
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Natália Cruz-Martins | + 8296 word(s) | 8296 | 2021-05-26 08:41:16 | | | |
| 2 | Conner Chen | Meta information modification | 8296 | 2021-06-01 11:18:56 | | |
Video Upload Options
The phytochemical constituents present in conifer extracts are nontoxic at therapeutic levels, with polyphenolic compounds having significant biological activities. Stilbenes, terpenes, alkaloids, lignins and flavanoids, such as quercetin, rutin, resveratrol, and the compounds PYC and enzogenol, are the phytochemical components of conifer extracts reported to have sedative, antidiabetic, anticancer and anesthetic effects. In addition, phytochemicals present in conifer extracts assist in the regulation of glucose and lipid metabolism, insulin secretion, stimulating β cells, the NF-kB signaling pathway, the inhibition of gluconeogenic enzymes, ROS protective action as well as targeting and modulating cytokines which affect neuron cells and reduce oxidative stress.
1. Background
Medicinal plants have long been used as a source for traditional remedies in nearly all cultures [1]. Nature provides an endless supply of novel phytochemicals, which are referred to as natural products (NPs), and natural product drug development is a difficult task for developing new leads [2]. Traditional medicines (TMs) are valuable because they use natural products; for example, Ayurveda, Kampo, traditional Chinese medicine (TCM), traditional Korean medicine (TKM) and Unani use natural products and have been practiced for thousands of years, blossoming into well-regulated medical systems [3]. As time passed and medication progressed, synthetic drugs, such as enoxaparin, aspirin, warfarin, ibuprofen, naproxen, clopidogrel and diclofenac, became available over the counter and were linked to mild (headaches, back pain) to severe side effects (difficulty breathing, excessive bleeding, and hemorrhage) [4]. There are no doubts that the use of natural products has markedly improved certain forms of cancer, diabetes, hypertension, pain, memory deficit, Alzheimer’s disease (AD), and migraine [5], and their further use should be continued in order to meet the urgent need for effective drugs to treat human diseases [6].
Coniferous plants, such as plants belonging to the Taxus, Cupressus, Picea, Pinus, Cedrus, Araucaria genera, are found worldwide and have shown several beneficial activities against diseases, highlighting the importance of conifers in drug development [7]. Conifers are woody, have needle-shaped single-veined leaves, and consist of male and female unisexual cones with bract scales [8]. They comprise eight families (Pinaceae, Araucariaceae, Cupressaceae, Podocarpaceae, Cephalotaxaceae, Taxaceae, Phyllocladaceae, Sciadopityaceae), 70 genera, and 630 species [9]. A number of genera include a vast number of species, such as Pinus (110), Podocarpus (105), Juniperus (55), Abies (50), Picea (35), Dacrydium (21), Taxodium (29) Pseudotsuga (22), Agathis (22), Araucaria (19), and Taxus (19), whereas there are some genera which contain a lower number of species, including Larix (10), Cedrus (4), Torreya (6) and Cryptomeria (1) [10]. They can be found in abundance in tropical lowland and submontane forests (Figure 1).
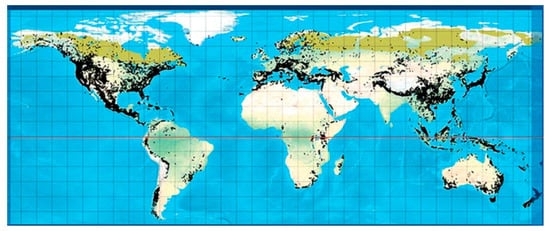
Figure 1. Global distribution of all conifers [11].
They contain secondary metabolites that combat pathogens and activate the plant’s defense mechanism [12]. The three major phytochemical groups, viz. terpenoids (resin acids and terpenes), alkaloids (piperidines) and polyphenols (phenolic acids, flavonoids, proanthocyanidins, lignans, acetophenones, and stilbenes) [13], present in the species of conifer trees are shown in Table 1, and their phytochemical compounds’ structures are shown in Figure 2. It is very important to understand the evolutionary pathway of Gymnosperms, in accordance with Sporne’s proposal, wherein the conifers represent the core, and the teachings that we can receive from it, comparing the morphological characters and their correlation with the chemical ones [14].
Table 1. Phytochemical constituents present in conifers.
| Nitrogen Compounds | |||
| Alkaloids | Vellosimine, 1,6-dehydropinidine, cis-pinidine, 1,6-dehydropinidinone, epipinidinone, cis-pinidinol, trans-pinidine, euphococcinine, α-pipecoline 1, (−)-pinidine | P. mariana, P. abies, P. sabiniana, P. torreyana, | [13][15][16] |
| Lignans | Lariciıesinol, taxiresinol, 3’-demethylisolariciresino1-9’-hydroxyisopropylethe, isolariciresinol, deoxypodophyllotoxin, (−)-secoisolariciresinol, 3, 3-demethylisolariciresinol, isotaxiresinol 2, α-conidendrin, (+)-pinoresinol, (−)-matairesinol, arctiin, dibenzylbutyrolactol, (−)-wikstromol, (−)-traxillagenin, (−)-arctigenin, traxillaside, 4′-deme-thyltraxillagenin, [(2R,3R)-2-(4′’-hydroxy-3′’-methoxybenzyl)-3-(4′-hydroxy-3′,5′dimethoxybenzyl)-butyrolactone] | T. baccata, J. taxifolia, J. sabina, J. virginiana, J. virginiana, P. roxburghii, Cedrus deodara, T. nucifera | [17][18][19][20][21][22][23] |
| Polyphenols: Flavonoids | |||
| Flavanonols | Taxifolin, cedeodarin | C. deodara, L. simbraca, P. roxburghii, P. mariana, P. abies, A. pindrow, A. excelsa; P. pinea, P. halepensis, P. pinaster, P. gerardiana | [21][24][25][26][27][28] |
| Flavones | Pilosanol B, luteolin, apigenin, apigenin 6-C-b-glucopyranoside | P. mariana, A. excelsa, P. abies, P. sylvestris, P. menziesii, P. menziesii, J. communis, A. angustifolia, L. deciduas | [15][26][27][29][30][31] |
| Biflavones | Bilobetin, cupressuflavone II-7-O-methyl-robustaflavone | T. wallichiana, C. macrocarpa, A. angustifolia | [31][32][33][34] |
| Flavonols | Quercetin, dihydroquercetin, rutin, kaempferol, dihydrokaempferol | J. communis, J. oxycedrus, P. gerardiana, P. roxburghii, P. wallichiana, A. angustifolia, P. abies, L. deciduas, P. sylvestris, P. menziesii, M. glyptostroboides, J. excelsa, P. mariana, J. foetidissima | [15][21][29][30][31][35][36][37][38] |
| Flavan-3-ols | Monomers: (−)-epicatechin, (−)-epicatechin-3-gallate, (+)-catechin, sennidin A, (−)-epigallocatechin, | P. pinaster, P. pinea, P. halepensis, P. roxburghii, P. wallichiana, P. gerardiana, J. foetidissima, A. angustifolia, P. abies, L. deciduas, P. sylvestris, J. communis, P. menziesii, J. oxycedrus, M. glyptostroboides, J. excelsa | [15][21][27][28][29][31][35][36][37][38] |
| Polymers: Procyanidin B1, B2, procyanidin A2, | P. halepensis, P. pinea, P. pinaster | [28] | |
| Phenolic acids | |||
| Benzoic acids | p-hydroxybenzoic acid, 2,5-dihydroxobenzoic acid, gallic acid, 4-hydroxybenzoic acid, protocatechuic acid, ellagic acid | P. abies, L. deciduas, P. sylvestris, P. menziesii, P. kesiya, J. communis, A. excelsa, P. roxburghii, P. wallichiana, P. gerardiana, L. deciduas, J. communis | [21][26][29] |
| Hydroxycinnamic acid | Caffeic acid, t-cinnamic Acid, p-coumaric acid, vanillic acid, ferulic acid, salicylic acid, sinapic acid, syringic acid, chlorogenic acid, 5-caffeoylquinic acid, caffeic acid 4-O-glucoside | P. abies, L. deciduas, P. sylvestris, T. baccata, P. mariana, P. pinaster, P. kesiya, L. deciduas, J. communis, P. menziesii, M. glyptostroboides | [15][39][27][29][30][37] |
| Stilbenes | trans-resveratrol, resveratrol, trans-pinosylvin, cis-stilbenes, pinosylvin, dihydro-monomethyl, trans-stilbenes, trans-piceatannol, trans-piceid, trans-isorhapontin, trans-isorhapontigenin, phenanthrenes, astringin, trans-astringin | P. mariana, P. abies, J. communis, P. pinaster, P. sylvestris, P. strobes, P. roxburghii, P. wallichiana, P. gerardiana, P. merkusii | [8][15][27][40][41][42][43][44][45][46] |
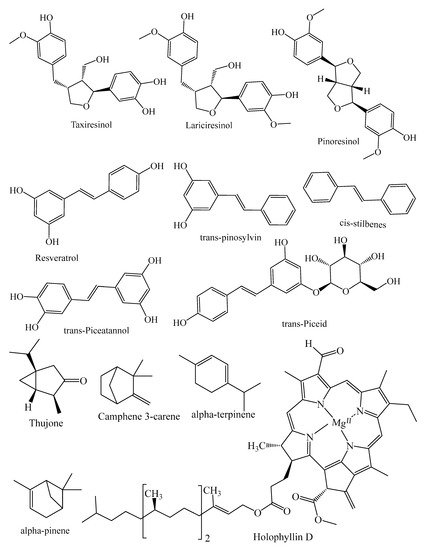
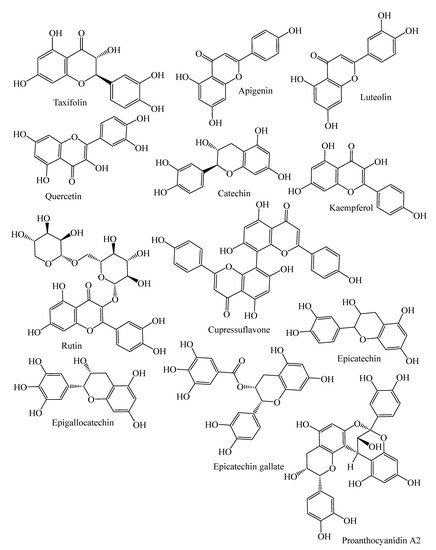
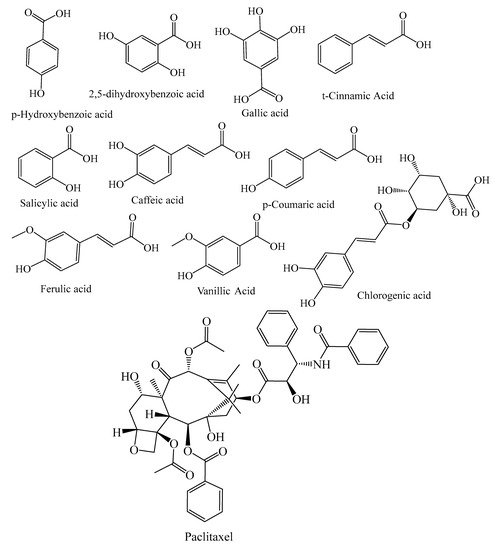
Figure 2. Structures of phytochemical compounds present in different conifer spp.
2. Conifers Phytochemicals Components
2.1. Terpenes
Terpenes are isoprenoids not containing nitrogen and sulfur and seem to be the main and largest group of natural phytochemicals group in conifers [12]. The terpenoids can be classified as C5 (hemiterpenes), C15 (sesquiterpenes), C20 (diterpenes), C10 (monoterpenes), C25 (sesterpenes), C40 (tetraterpenes), C40 (polyterpenes), and C30 (triterpenes) on the basis of C5 units [47]. Table 1 represents the most common mono-, sesqui- and diterpenes present in conifers. In particular, monoterpenes have been extensively studied, especially for their antiviral properties. Further, Porres-Martínez et al. (2016) reported their biological activities, including the anti-inflammatory, anticancer, antioxidant, and neuroprotective effects [48]. However, taxol diterpene derived from Taxus spp. have potential against malaria and cancer [47]. Sesquiterpenes have antiseptic, antimicrobial and disinfectant properties [49]. Kopaczyk et al. (2020) showed that the antioxidant activity of terpenes can prevail over oxidative stress aggravated by internal and external stimuli [12].
2.2. Alkaloids
There are several classes of alkaloids which are classified on the basis of the heterocyclic ring system and biosynthetic precursor which are of great interest. The alkaloids comprise quinolizidines, indoles, tropanes, pyrrolidines, pyrrolizidines, imidazoles, piperidines and isoquinoline purines [50]. There are numerous studies on the biological activity and medicinal uses of alkaloids [51]. In addition, alkaloids have been shown to have antitumor, anti-hyperglycemic and antibacterial activities [13]. Virjamo et al. (2020) reported that among the piperidine compounds of P. abies, only 1,6-dehydropinidine exhibited antibacterial effects by using a larger number of strains, whereas cis-pinidine was revealed to be toxic for vertebrates, which may only act in defense against herbivores [13].
2.3. Polyphenols
Polyphenols are of major relevance and perform a range of functions from skeletal constituents in various tissues to pigmentation in many plant organs [52]. They act as natural antioxidants, being able to inhibit lipid peroxidation, carcinogenesis, antimicrobial activity, direct capillary constrictive action, phytohormones, and have also the ability to stabilize ascorbic acid [53]. Flavonoids (isoflavones, flavonols, flavanonols, flavones, tannins, flavanones, anthocyanidins), stilbenes (resveratrol), phenolic acids (hydroxybenzoic and hydroxycinnamic acids), lignans, can all be found in plants [54]. Polyphenols, especially flavonoids, such as rutin, quercetin, apigenin, and epicatechin, are widely found in conifers. The genera Araucaria, Pinus, Cedrus, etc. are reported for their antimicrobial, anticancer, antidiabetic, neuroprotective [31] and anti-inflammatory properties and can be used in the treatment of neurodegenerative diseases, as well as being helpful in reducing αβ toxicity and neuronal dysfunction [55].
2.3.1. Flavonoids
Flavonoids are the most abundant phenol group in nature, present in a wide range of conifers [56]. Flavonoids have a central three-ring structure, but the different subclasses vary due to the centrally located heterocyclic ring structure (C-ring), which connects the two benzene rings [57]. To date, more than 6000 flavonoids have been recorded in several studies from plants. Flavonoids are aglycones in their basic structure, but most of them are glycosides in plants [58]. The subclasses of flavonoids found in the leaves, barks and seeds of conifers are represented in Table 1.
2.3.2. Lignans
Lignans are phenylpropanoids dimers made up of two coniferyl or sinapyl alcohol units bound together at the tails [59]. Isolariciresinol, taxiresinol, lariciresinol, pinoresinol, and their glycosides are examples of such compounds. There is a growing interest in lignans, especially because of their chemotherapeutic ability [60]. The most commonly present lignin compounds in conifer spp. are shown in Table 1.
2.3.3. Stilbenes
Stilbenes are produced by a number of conifer species, including Pinus sylvestris and Picea abies. Briefly, stilbenes are phenolic compounds with a heterologous bridge connecting two aromatic rings [61]. Many other compounds, such as trans-pinosylvin, cis-stilbene, resveratrol and piceatannol, have been isolated from the barks of conifer species (Table 1). For stilbenes, excellent antimicrobial effects have been reported [56].
