patients with Acute Coronary Syndrome. Antioxidants 2018, 7, 99.
- Dhalla, A.K.; Hill, M.; Singal, P.K. Role of oxidative stress in the transition of hypertrophy to heart failure. J. Am. Coll. Cardiol. 1996, 28, 506–514.
- Senior, R.; Basu, S.; Kinsey, C.; Schaeffer, S.; Lahiri, A. Carvidilol prevents remodeling in patients with left ventricular dysfunction after acute myocardial infarction. Am. Heart J. 1999, 137, 646–652.
- Khaper, N.; Singal, P.K. Effects of after load reducing drugs on the pathogenesis of antioxidant changes and congestive heart failure in rats. J. Am. Coll. Cardiol. 1997, 219, 856–861.
- Wang, T.J.; Larson, M.G.; Levy, D.; Vasan, R.S.; Leip, E.P.; Wolf, P.A.; D’Agostino, R.B.; Kannel, W.B.; Benjamin, E.J. Temporal relations of atrial fibrillation and congestive heart failure and their joint influence on mortality: The Framingham Heart Study. Circulation 2003, 107, 2920–2925.
- Maisel, W.H.; Stevenson, L.W. Atrial fibrillation in heart failure: Epidemiology, pathophysiology, and rationale for therapy. Am. J. Cardiol. 2003, 91, 2D–8D.
- Hynes, B.J.; Luck, J.C.; Wolbrette, D.L.; Bhatta, L.; Khan, L.; Samii, S.; Naccarelli, G.V. Atrial fibrillation in patients with heart failure. Curr. Opin. Cardiol. 2003, 18, 32–38.
- Zozina, V.I.; Covantev, S.; Goroshko, O.A.; Krasnykh, L.M.; Kukes, V.G. Coenzyme Q10 in Cardiovascular and Metabolic Diseases: Current State of the Problem. Curr. Cardiol. Rev. 2018, 14, 164–174.
- Kumar, A.; Kaur, H.; Devi, P.; Mohan, V. Role of coenzyme Q10 (CoQ10) in cardiac disease, hypertension and Meniere-like syndrome. Pharmacol. Ther. 2009, 124, 259–268.
- de Frutos, F.; Gea, A.; Hernandez-Estefania, R.; Rabago, G. Prophylactic treatment with coenzyme Q10 in patients undergoing cardiac surgery: Could an antioxidant reduce complications? A systematic review and meta-analysis. Interact. Cardiovasc. Thorac. Surg. 2015, 20, 254–259.
- Zhao, Q.; Kebbati, A.H.; Zhang, Y.; Tang, Y.; Okello, E.; Huang, C. Effect of coenzyme Q10 on the incidence of atrial fibrillation in patients with heart failure. J. Investig. Med. 2015, 63, 735–739.
- Senes, M.; Erbay, A.R.; Yilmaz, F.M.; Topkaya, C.; Zengi, O.; Dogan, M.; Yucel, D. Coenzyme Q10 and high-sensitivity C-reactive protein in ischemic and idiopathic dilated cardiomyopathy. Clin. Chem. Lab. Med. 2008, 46, 382–386.
- Manzoli, U.; Rossi, E.; Littarru, G.P.; Frustaci, A.; Lippa, S.; Oradei, A.; Aureli, V. Coenzyme Q10 in dilated cardiomyopathy. Int. J. Tissue React. 1990, 12, 173–178.
- Soongswang, J.; Sangtawesin, C.; Durongpisitkul, K. The effect of coenzyme Q10 on idiopathic chronic dilated cardiomyopathy in children. Pediatr. Cardiol. 2005, 26, 361–366.
- Kocharian, A.; Shabanian, R.; Rafiei-Khorgami, M.; Kiani, A.; Heidari-Bateni, G. Coenzyme Q10 improves diastolic function in children with idiopathic dilated cardiomyopathy. Cardiol. Young 2009, 19, 501–506.
- Langsjoen, P.H.; Langsjoen, A.; Willis, R.; Folkers, K. Treatment of hypertrophic cardiomyopathy with coenzyme Q10. Mol. Asp. Med. 1997, 18, S145–S151.
- Adarsh, K.; Kaur, H.; Mohan, V. Coenzyme Q10 (CoQ10) in isolated diastolic heart failure in hypertrophic cardiomyopathy (HCM). Biofactors 2008, 32, 145–149.
- Conklin, K.A. Coenzyme Q10 for prevention of anthracycline-induced cardiotoxicity. Integr. Cancer Ther. 2005, 4, 110–130.
- Greenlee, H.; Shaw, J.; Lau, Y.I.; Naini, A.; Maurer, M. Lack of effect of coenzyme Q10 on doxorubicin cytotoxicity in breast cancer cell cultures. Integr. Cancer Ther. 2012, 11, 243–250.
- Mustafa, H.N.; Hegazy, G.A.; Awdan, S.A.E.; AbdelBaset, M. Protective role of COQ10 or L-carnitine on the integrity of the myocardium in doxorubicin induced toxicity. Tissue Cell 2017, 49, 410–426.
- Moskowitz, M.A.; Lo, E.H.; Iadecola, C. The science of stroke: Mechanisms in search of treatments. Neuron 2010, 67, 181–198.
- Kleinig, T.J.; Vink, R. Suppression of inflammation in ischemic and hemorrhagic stroke: Therapeutic options. Curr. Opin. Neurol. 2009, 22, 294–301.
- Simani, L.; Ryan, F.; Hashemifard, S.; Hooshmandi, E.; Madahi, M.; Sahraei, Z.; Razaei, O.; Heydari, K.; Ramezami, M. Serum Coenzyme Q10 is associated with clinical neurological outcomes in acute stroke patients. J. Mol. Neurosci. 2018, 31, 1–6.
- Allen, C.; Bayraktutan, U. Oxidative stress and its role in the pathogenesis of ischaemic stroke. Int. J. Stroke 2009, 4, 461–470.
- Nasoohi, S.; Simani, L.; Khodagholi, F.; Nikseresht, S.; Faizi, M.; Naderi, N. Coenzyme Q10 supplementation improves acute outcomes of stroke in rats pretreated with atorvastatin. Nutr. Neurosci. 2017, 22, 264–272.
- Liu, G.; Geng, J. Glial fibrillary acidic protein as a prognostic marker of acute ischemic stroke. Hum. Exp. Toxicol. 2018, 37, 1048–1053.
- Sharpe, P.C.; Mulholland, C.; Trinick, T. Ascorbate and malondialdehyde in stroke patients. Ir. J. Med. Sci. 1994, 163, 488–491.
- Milanlioglu, A.; Aslan, M.; Ozkol, H.; Çilingir, V.; Nuri Aydın, M.; Karadas, S. Serum antioxidant enzymes activities and oxidative stress levels in patients with acute ischemic stroke: Influence on neurological status and outcome. Wien. Klin Wochenschr. 2016, 128, 169–174.
- Ramezani, M.; Sahraei, Z.; Simani, L.; Heydari, K.; Shahidi, F. Coenzyme Q10 supplementation in acute ischemic stroke: Is it beneficial in short-term administration? Nutr. Neurosci. 2018, 1–6.





