
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Judyta Cielecka-Piontek | + 5529 word(s) | 5529 | 2021-01-20 07:20:55 | | | |
| 2 | Vivi Li | + 4 word(s) | 5533 | 2021-02-07 03:43:48 | | |
Video Upload Options
Cannabis sativa L. turned out to be a valuable source of chemical compounds of various structures, showing pharmacological activity. The most important groups of compounds include phytocannabinoids and terpenes. The pharmacological activity of Cannabis (in epilepsy, sclerosis multiplex (SM), vomiting and nausea, pain, appetite loss, inflammatory bowel diseases (IBDs), Parkinson’s disease, Tourette’s syndrome, schizophrenia, glaucoma, and coronavirus disease 2019 (COVID-19), which has been proven so far, results from the affinity of these compounds predominantly for the receptors of the endocannabinoid system (the cannabinoid receptor type 1 (CB1), type two (CB2), and the G protein-coupled receptor 55 (GPR55) but, also, for peroxisome proliferator-activated receptor (PPAR), glycine receptors, serotonin receptors (5-HT), transient receptor potential channels (TRP), and GPR, opioid receptors. The synergism of action of phytochemicals present in Cannabis sp. raw material is also expressed in their increased bioavailability and penetration through the blood–brain barrier.
1. Introduction
Cannabis sativa L. is one of the most ancient plant species used by humans for many purposes. Besides medicinal use, it also serves as a fiber; food; and is an important raw material for the production of rope, textiles, and paper. Surprisingly, the first human use of Cannabis is reported to be 10,000 years ago, at the end of the Ice Age [1]. The most ancient Chinese Pharmacopoea (written in the first century before the current era), the “Shen Nung Pen Ts’ao Ching”, is the first historical evidence of the use of Cannabis in traditional medicine. It includes all the traditional remedies used and orally bequeathed for over two thousand years when it was emperor Shen Nung’s (2700 years before the current era) reign [2]. The Cannabis plant first arrived in Europe with Scythians or proto-Scythians moving from Central Asia about 3500 years ago [3]. Modern reports on medical marijuana date back to the nineteenth century, when the Irish doctor William Brooke O’Shaughnessy performed experiments on the pharmacological and toxic properties of Cannabis [4][5]. He suggested Indian hemp as a treatment for tetanus and other convulsive diseases. In 1851, for the first time, Cannabis was included in the third edition of the United States Pharmacopoeia (USP) with the use of Cannabis flowers as an analgesic, anticonvulsant, and hypnotic [6]. In the second half of the nineteenth century, there was a significant increase in the use of Cannabis in medicine and research on its phytochemistry and pharmacology. At the beginning of the twentieth century, as the recreational use of Cannabis grew, in 1937, the “Marijuana Tax Act” was introduced, and in 1941, Cannabis was removed from the twelfth edition of the U.S. Pharmacopeia by the American Medical Association [5]. In the 1960s, the use of recreational Cannabis increased throughout the Western world. The discovery of the chemical structure of Δ9-tetrahydrocannabinol (Δ9-THC), which was identified by Gaoni and Mechoulam, boosted multidirectional research on Cannabis [7].
The botanical definition of Cannabis sativa L. has been a goal of many studies due to its morphological and chemical differentiation. The currently accepted classification distinguishes two subspecies—namely, ssp. sativa and ssp. indica. Within each of them, there are two main varieties, cultivated and wild. The most important from a medicinal point of view is C. sativa ssp. sativa var. sativa (so-called C. sativa) and C. sativa ssp. indica var. indica (so-called C. indica). C. sativa ssp. sativa var. spontanea (the so-called C. ruderalis) is rarely found and much less exploited. The plant develops both male and female specimens. Dried inflorescences from female plants (Cannabis flos) are known as marijuana. It contains resin, which is a source of phytocannabinoids. Purified resin is known as hashish. So-called C. indica is usually grown for recreational use, whereas so-called C. sativa has recently gained much attention as a source of medical marijuana.
The possibility of interactions between components of various Cannabis products is high due to the richness of the compounds. Hitherto, over 100 cannabinoids have been identified in the Cannabis sp., and the best known, famous, and responsible for its pharmacological activity are the psychoactive Δ9-tetrahydrocannabinol (THC) and cannabidiol (CBD) [8]. These cannabinoids are produced in the plant in acid form and require decarboxylation, which is caused mainly by a high temperature. There are also other cannabinoids in the plant, such as cannabigerol (CBG), cannabichromene (CBC), cannabidivarin (CBDV), and cannabinol (CBN). However, the Cannabis sp. comprises not only cannabinoids. The presence of over 600 compounds has been reported so far, with over 150 different terpenes—including monoterpenes, sesquiterpenes, and, also, di- and triterpenes, as well as sugars, steroids, fatty acids, non-cannabinoid phenols, flavonoids, phenylpropanoids, alkanes, and nitrogenous compounds [9][10].
The grounds for the conclusion that there are interactions between Cannabis plant components is that the Cannabis flos extract has a higher potency than the individual isolated cannabinoids, which is called the “entourage effect” [8]. As a result, a more efficient pharmacological effect can be obtained and a higher therapeutic efficacy among patients. Individual cannabinoids can interact synergistically with each other, which is called the “intra-entourage effect”. The enhanced therapeutic effect of cannabinoids through other plant secondary metabolites such as terpenoids is called the “inter-entourage effect”. Terpenoids significantly alter the biological activity of cannabinoids.
Cannabis potency was initially defined by the content of THC. Over the years, the THC content in products available worldwide has elevated [11]. It is worth emphasizing that the use of products with high THC contents and low CBD contents may lead to side effects. A case-control study showed that the administration of Cannabis rich in THC might be associated with a raised risk of psychosis in patients, mostly when the CBD level was relatively low [12]. The THC/CBD ratio is significant in determining the potency of the Cannabis plant. CBD does not cause euphoria. Scientists even confirmed its antipsychotic and anxiolytic effect and the reduction of some side effects of THC [13]. Using CBD and THC together is considered to have a milder effect. Additionally, pretreatment with CBD before the administration of THC seems to be beneficial. In one study, it prevented the transient psychotic symptoms induced by THC [14]. That is why it is so crucial to antagonize the psychotic and other side effects of THC by using preparations containing both THC and CBD.
2. Pharmacological Application
The effectiveness of extracts or drugs from Cannabis sp. or synthetic cannabinoids in diseases (Figure 1.) has been shown below. Data indicating the clinical effectiveness of cannabinoid compounds in such a wide array of disease units is an argument for their multitarget action. The ability to antagonize or agonize CB1 and CB2 receptors results in the stimulation or inhibition of the endocannabinoid system, both in the peripheral tissues and in the central nervous system. As indicated below, Cannabis sp. extracts have significant therapeutic potential. Still, due to the need to continue further clinical trials, they should be used as one of the last therapeutic procedures.