2.3.4. Tannins
Tannins are polyphenolic compounds that can be in a wide range of plants. Tannins are colored pigments, astringent and are characterized by a bitter taste [62]. Tannin-rich conifer bark extracts have antimicrobial properties and high potential in preventing lipids from oxidation in the liposome model [63]. As a result, the tannins can be divided into four main classes based on their structural characteristics: gallotannins, complex tannins, ellagitannins and condensed tannins [64]. Condensed tannins (CTs), also well-known as proanthocyanidins, are prevalent in P. abies and P. sylvestris tree bark [62]. Condensed tannins are considered as polymers or oligomers of flavan3-ol units connected by C-C bonds that are hydrolysis resistant [65]. Procyanidins (PCs) and prodelphinidins (PDs) are the most popular PAs. Catechin and other epicatechin units make up PCs. Epigallocatechin units make up PDs [66].
Tannins exhibit antioxidant activity through various pathways, including free radical scavenging, transition metal chelation, and inhibition of pro-oxidative enzymes [67], besides having the capability to bind and form complexes with proteins and other compounds, and being responsible for their biological activity [68]. Tannins also act as antimicrobial agents, inhibiting extracellular microbial enzymes, depriving microbial growth substrates, and exerting a direct action on microbial metabolism, such as the denaturation of cell membrane proteins [69]. In the food industry, they could be used as functional coatings, adhesives, preservatives and as flavor compounds [56]. In a study, pine and spruce bark-derived PA-rich extracts revealed good potential for use in the food industry to develop preservative agents and to prevent lipid peroxidation in food items containing fatty-acids [63].
3. Traditional Medicinal Uses
Since prehistoric times, coniferous plants have been used as a medicinal source. Plant-based research has received more attention in recent years, and the literature supports the possible use of medicinal plants in conventional processes [70]. T. orientalis leaves and stems are utilized in traditional medicine to cure nervous system disorders, insomnia, heart palpitations, hemorrhage and fever. Fresh cedar leaves steeped for seven days in a 60% alcohol solution are often used by traditional Chinese physicians to encourage hair growth [71]. Cupressus spp. leaves, cones, stem bark have also been revealed to be useful in the treatment of hemorrhoids, bleeding varicose veins, asthma cough, spasms, diarrhea, rheumatism, common colds, piles, urinary tract ailments and vaginal discharge [7]. Different parts of the Pinus spp. bark, leaf, cone, and resin are also prescribed to treat cold-influenza, cough, tuberculosis, and bronchitis as a diaphoretic, rubefacient, antiseptic, diuretic, stimulant and febrifuge, while resin is also used in wound healing and injury [72]. The extract from Juniperus spp. leaves, berries and bark has also been used for the treatment of chronic eczema, hyperglycemia, obesity, tuberculosis, bronchitis, and pneumonia. The female cones, wood and leaves of J. foetidissima, J. communis and J. excelsa are used as a tonic for gout and rheumatism, a carminative, a diuretic, a treatment for urinary tract infection and stomach ache, an expectorant, a stimulant, an emmenagogue, and a treatment for the common cold [7].
Different parts of Taxus spp. have precise ethnomedicinal uses; for example, the leaves’ juice is used to cure cancer and bronchitis; bark juice and other parts, such as the leaf, are used for asthma and bronchitis, while trunk oil and cones are used to treat sheep diseases, bad breath, halitosis, inflammatory diseases of the lower urinary tract, renal stones, urinary infection, rheumatism dyspeptic complaints, hemorrhoids and cancer [7][73], and powdered dried leaves are considered to be effective in epilepsy, asthma, headache, diarrhea, bronchitis and hiccoughs [74]. A decoction developed from the bark is used to relieve pain from the muscles, knees, and rheumatoid arthritis, whereas a decoction made from the leaves is used to treat liver issues [75].
4. Conifers Extracts Rich in Phytochemical with Putative Health Effects
4.1. Oxidative Stress
The role of oxidative stress in the progression of degenerative ageing is well understood. Diabetes mellitus, coronary heart disease, cancer, inflammation, stroke, neurological conditions (e.g., AD), and ageing have all been linked to reactive oxygen species (ROS) generation [37]. Both enzymatic and non-enzymatic reactions are involved in the synthesis of ROS. ROS are known to be generated by enzymatic reactions present in many cell processes, including the respiratory chain system, prostaglandin synthesis and phagocytosis [76]. All ROS are produced through enzymatic action, including NADPH oxidase, xanthine oxidase and peroxidase in many cell processes, in whom the superoxide radical (O2●−) is generated [77]. Different ROS, formed during enzymatic reactions and with the action of enzymes (amino acid oxidase and xanthine oxidase), include hypochlorous acid (HOCl), hydrogen peroxide (H2O2), peroxynitrite (ONOO-), and hydroxyl radicals (OH●) [78]. The “Fenton reaction” between O2− and H2O2 takes place in the presence of Fe2+ or Cu+, which work as catalysts, producing OH, the most reactive free radical species [79]. The production of ROS has been related to non-enzymatic interactions between oxygen and organic compounds, as well as when cells are in contact with ionizing radiation during mitochondrial respiration [79][80]. Extensive research is currently needed to discover phytochemical compounds with the ability to boost the immune system and reduce oxidative stress [81]. The quest for new antioxidant molecules is a crucial part of promoting healthy ageing and combating oxidative stress [78]. Flavonoids, phenolic acids, vitamins and carotenoids are examples of natural compounds with antioxidant potential that have antagonistic effects on degenerative and inflammatory processes throughout the body, have beneficial effects on the immune and digestive systems, prevent ROS-related chronic problems and improve the overall quality of life, as shown in Figure 3 [73].
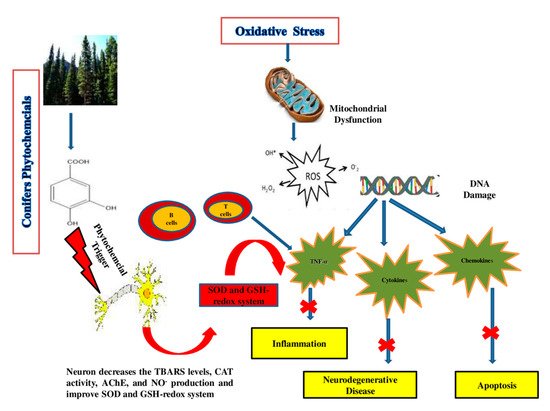
Figure 3. Action mechanism of conifers’ phytochemical compounds in oxidative stress, apoptosis, and neurodegenerative diseases. The phytochemicals’ multi-target effects in the brain include mitochondrial protection, anti-aggregation, anti-oxidant, anti-apoptotic and anti-inflammatory activity.
4.2. Synergism between ROS and other Diseases
The overproduction of ROS has been linked to a variety of chronic diseases, including cardiovascular, inflammatory and neurodegenerative diseases, and even cancer. The following sections explain on the relationship between ROS and chronic diseases.
4.2.1. Antioxidant Activity
In chronic obstructive pulmonary diseases (COPD), ROS play an important role [82]. The bark, wood, needle, and cone extracts of the Pinaceae family are high in polyphenolic compounds (Pycnogenol), primarily procyanidins, stilbenes, tannins and phenolic acids, and have significant antioxidant activity against ROS [83]. The antioxidant activity of conifer extracts has been confirmed by DPPH, FRAP and reducing power assays [40].
Flavonoids’ ability to scavenge free radicals can protect the human body from oxidative damage, which accelerates the ageing process [84]. Pycnogenol®, a polyphenol-rich compound extracted from the bark of P. pinaster, P. glauca, and P. mariana, has shown the ability to boost plasma antioxidant capacity and ameliorate pulmonary function and asthma traits [40][85]. In a study by Senthilmohan et al. (2003), 6–12 weeks of supplementation of Enzogenol®, and proanthocyanidin-rich flavonoid extracted from Pinus radiata bark in combination with vitamin C reduced DNA and protein oxidative damage in 55–75-year-old people [86]. In vivo studies have reported that the intake of quercetin alone did not protect DNA, but the combination of flavonoids (quercetin and myricetin) and isoflavonoids provides protection against DNA damage [87]. It has been found that the stilbenoid component resveratrol and piceatannol obtained from conifer spp. have more potent biological activities, namely as antioxidants [88]. Terpenoids and phenolic compounds, such as pinene, lycopene, camphene, gallocatechin, lutein, limonene and catechin found in the extract of conifer bark and needles have also been reported for their antioxidant potential by DPPH, FRAP, H2O2, ABTS assays, as shown in Table 2 [89].
4.2.2. Anti-Inflammatory Activity
When contagious microorganisms such as fungi, bacteria and viruses come into contact with the body, they remain in specific tissues and flow into the bloodstream, causing inflammation [90]. This also occurs as an end result of tissue damage, cancer, cell death, degeneration and ischemia [91][92][93]. In most cases, both the innate and adaptive immune responses are responsible for inflammation development [94]. The primary protection against invading foreign microbodies and cancer cells is the innate immune system, which involves macrophages, dendritic cells, and mast cells [90]. In the adaptive immune system, specialized cells (B and T cells) remove foreign pathogens and cancer cells by generating specific receptors and antibodies [95]. Cytokines such as interleukins, interferons, tumor necrosis factor, eicosanoids (leukotrienes and prostaglandins), modulatory inflammation-transcription nuclear factor (NF-ĸB) and chemokines (monocyte chemoattractant protein 1), are the inflammatory mediators and cellular pathways that have been extensively studied in relation to human pathological conditions [96]. Tumor necrosis factor-α (TNF) is a pro-inflammatory cytokine that is secreted by a variety of cells and has a variety of cellular effects [97]. It has also been linked to a variety of human illnesses, including cancer, mental and skin disorders, immune and inflammatory diseases. IL-1 is another cytokine that primarily has a pro-inflammatory effect [98]. It raises the levels of pro-inflammatory cytokines, including IL-1, TNF and IL-6 [99]. On the other hand, IL-1 has been linked to anti-inflammatory properties. Likewise, IL-1α and IL-6 originating from activated mast cells in the innate immune response also boost acute phase protein synthesis and thus show some anti-inflammatory effects [100]. The cytokine family members, including IL-12, IL-27, IL-23 and IL-35, function as a pro- and anti-inflammatory response [96][101][102]. On the other hand, IL-10 has been recognized as an effective anti-inflammatory cytokine, and helps in preventing several pro-inflammatory mediators from further action [103]. It protects tissue from homeostasis, defends against injury and damage caused by an overactive inflammatory response [103][104][105]. TNF-α accelerates PGE2 synthesis changes caused by edema and the flow of blood [34]. The extraction of plant materials is the first step in deciding the plant biological activities. The is a high probability of synergism between bioactive components when a whole extract is used, which could be lost if each and every component is isolated [106]. This form of synergism has been documented in numerous medical studies, generally for anti-inflammatory function [90]. There are different types of extraction and separation processes, such as:
1. Soxhlet extraction: The Soxhlet extraction method is a more efficient extraction method with high extraction yield and requires less solvent and time. This method requires electricity and solvents such as methanol, petroleum ether, and acetonitrile for the extraction process. However, sometimes high temperature and long extraction time enhance the possibility of thermal degradation and the loss of bioactive compound fraction activity [107].
2. Percolation: Extraction yield is better in percolation than maceration; in this process, pre-soaked plant material is added to a container, which allows the constantly controlled removal of the extract via a valve at the bottom and adding fresh solvent from the top.
3. Maceration: Maceration is carried out at room temperature by soaking the material with the solvent with eventual stirring. It has the advantage of moderate extraction conditions but suffers from high solvent consumption, long extraction times and low extraction yields. It could be used for the extraction of thermo labile components.
4. Ultrasound-assisted extraction: In UAE, the plant material, usually in a glass container, is covered by the extraction solvent and put into an ultrasonic bath. It decreases extraction time and improves extraction yields due to mechanical stress, which induces cavitation and cellular breakdown, and has gained increasing popularity [108]. For the isolation of extract from the solvent, the distillation process and many evaporators are used. After isolation to concentrate the extract, many researchers used a rotary evaporator, a normal air-drying process and distillation methods. Generally, to separate different solvent extracts, a separatory funnel is required [109].
Anti-inflammation is one of the main recorded effects of conifer phytochemicals among the numerous biological activities that have been studied so far. Table 3 reported the anti-inflammatory effect of conifer phytochemicals in in vivo and in vitro models.
Cupressuflavone (CUF) isolated from C. macrocarpa has the ability to reduce the levels of several cytokines, including IL-1b, IL-6, TNF-α and PGE2, in plasma dose-dependently, and thus acts as an anti-inflammatory agent [34]. Triterpenoids and abietane type’s compounds extracted from Abies chensiensis show anti-inflammatory effects against NO production in RAW 264.7 macrophage cells [110]. It has been found that tasumatrol, deacetylbaccatin, paclitaxel and many other terpenoids extracted from Taxus spp. are effective in the anti-inflammatory process initiated through the carrageenan and cotton pellets induced edema model [39][111]. Kyung-Jae Cha. (2016) reported that in atopic dermatitis, the Picea wilsonii mast extract is useful and potent only in the inhibition of the production of the inflammatory cytokines IL-6, MCP-1 and IL-13, without significant change in IL-8 production induced in human adult low-calcium high-temperature (HaCaT) cell lines [100]. T. occidentalis mother tincture-containing terpenoids (thujone), polyphenols and flavanoids have potential in reducing ulcerative colitis inflammation in the mouse intestine and rectum by decreasing the stimulation of the pro-inflammatory cytokines IL6 and TNF-α induced by 2,4,6-trinitrobenzenesulfonic acid (TNBS) [112]. THP-1 cell adhesion to TNF was suppressed by enzogenol at a concentration of 5–25 g/mL onto TNF-α-activated human umbilical vein endothelial cells (HUVEC) by reducing integrin β2 induction and inhibiting monocyte trans-endothelial migration [113]. The anti-inflammatory and platelet-inhibitory effects of pycnozenol, extracted from Pinus maritime bark extract, inhibited the activity of cyclooxygenase (COX)-1 and COX-2 present in human plasma [114]. Inflammation has been attributed to cancer and neurodegenerative diseases [34].
4.2.3. Anticancer Activity
According to the report by the World Health Organization, cancer was a major cause of death in 2018, with a death rate of 9.6 million people [115]. Hippocrates, before 370 B.C., coined the word “cancer” to describe carcinoma tumors [116]. On the basis of evidence, bone cancer was identified in Ancient Egyptian mummies in around 1600 B.C., and cancer of the breast was identified in 1500 B.C., although there is no record of a cure for cancer [117]. Giovanni’s research laid the foundation for scientific cancer techniques in 1761, when he performed the first autopsies on dead human bodies to determine the connection between a patient’s disease and their death, as well as pathologic studies [118]. Cancer has been identified as the chief matter of public health concern around the world [119]. Surgery, radiotherapy, and chemotherapy are some of the conventional cancer treatments [120][121]. On the other hand, despite the use of a variety of synthetic drugs for cancer treatment and the successful completion of different management schedules, current therapies are not able to achieve the desired results, as tumor relapse and metastasis are common [122]. Nature contains various chemicals and pharmacologically active substances which act as anticancer drugs [123]. Recently, many of the phytochemicals and synthetic analogs, such as HS-1793 (resveratrol), have been identified as inhibiting the growth of cells and inducing apoptotic cell death, helping to cure cancer [124]. While only a few phytochemical compounds obtained from natural products have been developed into clinically active drugs, their bioactive components may be used as a basis for the development of more successful analogues and prodrugs using chemical techniques such as metabolomics, complete or combinatorial fabrication, and biosynthetic pathway modification [125]. Many phytochemical compounds are highly efficient in inducing apoptosis and cytotoxicity by modulating different MAPK andAKT/PI3K pathways, and suppress cancer cells line invasion and migration potential along with the stimulation of senescence phenotype, regulation of Bax or p53 genes, cell cycle arrest and modulation of IL-8, IFN-γ, TNF, IL-6 [126]. Many of the compounds derived from conifers’ bark and leaves act as antitumor drugs, such as paclitaxel (PTX) (trade name Pycnogenol® and Taxol®), a diterpene found in the crude extract of P. pinaster and Taxus brevifolia bark [127]. Paclitaxel, as well as its analogues docetaxel (taxoteres) and jevtanas (cabazitaxel), are examples of chemotherapeutic synthetic analogues derivative from plants that have been formulated and validated clinically [125]. By binding microtubules, PTX and other microtubule-targeting agents (MTAs) induce cellular death [128]. Microtubules are tubulin heterodimers that play a role in disease and perform numerous cellular functions including transport, force production in cell division, and structural support [129]. During the G2 phase of the cell cycle, tubulin is produced, and microtubules are assembled. Microtubule stabilizing agents, such as PTX, bind to α/β tubulin in order to disassemble microtubules. As a result, they cause cell death and are used as an anti-cancer agent [130]. In general, cells exposed to PTX are stuck in the G2/M phase, resulting in death due to failure to move through the cell cycle [131][132]. Recently, a study published in 2017 found that pycnogenol and PTX at doses of 20 g/mL and 0.5 μM cause DNA and mitochondrial damage in cancer breast cell line (MDA-MB) in 24 h, and concluded that it is possibly a target drug for cancer treatment through DNA and mitochondrial damage mechanisms [127]. From different conifer species crude extracts, the anticancer activities on different cancer cell lines are summarized in Table 4.