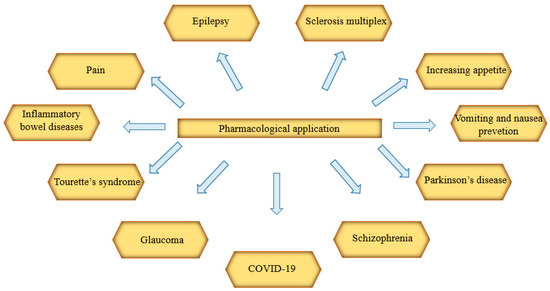
Figure 1. Pharmacological applications of extracts or drugs from Cannabis sp. or synthetic cannabinoids in diseases. COVID-19: coronavirus disease 2019.
2.1. Epilepsy
Epilepsy is believed to be the most common noninfectious neurological disease. Moreover, one-third of patients suffer from seizures resistant to standard antiepileptic drugs. Antiepileptic drugs have been used for over 160 years. They prevent seizures, but there is no evidence that they have disease-modifying properties. They affect the mechanisms causing the brain’s excitability. Antiepileptic drugs disrupt the creation and spread of epileptic hyperactivity. These drugs act on the following mechanisms: (i) modulation of voltage-dependent sodium channels (carbamazepine and phenytoin), calcium channels (ethosuximide), and potassium channels (retigabine and ezogabine); (ii) enhancement of inhibited GABA transmission (benzodiazepines, tiagabine, and vigabatrin); (iii) weakening of the stimulant glutamate transmission (perampanel); and (iv) presynaptic modification of neurotransmitter release (levetiracetam, brivaracetam, gabapentin, and pregabalin) [15]. Drugs introduced before 1989 are called “first-generation” drugs, and those introduced later are named “second-generation” drugs. Medicines that have been recently approved (brivaracetam, eslicarbazepine acetate, lacosamide, perampanel, rufinamide, and stiripentol) that are improvements to classic drugs or rely on novel mechanisms are called “third-generation” drugs [16]. Since 2018, the last group has also included CBD.
Drug-resistant epilepsy is diagnosed when symptom control cannot be achieved despite the use of at least two different antiepileptic drugs in an appropriate dose [17]. Epilepsy with the greatest resistance to treatment includes Lennox-Gastaut syndrome (LGS), Dravet syndrome (DS), fever-related epilepsy syndrome, and epilepsy in tuberous sclerosis syndrome (TSC) [18]. The patient’s quality of life and cognitive and psychosocial problems significantly deteriorate.
Due to the occurrence of drug-resistant epilepsy and severe side effects associated with the use of traditional antiepileptic drugs, preparations with higher safety and effectiveness are being sought. The antiepileptic use of Cannabis dates back to ancient times. It is generally accepted that Cannabis benefits in anticonvulsant therapy are linked to CBD. However, the mechanism of CBD action in the treatment of epilepsy is not fully understood. The orphan G-protein coupled receptor, GPR55 antagonism, is mentioned as the mechanism. Recent studies have shown that CBD significantly reduces the number of seizures. Moreover, it seems that only mild side effects occur. Among these, increased liver enzymes levels are improved with a continuity of therapy or by dose reduction. However, it should be emphasized that CBD is most often used in studies with standard antiepileptic therapy, so it is difficult to assess whether it has antiepileptic effects or only enhances the effects of traditional drugs [19]. The FDA approved a 99% CBD extract called Epidiolex in 2018 for the treatment of seizures associated with LGS, DS, or TSC in patients one years of age and older [20].
In recent years, there has been an increased interest in the use of Cannabis as part of the treatment of childhood epilepsy, among others, due to numerous media reports on children whose symptoms improved with Cannabis. Many parents of children struggling with drug-resistant epilepsy turn to alternative therapeutic measures, sometimes with doctors’ help and sometimes on their own [17]. A lot of attention was focused on a Colorado family who was looking for alternative treatments for a girl suffering from DS, Charlotte Figi. Her supportive treatment included a CBD-rich Cannabis strain that was later named “Web Charlotte.” After three months of using it, the number of seizures dropped from 300 per week to 30 per month. However, after 20 months of treatment, the monthly number of attacks was about three, and the girl started talking and walking [20].
The effectiveness of CBD against the drug-resistant seizures that occur in DS has been studied. DS is a type of childhood epilepsy with a low efficacy of traditional antiepileptic drugs and a high mortality rate. A randomized, double-blind clinical trial with a placebo control was performed. The groups were randomly selected among 120 children and young adults, and the patients were subjected to standard antiepileptic treatment. Participants were administered 20-mg/kg body weight CBD daily as an oral solution or placebo. The results were reported after 14 weeks of treatment and are presented in Table 1 [21]. CBD is more effective than a placebo in reducing epileptic seizures in patients suffering from DS. At the same time, such therapy carries a greater risk of side effects [21].
Table 1. Results of the efficacy of CBD in the treatment of epileptic seizures in Dravet’s syndrome in children [21].
| Test Group | Control Group | |
|---|---|---|
| Number of patients | 61 | 59 |
| Applied treatment | 20 mg/kg body weight of cannabidiol daily | Placebo |
| Number of seizures per month after four weeks of standard treatment | 12.4 | 14.9 |
| Number of seizures after 14 weeks of study treatment | 5.9 | 14.1 |
| The proportion of patients in whom the number of seizures decreased by at least 50% | 43% | 27% |
| Percentage of patients who were completely seizure-free | 5% | 0% |
In a phase 3 clinical trial, 225 LGS patients aged 2–55 years with two or more seizures per week were administered CBD oral solution—10 mg/kg body weight per day or twice, in addition to a conventional antiepileptic regimen to patients in the test group and placebo for 14 weeks. The median percent reduction in seizure frequency from the baseline was 37.2% in the CBD 10 mg/kg/day group, 41.9% in the CBD 20 mg/kg/day group, and 17.2% in the placebo group [22]. The reduction in seizure frequency was dose-related.
Among 171 patients aged 2–55 years diagnosed with LGS, whose seizure frequency over four weeks was at least two per week and who did not respond to treatment with at least two antiepileptic drugs (the most common were lamotrigine, valproate, and clobazam), 86 patients received CBD at a dose of 20 mg/kg/day for 14 weeks; the rest obtained a placebo [23]. After 14 weeks of treatment, the monthly seizure frequency had decreased by a median of 43.9% from the baseline in the CBD group. A 50% or more reduction in seizure frequency was reported in 44% of patients in the CBD group and 24% of the placebo group. Serious adverse events (pneumonia, viral infection, increase in alanine aminotransferase, increase in aspartate aminotransferase, and increase in γ-glutamyltransferase) occurred in 23.26% of patients in the CBD-treated group and in 4.71% in the placebo group. Serious treatment-emergent (increased levels of alanine aminotransferase, aspartate aminotransferase, and γ-glutamyltransferase) occurred in four patients in the CBD group. Instead, the most common not serious adverse events (vomiting, diarrhea, loss of appetite, and somnolence) occurred in 61.63% of the CBD group and 50.59% of the patients in the placebo group. The study showed many side effects in patients using CBD, but in this group, the reduction in seizure frequency was greater than in the placebo group.
2.2. Sclerosis Multiplex
Multiple sclerosis is the most common inflammatory demyelinating disease of the central nervous system. It arises as a result of an autoimmune response directed against myelin, which, in consequence, leads to worsening neurodegeneration and disability [24]. Common symptoms of multiple sclerosis include vision problems, pain, muscle weakness, balance problems, and paralysis [25]. In addition, patients who suffer from spasticity, which is defined as the activation of muscles without the patient’s will, are under increased risk of complications and are subject to pain and problems with falling asleep [26]. These symptoms are caused by progressive abnormal neuronal transmission. The disease is initially characterized by long asymptomatic periods abruptly interrupted by relapses, usually lasting several days or weeks [25]. The beginning of modern treatment of multiple sclerosis can be associated with the introduction of interferon beta and glatiramer acetate in the treatment of multiple sclerosis with relapsing remission [27]. Then, monoclonal antibodies were introduced, such as natalizumab, and, later, also alemtuzumab and ocrelizumab. Oral medicines such as fingolimod, teriflunomide, dimethyl fumarate, and cladribine have also been developed.
New therapeutic options are still being sought, especially for continuously progressive forms of the disease. Currently, the available therapies may delay disease progression and reduce the number of attacks but do not improve the patients’ quality of life [28][24].
Numerous clinical and preclinical studies give hope that cannabinoids derived from Cannabis sativa L. can be used to control chronic pain and spasticity. Preclinical studies have also demonstrated the potential use of endocannabinoids’ neuroprotective properties and partial inhibition of disease progression [24]. Nabiximols, THC, and oral Cannabis extract are believed to be helpful in the treatment of spasticity. Sativex® is currently the only pharmaceutical product approved for this indication. However, Sativex® is not a first-line treatment. It is indicated for the relief of moderate-to-severe spasticity symptoms in adult sclerosis multiplex (SM) patients who are unresponsive to other spasticity-relieving medicinal products and show a clinically significant improvement in spasticity symptoms in the initial stage of therapy. Medical marijuana reduces the pain and spasticity in SM but negatively affects balance and posture. Nabiximols may also be effective in disturbance of the normal bladder function, which is common in this condition [25]. The effects of individual cannabinoids on pain and muscle spasticity in SM patients are shown in Table 2.
Table 2. Cannabinoid therapies and their impacts on the symptoms of multiple sclerosis determined in clinical trials [25]. ↓: decrease.
| The Form | Effects on Symptoms |
|---|---|
| smoked marijuana | spasticity ↓, pain ↓ |
| nabiximols | spasticity ↓, pain ↓ |
| oral Cannabis extract | muscle spasticity ↓ |
| dronabinol | spasticity ↓, pain ↓ |
| nabilone (+gabapentin) | pain ↓ |
Studies of the use of medical marijuana in the treatment of SM suggest a reduction in spasticity after using phytocannabinoids, but it is not statistically significant. However, there was a clinically significant subjective reduction in spasticity reported by patients. Cannabinoids appear to be well-tolerated by patients using them as add-on therapy. It enables them to improve their score on the Ashworth Scale, which is a measure of spasticity by measuring the resistance that occurs when soft tissue is passively stretched. However, many studies give contradictory results, often without statistical significance. Unfortunately, the use of Cannabis harms cognitive functions, and as many as 40–60% of SM patients suffer from it, so when starting cannabinoid therapy, the patient’s cognitive functions should be monitored. Medical marijuana has a positive impact on SM symptoms. However, medical marijuana does not slow the disease’s progression [28].
2.3. Vomiting and Nausea Prevention
Approximately 45–61% of cancer patients suffer from chemotherapy-induced nausea and vomiting (CINV). In antiemetic prophylaxis during chemotherapy, the following are used: (i) drugs from the group of 5-HT3 receptor antagonists—ondansetron, granisetron, tropisetron, dolasetron, palonosetron; (ii) drugs from the group of neurokinin-1 receptor antagonists—aprepitant; (iii) corticosteroids—the most commonly recommended dexamethasone or methylprednisolone; and (iv) complementary drugs: dopamine receptor antagonists (metoclopramide), phenothiazine derivatives (chlorpromazine), antihistamines, and butyrophenone derivatives (haloperidol and droperidol) [29]. The guidelines propose the use of antiemetic drugs in monotherapy or according to a schedule of two-to-three drugs, depending on the degree of risk of nausea and vomiting and the individual sensitivity of the patient. If the antiemetic prophylaxis proves ineffective, additional antiemetic drugs are used when needed. An acute episode due to the stimulation of gastrointestinal receptors occurs up to 24 h after the initiation of chemotherapy, and delayed CINV occurs within one–five days, mainly due to the activation of receptors located in the brain. Statistically, the delayed effect is more frequent, and the incidence of nausea outweighs the incidence of vomiting [30].
According to studies, the incidence of nausea and vomiting can be reduced by activating the CB1 receptor with THC [30]. As a result of receptor stimulation, the proemetic effects of dopamine and serotonin are abolished [31]. In the fight against CINV, oral cannabinoid preparations are more effective than a placebo. However, compared with other antiemetics, studies lead to different conclusions. Some of them suggest the superiority of THC, others, a similar effectiveness, and still others show that the best effect is obtained when combining traditional drugs with cannabinoids [26].
After reviewing randomized controlled trials, the conclusions are that administering cannabinoids to patients during chemotherapy allows for 70% control of nausea and 66% control of vomiting, while, after administration of a placebo, the results reached 57% and 36%, respectively [32]. During the research carried out in the seventies and eighties, THC’s superiority in the prevention of nausea and vomiting occurring during radio- and chemotherapy over the placebo and antiemetic drugs used at that time was found. Currently, however, we are already in possession of more effective drugs for this indication. The comparisons show that newer drugs can control nausea in 90% of patients, compared to 30% with cannabinoids. For this reason, they are not used in the first line of treatment but only as an adjunct or second-line treatment [116]. Cesamet® or Marinol® can be used in the treatment of CINV [28].
2.4. Pain
The endocannabinoid system is involved in pain control, and therefore, Cannabis may be used to relieve it [28]. The most common medical use of Cannabis is pain management, but it is not equally effective for all types of pain. Most likely, it results from different mechanisms of pain formation. Studies show that cannabinoids cannot combat acute pain, but they can only relieve chronic pain to a limited extent [33]. The pharmacotherapy of chronic pain is incredibly difficult, as is the pain that continues despite the tissue’s healing.