Table 2. Antioxidant capacity of extracts obtained from different conifer spp.
| Conifer spp. | Part Used | Compounds | Nature of Extract | Radical Scavenging Assay | Dose/Concentration | Main Effects | References |
|---|---|---|---|---|---|---|---|
| Aurocaria cookii | Leaves | Phenolic compounds | Methanol, chloroform, petroleum ether | DPPH | 1000 μg/mL | Methanol extract shows the best antioxidant activity with 63% inhibition, higher than the other two compounds | [133] |
| A. excelsa | Needle | Flavanoids | Methanol | DPPH | 50–72.5 μg/mL | Methanol/water extract shows antioxidant activity | [26] |
| C. deodara | Heart wood | Tannins, flavonoids, and phenolic compounds | Water/alcohol | DPPH, superoxide radical-scavenging activity, ABTS | DPPH-IC50 (μg/mL): 61.89 (water extract), 75.79 (alcohol extract) superoxide radical-scavenging activity— IC50 (μg/mL): 87.76 (water extract), 121.55 (alcohol extract). ABTS-IC50 (μg/mL): 115.29 (water extract), 122.42 (alcohol extract). |
DPPH radical-scavenging activity and the reducing power of C. deodara were potent in water and alcohol extract | [134] |
| C. japonica | All parts | Phenolic compounds | Methanol | ORAC, SOD |
4.09–7.64 TE/mg 3.63–4.06μg/mL |
The methanol extracts from each part of C. japonica except for pollen showed strong activities in the bioactivity assays. | [135] |
| J. communis | Berry | Flavanoids (quercetin rutin, apigenin) chlorogenic acid |
Alcohol/Water | DPPH | EC50 1.42 mg/mL against standard Ascorbic acidEC50 value of 0.365 mg/mL | The antioxidant activity was confirmed as 81.63 ± 0.38% by the DPPH assay. | [30] |
| L. laricina | Bark | Phenolic compounds | Ethanol/Water | ORAC | IC50 0.878 μg/mL. | Bark extract of LL shows significant antioxidant activity | [40] |
| Metasequoia glyptostroboides | Cone | Terpenoid | Ethyl acetate | DPPH, NO, superoxide, and H2O2 | 5–250 μg /mL | Sugiol derived from cone extract show good antioxidant activity—78.38, 72.42, 74.45 and 85.04%, respectively. | [136] |
| Picea abies | Bark | Atilbenoids | Ethanol/Water | DPPH | 49.74 μg/mL | UVA-induced modification of the stilbene-rich inner bark extracts increased the antioxidant activity as UVA irradiation decreased the capacity of the extracts to prevent lipid oxidation in the liposome system method | [42] |
| P. smithiana | Leaves | Phenolic compounds | Methanol | DPPH | IC50 (μg/mL)- | Results of the DPPH radical scavenging activity and FRAP study determine that methanol extracts of leaf displayed the highest antiradical efficiency | [137] |
| 228 | |||||||
| FRAP | 494 | ||||||
| Reducing Power assay | 978 | ||||||
| Pinus gerardiana | Bark | Phenolic compounds | Ethanol | DPPH | IC50 value μg/mL | P. gerardiana shows promising H2O2 radical scavenging activity | [89] |
| 102.8 | |||||||
| H2O2 | 81.83 | ||||||
| NO2 | 109.2 | ||||||
| P. halepensis | Bark | Phenolic compounds | Ethanol/Water | IC50 (μg/mL). Ethanol and the water | Ethanol and water extract of bark exhibited significant free radical neutralization capacities, at conc. 0.5–8 μg/mL | [138] | |
| DPPH | 3.28, 3.26 | ||||||
| ABTS | 3.1, 3.59 | ||||||
| P. pinaster | Bark | Phenolic compounds | Ethanol/Water | PB (50%) and (90%) IC50 value μg/mL | PP bark extracts formed from PB 50% (50% ethanol) have maximum (DPPH, ABTS) radical scavenging activity while FRAP shows activity with (PB 90%) | [27] | |
| DPPH | 49.74 | ||||||
| ABTS | 59.41 | ||||||
| FRAP | 101.3 | ||||||
| P. roxburghii | Bark | Phenolic compounds | Ethanol | IC50 value μg/mL | Pine extract shows significant antioxidant activity | [89] | |
| DPPH | 97.54 | ||||||
| H2O2 | 86.90 | ||||||
| NO2 | 111.38 | ||||||
| P. wallichiana | Bark | Phenolic compounds | Ethanol | IC50 (μg/mL) | Pine extract shows significant radical scavenging activity | [89] | |
| DPPH | 111.40 | ||||||
| H2O2 | 84.18 | ||||||
| NO2 | 98.5 | ||||||
| Thuja occidentalis | Leaves | Flavonoids, phenols | Methanol | DPPH, FRAP | 20–100 μg/mL | Crude extract shows significant antioxidant activity | [139] |
| T. occidentalis | Non-woody branches with leaves |
Polyphenol, flavonoids | Mother tincture (MT) | DPPH, ORAC, NO | 25 or 50 mg/kg | T. occidentalis mother tincture displayed 88.3% antioxidant activity by DPPH and about 78% by NO assay | [112] |
| Taxus baccata | Leaves and cones |
Flavonoids, phenols | Methanol | DPPH | IC50 (μg/mL) 105.41, 518.51 leaves and cones resp. | Acetone and ethyl acetate extract of leaves show good scavenging activity | [140] |
| Water | DPPH | 533.66, >1000 leaves and cones resp. | |||||
| Acetone | DPPH | 25.24, 81.43 leaves and cones resp. | |||||
| Ethyl acetate | DPPH | 29.84, 180.26 leaves and cones resp. | |||||
| Petroleum ether | DPPH | 438.92, > 1000 leaves and cones resp. | |||||
| T. wallichiana | Leaves | Terpenoids, flavonoids | IC50 values (μg/mL) | The maximum DPPH activity was observed in methanol extract (91.25%), followed by water (87.64%), ethanol (85.23%), and ethyl acetate (83.27%) at the highest concentration (700μg/ml) | [141] | ||
| Methanol | Superoxide radical | 170.30 | |||||
| DPPH | 212.00 | ||||||
| LPO | 126.09 | ||||||
| Hydroxyl radical | 82.34 | ||||||
| Ethyl acetate | Superoxide radical | 297.55 | |||||
| DPPH | 301.80 | ||||||
| LPO | 151.96 | ||||||
| Hydroxyl radical | 199.05 | ||||||
| Water | Superoxide radical | 257.00 | |||||
| DPPH | 258.29 | ||||||
| Hydroxyl radical | 175.33 | ||||||
| T. wallichiana | Leaf, stem | Polyphenols, flavanoids, terpenoids | Methanol | DPPH FRAP |
IC50 value (μg/mL.) Leaves (23.18) Stem (56.75) |
DPPH and FRAP activity of TW leaves and stem extract have high antioxidant activities. | [142] |
PB-Pine bark; TW-Taxus wallichiana.
Table 3. Anti-inflammatory capacity of different conifers spp.
| Conifer spp. | Part Used | Nature of Extract | Compounds | Major Method(s) of Testing | Dose. Conc | Main Effect | References |
|---|---|---|---|---|---|---|---|
| Abies chensiensis | Twigs and leaves | Ethanol | Terpenoids | Induce lipopolysaccharide to produce inflammation in RAW 264.7 macrophage cells | 0.2–50.0 μM | 4 compounds—3α-hydroxyl-8,14,22Z,24-tetraenlanosta-26,23-olide; (5R,20R)-8(14→13R)-abeo-17,13-friedo-3-oxolanosta-8,14(30),22Z,24-tetraen-26,23-olide; 8,14,22Z,24-tetraen-3-oxolanosta-26,23-olide; and (23R, 25R)-3,4-seco-9β H-lanosta-4 (28),7-dien-16α-hydroxyl-26,23-olid-3-oate—extracted from extracts showed significant anti-inflammatory activities of inhibition against NO formation with IC50 value of 15.9, 18.7, 20.18, and 10.9 μM |
[110] |
| A. georgei | Aerial parts | Chloroform, ethyl acetate, n-butanol | Flavanoids | dimethylbenzene-induced ear oedema in mice | 200 mg/kg | AG ethyl acetate extract shows 18% inhibition against dimethylbenzene-induced ear edema in mice while carrageenin-induced paw edema in rats shows inhibition ratios 28.2% and 35.6%, after 2 and 6h, respectively. | [143] |
| Carrageenin-induced paw oedema rat | 140 mg/kg | ||||||
| A. webbiana | Leaves | Methanol/Petroleum ether extract | Flavanoids | Carrageenan-induced rat hind paw edema model in Albino mice | 400 mg/kg | Plant leaves extract possesses significant anti-inflammatory properties | [144] |
| Agathis robusta | Leaves | Methanol | Flavanoids, tannins and saponins | Heat induced hemolytic method in human red blood cell (HRBC) membrane | 400 μg/kg | Leaves extract shows good antiinflammatory activity | [145] |
| Cedrus deodara | Stem bark | Methanol | Deodarin, quercetin, taxifolin | Carrageenin-induced paw edema in Albino rat | 100 mg/kg | Anti-inflammatory activity with 43.47% inhibition | [146] |
| Cupressus macrocarpa | Leaves | Methanol | Cupressuflavone (CUF) | Carrageenan-induced paw edema model in Mice | 40, 80, and 160 mL/kg |
CUF demonstrated antiinflammatory activity by inhibiting paw edema with 55, 60, and 64%, by decreasing the plasma pro-inflammatory mediators PGE2, IL-6, TNF-a and IL-1b |
[34] |
| Juniperus communis | Berry | Alcohol/Water | Flavanoids (quercetin rutin, apigenin) chlorogenic acid | Acute-dextran and kaolin subacute inflammation induced in Wistar Rat | 10 mL/kg | The antiinflammatory action of the juniper extract, administered as a microemulsion in acute-dextran model was increased when compared to kaolin subacute inflammation induced model. | [30] |
| J. oxycedrus | Berry | Ethanol, n-butanol | Flavonoids (amentoflavone, cupressuflavone, hinokiflavone, and rutin) | Carrageenan-induced hind paw edema model in mice | 100 mg/kg | Ethanol extract of Joso berries displayed remarkable inflammatory inhibition ranging between 24.5% and 23.7% at 100 mg/kg in carrageenan-induced edema model | [147] |
| J. foetidissima | Berry | Ethanol | Flavonoids (amentoflavone, cupressuflavone, hinokiflavone, and rutin) | carrageenan-induced hind paw edema model in mice | 100 mg/kg | JFB extract at a dose of 100 mg/kg. shows high antiinflammatory effect 26.9% | [147] |
| Pinus gerardiana, P. roxburghii, P. wallichiana | Bark | Ethanol | Flavanoid, tannin | against albumin denaturation, HRBC membrane stabilization assay | 2500 μg/mL | P. roxburghii extract showed highest (%) of inhibition and protection i.e 86.54 and 89.92 against albumin denaturation and HRBC membrane stabilization. However, P. wallichiana have least inhibition and protection percentage, i.e., 76.54 and 81.2% | [89] |
| Taxus baccata | Aerial parts | Methanol | Terpenoids | ear edema induced in mice | 3.2 mg/ear | T. baccata extract displayed best activity | [39] |
| T. baccata | Bark | Ethanol | Alkaloids, terpenoids, flavonoids | carrageenan-induced paw edema in Wistar Albino rat | 200 mg/kg | Percentage of inhibition is 44% at a dose of 200 mg/kg | [148] |
| T. baccata | Heart wood | Ethanol | Taxoids, lignans | carrageenan-induced hind paw edema model inS wiss albino mice | 30–100 mg/kg | TBW shows significant antinociceptive and anti-inflammatory activities | [17] |
| T. wallichiana | Bark | Methanol | Tasumatrol B, 1,13-diacetyl-10-deacetylbaccatin III (10-DAD) and 4-deacetylbaccatin III (4-DAB) | carrageenan-induced paw edema and Cotton-pellet oedema model in Wistar rats and Swiss albino mice | 20 and 40 mg/kg; 40 mg/kg | In a carrageenan-induced inflammation model, tasumatrol B at a dose of 20 mg/kg showed significant activity, while in a cotton-pellet edema model tasumatrol B was found to be highly significant at the dose of 40 mg/kg. | [111] |
| Thuja occidentalis | Non-woody branches with leaves | Mother tincture (MT) | Polyphenols, flavonoids | Administered 2,4,6-trinitrobenzenesulfonic acid to induce intrarectal colitis in mice | 25 or 50 mg/kg | MT manage to relieve intestinal inflammation experimentally induce by TNBS in 7 days. | [112] |
JFB—Juniper foetidissima berry; AG—Abies georgei; TBW—Taxus baccata heart wood.