Neuropathic pain is a type of chronic pain that occurs following damage to the peripheral or central nervous system. It appears as a result of an injury, disease, cancer, immune disorder, or an effect of certain drugs. Often, patients then experience allodynia, hyperalgesia, depression, sleep problems, and anxiety, and their contact with society is usually limited. According to Special Interest Group on Neuropathic Pain, the guidelines for the treatment of neuropathic pain include: (i) first-line therapy: gabapentinoids—gabapentin and pregabalin, tricyclic antidepressants—amitriptyline, and serotonin-norepinephrine reuptake inhibitors—duloxetine and venlafaxine; (ii) second-line therapy: opioids—tramadol and tapentadol and topical treatment—lidocaine and capsaicin; and (iii) third-line therapy: strong opioids—morphine and oxycodone and a neurotoxin—botulinum toxin [34]. Due to the side effects of using available drugs in the treatment of neuropathic pain, and the fact that more than 50% of people do not achieve the expected improvement, new therapeutic agents are sought continuously [35].
2.5. Increasing Appetite
Anorexia may appear in the course of such chronic diseases as AIDS, cancer, or anorexia nervosa [28]. Progestational agents (megestrol) are often used to increase the appetite, and in the short-term, the use of corticosteroids, the oral ghrelin mimetic anamorelin, can be used [36]. However, there are many limitations to their use. Hormonal manipulation may be contraindicated in many patients, and long-term corticosteroid therapy may lead to metabolic changes, more fractures, cataracts, gastrointestinal discomfort, and changes in mood or behavior. Seven patients with hematogenous metastatic melanoma and liver metastases suffering from extensive loss of appetite and nausea were administered dronabinol (synthetic delta-tetrahydrocannabinol) capsules for four weeks [37]. Most of the patients reported increased appetite and decreased nausea. These results lasted for several weeks. A more extensive, randomized study showed that oral Dronabinol could help stimulate appetite in advanced cancer patients and slow weight loss. The study involved 469 people who had lost weight due to advanced cancer. They were divided into three groups. The first received 5 mg of Dronabinol daily, the second 800 mg of megestrol daily, and the third received both drugs. In the group of people receiving only megestrol, their appetite was increased by 75%, while their weight by 11%. In those receiving only Dronabinol, these values were 49% and 3%, respectively, and these differences were statistically significant. On the other hand, the administration of both drugs did not provide additional benefits. Numerous animal studies have also confirmed an increase in appetite and the amount of food consumed after the administration of cannabinoids. Studies in the 1970s showed that smoking marijuana increases the number of calories you eat. It seems crucial that, in people affected by cancer, medical marijuana will have antiemetic and appetite-increasing effects [38]. Marinol® has been approved as a drug increasing appetites in patients with AIDS and cancer [39].
Cannabinoids have been established as possible for use in the third line of neuropathic pain treatment, right after antidepressants, anticonvulsants, and opioids. Health Canada has also implemented guidelines recommending Cannabis smoking to combat noncancerous chronic pain. Ultimately, it was established that there is substantial evidence of a positive effect of Cannabis use in chronic pain patients [33]. The best results in the treatment of neuropathic pain are achieved with low doses of THC/CBD or high doses of CBD [35].
2.6. Inflammatory Bowel Diseases
Inflammatory bowel diseases include ulcerative colitis and Crohn’s disease. Although the etiology of these diseases has not been elucidated so far, it is assumed that directing the human immune system against its own intestinal microflora contributes to their formation. In Crohn’s disease, conventional medication includes aminosalicylates, corticosteroids, thiopurines, methotrexate, and thalidomide. Antitumor necrosis factors (infliximab, adalimumab, and certolizumab) and other biologics such as antibodies (vedolizumab and ustekinumab) are also used [40].
The endocannabinoid system is probably responsible for maintaining intestinal homeostasis, because when disturbances occur, it triggers the synthesis of effector molecules so that it may be involved in intestinal inflammation. The presence of the endocannabinoid system within the entire gastrointestinal tract has also been demonstrated. Cannabis has been used in enteritis since ancient times, but unfortunately, research into its use in this indication is rare. Survey studies have shown that patients use Cannabis to self-medicate to relieve inflammatory bowel disease symptoms such as abdominal pain, loss of appetite, and diarrhea. Unfortunately, most studies are characterized by insufficient statistical significance and a problem with the selection of an appropriate placebo, as the effects of Cannabis on the central nervous system are difficult to mask [41].
In an anonymous survey, inflammatory bowel diseases' (IBD) patients reported that Cannabis relieved abdominal pain (83.9%), abdominal cramps (76.8%), arthralgia (48.2%), and, to a lesser extent, diarrhea (28.6%) [42]. A prospective, placebo-controlled study was conducted to test the ability of Cannabis to remission Crohn’s disease. The study involved 21 patients who did not respond to standard treatment, and their Crohn’s disease activity index (CDAI) was over 200. In the study group, patients received a cigarette containing 115 mg of THC twice a day, while, in the second group, patients received a placebo in the form of THC-free Cannabis flowers. Their conditions were assessed during the eight-week treatment and, also, two weeks after its completion. Forty-five percent (5/11) of the test group achieved complete remission (CDAI decrease <150), while, in the placebo group, this effect was found in 10% (1/10) of people. In contrast, a decrease in the CDAI score by a minimum of 100 was observed in 10/11 people receiving Cannabis and 4/10 in the placebo group. It is also worth noting the lack of reported adverse effects of the applied treatment [43].
2.7. Parkinson’s Disease
Parkinson’s disease is a neurological condition involving the degeneration of nigrostriatal dopamine neurons. The disease’s symptoms are primarily muscle stiffness, tremor at rest, bradykinesia, and lack of postural stability. Additionally, depression, anxiety, orthostatic hypotension, constipation, fatigue, and sleep problems may appear. Modern pharmacotherapy consists of supplementing the deficiency of dopamine and symptomatic treatment. In patients with Parkinson’s disease, treatment is symptomatic and focuses on improving the motor and nonmotor symptoms [44]. No disease-modifying drugs are available. Drugs that affect the motor impairment include levodopa, dopamine agonists (ropinirole and pramipexole), monoamine oxidase inhibitors (selegiline and rasagiline), catechol-O-methyltransferase inhibitors (entacapone), anticholinergic drugs (trihexyphenidyl), and choline inhibitors. Depending on the symptoms, antipsychotics or antidepressants are also used. According to this procedure, positive effects on the motor symptoms are obtained, but also, side effects, such as dyskinesia, appear. For this reason, a solution that would holistically approach the treatment of this disease is continuously being sought. Components of the endocannabinoid system are involved in regulating motor functions and dopamine activity. Therefore, Cannabis seems to be an attractive therapeutic option in the treatment of movement and neurodegenerative disorders [45]. Studies have found medical marijuana to help combat Parkinson’s disease’s motor symptoms, such as stiffness, tremors, and bradykinesia. In addition, it provides pain relief and reduces sleep disturbance in patients [46].
2.8. Tourette’s Syndrome
Tourette’s syndrome’s main symptoms are numerous motor tics and the presence of at least one vocal tic. This disease affects approximately 1% of the population, and men are affected by the disease three-to-four times more often than women. The therapy of Tourette’s syndrome may consist of neuropsychological interventions; pharmacotherapy: (i) drugs that block or lessen dopamine—haloperidol and risperidone, (ii) botulinum injections, (iii) attention-deficit hyperactivity disorder medications—methylphenidate and dextroamphetamine, (iv) central adrenergic inhibitors—clonidine and guanfacine, (v) antidepressants—fluoxetine, and (vi) antiseizure medications: topiramate; and deep brain stimulation [47].
The treatments available today often fail to give the desired results or cause serious side effects, and many patients are looking for alternatives or additive supplements to their standard therapy to relieve their symptoms [48]. They reach for dietary supplements or stimulants such as alcohol, nicotine, or marijuana, among others. Two patients using Sativex® showed a clear improvement in motor and vocal tics [28]. However, there is still very limited evidence for THC’s efficacy in relieving the symptoms of Tourette’s syndrome, only from small clinical trials [49]
2.9. Schizophrenia
Schizophrenia is a mental illness emerging most frequently in early adulthood. It involves so-called positive and negative symptoms and cognitive dysfunction. The positive symptoms include hallucinations, paranoia, and delusions, while the negative symptoms include emotional withdrawal, lack of motivation, and decreased affect. The main drugs in the treatment of schizophrenia are antipsychotics (neuroleptics): (i) first generation—haloperidol, perazine, and chlorpromazine; (ii) second generation—olanzapine, quetiapine, and risperidone; and (iii) third generation—aripiprazole, brexpiprazole, and cariprazine. Complementary, non-neuroleptic drugs are also used—antidepressants, sedatives, hypnotics, and mood stabilizers [50].
Studies have shown an increased use of marijuana in people with schizophrenia from the general population. It has been proven that these patients’ endocannabinoid systems are disrupted. The number of their CB1 receptors is increased. Besides, consuming Cannabis sp. elevates the risk of developing schizophrenia. Their harmfulness, however, depends on the content of THC and CBD, because THC promotes psychosis while CBD has an antipsychotic effect. The long-term use of Cannabis disrupts neuronal synchronization in a similar way to people with schizophrenia. Cannabis adversely affects the positive symptoms, and its use leads to more frequent relapses of the disease after achieving remission. However, according to Cannabis use, an improvement in the negative symptoms was found [25].
2.10. Glaucoma
Glaucoma is an eye disorder characterized by optic neuropathy with visual field loss. It is often accompanied by increased pressure in the eyeball and results in the irreversible decrement of vision. Glaucoma can be treated with medication (eye drops), laser therapy, or surgery. The eye drops used: beta blockers—timolol, levobunolol, and carteolol; prostaglandins—latanoprost, travoprost, and tafluprost; alpha-adrenergic agonists—brimonidine and apraclonidine; carbonic anhydrase inhibitors—dorzolamide and brinzolamide; and cholinergic drugs—pilocarpine and carbachol [51].
Studies using oral, intravenous, and inhaled medical marijuana have shown that Δ9-THC reduces the intraocular pressure, which is a risk factor for glaucoma. The patients (60–65%) experience a 25% reduction in intraocular pressure [52]. Moreover, THC has been shown to have neuroprotective potential [53]. The intraocular pressure lowering effect is probably related to the reduction of water production. However, this condition does not last long, necessitating frequent administration of the drug. Another disadvantage is the blood pressure reduction of marijuana and its potential ability to reduce ocular perfusion [54]. There have also been studies using THC in the form of eye drops, but these were less effective. This approach would avoid systemic side effects but require the development of a more effective delivery system [52]. Unfortunately, the advantage of marijuana in terms of safety and effectiveness over currently available glaucoma treatments has not been demonstrated so far [26].
2.11. COVID-19
At the end of 2019, the first-identified cases of a pneumonia of unknown origin emerged from China. The National Health Commission of China reported on the epidemic in early 2020 [55]. Initially, the virus was called “novel coronavirus 2019” (2019-nCoV) by the World Health Organization (WHO), but the name was changed to “severe acute respiratory syndrome coronavirus 2” (SARS-CoV-2) by the international committee of the Coronavirus Study Group (CSG), while the disease was designated “coronavirus disease 2019” by the WHO [56]. This virus is very contagious and has rapidly spread globally. On 11 March 2020, the epidemic of COVID-19 disease caused by the SARS-CoV-2 coronavirus was declared a pandemic by the WHO. Until then, more than 118,000 people across 114 countries, territories, and areas were infected by this virus [57]. During treatment, already known drugs are being used. Neither a vaccine nor a specific cure has been developed yet [58].
The virus is transmitted directly from human-to-human (via coughing, sneezing, and the spread of respiratory droplets or aerosols) or indirectly (via contaminated surfaces). Therefore, isolation is a crucial element in slowing down the spread of the novel virus [57]. Patients go through infection with varying severity, from asymptomatic to mild symptoms through severe stages to fatal cases. According to estimates, approximately 70% of COVID-19 patients are asymptomatic or with mild disease, while the remaining 30% are severe cases [59]. The disease progression may depend on the patient’s genetic differences in the immune system and the individual’s exposome [60].
A mild course of illness symptoms are cough, fever, headache, shortness of breath, muscle pain, sore throat, diarrhea, vomiting, and the most characteristic loss of taste and/or smell [59]. In the severe disease, dyspnea appears, respiratory frequency is ≥30/min, and blood oxygen saturation (SpO2) is ≤93%. The percentage of oxygen supplied is (fraction of inspired oxygen, FiO2) < 300, and/or lung infiltrates are >50% within 24 to 48 h. Severe pneumonia, Acute Respiratory Distress Syndrome, as well as extrapulmonary symptoms and systemic complications such as sepsis and septic shock are observed in patients
In the case of critically ill patients, high concentrations of interleukins (ILs)—IL-2, IL-7, and IL-10; granulocyte colony-stimulating factor (G-SCF); monocyte chemoattractant protein-1 (MCP1/CCL2); macrophage inflammatory protein 1 alpha (MIP1-α/CCL3); CXC-chemokine ligand (CXCL10/IP10); tumor necrosis factor (TNF-α); C-reactive protein (CRP); ferritin; and D-dimers in SARS-CoV-2 are observed in patients’ plasma [61]. The disease’s high severity is related to the cytokine storm, which is possibly induced by the interleukin-6 amplifier [62]. Respiratory failure, septic shock, and/or dysfunction or failure of multiple organs appear in the critical disease [63].