Table 4. In vivo and in vitro anticancer and cytotoxic studies of conifer extracts.
| Conifer spp. | Part Used | Nature of Extract | Compounds | In Vitro and in Vivo Model | Dose. Conc | Main Effects | References |
|---|---|---|---|---|---|---|---|
| Abies georgei | Aerial parts | Chloroform, ethyl acetate, n-butanol | Flavanoids | Human tumor cell lines-A549, QGY-7703, LOVO, 6T-CEM | 77.5, 11.1, 7.8, 32.8 μg/mL | AGC extract has potent tumour and antiproliferative effects in humor tumor cell lines | [143] |
| (Mice) S180 tumours cell lines | 100, 200 and 400 mg/kg | AGC also exhibited activity in tumour growth inhibition in a dose-dependent manner, with ratios of 46.7, 53.1 and 31.0% at doses of 100, 200 and 400 mg/kg, respectively | |||||
| Araucaria angustifolia | Female strobili | Water | Fatty acids and polyphenols | Laryngeal carcinoma HEp-2 cells | 100–500 μg/mL | AAE inhibit the activity of mitochondria complex I and induce redox stress and cytochrome c, which leads cleavage of nuclear proteins of larynx HEp-2 cancer cells | [149] |
| Cedrus deodara | Stem wood | Chloroform | Lignans (Matairesinol, dibenzylbutyrolactol, (−)-Wikstromol) | In vitro human cell lines (cervix, breast, colon, liver, CNS, prostrate) | In vitro cytotoxicity IC 50 value-Wikstromol (71.31–93.63) and Matairesinol (50.84–95.36) μg/mL | CD lignin mixture have potent to show a cytotoxic effect at the maximum in CNS and at the minimum in liver against cancer cell lines in a dose-dependent manner at 100 μg/mL from 49 to 95%. | [22] |
| Human T lymphoblast, acute lymphoblastic leukemia cell line, Molt-4 and human promyelocytic leukemia cell line (HL-60) | IC50 (μg/mL) 15 |
AP9-cd-induced endogenous NO production leads to the generation of peroxide and disruption of mitochondrial membrane potential, leading to apoptotic pathway activation Increase in sub-G0 fraction from 35 to 60% in 24 to 48h |
[150] | ||||
| In vivo swiss albino mice (K562 cells) | The lignin mixture displays anti-cancer effects by regulating annexin V binding, intracellular caspase activities and DNA fragmentation | ||||||
| C. deodara | Needle | Ethanol | Kaempferol, myricetin, isorhamnetin and quercetin | HepG2 cells | IC50 114.12 μg/mL | TFPNCD shows potent cytotoxicity by inhibiting the growth of HepG2 cells in a dose-dependent manner Regulates cell cycle and apoptosis |
[151] |
| Cryptomeria japonica | Leaves | Methanol | Flavonoids | Albino mice of Ehrlich Ascites Carcinoma (EAC). | 100–400 μg/gm | Tumor cell count as well as the amounts of ascetic tumour cells in packed cells were significantly reduced in infected mice treated with MC | [152] |
| Juniper communis | Berry | Methanol and water | Phenolic compounds | CaCo2 and HeLa carcinoma cell lines | IC50 1300–2500 μg/mL | Methanol and water extracts of JCB show potent antiproliferative activity against cancer cell lines | [153] |
| J. taxifolia | Leaves | Chloroform | Polyphenols and lignan | human leukemia (HL-60) cells | 2.5 μg/mL | 7α-hydroxysandaracopimaric acid, a diterpenoid compound obtained from J. taxifolia leaves, shows antitumor effects on HL-60 cells | [154] |
| J. phoenicea | Aerial parts | Chloroform | Polyphenols | IC50 values (μg/mL) | It is found that JPCF disrupts cell cycle progression in the G0/G1phase and shows apoptotic, antiproliferative and necrotic effects on cancer cells lines | [155] | |
| Human lung (A549) | 34.2 | ||||||
| Breast (MCF-7) | 24.5 | ||||||
| Liver (HepG2) cancer cells | 57.6 | ||||||
| J. foetidissima | Needle | Methanol | Quercetin, rutin | Rat brain tumor (C6) cell lines | IC50 values (μg/mL) 10.65 |
J. foetidissima needle extract showed significant antiproliferative activity | [38] |
| M. glyptostroboides | Leaf | Water | Polyphenols | PC12 cells | 25 μg/mL | M. glyptostroboides leaf extract shows a cytotoxic effect and prevents oxidative damage of neuronal PC12 cells, protecting them from apoptosis; it was also found to significantly inhibit the release of LDH, which may result from apoptosis or necrosis | [37] |
| Picea wilsonii | Whole plant | DMSO | ND | Human keratinocyte HaCaT cell lines | 1–3 g/mL | PwM extracts inhibit the production of MCP-1 IL-6, IL-13 and but do not inhibit IL-8 production | [100] |
| Pinus kesiya | Woody twig | Ethanol | Phenolic compounds and flavonoids | Human hepatocarcinoma (HepG2) cell lines | IC50 (μg/mL) 52.0 | PK Extract exhibited a potent cytotoxic effect in the HepG2 cell line | [156] |
| P. kesiya | Branch | Ethanol | Phenolic compounds and flavonoids | Human leukemic U937 cancer cells |
IC50: 299 μg/mL | PK ethanol extract possesses anticancer activity against U937 human leukemic cells via apoptosis | [157] |
| P. merkusii | Leaves | Methanol | Phenolic compounds | MCF-7, A549, HT 1080 and HepG2 Huh-7 cancer cell lines | IC50 (μg/mL) 4.5, 16, 4.1, 5.6, 9.5 |
PM methanol extract possesses anticancer activity against human cancer cell lines | [158] |
| T. baccata | Leaves, cones | Methanol | Phenolic compounds | HCT-116 human colon cancer and MDA-MB-231 human breast cancer cell lines | IC50 μg/mL Leaves: 14.43 and 4.59 cones: 49.69 and 133.53 |
Methanol extracts of leaves had better activity on HCT-116 cells than seed cone extract, with IC50 values of 14.3 for 24 h and 4.59 for 72 h. Meanwhile, extracts did not show any significant cytotoxic effects on the cancer cell lines | [140] |
| T. wallichiana | Heartwood | Methanol | Lignans 1 (taxiresinol 1) 2, 3 | colon, ovarian liver, and breast cancer cell lines | IC90 lignan 2 and 3 μg/mL Caco 2:0.08 and 0.056 and 0.251 |
Taxiresinol 1 shows anticancer activity against ovary, colon, liver and breast cancer cell lines, while lignans 2 and 3 were found to be most active against Caco-2 cell lines | [159] |
| T. yunnanensis | All parts | ND | α-Conidendrin | MCF-7 andMDA-MB-231 cancer cell lines | 40 μM | α-conidendrin have the potential to inhibit human breast cancer cell lines MDA-MB-231 and MCF-7, showing viability of 73 and 82%, respectively | [19] |
| P. roxburghii | Leaves | Water and ethanol | Phenolic compounds | A549 human lung cancer cell line | 111.2 and 112.7 μg/mL | PRL extract shows potent anticancer activity against cancer cell lines. | [160] |
| Taxus cuspidata | Branches and leaves | Water | Polysaccharides | MCF7 | IC50 μg/mL | Purified polysaccharides (Pe4) on HeLa cells had the highest inhibitory effect, and its IC50 value is 89.9, while (Pe1) shows the best cytotoxic capacity against cancer lines HepG2 and MCF7, with IC50 conc. 132.0 and 169.0 μg/mL, respectively | [161] |
| 169.0 | |||||||
| Hela | 89.9 | ||||||
| HepG2 | 132.0 | ||||||
| Thuja occidentalis | Leaves and non-woody branches | Mother tincture (MT) | Polyphenols including flavonoids | Caco-2 cells | 25 or 50 mg/kg | Caco-2 cells exposed to H2O2 and T. occidentalis MT proves its radical scavenging activity by reducing GSH level by 103% and 98% as compared to TNBS group; MT also managed to reduce the lipid peroxidation | [112] |
| T. occidentalis | Leaves | Ethanol | ND | Human NSCLC (A549) cell lines | IC50 μg/mL | Extract of TO shows both anticancer and antiproliferative activities against NSCLC (A549) cell lines in a dose-dependent manner. | [162] |
| 282 | |||||||
| Human normal embryonic cell lines (L-132) | 376 | ||||||
| T. occidentalis | ND | Mother tincture (MT) Thujone-rich fraction (TRF) |
Thujone | A375 human malignant melanoma cell line | 200 μg/mL | TRF as compared with TO MT on exposure to A375 cells exhibited highly cytotoxic, apoptotic and antiproliferative effects, but TRF shows a lower growth inhibitory response towards peripheral blood mononuclear cell (normal cells) | [163] |
ND—Not determined; AGC—Abies georgei chloroform extract; AAE—Araucaria angustifolia water extract; TFPNCD—total flavonoids from the pine needles of Cedrus deodara; PRL—P. roxburghii leaves.
4.2.4. Neurodegenerative Diseases
Neurodegenerative diseases (NDs) are more common among the elderly and may even lead to death, and so are a major threat in the 21st century [164]. AD, Parkinson’s disease (PD), Huntington’s disease (HD), amyotrophic lateral sclerosis (ALS), frontotemporal dementia, and the spinocerebellar ataxias are examples of ND [165], whose main features include nitrosative/oxidative stress, mitochondrial dysfunction, aggregated proteins accumulation, synapse loss, neuro-inflammation and decreased neuronal survival [166]. The progression of ND is also affected by genetic and surrounding ecological factors [164]. Indeed, it has been stated that the appropriate mechanism behind the cause of ND is mitochondrial dynamics variation, which elevates the oxidative damage, altering the biological activity of respiratory complexes, which results in brain energy dysfunction [167]. These stimuli trigger cellular stress, which leads to the synthesis and release of brain-derived neurotrophic factor (BDNF), as well as the activation of transcription factor CREB (cAMP response element-binding protein), with consequent expression of Arc (synaptic plasticity), PGC-1 (cellular energy metabolism), and APE1 (DNA repair enzyme), as well as the activation of the tropomyosin-related kinase (Trk B) receptor family and other downstream protein kinases [168].
Neurotrophins avoid neuron degeneration by binding to and activating the Trk receptor family, which is located in the plasma membrane [166]. Neurotrophins work by binding to and activating the Trk receptor family, which is found in the plasma membrane, to prevent neuron degeneration. Since neurotrosphins bind to Trk receptors, they create a microenvironment that promotes neuron development [169]. Various intracellular signaling pathways, such as ERK and PI3k/AKT, are regulated as a result of this binding, allowing cells to survive and aiding in the recovery of neurons from neurodegeneration. Additionally to signaling pathway activation, neurotrophins support Bcl-2 gene expression, which inhibits intracellular apoptosis [169]. Thus, early diagnosis of neurodegeneration may allow for early treatment, which may help to prevent the disease from progressing further [170]. Inhibition of the N-methyl-D-aspartate (NMDA) receptor can prevent or postpone AD. The drugs memantine and namzaric, which act as antagonists for the NMDA receptor, are used to treat AD patients [171].
Bioactive molecules have been recognized for their valuable biological effects, including neuroprotective properties, such as the ability to regulate mitochondria in a way that is distinct from TMs [172]. Branco et al. (2018) found that the flavonoid-rich A. angustifolia bracts extract (AAE) has neuroprotective properties by restoring rotenone-induced mitochondrial complex I, inhibiting the formation of lipid peroxidation and neuronal ROS, and through over expression of NDUFS7 protein and NDUFV2 gene levels in human dopaminergic SH-SY5Y cells [31]. Bark extract of P. pinaster shows protective effects against oxidative hemolysis induced by H2O2, the formation of thiobarbituric acid reactive products and lipid peroxidation [173]. In addition, it prevents oxidative damage to many proteins aggregation and may lessen the risk of several NDs, such as AD, PD and HD [174]. The neuro-protective potential of various conifer spp. crude extracts is summarized in Table 5.
Table 5. Conifers’ phytochemicals demonstrating neuroprotective potential in vitro and in vivo.
| Conifers spp. | Compounds with Neuroprotective Potential | Model | Effective Concentration | Relevant Bioactivities | Reference |
|---|---|---|---|---|---|
| Abies holophylla | Holophyllin-D | C6 glioma cells | 20 μM | Diterpenes compound holophyllin D shows neuroprotective potential in C6 glioma cells by inducing nerve growth factor | [175] |
| Araucara angustifolia | Catechin, epicatechin and rutin | Rat | 10 mg/mL | AAE has antioxidant and neuroprotective properties as it decreases the TBARS levels, CAT activity and NO production in the hippocampus region of the brain in rats. | [176] |
| A. angustifolia | Catechin, epicatechin, rutin, quercetin and apigenin | human dopaminergic SH-SY5Y cells | 5 μg/mL | Decrease in the production of neuron (ROS) and lipid peroxidation. | [31] |
| A. angustifolia | Quercetin | cockroach | 200–400 μg/g | Neurotoxicity modulates the behavior of insects by altering the dopaminergic pathways, as quercetin has the ability to induce selective inhibitory actions on NMDA and GABA receptors and inhibit the enzyme acetylcholinesterase (AChE) | [177] |
| Cedrus deodara | Cedrin | PC12 cells | 0.1, 1 and 10 μM | PC12 cells injured by amyloid β1–42 can be improved by cedrin. Cedrin can reduce (ROS) overproduction, enhance the activity of SOD and decrease MDA content and inhibition of oxidative stress, improvement of mitochondrial dysfunction and suppression of apoptosis in PC12 cells | [178] |
| Metasequoia glyptostroboides | Gallic acid, rutin, myricetin, kaempferol, quercitrin, epigallocatechin, epicatechin gallate epigallocatechin gallate and caffeic acid | Neuronal PC12 cells | 2 mg/mL | The extracts effectively reduced the hydrogen peroxide-induced lipid peroxidation in neuronal PC12 cells by decreasing intracellular ROS accumulation | [37] |
| Pinus densiflora | Catechin, quercetin dehydrate, astragalin and kaempferol | Mice | 50–100 mg/kg | Catechin displayed a potential effect protecting mouse brains from oxidative damage via the improvement of the antioxidant capacities of TAC, the GSH-redox system, SOD and CAT in the hippocampus region as well as the inactivation of cytokines such as NF-kB in pyramidal cells of the hippocampal CA1 region, while PNE shows antiamnesic properties and effects in Alzheimer’s, as it attenuated the increase in serum corticosterone level and up-regulation of GR hippocampal gene expression | [179][180] |
| P. eldarica | Needle extract | Mice | 50 mg/kg | Alkanes, sterols, terpenoids, and quercetin, which is found in P. eldarica, help in inducing sleep and alter the sleep–wake cycle partly via activation of GABA receptors | [181] |
| P. massoniana | Polyprenols | Mice | 25 mg/kg | Polyprenols significantly increased T-AOC, GSHPx, damaging peroxide components from cells in order to stop the lipid peroxidation chain reaction and avoid excessive hydrolysis to form NEP, MDA, SOD activity (remove free radicals) and β-site AβPP cleaving enzyme 1 (BACE1) expression, while NOS activity, MDA concentration, NO, concentration of Aβ1-42 and PS1 were reduced | [182] |
| P. pinaster | Pycnogenol (PYC) | Mice | 20 mg/kg | In the MPTP-induced mouse model, PYC could prevent dopaminergic neurons by reducing oxidative loads, suppressing glial cell activation, and inhibiting inflammatory responses | [85][183] |
| P. roxburghii | Quercetin, rutin, gallic acid | Wistar albino | 100–300 mg/kg | Quercetin and gallic acid, both present in stem bark, have been shown to inhibit neuronal toxicity and apoptosis by reversing mitochondrial dysfunction and free radical development | [184] |
| Thuja occidentalis | Water extract | Mice | 100 mg/kg | CNS depressant activity, anticonvulsant and muscle relaxant activity | [185] |
| Torreya nucifera, | Arctigenin | Rat Cortical cells | 0.01 µM to 10.0 µM. | Arctigenin significantly attenuated glutamate-induced neurotoxicity by inhibiting the binding of [3H]-kainate to its receptors | [23] |
| T. semen | Polyphenols, flavonoids | Mice | 0–10 mg/mL | TS increased the level of total glutathiones | [186] |
T-AOC—total antioxidative capacity; GSHPx—glutathione peroxidise; SOD—super oxide dismutase; NEP—neprilysin; MDA—malondialdehyde; NO—nitric oxide, NOS—nitric oxide synthase; PS1—presenilin 1, CAT—catalase.