The mechanism of SARS-CoV-2 attacking human cells is similar to that of SARS-CoV. Coronavirus proteins bind to angiotensin-converting enzyme 2 (ACE2) receptors, which allows them to introduce into the host’s cells viral RNA [64]. Extracts with a high content of CBD show the ability to downregulate enzymes: ACE2 and transmembrane serine protease 2 (TMPRSS2), which makes SARS-CoV-2 challenging to enter the human body through oral airways [65][66]. Therefore, it could be used as a prophylaxis before COVID-19. Cannabidiol exhibits direct antioxidant activity by influencing the level and activity of antioxidants and oxidants. It decreases reactive oxygen species (ROS) generation. Moreover, it affects the redox balance according to indirect mechanisms like the modulation of CBD, PPAR-γ, GPR55, and TRP channel receptors 5-HT1A [67]. Moreover, CBD as a PPAR-γ agonist causes pulmonary inflammation limitation, a decrease of fibrosis, and the inhibition of viral replication and immunomodulation. CBD also has anti-inflammatory properties due to the inhibition of cytokine production: IL-1α and β, IL-2, IL-6, interferon-gamma, inducible protein-10, monocyte chemoattractant protein-1, macrophage inflammatory protein-1α, and TNF-α [68][69][70]. Cannabis constituents THC and CBD also inhibit T-helper type 1 (Th1) cytokines and/or promote an in vitro and in vivo Th2 immune response [71]. Th1 and the inflammatory immune response profile predominate in COVID-19. Some viruses may take advantage of host inflammation, and others may be detrimental to host inflammation [71]. In COVID-19, hyperinflammatory immune responses can have serious consequences, such as increased disease severity and patients’ mortality. Therefore, the immunomodulatory effects of cannabinoids can help reduce the severity of symptoms. However, it should be remembered that Cannabis is an unconventional immunomodulatory agent. Its route of administration through smoking is contraindicated in patients suffering from COVID-19. Therefore, inhalation (extracts) or oral administration should be considered to increase patient safety [71]. Unfortunately, there is no reliable evidence in humans confirming effective anti-inflammatory doses for CBD [72].
References
- Okazaki, H.; Kobayashi, M.; Momohara, A.; Eguchi, S.; Okamoto, T.; Yanagisawa, S.; Okubo, S.; Kiyonaga, J. Early Holocene coastal environment change inferred from deposits at Okinoshima archeological site, Boso Peninsula, central Japan. Quat. Int. 2011, 230, 87–94.
- Jiang, H.-E.; Li, X.; Zhao, Y.-X.; Ferguson, D.K.; Hueber, F.; Bera, S.; Wang, Y.-F.; Zhao, L.-C.; Liu, C.-J.; Li, C.-S. A new insight into Cannabis sativa (Cannabaceae) utilization from 2500-year-old Yanghai Tombs, Xinjiang, China. J. Ethnopharmacol. 2006, 108, 414–422.
- Pisanti, S.; Bifulco, M. Medical Cannabis: A plurimillennial history of an evergreen. J. Cell. Physiol. 2019, 234, 8342–8351.
- MacGillivray, N. Sir William Brooke O’Shaughnessy (1808–1889), MD, FRS, LRCS Ed: Chemical pathologist, pharmacologist and pioneer in electric telegraphy. J. Med. Biogr. 2017, 25, 186–196.
- Zuardi, A.W. History of cannabis as a medicine: A review. Braz. J. Psychiatry 2006, 28, 153–157.
- Russo, E.B. History of cannabis and its preparations in saga, science, and sobriquet. Chem. Biodivers. 2007, 4, 1614–1648.
- Gaoni, Y.; Mechoulam, R. Isolation, structure, and partial synthesis of an active constituent of hashish. J. Am. Chem. Soc. 1964, 86, 1646–1647.
- Ferber, S.G.; Namdar, D.; Hen-Shoval, D.; Eger, G.; Koltai, H.; Shoval, G.; Shbiro, L.; Weller, A. The “entourage effect”: Terpenes coupled with cannabinoids for the treatment of mood disorders and anxiety disorders. Curr. Neuropharmacol. 2020, 18, 87–96.
- Namdar, D.; Voet, H.; Ajjampura, V.; Nadarajan, S.; Mayzlish-Gati, E.; Mazuz, M.; Shalev, N.; Koltai, H. Terpenoids and phytocannabinoids co-produced in Cannabis sativa strains show specific interaction for cell cytotoxic activity. Molecules 2019, 24, 3031.
- Booth, J.K.; Bohlmann, J. Terpenes in Cannabis sativa–From plant genome to humans. Plant Sci. 2019, 284, 67–72.
- ElSohly, M.A.; Mehmedic, Z.; Foster, S.; Gon, C.; Chandra, S.; Church, J.C. Changes in cannabis potency over the last 2 decades (1995–2014): Analysis of current data in the United States. Biol. Psychiatry 2016, 79, 613–619.
- Di Forti, M.; Marconi, A.; Carra, E.; Fraietta, S.; Trotta, A.; Bonomo, M.; Bianconi, F.; Gardner-Sood, P.; O’Connor, J.; Russo, M. Proportion of patients in south London with first-episode psychosis attributable to use of high potency cannabis: A case-control study. Lancet Psychiatry 2015, 2, 233–238.
- Niesink, R.J.M.; van Laar, M.W. Does cannabidiol protect against adverse psychological effects of THC? Front. Psychiatry 2013, 4, 130.
- Bhattacharyya, S.; Morrison, P.D.; Fusar-Poli, P.; Martin-Santos, R.; Borgwardt, S.; Winton-Brown, T.; Nosarti, C.; O’Carroll, C.M.; Seal, M.; Allen, P. Opposite effects of Δ-9-tetrahydrocannabinol and cannabidiol on human brain function and psychopathology. Neuropsychopharmacology 2010, 35, 764–774.
- Sills, G.J.; Rogawski, M.A. Mechanisms of action of currently used antiseizure drugs. Neuropharmacology 2020, 168, 107966.
- Chen, Z.; Brodie, M.J.; Kwan, P. What has been the impact of new drug treatments on epilepsy? Curr. Opin. Neurol. 2020, 33, 185–190.
- Elliott, J.; DeJean, D.; Clifford, T.; Coyle, D.; Potter, B.; Skidmore, B.; Alexander, C.; Repetski, A.E.; McCoy, B.; Wells, G.A. Cannabis for pediatric epilepsy: Protocol for a living systematic review. Syst. Rev. 2018, 7, 95.
- O’Connell, B.K.; Gloss, D.; Devinsky, O. Cannabinoids in treatment-resistant epilepsy: A review. Epilepsy Behav. 2017, 70, 341–348.