4.2.5. Alzheimer’s Disease (AD)
AD is a common neurodegenerative disease that affects 80% of the elderly population, accounting for about half of all dementia cases and ultimately results in death [187]. Its symptoms include failure to learn, gradual memory loss, and deterioration in behavior and neuronal function [188]. Regarding treatment, only five approved treatment options are licensed in the European Union for the treatment of AD, including rivastigmine, donepezil (cholinesterase inhibitors (ChEIs), galantamine, and memantine (NMDAR antagonist) [189]. An antimitotic agent paclitaxel widely used for the treatment of lung, ovarian and breast cancer has also been investigated as a possible treatment for AD [190]. It is mainly effective in the treatment of tauopathies, which are disorders caused by mutations in the tau protein, which is abundant in central nervous system (CNS) cells and acts by stabilizing microtubules [191]. The consumption of polyphenol-rich foods or beverages has been related to the prevention of AD in distinct studies [192]. The accumulation of amyloid-(A) in brain and leptomeningeal vessels causes cerebral amyloid angiopathy (CAA), which is also a central component of neuritic plaques in AD amyloid-(A) and has been related to the pathogenesis of two of the most common forms of dementia: AD and CAA. As a result, Aβ should be a top priority in the treatment of these diseases, which currently have no effective therapies [193]. Taxifolin, an antioxidant and anti-glycation flavonoid, reduces Aβ aggregation and its accumulation in the cerebrovascular system. In vitro studies have shown that taxifolin facilitates Aβ clearance in the brain, prevents Aβ fibril formation and CAA cognitive loss, and increases cerebral blood flow [194]. The methanol extract of P. roxburghii bark contains bioactive compounds, such as quercetin and gallic acid, which play important roles in neuroprotection by reversing mitochondrial dysfunction, free radical formation, and improving memory and cognition in rats, as well as reducing oxidative stress by improving acetylcholine levels. Furthermore, anti-AD activity has been documented in Pinus species, such as P. halepensis and P. massoniana [195]. Piceatannol, a compound derived from pine bark, has proven to be effective in preventing AD [196]. Resveratrol (RV), a stilbenoid, protects neurons from oxidative damage in a variety of ways, such as lowering lipid peroxidation and increasing intracellular antioxidant levels including antioxidant enzymes catalase (CAT), superoxide dismutase (SOD), glutathione peroxidase (GPx), and heme oxygenase 1 (HO-1) [197]. In this way, RV acts as an anti-AD agent by reducing neuroinflammation, inhibiting Aβ-plaque formation and tauopathy, and as a result inhibits neuronal death and improves memory [198]. Pycnogenol derived from P. pinaster bark has antioxidant, anti-inflammatory, and neuroprotective properties, including inhibition of amyloid-induced neuron apoptosis [199]. When the effect of pycnogenol was investigated in AD-related pathology in a β-amyloidosis mouse model, a decline in plaque numbers was found, while no changes were reported in the soluble β-amyloidosis levels, astrocytes, neurons, microglia, myelination pattern, morphology of axons and the gene expression of APP-processing enzymes [200]. Hence, it is suggested that pycnogenol has potential use in the prevention or in early stages of AD and mild cognitive impairment (MCI) [201]. Table 5 summarizes the neuroprotective potential of different conifers’ phytochemicals in AD.
4.2.6. Parkinson’s Disease
PD is second to AD in terms of the most prevalent progressive ND, with an estimated global prevalence of over 10,000,000 cases [202]. The selective loss of dopaminergic neurons in the substantia nigra pars compacta (SNpc) leads to PD. Briefly, PD occurs due to oxidative stress, dysfunction of mitochondrial complex-1, oxidative cell damage, neuronal excitotoxicity, calcium homeostasis, apoptosis, distressed energy metabolism, inflammation and protein aggregation, such as a-synuclein, apoptosis, and interaction between genetic and environmental causes [203]. Due to uncoordinated mouth and throat movements, PD causes bradykinesia, muscle rigidity, rest tremor, and the loss of postural control, as well as certain secondary symptoms, such as dementia, sialorrhea, soft voice, and trouble swallowing [204][205]. Oxidative stress generates ROS that causes oxidative damage, such as 4-hydroxynonenal (HNE), 26S proteasome and interferes with dopamine metabolism leading to PD [70]. Changes in protein ubiquitination and degradation have recently been related to dopaminergic cell death in PD [206]. Presynaptic protein α-synuclein (α-syn) influences the release of neurotransmitters from synaptic vesicles in the brain [207]. Currently, the treatment of PD includes drugs such as L-DOPA, which is catalyzed primarily by dopa decarboxylase in the brain, and some others such as ropinirole, selegiline, and rasagiline. Ropinirole has some adverse effects, including ankle oedema, vomiting, nausea, hypotension, insomnia, weight loss, hallucinations, psychosis, arrhythmia, dry mouth, nightmares, persistent diarrhea, somnolence and constipation, limiting their clinical applications [208]. As a result, the focus of rising interest in alternative treatments for ND, such as PD, has turned to natural products, which can provide alternatives due to their high effectiveness and few side effects [209]. Many plant extracts tend to stop α-syn from oligomerization and fibrillation, which is an emerging therapeutic mechanism in PD [172]. Methanol extract of J. communis at doses of 100 and 200 mg/kg was found to be effective in reducing catalepsy, enhancing locomotor activity (actophotometer), and increasing the level of reduced glutathione (GSH), protein level and muscle activity in rats [210]. In an in vitro study on Fisher F344 rats, Zhang et al. (2010) discovered that RV protect dopaminergic neurons from damage caused by MPP+, 6-OHDA, and also show efficacy against lipopolysaccharide-induced neurotoxicity by inhibiting nuclear factor kappa B (NF-κB) signaling and microglial activation [211].
4.2.7. Insomnia
Insomnia is a chronically debilitating disease that has become increasingly common, posing immense health and economic challenges for both individuals and the community [212]. Trouble falling asleep, staying asleep, fragmented sleep (repeatedly waking up at night or waking up early in the morning) are all symptoms of this condition [213]. While behavioral therapy, psychotherapy and light therapy have all been used to treat insomnia, the most common medications for insomnia are hypnotic drugs that target GABAA-benzodiazepine (BZD) receptors, such as diazepam and zolpidem [214]. However, several side effects have been identified, including cognitive impairment, resistance, headaches, nausea, and rebound insomnia [215][216]. Methanol extract of A. webbiana leaves showed potent synergistic effect in mice at dose of 100, 150, and 200 mg/kg, with sleep-inducing sedative drugs, diazepam (6 mg/kg), pentobarbitone sodium (50 mg/kg) and propylene glycol [144]. In addition, the major monoterpenoid components present in Pinus spp., α-pinene and 3-carene, have been reported to have hypnotic effects through GABAA-BZD receptors. 3-carene increases the length of sleep in mice given pentobarbital-induced sleep drugs by binding to the BZD site of the GABAA-BZD receptor α1 and ϒ2 [217].
References
- Mustafa, G.; Arif, R.; Atta, A.; Sharif, S.; Jamil, A. Bioactive Compounds from Medicinal Plants and Their Importance in Drug Discovery in Pakistan. Matrix Sci. Pharma 2017, 1, 17–26.
- Abdel-Razek, A.S.; El-Naggar, M.E.; Allam, A.; Morsy, O.M.; Othman, S.I. Microbial natural products in drug discovery. Processes 2020, 8, 470.
- Yuan, H.; Ma, Q.; Ye, L.; Piao, G. The traditional medicine and modern medicine from natural products. Molecules 2016, 21, 559.
- Nisar, B.; Sultan, A.; Rubab, S.L. Comparison of Medicinally Important Natural Products versus Synthetic Drugs-A Short Commentary. Nat. Prod. Chem. Res. 2018, 06, 308.
- Newman, D.J.; Cragg, G.M.; Snader, K.M. Natural products as sources of new drugs over the period 1981–2002. J. Nat. Prod. 2003, 66, 1022–1037.
- Galm, U.; Shen, B. Natural Product Drug Discovery: The Times Have Never Been Better. Chem. Biol. 2007, 14, 1098–1104.
- Akaberi, M.; Boghrati, Z.; Amiri, M.S.; Khayyat, M.H.; Emami, S.A. A Review of Conifers in Iran: Chemistry, Biology and their Importance in Traditional and Modern Medicine. Curr. Pharm. Des. 2020, 26, 1584–1613.
- Bhardwaj, K.; Islam, M.T.; Jayasena, V.; Sharma, B.; Sharma, S.; Sharma, P.; Kuča, K.; Bhardwaj, P. Review on essential oils, chemical composition, extraction, and utilization of some conifers in Northwestern Himalayas. Phyther. Res. 2020, 34, 2889–2910.
- Bhardwaj, K.; Dhanjal, D.S.; Sharma, A.; Nepovimova, E.; Kalia, A.; Thakur, S.; Bhardwaj, S.; Chopra, C.; Singh, R.; Verma, R.; et al. Conifer-derived metallic nanoparticles: Green synthesis and biological applications. Int. J. Mol. Sci. 2020, 21, 9028.
- Conifers. Available online: (accessed on 15 March 2021).
- Farjon, A. The Kew Review Conifers of the World. Kew Bull. 2018, 5974, 1–16.
- Kopaczyk, J.M.; Warguła, J.; Jelonek, T. The variability of terpenes in conifers under developmental and environmental stimuli. Environ. Exp. Bot. 2020, 180, 104197.
- Virjamo, V.; Fyhrquist, P.; Koskinen, A.; Lavola, A.; Nissinen, K.; Julkunen-Tiitto, R. 1,6-Dehydropinidine Is an Abundant Compound in Picea abies (Pinaceae) Sprouts and 1,6-Dehydropinidine Fraction Shows Antibacterial Activity against Streptococcus equi Subsp. equi. Molecules 2020, 25, 4558.
- Mill, R.R.; Chase, M.W. A new classification and linear sequence of extant gymnosperms. Phytotaxa 2011, 19, 55–70.
- St-Pierre, A.; Blondeau, D.; Bourdeau, N.; Bley, J.; Desgagné-Penix, I. Chemical Composition of Black Spruce (Picea mariana) Bark Extracts and Their Potential as Natural Disinfectant. Ind. Biotechnol. 2019, 15, 219–231.
- Tawara, J.N.; Blokhin, A.; Foderaro, T.A.; Stermitz, F.R.; Hope, H. Toxic Piperidine Alkaloids from Pine (Pinus) and Spruce (Picea) Trees. New Structures and a Biosynthetic Hypothesis. J. Org. Chem. 1993, 58, 4813–4818.
- Küpeli, E.; Erdemoǧlu, N.; Yeşilada, E.; Şener, B. Anti-inflammatory and antinociceptive activity of taxoids and lignans from the heartwood of Taxus baccata L. J. Ethnopharmacol. 2003, 89, 265–270.
- Juyal, D.; Thawani, V.; Thaledi, S.; Joshi, M. Ethnomedical properties of Taxus wallichiana Zucc. (Himalayan yew). J. Tradit. Complement. Med. 2014, 4, 159–161.
- Hafezi, K.; Hemmati, A.A.; Abbaszadeh, H.; Valizadeh, A.; Makvandi, M. Anticancer activity and molecular mechanisms of α-conidendrin, a polyphenolic compound present in Taxus yunnanensis, on human breast cancer cell lines. Phyther. Res. 2020, 34, 1397–1408.
- Ivanova, D.I.; Tashev, A.N.; Nedialkov, P.T.; Ilieva, Y.E.; Atanassova, T.N.; Olech, M.; Nowak, R.; Angelov, G.; Tsvetanova, F.V.; Iliev, I.A.; et al. Antioxidant and antiproliferative activity of Juniperus L. Species of Bulgarian and foreign origin and their anticancer metabolite identification. Bulg. Chem. Commun. 2018, 50, 144–150.
- Kanchan, B.; Prerna, B.; Simran, K. Medicinal value of secondary metabolites of pines grown in Himalayan region of India. Res. J. Biotechnol. 2020, 15, 131–140.
- Singh, S.K.; Shanmugavel, M.; Kampasi, H.; Singh, R.; Mondhe, D.M.; Rao, J.M.; Adwankar, M.K.; Saxena, A.K.; Qazi, G.N. Chemically standardized isolates from Cedrus deodara stem wood having anticancer activity. Planta Med. 2007, 73, 519–526.
- Jang, Y.P.; Kim, S.R.; Choi, Y.H.; Kim, J.; Kim, S.G.; Markelonis, G.J.; Oh, T.H.; Kim, Y.C. Arctigenin protects cultured cortical neurons from glutamate-induced neurodegeneration by binding to kainate receptor. J. Neurosci. Res. 2002, 68, 233–240.
- Asmi, K.S.; Lakshmi, T.; Balusamy, S.R.; Parameswari, R. Therapeutic aspects of taxifolin—An update. J. Adv. Pharm. Educ. Res. 2017, 7, 187–189.
- Hammerbacher, A.; Kandasamy, D.; Ullah, C.; Schmidt, A.; Wright, L.P.; Gershenzon, J. Flavanone-3-hydroxylase plays an important role in the biosynthesis of spruce phenolic defenses against bark beetles and their fungal associates. Front. Plant Sci. 2019, 10, 1–15.
- Michael, H.N.; Awad, H.M.; El-Sayed, N.H.; Paré, P.W. Chemical and antioxidant investigations: Norfolk pine needles (Araucaria excelsa). Pharm. Biol. 2010, 48, 534–538.
- Ferreira-Santos, P.; Genisheva, Z.; Botelho, C.; Santos, J.; Ramos, C.; Teixeira, J.A.; Rocha, C.M.R. Unravelling the biological potential of Pinus pinaster bark extracts. Antioxidants 2020, 9, 334.
- Gascón, S.; Jiménez-Moreno, N.; Jiménez, S.; Quero, J.; Rodríguez-Yoldi, M.J.; Ancín-Azpilicueta, C. Nutraceutical composition of three pine bark extracts and their antiproliferative effect on Caco-2 cells. J. Funct. Foods 2018, 48, 420–429.
- Dziedzinski, M.; Kobus-Cisowska, J.; Szymanowska, D.; Stuper-Szablewska, K.; Baranowska, M. Identification of polyphenols from coniferous shoots as natural antioxidants and antimicrobial compounds. Molecules 2020, 25, 3527.
- Fierascu, I.; Ungureanu, C.; Avramescu, S.M.; Cimpeanu, C.; Georgescu, M.I.; Fierascu, R.C.; Ortan, A.; Sutan, A.N.; Anuta, V.; Zanfirescu, A.; et al. Genoprotective, antioxidant, antifungal and anti-inflammatory evaluation of hydroalcoholic extract of wild-growing Juniperus communis L. (Cupressaceae) native to Romanian southern sub-Carpathian hills. BMC Complement. Altern. Med. 2018, 18, 1–15.
- Branco, C.S.; Duong, A.; Machado, A.K.; Wu, A.; Scola, G.; Andreazza, A.C.; Salvador, M. Araucaria angustifolia (Bertol.) Kuntze has neuroprotective action through mitochondrial modulation in dopaminergic SH-SY5Y cells. Mol. Biol. Rep. 2019, 46, 6013–6025.
- Nisar, M.; Khan, I.; Ahmad, B.; Ali, I.; Ahmad, W.; Choudhary, M.I. Antifungal and antibacterial activities of Taxus wallichiana Zucc. J. Enzym. Inhib. Med. Chem. 2008, 23, 256–260.
- Freitas, A.M.; Almeida, M.T.R.; Andrighetti-Fröhner, C.R.; Cardozo, F.T.G.S.; Barardi, C.R.M.; Farias, M.R.; Simões, C.M.O. Antiviral activity-guided fractionation from Araucaria angustifolia leaves extract. J. Ethnopharmacol. 2009, 126, 512–517.
- Al-Sayed, E.; Gad, H.A.; El-Shazly, M.; Abdel-Daim, M.M.; Nasser Singab, A. Anti-inflammatory and analgesic activities of cupressuflavone from Cupressus macrocarpa: Impact on pro-inflammatory mediators. Drug Dev. Res. 2018, 79, 22–28.
- Ferrentino, G.; Haman, N.; Morozova, K.; Tonon, G.; Scampicchio, M. Phenolic compounds extracted from spruce (Picea abies) by supercritical carbon dioxide as antimicrobial agents against gram-positive bacteria assessed by isothermal calorimetry. J. Therm. Anal. Calorim. 2020.
- Hoon, L.Y.; Choo, C.; Watawana, M.I.; Jayawardena, N.; Waisundara, V.Y. Evaluation of the total antioxidant capacity and antioxidant compounds of different solvent extracts of Chilgoza Pine nuts (Pinus gerardiana). J. Funct. Foods 2015, 18, 1014–1021.
- Lee, S.J.; Lee, S.Y.; Hur, S.J.; Bae, Y., II; Jeong, C.H. Neuroprotective and antioxidant effects of Metasequoia glyptostroboides leaf extract. Curr. Top. Nutraceutical Res. 2016, 14, 67–72.
- Sahin Yaglioglu, A.; Eser, F. Screening of some Juniperus extracts for the phenolic compounds and their antiproliferative activities. S. Afr. J. Bot. 2017, 113, 29–33.
- Osuna-Torres, L.; García-Martí, X.; Ventura-Zapata, E.; López-Upton, J.; Zamilpa-Alvarez, A.; González-Cortazar, M.; Herrera-Ruiz, M.; Tapia-Barrera, N. Taxus globosa Schltdl. (Mexican yew) and Taxus baccata L. (European yew): Intra and interspecies analysis of taxol content and biological activity according to different sources. For. Syst. 2015, 24, 16.
- Legault, J.; Girard-Lalancette, K.; Dufour, D.; Pichette, A. Antioxidant potential of bark extracts from boreal forest conifers. Antioxidants 2013, 2, 77–89.
- Lantto, T.A.; Colucci, M.; Závadová, V.; Hiltunen, R.; Raasmaja, A. Cytotoxicity of curcumin, resveratrol and plant extracts from basil, juniper, laurel and parsley in SH-SY5Y and CV1-P cells. Food Chem. 2009, 117, 405–411.
- Välimaa, A.L.; Raitanen, J.E.; Tienaho, J.; Sarjala, T.; Nakayama, E.; Korpinen, R.; Mäkinen, S.; Eklund, P.; Willför, S.; Jyske, T. Enhancement of Norway spruce bark side-streams: Modification of bioactive and protective properties of stilbenoid-rich extracts by UVA-irradiation. Ind. Crops Prod. 2020, 145, 112150.
- Raiber, S.; Schröder, G.; Schröder, J. Molecular and enzymatic characterization of two stilbene synthases from Eastern white pine (Pinus strobus) A single Arg/His difference determines the activity and the pH dependence of the enzymes. FEBS Lett. 1995, 361, 299–302.
- Hovelstad, H.; Leirset, I.; Oyaas, K.; Fiksdahl, A. Screening analyses of pinosylvin stilbenes, resin acids and lignans in Norwegian conifers. Molecules 2006, 11, 103–114.
- Francezon, N.; Meda, N.S.B.R.; Stevanovic, T. Optimization of bioactive polyphenols extraction from Picea mariana bark. Molecules 2017, 22, 2118.
- Latva-Mäenpää, H. Bioactive and Protective Polyphenolics From Roots and Stumps of Conifer Trees (Norway Spruce and Scots Pine); Helsingin Yliopisto: Helsinki, Finland, 2017; ISBN 9789515134653.
- Singh, B.; Sharma, R.A. Plant terpenes: Defense responses, phylogenetic analysis, regulation and clinical applications. 3 Biotech 2015, 5, 129–151.
- Porres-Martínez, M.; González-Burgos, E.; Carretero, M.E.; Pilar Gómez-Serranillos, M. In vitro neuroprotective potential of the monoterpenes α-pinene and 1,8-cineole against H2O2-induced oxidative stress in PC12 cells. Z. fur Naturforsch. Sect. C J. Biosci. 2016, 71, 191–199.
- Da Silveira E Sá, R.D.C.; Andrade, L.N.; De Sousa, D.P. Sesquiterpenes from essential oils and anti-inflammatory activity. Nat. Prod. Commun. 2015, 10, 1767–1774.
- Dey, P.; Kundu, A.; Kumar, A.; Gupta, M.; Lee, B.M.; Bhakta, T.; Dash, S.; Kim, H.S. Analysis of Alkaloids (Indole Alkaloids, Isoquinoline Alkaloids, Tropane Alkaloids); Elsevier Inc.: Amsterdam, The Netherlands, 2020; ISBN 9780128164556.
- Thawabteh, A.; Juma, S.; Bader, M.; Karaman, D.; Scrano, L.; Bufo, S.A.; Karaman, R. The biological activity of natural alkaloids against herbivores, cancerous cells and pathogens. Toxins 2019, 11, 656.
- Ignat, I.; Volf, I.; Popa, V.I. A critical review of methods for characterisation of polyphenolic compounds in fruits and vegetables. Food Chem. 2011, 126, 1821–1835.
- Tanase, C.; Boz, I.; Stingu, A.; Volf, I.; Popa, V.I. Physiological and biochemical responses induced by spruce bark aqueous extract and deuterium depleted water with synergistic action in sunflower (Helianthus annuus L.) plants. Ind. Crops Prod. 2014, 60, 160–167.
- Tanase, C.; Cosarcă, S.; Muntean, D.L. A critical review of phenolic compounds extracted from the bark of woody vascular plants and their potential biological activity. Molecules 2019, 24, 1182.
- El Omari, N.; Ezzahrae Guaouguaou, F.; El Menyiy, N.; Benali, T.; Aanniz, T.; Chamkhi, I.; Balahbib, A.; Taha, D.; Shariati, M.A.; Zengin, G.; et al. Phytochemical and biological activities of Pinus halepensis mill., and their ethnomedicinal use. J. Ethnopharmacol. 2021, 268, 113661.
- Metsämuuronen, S.; Sirén, H. Bioactive phenolic compounds, metabolism and properties: A review on valuable chemical compounds in Scots pine and Norway spruce. Phytochem. Rev. 2019, 18, 623–664, ISBN 0123456789.
- Rodríguez-García, C.; Sánchez-Quesada, C.; Gaforio, J.J.; Gaforio, J.J. Dietary flavonoids as cancer chemopreventive agents: An updated review of human studies. Antioxidants 2019, 8, 137.
- Tsao, R. Chemistry and Biochemistry of Dietary Polyphenols. Nutrients 2010, 2, 1231–1246.
- Nanda, S.; Mohanty, J.N.; Mishra, R.; Joshi, R.K. Metabolic Engineering of Phenylpropanoids in Plants. In Transgenesis and Secondary Metabolism; Springer: Berlin/Heidelberg, Germany, 2017; pp. 485–510.
- Saleem, M.; Kim, J.; Ali, S.; Sup, Y. An update on bioactive plant lignans. Nat. Prod. Rep. 2005, 22, 696–716.
- García-Pérez, M.E.; Royer, M.; Herbette, G.; Desjardins, Y.; Pouliot, R.; Stevanovic, T. Picea mariana bark: A new source of trans-resveratrol and other bioactive polyphenols. Food Chem. 2012, 135, 1173–1182.
- Salminen, J.; Karonen, M. Chemical ecology of tannins and other phenolics: We need a change in approach. Br. Ecol. Soc. 2011, 25, 325–338.
- Raitanen, J.E.; Järvenpää, E.; Korpinen, R.; Mäkinen, S.; Hellström, J.; Kilpeläinen, P.; Liimatainen, J.; Ora, A.; Tupasela, T.; Jyske, T. Tannins of conifer bark as Nordic piquancy—sustainable preservative and aroma? Molecules 2020, 25, 567.
- Koche, D.; Shirsat, R.; Kawale, M. An overview of major classes of phytochemicals: Their type and role in disease prevention. Hislopia J. 2016, 9, 2016.
- Prothmann, J.; Sun, M.; Spégel, P.; Sandahl, M.; Turner, C.; Scheuba, J.; Wronski, V.K.; Rollinger, J.M.; Grienke, U.; Santos-Buelga, C.; et al. Relationship between phenolic compounds, anthocyanins content and antioxidant activity in colored barley germplasm. J. Agric. Food Chem. 2017, 53, 1713.
- Matthews, S.; Mila, I.; Scalbert, A.; Donnelly, D.M.X. Extractable and non-extractable proanthocyanidins in barks. Phytochemistry 1997, 45, 405–410.
- Koleckar, V.; Kubikova, K.; Rehakova, Z.; Kuca, K.; Jun, D.; Jahodar, L.; Opletal, L. Condensed and Hydrolysable Tannins as Antioxidants Influencing the Health. Mini-Rev. Med. Chem. 2008, 8, 436–447.
- De Bruyne, T.; Pieters, L.; Deelstra, H.; Vlietinck, A. Condensed vegetable tannins: Biodiversity in structure and biological activities. Biochem. Syst. Ecol. 1999, 27, 445–459.
- Scalbert, A. Antimicrobial properties of tannins. Phytochemistry 1991, 30, 3875–3883.
- Bhangale, J.O.; Acharya, S.R. Anti-Parkinson Activity of Petroleum Ether Extract of Ficus religiosa (L.) Leaves. Adv. Pharmacol. Sci. 2016, 2016, 9436106.
- Brijesh, K.; Ruchi, R.; Sanjita, D.; Saumya, D.; June, A. Phytoconstituents and Therapeutic potential of Thuja occidentalis. Res. J. Pharm. Biol. Chem. Sci. 2012, 3, 354–362.
- Shuaib, M.; Ali, M.; Ahamad, J.; Naquvi, K.J.; Ahmad, M.I. Pharmacognosy of Pinus roxburghii: A Review. Phytochemistry 2006, 2, 262–268.
- Poudel, R.C.; Gao, L.M.; Möller, M.; Baral, S.R.; Uprety, Y.; Liu, J.; Li, D.Z. Yews (Taxus) along the Hindu Kush-Himalayan region: Exploring the ethnopharmacological relevance among communities of Mongol and Caucasian origins. J ethnopharmacol. 2013, 147, 190–203.
- Kunwar, R.M.; Shrestha, K.P.; Bussmann, R.W. Traditional herbal medicine in Far-west Nepal: A pharmacological appraisal. J. Ethnobiol. Ethnomed. 2010, 6, 1–18.
- Sharma, H.; Garg, M. A review of traditional use, phytoconstituents and biological activities of Himalayan yew, Taxus wallichiana. J. Integr. Med. 2015, 13, 80–90.
- Di Meo, S.; Reed, T.T.; Venditti, P.; Victor, V.M. Role of ROS and RNS Sources in Physiological and Pathological Conditions. Oxid. Med. Cell. Longev. 2016, 2016, 1245049.
- Phaniendra, A.; Jestadi, D.B.; Periyasamy, L. Free Radicals: Properties, Sources, Targets, and Their Implication in Various Diseases. Indian J. Clin. Biochem. 2015, 30, 11–26.
- Kumar, H.; Bhardwaj, K.; Nepovimova, E.; Kuča, K.; Dhanjal, D.S.; Bhardwaj, S.; Bhatia, S.K.; Verma, R.; Kumar, D. Antioxidant functionalized nanoparticles: A combat against oxidative stress. Nanomaterials 2020, 10, 1334.
- Collin, F. Chemical basis of reactive oxygen species reactivity and involvement in neurodegenerative diseases. Int. J. Mol. Sci. 2019, 20, 2407.
- Genestra, M. Oxyl radicals, redox-sensitive signalling cascades and antioxidants. Cell. Signal. 2007, 19, 1807–1819.
- Ricordi, C.; Garcia-Contreras, M.; Farnetti, S. Diet and Inflammation: Possible Effects on Immunity, Chronic Diseases, and Life Span. J. Am. Coll. Nutr. 2015, 34, 10–13.
- Boukhenouna, S.; Wilson, M.A.; Bahmed, K.; Kosmider, B. Reactive oxygen species in chronic obstructive pulmonary disease. Oxid. Med. Cell. Longev. 2018, 2018, 5730395.
- Packer, L.; Rimbach, G.; Virgili, F. Antioxidant activity and biologic properties of a procyanidin-rich extract from pine (Pinus maritima) bark, pycnogenol. Free Radic. Biol. Med. 1999, 27, 704–724.
- Lobo, V.; Patil, A.; Phatak, A.; Chandra, N. Free radicals, antioxidants and functional foods: Impact on human health. Pharmacogn. Rev. 2010, 4, 118–126.
- Iravani, S.; Zolfaghari, B. Pharmaceutical and nutraceutical effects of Pinus pinaster bark extract. Res. Pharm. Sci. 2011, 6, 1–11.
- Senthilmohan, S.T.; Zhang, J.; Stanley, R.A. Effects of flavonoid extract Enzogenol with vitamin C on protein oxidation and DNA damage in older human subjects. Nutr. Res. 2003, 23, 1199–1210.
- Azqueta, A.; Collins, A. Polyphenols and DNA damage: A mixed blessing. Nutrients 2016, 8, 785.
- Kukreja, A.; Wadhwa, N. Therapeutic Role of Resveratrol and Piceatannol in Disease Prevention. J. Blood Disord. Transfus. 2014, 5, 9.
- Sharma, A.; Goyal, R.; Sharma, L. Potential biological efficacy of Pinus plant species against oxidative, inflammatory and microbial disorders. BMC Complement. Altern. Med. 2016, 16, 1–11.
- Azab, A.; Nassar, A.; Azab, A.N. Anti-inflammatory activity of natural products. Molecules 2016, 21, 1321.
- Artis, D.; Spits, H. The biology of innate lymphoid cells. Nature 2015, 517, 293–301.
- Fernandes, J.V.; Cobucci, R.N.O.; Jatobá, C.A.N.; de Medeiros Fernandes, T.A.A.; de Azevedo, J.W.V.; de Araújo, J.M.G. The Role of the Mediators of Inflammation in Cancer Development. Pathol. Oncol. Res. 2015, 21, 527–534.
- Heppner, F.L.; Ransohoff, R.M.; Becher, B. Immune attack: The role of inflammation in Alzheimer disease. Nat. Rev. Neurosci. 2015, 16, 358–372.
- Rock, K.L.; Rock, K.L. Innate and adaptive immune responses to cell death. Immunol. Rev. 2011, 243, 191–205.
- Waisman, A.; Liblau, R.S.; Becher, B. Innate and adaptive immune responses in the CNS. Lancet Neurol. 2015, 14, 945–955.
- Vignali, D.A.A.; Kuchroo, V.K. Review IL-12 family cytokines: Immunological playmakers. Nat. Immunol. 2012, 13, 722–728.
- Montgomery, S.L.; Bowers, W.J. Tumor Necrosis Factor-alpha and the Roles it Plays in Homeostatic and Degenerative Processes Within the Central Nervous System. J. Neuroimmune Pharmacol. 2012, 7, 42–59.
- Fenton, M.J. Review: Transcriptional and post-transcriptional regulation of interleukin 1 gene expression. Int. J. Immunopharm. 1992, 14, 401–411.
- Rider, P.; Carmi, Y.; Voronov, E.; Apte, R.N. Interleukin-1α. Semin. Immunol. 2013, 25, 430–438.
- Cha, K.-J. The Anti-Inflammatory Effects of Picea wilsonii Mast on HaCaT Cells. Korean J. Clin. Lab. Sci. 2016, 48, 365–370.