- Zaheer, S.; Kumar, D.; Khan, M.T.; Giyanwani, P.R.; Kiran, F.N.U. Epilepsy and cannabis: A literature review. Cureus 2018, 1–7.
- Sekar, K.; Pack, A. Epidiolex as adjunct therapy for treatment of refractory epilepsy: A comprehensive review with a focus on adverse effects. F1000Research 2019, 8, F1000 Faculty Rev-234.
- Devinsky, O.; Cross, J.H.; Laux, L.; Marsh, E.; Miller, I.; Nabbout, R.; Scheffer, I.E.; Thiele, E.A.; Wright, S. Trial of cannabidiol for drug-resistant seizures in the Dravet syndrome. N. Engl. J. Med. 2017, 376, 2011–2020.
- Devinsky, O.; Patel, A.D.; Cross, J.H.; Villanueva, V.; Wirrell, E.C.; Privitera, M.; Greenwood, S.M.; Roberts, C.; Checketts, D.; VanLandingham, K.E. Effect of cannabidiol on drop seizures in the Lennox–Gastaut syndrome. N. Engl. J. Med. 2018, 378, 1888–1897.
- Thiele, E.A.; Marsh, E.D.; French, J.A.; Mazurkiewicz-Beldzinska, M.; Benbadis, S.R.; Joshi, C.; Lyons, P.D.; Taylor, A.; Roberts, C.; Sommerville, K. Cannabidiol in patients with seizures associated with Lennox-Gastaut syndrome (GWPCARE4): A randomised, double-blind, placebo-controlled phase 3 trial. Lancet 2018, 391, 1085–1096.
- Chiurchiù, V.; van der Stelt, M.; Centonze, D.; Maccarrone, M. The endocannabinoid system and its therapeutic exploitation in multiple sclerosis: Clues for other neuroinflammatory diseases. Prog. Neurobiol. 2018, 160, 82–100.
- Suryadevara, U.; Bruijnzeel, D.M.; Nuthi, M.; Jagnarine, D.A.; Tandon, R.; Bruijnzeel, A.W. Pros and cons of medical cannabis use by people with chronic brain disorders. Curr. Neuropharmacol. 2017, 15, 800–814.
- Belendiuk, K.A.; Baldini, L.L.; Bonn-Miller, M.O. Narrative review of the safety and efficacy of marijuana for the treatment of commonly state-approved medical and psychiatric disorders. Addict. Sci. Clin. Pract. 2015, 10, 1–10.
- Tintore, M.; Vidal-Jordana, A.; Sastre-Garriga, J. Treatment of multiple sclerosis—Success from bench to bedside. Nat. Rev. Neurol. 2019, 15, 53–58.
- Mouhamed, Y.; Vishnyakov, A.; Qorri, B.; Sambi, M.; Frank, S.M.S.; Nowierski, C.; Lamba, A.; Bhatti, U.; Szewczuk, M.R. Therapeutic potential of medicinal marijuana: An educational primer for health care professionals. Drug. Healthc. Patient Saf. 2018, 10, 45.
- Beauchemin, M.; Sung, L.; Hershman, D.L.; Weng, C.; Dupuis, L.L.; Schnall, R. Guideline concordant care for prevention of acute chemotherapy-induced nausea and vomiting in children, adolescents, and young adults. Support. Care Cancer 2020, 28, 1–9.
- Badowski, M.E. A review of oral cannabinoids and medical marijuana for the treatment of chemotherapy-induced nausea and vomiting: A focus on pharmacokinetic variability and pharmacodynamics. Cancer Chemother. Pharmacol. 2017, 80, 441–449.
- May, M.B.; Glode, A.E. Dronabinol for chemotherapy-induced nausea and vomiting unresponsive to antiemetics. Cancer Manag. Res. 2016, 8, 49.
- Narain, T.; Farrah, K. Nabilone for Non-Chemotherapy Associated Nausea and Vomiting and Weight Loss Due to Medical Conditions: A Review of Clinical Effectiveness and Guidelines; Canadian Agency for Drugs and Technologies in Health: Ottawa, ON, Canada, 2017.
- Vučković, S.; Srebro, D.; Vujović, K.S.; Vučetić, Č.; Prostran, M. Cannabinoids and pain: New insights from old molecules. Front. Pharmacol. 2018, 9, 1259.
- Cavalli, E.; Mammana, S.; Nicoletti, F.; Bramanti, P.; Mazzon, E. The neuropathic pain: An overview of the current treatment and future therapeutic approaches. Int. J. Immunopathol. Pharmacol. 2019, 33, 1–10.
- Casey, S.L.; Vaughan, C.W. Plant-based cannabinoids for the treatment of chronic neuropathic pain. Medicines 2018, 5, 67.
- Childs, D.S.; Jatoi, A. A hunger for hunger: A review of palliative therapies for cancer-associated anorexia. Ann. Palliat. Med. 2019, 8, 50.
- Zutt, M.; Hänßle, H.; Emmert, S.; Neumann, C.; Kretschmer, L. Dronabinol for supportive therapy in patients with malignant melanoma and liver metastases. Hautarzt 2006, 57, 423.
- Abrams, D.I.; Guzman, M. Cannabis in cancer care. Clin. Pharmacol. Ther. 2015, 97, 575–586.
- Hall, W. Focus: Addiction: US Policy Responses to Calls for the Medical Use of Cannabis. Yale J. Biol. Med. 2015, 88, 257.
- Shi, H.Y.; Ng, S.C. The state of the art on treatment of Crohn’s disease. J. Gastroenterol. 2018, 53, 989–998.
- Hasenoehrl, C.; Storr, M.; Schicho, R. Cannabinoids for treating inflammatory bowel diseases: Where are we and where do we go? Expert Rev. Gastroenterol. Hepatol. 2017, 11, 329–337.
- Storr, M.; Devlin, S.; Kaplan, G.G.; Panaccione, R.; Andrews, C.N. Cannabis use provides symptom relief in patients with inflammatory bowel disease but is associated with worse disease prognosis in patients with Crohn’s disease. Inflamm. Bowel Dis. 2014, 20, 472–480.
- Naftali, T.; Schleider, L.B.-L.; Dotan, I.; Lansky, E.P.; Benjaminov, F.S.; Konikoff, F.M. Cannabis induces a clinical response in patients with Crohn’s disease: A prospective placebo-controlled study. Clin. Gastroenterol. Hepatol. 2013, 11, 1276–1280.
- Armstrong, M.J.; Okun, M.S. Diagnosis and treatment of Parkinson disease: A review. JAMA 2020, 323, 548–560.
- Babayeva, M.; Assefa, H.; Basu, P.; Chumki, S.; Loewy, Z. Marijuana compounds: A nonconventional approach to Parkinson’s disease therapy. Park. Dis. 2016, 2016, 1279042.