- Langrish, C.L.; Mckenzie, B.S.; Wilson, N.J.; Kastelein, R.A.; Cua, D.J. IL-12 and IL-23: Master regulators of innate and adaptive immunity. Immunol. Rev. 2004, 202, 96–105.
- Duvallet, E.; Semerano, L.; Assier, E.; Falgarone, G.; Duvallet, E.; Semerano, L.; Assier, E.; Falgarone, G.; Duvallet, E.; Semerano, L.; et al. Interleukin-23: A key cytokine in inflammatory diseases. Ann. Med. 2011, 3890, 503–511.
- Sabat, R. Cytokine & Growth Factor Reviews IL-10 family of cytokines. Cytokine Growth Factor Rev. 2010, 21, 315–324.
- Ng, T.H.S.; Britton, G.J.; Hill, E.V.; Verhagen, J.; Burton, B.R.; Wraith, D.C. Regulation of adaptive immunity; the role of interleukin-10. Front. Immunol. 2013, 4, 1–14.
- Kwilasz, A.J.; Grace, P.M.; Serbedzija, P.; Maier, S.F.; Watkins, L.R. Neuropharmacology The therapeutic potential of interleukin-10 in neuroimmune diseases. Neuropharmacology 2014, 2, 55–69.
- Atanasov, A.G.; Waltenberger, B.; Pferschy-Wenzig, E.M.; Linder, T.; Wawrosch, C.; Uhrin, P.; Temml, V.; Wang, L.; Schwaiger, S.; Heiss, E.H.; et al. Discovery and resupply of pharmacologically active plant-derived natural products: A review. Biotechnol. Adv. 2015, 33, 1582–1614.
- Zhang, Q.W.; Lin, L.G.; Ye, W.C. Techniques for extraction and isolation of natural products: A comprehensive review. Chin. Med. 2018, 13, 1–26.
- Bucar, F.; Wube, A.; Schmid, M. Natural product isolation-how to get from biological material to pure compounds. Nat. Prod. Rep. 2013, 30, 525–545.
- Gopalasatheeskumar, K. Significant Role of Soxhlet Extraction Process in Phytochemical. Mintage J. Pharm. Med. Sci. 2018, 7, 43–47.
- Zhao, Q.Q.; Wang, S.F.; Li, Y.; Song, Q.Y.; Gao, K. Terpenoids with anti-inflammatory activity from Abies chensiensis. Fitoterapia 2016, 111, 87–94.
- Qayum, M.; Nisar, M.; Shah, M.R.; Adhikari, A.; Kaleem, W.A.; Khan, I.; Khan, N.; Gul, F.; Khan, I.A.; Zia-Ul-Haq, M.; et al. Analgesic and antiinflammatory activities of taxoids from Taxus wallichiana Zucc. Phyther. Res. 2012, 26, 552–556.
- Stan, M.S.; Voicu, S.N.; Caruntu, S.; Nica, I.C.; Olah, N.K.; Burtescu, R.; Balta, C.; Rosu, M.; Herman, H.; Hermenean, A.; et al. Antioxidant and anti-inflammatory properties of a Thuja occidentalis mother tincture for the treatment of ulcerative colitis. Antioxidants 2019, 8, 416.
- Kim, D.S.; Kim, M.S.; Kang, S.W.; Sung, H.Y.; Kang, Y.H. Pine bark extract enzogenol attenuated tumor necrosis factor-α- induced endothelial cell adhesion and monocyte transmigration. J. Agric. Food Chem. 2010, 58, 7088–7095.
- Schäfer, A.; Chovanová, Z.; Muchová, J.; Sumegová, K.; Liptáková, A.; Högger, P. Inhibition of COX-1 and COX-2 activity by plasma of human volunteers after ingestion of French maritime pine bark extract (Pycnogenol). Biomed. Pharmacother. 2005, 60, 5–9.
- Latest Global Cancer Data_ Cancer Burden Rises to 18 2018. WHO. Available online: (accessed on 15 February 2021).
- Yan, S.H. An early history of human breast cancer: West meets East. Chin. J. Cancer 2013, 32, 475–477.
- Sudhakar, A. History of Cancer, Ancient and Modern Treatment Methods. J. Cancer Sci. Ther. 2009, 01, i–iv.
- Ghosh, S.K. Giovanni Battista Morgagni (1682–1771): Father of pathologic anatomy and pioneer of modern medicine. Anat. Sci. Int. 2017, 92, 305–312.
- Cancer. Available online: (accessed on 20 February 2021).
- Ma, X.; Wang, Z. Anticancer drug discovery in the future: An evolutionary perspective. Drug Discov. Today 2009, 14, 1136–1142.
- Widmer, N.; Bardin, C.; Chatelut, E.; Paci, A.; Beijnen, J.; Levêque, D.; Veal, G.; Astier, A. Review of therapeutic drug monitoring of anticancer drugs part two—Targeted therapies. Eur. J. Cancer 2014, 50, 2020–2036.
- Kuczynski, E.A.; Sargent, D.J.; Grothey, A.; Kerbel, R.S. Drug rechallenge and treatment beyond progression-implications for drug resistance. Nat. Rev. Clin. Oncol. 2013, 10, 571–587.
- Lichota, A.; Gwozdzinski, K. Anticancer activity of natural compounds from plant and marine environment. Int. J. Mol. Sci. 2018, 19, 3533.
- Kim, J.A.H.; Kim, D.H.; Hossain, M.A.; Kim, M.Y.; Sung, B.; Yoon, J.H.; Suh, H.; Jeong, T.C.; Chung, H.Y.; Kim, N.D. HS-1793, a resveratrol analogue, induces cell cycle arrest and apoptotic cell death in human breast cancer cells. Int. J. Oncol. 2014, 44, 473–480.
- Sharifi-Rad, J.; Ozleyen, A.; Tumer, T.B.; Adetunji, C.O.; El Omari, N.; Balahbib, A.; Taheri, Y.; Bouyahya, A.; Martorell, M.; Martins, N.; et al. Natural products and synthetic analogs as a source of antitumor drugs. Biomolecules 2019, 9, 679.
- Tafrihi, M.; Imran, M.; Tufail, T.; Gondal, T.A.; Caruso, G.; Sharma, S.; Sharma, R.; Atanassova, M.; Atanassov, L.; Valere, P.; et al. The Wonderful Activities of the Genus Mentha: Not Only Antioxidant Properties. Molecules 2021, 26, 1118.
- Birinci, H.; Şen, B.; Sayğılı, S.; Ölmez, E.; Uluer, E.T.; Özbilgin, K. The Effect of Pycnogenol and Paclitaxel on DNA Damage in Human Breast Cancer Cell Line. Proceedings 2017, 1, 1023.
- Dinić, J.; Ríos-Luci, C.; Karpaviciene, I.; Cikotiene, I.; Fernandes, M.X.; Pešić, M.; Padrón, J.M. CKT0353, a novel microtubule targeting agent, overcomes paclitaxel induced resistance in cancer cells. Investig. New Drugs 2020, 38, 584–598.
- Binarová, P.; Tuszynski, J. Tubulin: Structure, Functions and Roles in Disease. Cells 2019, 8, 1294.
- Zhang, D.; Kanakkanthara, A. Beyond the paclitaxel and vinca alkaloids: Next generation of plant-derived microtubule-targeting agents with potential anticancer activity. Cancers 2020, 12, 1721.
- Harshita; Barkat, M.A.; Beg, S.; Pottoo, F.H.; Ahmad, F.J. Nanopaclitaxel therapy: An evidence based review on the battle for next-generation formulation challenges. Nanomedicine 2019, 14, 1323–1341.
- Ganguly, A.; Yang, H.; Cabral, F. Paclitaxel-dependent cell lines reveal a novel drug activity. Mol. Cancer Ther. 2010, 9, 2914–2923.
- Banerjee, S.; Das, A.; Chakraborty, P.; Suthindhiran, K.; Jayasri, M.A. Antioxidant and antimicrobial activity of Araucaria cookii and Brassaia actinophyla. Pak. J. Biol. Sci. 2014, 17, 715–719.
- Jain, S.; Kumar, D.; Malviya, N.; Jain, A.; Jain, S.; Jain, V. Estimation of total phenolic, tannins, and flavonoid contents and antioxidant activity of Cedrus deodara heart wood extracts. Egypt. Pharm. J. 2015, 14, 10.
- Horiba, H.; Nakagawa, T.; Zhu, Q.; Ashour, A.; Watanabe, A.; Shimizu, K. Biological activities of extracts from different parts of cryptomeria japonica. Nat. Prod. Commun. 2016, 11, 1337–1342.
- Bajpai, V.K.; Sharma, A.; Kang, S.C.; Baek, K.H. Antioxidant, lipid peroxidation inhibition and free radical scavenging efficacy of a diterpenoid compound sugiol isolated from Metasequoia glyptostroboides. Asian Pac. J. Trop. Med. 2014, 7, 9–15.
- Bhagat, M.; Gupta, S.; Sudan, R. In vitro Evaluation of Antioxidant Activity of Picea smithiana Growing in Bhaderwah Region of Jammu and Kashmir. Cell. Life Sci. J. 2017, 2.
- Salhi, N.; Bouyahya, A.; El Guourrami, O.; El Jemli, M.; Bourais, I.; Zellou, A.; Cherrah, Y.; El Abbes Faouzi, M. Investigation of in vitro and in vivo antioxidant and antidiabetic activities of Pinus halepensis extracts. J. Herbmed Pharmacol. 2021, 10, 123–131.
- Tekaday, D.; Antony, B.; Jain, S. Antimicrobial, antioxidant and phytochemical investigation of Thuja occidentalis (Arbor vitae) leave extract. GSC Biol. Pharm. Sci. 2020, 12, 108–116.
- Milutinović, M.G.; Stanković, M.S.; Cvetković, D.M.; Topuzović, M.D.; Mihailović, V.B.; Marković, S.D. Antioxidant and anticancer properties of leaves and seed cones from European yew (Taxus baccata L.). Arch. Biol. Sci. 2015, 67, 525–534.
- Bhat, M.A.; Ganie, S.A.; Dar, K.B.; Ali, R.; Hamid, R. In Vitro antioxidant potential and hepatoprotective activity of Taxus Wallichiana. Asian J. Pharm. Clin. Res. 2018, 11, 237–243.
- Subba, B. Analysis of Phytochemical Constituents and Biological Activity of Taxus Wallichiana Zucc. Dolakha District of Nepal. Int. J. Appl. Sci. Biotechnol. 2018, 6, 110–114.
- Yang, X.-W.; Zeng, H.-W.; Liu, X.-H.; Li, S.-M.; Xu, W.; Shen, Y.-H.; Zhang, C.; Zhang, W.-D. Anti-inflammatory and anti-tumour effects of Abies georgei extracts. J. Pharm. Pharmacol. 2008, 60, 937–941.
- Nayak, S.S.; Ghosh, A.K.; Debnath, B.; Vishnoi, S.P.; Jha, T. Synergistic effect of methanol extract of Abies webbiana leaves on sleeping time induced by standard sedatives in mice and anti-inflammatory activity of extracts in rats. J. Ethnopharmacol. 2004, 93, 397–402.
- Bisht, B.; Nainwal, P.; Saini, P. Evaluation of in vitro anti-inflammatory activity of Agathis robusta. J. Pharma. Res. 2012, 2, 1304–1306.
- Journal, A.I. An Indian Journal Note. Anal. Chem. 2007, 6, 4–8.
- Orhan, N.; Akkol, E.; Ergun, F. Evaluation of antiinflammatory and antinociceptive effects of some juniperus species growing in Turkey. Turk. J. Biol. 2012, 36, 719–726.
- Science, A. Assessment of Anti-Inflammatory Activity of Taxus baccata Linn. Bark Extract Satyajit Dutta * G. Mariappan ** Dipankar Sarkar ** Piyali Sarkar ** Table 1: Effect of Taxus baccata (L) bark extracts on Carrageenan-induced paw edema method in rats. Anc. Sci. Life 2010, 29, 19–21.
- Branco, C.D.S.; De Lima, É.D.; Rodrigues, T.S.; Scheffel, T.B.; Scola, G.; Laurino, C.C.F.C.; Moura, S.; Salvador, M. Mitochondria and redox homoeostasis as chemotherapeutic targets of Araucaria angustifolia (Bert.) O. Kuntze in human larynx HEp-2 cancer cells. Chem. Biol. Interact. 2015, 231, 108–118.
- Shashi, B.; Jaswant, S.; Madhusudana, R.J.; Kumar, S.A.; Nabi, Q.G. A novel lignan composition from Cedrus deodara induces apoptosis and early nitric oxide generation in human leukemia Molt-4 and HL-60 cells. Nitric Oxide Biol. Chem. 2006, 14, 72–88.
- Shi, X.; Liu, D.; Zhang, J.; Hu, P.; Shen, W.; Fan, B.; Ma, Q.; Wang, X. Extraction and purification of total flavonoids from pine needles of Cedrus deodara contribute to anti-tumor in vitro. BMC Complement. Altern. Med. 2016, 16, 1–9.
- Basu, L.R.; De, A.; Sarkar, P.; Karak, P.; Dastidar, S.G. Possibilities of developing novel potent antitumor agents from the leaves of Cryptomaria japonica. Int. J. Phytomed. 2016, 8, 404–410.
- Fernandez, A.; Cock, I.E. The therapeutic properties of juniperus communis L.: Antioxidant capacity, bacterial growth inhibition, anticancer activity and toxicity. Pharmacogn. J. 2016, 8, 273–280.
- Muto, N.; Tomokuni, T.; Haramoto, M.; Tatemoto, H.; Nakanishi, T.; Inatomi, Y.; Murata, H.; Inada, A. Isolation of apoptosis- and differentiation-inducing substances toward human promyelocytic leukemia HL-60 cells from leaves of Juniperus taxifolia. Biosci. Biotechnol. Biochem. 2008, 72, 477–484.
- Barnawi, I.O.; Nasr, F.A.; Noman, O.M.; Alqahtani, A.S.; Al-Zharani, M.; Alotaibi, A.A.; Daradka, H.M.; Al-Mishari, A.A.; Alobaid, W.A.; Alqahtani, A.; et al. Induction of apoptosis and cell cycle arrest by chloroform fraction of Juniperus phoenicea and chemical constituents analysis. Open Chem. 2021, 19, 119–127.
- Machana, S.; Weerapreeyakul, N.; Barusrux, S.; Nonpunya, A.; Sripanidkulchai, B.; Thitimetharoch, T. Cytotoxic and apoptotic effects of six herbal plants against the human hepatocarcinoma (HepG2) cell line. Chin. Med. 2011, 6, 2–9.
- MacHana, S.; Weerapreeyakul, N.; Barusrux, S.; Thumanu, K.; Tanthanuch, W. FTIR microspectroscopy discriminates anticancer action on human leukemic cells by extracts of Pinus kesiya; Cratoxylum formosum ssp. pruniflorum and melphalan. Talanta 2012, 93, 371–382.
- Thu, N.B.; Trung, T.N.; Ha, D.T.; Khoi, N.M.; Hung, T.V.; Hien, T.T.; Namhui, Y.; Bae, K. Screening of Vietnamese medicinal plants for cytotoxic activity. Nat. Prod. Sci. 2010, 16, 43–49.
- Chattopadhyay, S.K.; Kumar, T.R.S.; Maulik, P.R.; Srivastava, S.; Garg, A.; Sharon, A.; Negi, A.S.; Khanuja, S.P.S. Absolute configuration and anticancer activity of taxiresinol and related lignans of Taxus wallichiana. Bioorg. Med. Chem. 2003, 11, 4945–4948.
- Kaushik, P.; Lal Khokra, S.; Rana, A.C.; Kaushik, D. Evaluation of anticancer activity of Pinus roxburghii sarg. Against IMR-32 human neuroblastoma cancer cell line. Int. J. Pharm. Clin. Res. 2015, 7, 105–108.
- Jiang, P.; Zhang, Q.; Zhao, Y.; Xiong, J.; Wang, F.; Zhang, T.; Zhang, C. Extraction, Purification, and Biological Activities of Polysaccharides from Branches and Leaves of Taxus cuspidata S. Et Z. Molecules 2019, 24, 2926.
- Mukherjee, A.; Sikdar, S.; Bishayee, K.; Paul, A.; Ghosh, S.; Boujedaini, N.; Khuda-Bukhsh, A.R. Ethanolic extract of Thuja occidentalis blocks proliferation of A549 cells and induces apoptosis in vitro. J. Chin. Integr. Med. 2012, 10, 1451–1459.
- Khuda-Bukhsh, A.R.; Biswas, R.; Mandal, S.K.; Dutta, S.; Bhattacharyya, S.S.; Boujedaini, N. Thujone-rich fraction of Thuja occidentalis demonstrates major anti-cancer potentials: Evidences from in vitro studies on A375 cells. Evid. Based Complement. Altern. Med. 2011, 2011, 568148.
- Velmurugan, B.K.; Rathinasamy, B.; Lohanathan, B.P.; Thiyagarajan, V.; Weng, C.F. Neuroprotective role of phytochemicals. Molecules 2018, 23, 2485.
- Gitler, A.D.; Dhillon, P.; Shorter, J. Neurodegenerative disease: Models, mechanisms, and a new hope. DMM Dis. Model. Mech. 2017, 10, 499–502.
- Venkatesan, R.; Ji, E.; Kim, S.Y. Phytochemicals that regulate neurodegenerative disease by targeting neurotrophins: A comprehensive review. Biomed Res. Int. 2015, 2015, 814068.
- Johri, A.; Beal, M.F. Mitochondrial dysfunction in neurodegenerative diseases. J. Pharmacol. Exp. Ther. 2012, 342, 619–630.
- Yang, J.L.; Lin, Y.T.; Chuang, P.C.; Bohr, V.A.; Mattson, M.P. BDNF and exercise enhance neuronal DNA repair by stimulating CREB-mediated production of apurinic/apyrimidinic endonuclease 1. NeuroMol. Med. 2014, 16, 161–174.
- Huang, E.J.; Reichardt, L.F. Neurotrophins: Roles in neuronal development and function. Annu. Rev. Neurosci. 2001, 24, 677–736.
- Agrawal, M.; Biswas, A.; Levy, C.E. Molecular diagnostics of neurodegenerative disorders. Front. Mol. Biosci. 2015, 2, 1–10.
- Olivares, D.; Deshpande, V.K.; Shi, Y.; Lahiri, D.K.; Greig, N.H.; Rogers, J.T.; Huang, X. N-Methyl D-Aspartate (NMDA) Receptor Antagonists and Memantine Treatment for Alzheimer’s Disease, Vascular Dementia and Parkinson’s Disease. Curr. Alzheimer Res. 2012, 9, 746–758.
- Briffa, M.; Ghio, S.; Neuner, J.; Gauci, A.J.; Cacciottolo, R.; Marchal, C.; Caruana, M.; Cullin, C.; Vassallo, N.; Cauchi, R.J. Extracts from two ubiquitous Mediterranean plants ameliorate cellular and animal models of neurodegenerative proteinopathies. Neurosci. Lett. 2017, 638, 12–20.
- Physiology, G.; Waczulikova, I.; Kilanczyk, E.; Bryszewska, M. The effect of Pycnogenol on the erythrocyte membrane fluidity. Gen. Physiol. Biophys. 2004, 23, 39–51.
- Voss, P.; Horakova, L.; Jakstadt, M.; Kiekebusch, D.; Grune, T. Ferritin oxidation and proteasomal degradation: Protection by antioxidants. Free Radic. Res. 2006, 40, 673–683.
- Kim, C.S.; Subedi, L.; Kim, S.Y.; Choi, S.U.; Kim, K.H.; Lee, K.R. Diterpenes from the Trunk of Abies holophylla and Their Potential Neuroprotective and Anti-inflammatory Activities. J. Nat. Prod. 2016, 79, 387–394.
- Machado, F.D.; Kuo, J.; Ongaratti, B.R.; Medeiros, N.D.; Salvador, M.; Dani, C.; Funchal, C. Antioxidant and neuroprotective potential of extract of Brazilian pine Araucaria angustifolia bracts against oxidative stress induced by sodium azide in hippocampus. Integr. Pharmacol. Toxicol. Genotoxicol. 2015, 1, 16–20.
- Thiago, C.; Patrícia de Brum, V.; Patrícia Gomes da, S.; Marines de Avila, H.; Graziele Daiane, S.; Michele Stach, C.; Antônio Batista, P.; Sidnei, M.; Andreas Sebastian, M.; Chariston André Dal, B. Mechanism of the Entomotoxic Activity Induced by Araucaria Angustifolia Methanolic Extract in Nauphoeta Cinerea Lobster Cockroaches. J. Bot. Res. 2017, 1, 38–49.
- Zhao, Z.; Dong, Z.; Ming, J.; Liu, Y. Cedrin identified from Cedrus deodara (Roxb.) G. Don protects PC12 cells against neurotoxicity. Nat. Prod. Res. 2018, 6419, 1455–1458.
- Lee, J.S.; Kim, H.G.; Lee, H.W.; Han, J.M.; Lee, S.K.; Kim, D.W.; Saravanakumar, A.; Son, C.G. Hippocampal memory enhancing activity of pine needle extract against scopolamine-induced amnesia in a mouse model. Sci. Rep. 2015, 5, 1–10.
- Lee, J.S.; Kim, H.G.; Lee, H.W.; Kim, W.Y.; Ahn, Y.C.; Son, C.G. Pine needle extract prevents hippocampal memory impairment in acute restraint stress mouse model. J. Ethnopharmacol. 2017, 207, 226–236.
- Forouzanfar, F.; Ghorbani, A.; Hosseini, M. Hydroalcoholic extract of needles of Pinus eldarica enhances pentobarbital-induced sleep: Possible involvement of GABAergic system. Avicenna J. Phytomed. 2016, 6, 449.
- Wang, C.; He, L.; Yan, M. Effects of polyprenols from pine needles of Pinus massoniana on ameliorating cognitive impairment in a D -galactose-induced mouse model. Age 2014, 36, 9676.
- Khan, M.M.; Kempuraj, D.; Thangavel, R.; Zaheer, A. Protection of MPTP-induced neuroinflammation and neurodegeneration by Pycnogenol. Neurochem. Int. 2013, 62, 379–388.
- Kabra, A.; Baghel, U.S.; Hano, C.; Martins, N.; Khalid, M.; Sharma, R. Neuroprotective potential of Myrica esulenta in Haloperidol induced Parkinson’s disease. J. Ayurveda Integr. Med. 2020, 11, 448–454.
- Lokesh, D.; Amitabha, D.; Sachin, A.; Avijeet, J. Neuropharmacological Exploration of Thuja Occidentalis Linn. Int. Res. J. Pharm. 2011, 2, 143–148.
- Lee, S.; Choi, C.; Kim, J.; Lim, S.; Jung, H. The Antioxidant Activities and Neuroprotective Effects of Hot Water Extracts from Torreyae Semen. Korea J. Herbol. 2017, 32, 41–48.
- Deture, M.A.; Dickson, D.W. The neuropathological diagnosis of Alzheimer’s disease. Mol. Neurodegener. 2019, 14, 1–18.
- Jahn, H. Memory loss in Alzheimer’s disease. Dialogues Clin. Neurosci. 2013, 15, 445–454.
- Tanvir Kabir, M.; Sahab Uddin, M.; Al Mamun, A.; Jeandet, P.; Aleya, L.; Mansouri, R.A.; Md Ashraf, G.; Mathew, B.; Bin-Jumah, M.N.; Abdel-Daim, M.M. Combination drug therapy for the management of alzheimer’s disease. Int. J. Mol. Sci. 2020, 21, 3272.
- Durães, F.; Pinto, M.; Sousa, E. Old drugs as new treatments for neurodegenerative diseases. Pharmaceuticals 2018, 11, 44.
- Barbier, P.; Zejneli, O.; Martinho, M.; Lasorsa, A.; Belle, V.; Smet-Nocca, C.; Tsvetkov, P.O.; Devred, F.; Landrieu, I. Role of tau as a microtubule-associated protein: Structural and functional aspects. Front. Aging Neurosci. 2019, 10, 1–14.
- Cory, H.; Passarelli, S.; Szeto, J.; Tamez, M.; Mattei, J. The Role of Polyphenols in Human Health and Food Systems: A Mini-Review. Front. Nutr. 2018, 5, 1–9.
- Rensink, A.A.M.; De Waal, R.M.W.; Kremer, B.; Verbeek, M.M. Pathogenesis of cerebral amyloid angiopathy. Brain Res. Rev. 2003, 43, 207–223.
- Tanaka, M.; Saito, S.; Inoue, T.; Satoh-Asahara, N.; Ihara, M. Novel therapeutic potentials of taxifolin for amyloid-β-associated neurodegenerative diseases and other diseases: Recent advances and future perspectives. Int. J. Mol. Sci. 2019, 20, 2139.
- Sharma, L.; Sharma, A.; Goyal, R.; Alam, J. Pinus roxburghii Sarg. Ameliorates alzheimer’s disease-type neurodegeneration and cognitive deficits caused by intracerebroventricular-streptozotocin in rats: An in vitro and in vivo study. Indian J. Pharm. Sci. 2020, 82, 861–870.
- Hassaan, Y.; Handoussa, H.; El-Khatib, A.H.; Linscheid, M.W.; El Sayed, N.; Ayoub, N. Evaluation of plant phenolic metabolites as a source of Alzheimer’s drug leads. Biomed Res. Int. 2014, 2014, 843263.
- Arbo, B.D.; André-Miral, C.; Nasre-Nasser, R.G.; Schimith, L.E.; Santos, M.G.; Costa-Silva, D.; Muccillo-Baisch, A.L.; Hort, M.A. Resveratrol Derivatives as Potential Treatments for Alzheimer’s and Parkinson’s Disease. Front. Aging Neurosci. 2020, 12, 1–15.
- Ahmed, T.; Javed, S.; Javed, S.; Tariq, A.; Šamec, D.; Tejada, S.; Nabavi, S.F.; Braidy, N.; Nabavi, S.M. Resveratrol and Alzheimer’s Disease: Mechanistic Insights. Mol. Neurobiol. 2017, 54, 2622–2635.
- Maimoona, A.; Naeem, I.; Saddiqe, Z.; Jameel, K. A review on biological, nutraceutical and clinical aspects of French maritime pine bark extract. J. Ethnopharmacol. 2011, 133, 261–277.
- Peng, Q.L.; Zard, A.R.B.; Lau, B.H.S. Pycnogenol protects neurons from amyloid- b peptide-induced apoptosis. Mol. Brain Res. 2002, 104, 55–65.
- Paarmann, K.; Prakash, S.R.; Krohn, M.; Möhle, L.; Brackhan, M.; Brüning, T.; Eiriz, I.; Pahnke, J. French maritime pine bark treatment decelerates plaque development and improves spatial memory in Alzheimer’s disease mice. Phytomedicine 2019, 57, 39–48.
- Fang, C.; Lv, L.; Mao, S.; Dong, H.; Liu, B. Cognition Deficits in Parkinson’s Disease: Mechanisms and Treatment. Parkinsons Dis. 2020, 2020, 2076942.
- Dias, V.; Junn, E.; Mouradian, M.M. The role of oxidative stress in parkinson’s disease. J. Parkinsons Dis. 2013, 3, 461–491.
- Chen, L.; Ding, Y.; Cagniard, B.; Van Laar, A.D.; Mortimer, A.; Chi, W.; Hastings, T.G.; Un, J.K.; Zhuang, X. Unregulated cytosolic dopamine causes neurodegeneration associated with oxidative stress in mice. J. Neurosci. 2008, 28, 425–433.
- Zoccarato, F.; Toscano, P.; Alexandre, A. Dopamine-derived dopaminochrome promotes H2O2 release at mitochondrial Complex I: Stimulation by rotenone, control by Ca2+, and relevance to Parkinson disease. J. Biol. Chem. 2005, 280, 15587–15594.
- Ebrahimi-Fakhari, D.; Wahlster, L.; McLean, P.J. Protein degradation pathways in Parkinson’s disease: Curse or blessing. Acta Neuropathol. 2012, 124, 153–172.
- Javed, H.; Nagoor Meeran, M.F.; Azimullah, S.; Adem, A.; Sadek, B.; Ojha, S.K. Plant Extracts and Phytochemicals Targeting α-Synuclein Aggregation in Parkinson’s Disease Models. Front. Pharmacol. 2019, 9, 1555.
- Corona, J.C. Natural Compounds for the Management of Parkinson’s Disease and Attention-Deficit/Hyperactivity Disorder. Biomed Res. Int. 2018, 2018, 4067597.
- Ríos, J.L.; Onteniente, M.; Picazo, D.; Montesinos, M.C. Medicinal Plants and Natural Products as Potential Sources for Antiparkinson Drugs. Planta Med. 2016, 82, 942–951.
- Bais, S.; Gill, N.S.; Kumar, N. Neuroprotective Effect of Juniperus communis on Chlorpromazine Induced Parkinson Disease in Animal Model. Chin. J. Biol. 2015, 2015, 1–7.
- Zhang, F.; Shi, J.S.; Zhou, H.; Wilson, B.; Hong, J.S.; Gao, H.M. Resveratrol protects dopamine neurons against lipopolysaccharide-induced neurotoxicity through its anti-inflammatory actions. Mol. Pharmacol. 2010, 78, 466–477, reprinted in Mol. Pharmacol. 2010, 78, 981.
- Fang, X.S.; Hao, J.F.; Zhou, H.Y.; Zhu, L.X.; Wang, J.H.; Song, F.Q. Pharmacological studies on the sedative-hypnotic effect of Semen Ziziphi spinosae (Suanzaoren) and Radix et Rhizoma Salviae miltiorrhizae (Danshen) extracts and the synergistic effect of their combinations. Phytomedicine 2010, 17, 75–80.
- Akram, M.; Daniyal, M.; Munir, N.; Mohiuddin, E.; Sultana, S. Medicinal Plants Combating Against Insomnia: A Green Anti-Insomnia Approach. J. Nerv. Ment. Dis. 2019, 207, 927–935.
- Gooneratne, N.S.; Vitiello, M.V. Sleep in Older Adults. Normative Changes, Sleep Disorders, and Treatment Options. Clin. Geriatr. Med. 2014, 30, 591–627.
- Atkin, T.; Comai, S.; Gobbi, G. Drugs for insomnia beyond benzodiazepines: Pharmacology, clinical applications, and discovery. Pharmacol. Rev. 2018, 70, 197–245.
- Sateia, M.J.; Buysse, D.J.; Krystal, A.D.; Neubauer, D.N. Adverse effects of hypnotic medications. J. Clin. Sleep Med. 2017, 13, 839.
- Woo, J.; Yang, H.; Yoon, M.; Gadhe, C.G.; Pae, A.N.; Cho, S.; Justin Lee, C. 3-Carene, a phytoncide from pine tree has a sleep-enhancing effect by targeting the GABAA-benzodiazepine receptors. Exp. Neurobiol. 2019, 28, 593–601.