- Lotan, I.; Treves, T.A.; Roditi, Y.; Djaldetti, R. Cannabis (medical marijuana) treatment for motor and non–motor symptoms of Parkinson disease: An open-label observational study. Clin. Neuropharmacol. 2014, 37, 41–44.
- Novotny, M.; Valis, M.; Klimova, B. Tourette syndrome: A mini-review. Front. Neurol. 2018, 9, 139.
- Kanaan, A.S.; Jakubovski, E.; Müller-Vahl, K. Significant tic reduction in an otherwise treatment-resistant patient with Gilles de la Tourette syndrome following treatment with nabiximols. Brain Sci. 2017, 7, 47.
- Abramovici, H. Information for Health Care Professionals: Cannabis (Marihuana, Marijuana) and the Cannabinoids; Health Canada: Ottawa, ON, Canada, 2018.
- Stępnicki, P.; Kondej, M.; Kaczor, A.A. Current concepts and treatments of schizophrenia. Molecules 2018, 23, 2087.
- Weinreb, R.N.; Aung, T.; Medeiros, F.A. The pathophysiology and treatment of glaucoma: A review. JAMA 2014, 311, 1901–1911.
- Sun, X.; Xu, C.S.; Chadha, N.; Chen, A.; Liu, J. Focus: Addiction: Marijuana for Glaucoma: A Recipe for Disaster or Treatment? Yale J. Biol. Med. 2015, 88, 265.
- Adelli, G.R.; Bhagav, P.; Taskar, P.; Hingorani, T.; Pettaway, S.; Gul, W.; ElSohly, M.A.; Repka, M.A.; Majumdar, S. Development of a Δ9-tetrahydrocannabinol amino acid-dicarboxylate prodrug with improved ocular bioavailability. Investig. Ophthalmol. Vis. Sci. 2017, 58, 2167–2179.
- Bhartiya, S.; Ichhpujani, P. Complementary and alternate management of glaucoma: The verdict so far. J. Curr. Glaucoma Pract. 2014, 8, 54.
- Wang, C.; Horby, P.W.; Hayden, F.G.; Gao, G.F. A novel coronavirus outbreak of global health concern. Lancet 2020, 395, 470–473.
- Guo, Y.-R.; Cao, Q.-D.; Hong, Z.-S.; Tan, Y.-Y.; Chen, S.-D.; Jin, H.-J.; Tan, K.-S.; Wang, D.-Y.; Yan, Y. The origin, transmission and clinical therapies on coronavirus disease 2019 (COVID-19) outbreak–an update on the status. Mil. Med. Res. 2020, 7, 1–10.
- Vannabouathong, C.; Devji, T.; Ekhtiari, S.; Chang, Y.; Phillips, S.A.; Zhu, M.; Chagla, Z.; Main, C.; Bhandari, M. Novel coronavirus COVID-19: Current evidence and evolving strategies. J. Bone Joint Surg. Am. 2020, 102, 734.
- Onaivi, E.S.; Sharma, V. Cannabis for COVID-19: Can cannabinoids quell the cytokine storm? Future Sci OA 2020, 1–4.
- Cascella, M.; Rajnik, M.; Cuomo, A.; Dulebohn, S.C.; Di Napoli, R. Features, evaluation and treatment coronavirus (COVID-19). In Statpearls [Internet]; StatPearls Publishing: Treasure Island, FL, USA, 2020.
- Tahamtan, A.; Tavakoli-Yaraki, M.; Salimi, V. Opioids/cannabinoids as a potential therapeutic approach in COVID-19 patients. Expert Rev. Respir. Med. 2020, 14, 965–967.
- Tang, Y.; Liu, J.; Zhang, D.; Xu, Z.; Ji, J.; Wen, C. Cytokine storm in COVID-19: The current evidence and treatment strategies. Front. Immunol. 2020, 11, 1708.
- Hojyo, S.; Uchida, M.; Tanaka, K.; Hasebe, R.; Tanaka, Y.; Murakami, M.; Hirano, T. How COVID-19 induces cytokine storm with high mortality. Inflamm. Regen. 2020, 40, 1–7.
- Zaim, S.; Chong, J.H.; Sankaranarayanan, V.; Harky, A. COVID-19 and multi-organ response. Curr. Probl. Cardiol. 2020, 100618.
- Rossi, F.; Tortora, C.; Argenziano, M.; Di Paola, A.; Punzo, F. Cannabinoid Receptor Type 2: A Possible Target in SARS-CoV-2 (CoV-19) Infection? Int. J. Mol. Sci. 2020, 21, 3809.
- Ragia, G.; Manolopoulos, V.G. Inhibition of SARS-CoV-2 entry through the ACE2/TMPRSS2 pathway: A promising approach for uncovering early COVID-19 drug therapies. Eur. J. Clin. Pharmacol. 2020, 76, 1–8.
- Esposito, G.; Pesce, M.; Seguella, L.; Sanseverino, W.; Lu, J.; Corpetti, C.; Sarnelli, G. The potential of cannabidiol in the COVID-19 pandemic: A hypothesis letter. Br. J. Pharmacol. 2020, 177, 4967–4970.
- Atalay, S.; Jarocka-Karpowicz, I.; Skrzydlewska, E. Antioxidative and anti-inflammatory properties of cannabidiol. Antioxidants 2020, 9, 21.
- Byrareddy, S.N.; Mohan, M. SARS-CoV2 induced respiratory distress: Can Cannabinoids be added to anti-viral therapies to reduce lung inflammation? Brain. Behav. Immun. 2020, 87, 120–121.
- Khodadadi, H.; Salles, É.L.; Jarrahi, A.; Chibane, F.; Costigliola, V.; Yu, J.C.; Vaibhav, K.; Hess, D.C.; Dhandapani, K.M.; Baban, B. Cannabidiol modulates cytokine storm in acute respiratory distress syndrome induced by simulated viral infection using synthetic RNA. Cannabis Cannabinoid Res. 2020, 5, 197–201.
- Kaplan, B.L.F.; Springs, A.E.B.; Kaminski, N.E. The profile of immune modulation by cannabidiol (CBD) involves deregulation of nuclear factor of activated T cells (NFAT). Biochem. Pharmacol. 2008, 76, 726–737.
- Mamber, S.W.; Krakowka, S.; Osborn, J.; Saberski, L.; Rhodes, R.G.; Dahlberg, A.E.; Pond-Tor, S.; Fitzgerald, K.; Wright, N.; Beseme, S. Can Unconventional Immunomodulatory Agents Help Alleviate COVID-19 Symptoms and Severity? Msphere 2020, 5, e00288-20.
- Sexton, M. Cannabis in the Time of Coronavirus Disease 2019: The Yin and Yang of the Endocannabinoid System in Immunocompetence. J. Altern. Complement. Med. 2020, 26, 444–448.




