
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Alessia Catalano | + 5220 word(s) | 5220 | 2021-01-27 07:56:11 | | | |
| 2 | Rita Xu | -1780 word(s) | 3440 | 2021-02-03 09:23:14 | | |
Video Upload Options
Diarylureas are tyrosine kinase inhibitors well known in the art as anticancer agents, which might be useful tools for a reposition as antimicrobials.
1. Introduction
Today, bacterial resistance represents a critical problem for human health and the care of patients with numerous diseases, thus urgently calling for the immediate development and use in therapy of drugs against multi-resistant pathogens [1]. This has encouraged the research and investigation of various drug discovery approaches: one of them is drug repurposing, which is a strategy to establish new applications for approved or investigational drugs, aside from the original medical indications. Drug repurposing is becoming more appealing because it allows for overcoming the restrictions related to the expense and delay of the discovery and development of new drugs [2][3]. In the last twenty years, humans have encountered five pandemic diseases, severe acute respiratory syndrome (SARS), Avian flu, Ebola, Middle East respiratory syndrome (MERS) [4], and coronavirus disease 2019 (COVID-19) [5][6][7]. Recently, a new variant of COVID-19 was also described [8]. It has been reported that the disease prognosis of COVID-19 is largely influenced by multi-organ involvement. The multi-organ dysfunction is characterized by acute lung and liver failure, acute kidney injury, cardiovascular disease, and a wide spectrum of hematological abnormalities and neurological disorders. The most important mechanisms are related to the direct and indirect pathogenic features induced by SARS-CoV2. Moreover, organ failure may be induced by the cytokine storm, as a result of increased levels of inflammatory mediators, endothelial dysfunction, coagulation abnormalities, and the infiltration of inflammatory cells into the organs [9][10]. Medical treatment options for COVID-19 include antiviral drugs, chloroquine/hydroxychloroquine, antibiotics, beta-blockers, steroidal and nonsteroidal anti-inflammatory drugs, and some others [11][12]. In this framework, new multitarget molecules could be useful to counteract the effects of the virus at various levels and in different ways. The repositioning of anticancer diarylureas for new indications, such as antimicrobial [13], antiviral, and anti-inflammatory, could be a very interesting option for obtaining multitarget agents useful for the treatment or prevention of pandemics. Diarylureas or bis-arylureas are generally known as anticancer agents [14], for instance sorafenib, regorafenib, linifanib, tivozanib, and ripretinib, which have been recently reviewed by our research group [15]. Nevertheless, it is reported that they have also a lot of different and important biological activities [16][17]. Indeed, they are antimicrobials, particularly active in schistosomiasis and malaria [18][19][20], anti-inflammatory, antiplatelet, and antiviral agents [21][22], and allosteric modulators of the cannabinoid CB1 receptor [23], VLA-4 integrin antagonists [24], and EGFR inhibitors [25]. Taking into account the multiple factors involved in the pathogenesis of COVID-19, all the activities of diarylureas make them promising tools for the treatment of this disease. It is known that the novel coronavirus pneumonia is closely associated with inflammatory storms. Indeed, the patients with severe COVID-19 have a “cytokine storm” syndrome, given the high-levels of pro-inflammatory cytokines found in these patients [26]. Moreover, there is an increasing recognition of a prothrombotic state in COVID-19 [27] and neuropsychiatric diseases such as depression and anxiety [28]. The expression levels of angiotensin converting enzyme 2 (ACE2), acetylcholinesterase (AChE), interleukin-6 (IL-6), based on their involvement in both environmental responses and comorbid conditions, such as hypertension and type 2 diabetes mellitus, may substantially raise the respiratory syndrome coronavirus 2 (SARS-CoV-2) mortality [29].
2. Diarylureas with Antimicrobial Activity
Antimicrobials are small molecules that can inhibit or kill bacteria. However, some bacteria can grow and survive despite antimicrobial pressures, a characteristic known as antimicrobial resistance. In clinical settings, resistant bacterial infections decrease the achievable treatment options and increase morbidity and mortality, compared with those caused by susceptible microbes [30]. Moreover, traditional bactericidal antibiotics kill only actively growing bacterial cells, whereas non-growing metabolically inactive cells are tolerant to and therefore “persist” in the presence of legacy antibiotics. Thus, the discovery of new antibiotics may be envisaged. Triclocarban (TCC, Tables 1 and 2) is a diarylurea used as an antimicrobial agent in feminine care products, such as bar soaps, deodorants, detergents, and other disinfectants [31]. Besides its antimicrobial activity, TCC has been acknowledged as an endocrine disruptor, i.e., “an exogenous substance or mixture that alters function(s) of the endocrine system and consequently causes side effects in an intact organism, or its progeny, or (sub)populations” (World Health Organization 2013) resulting in hormonal effects [32]. Moreover, TCC demonstrated anti-inflammatory effects by inhibiting soluble epoxide hydrolase (sEH) in a lipopolysaccharide (LPS)-challenged murine model [33]. It has been demonstrated that TCC alter cardiac functions [34] and is associated to potential adverse birth outcomes in neonates, in particular decreased gestational age at birth [35]. The biodegradation of TCC is very slow as a result of its three chlorine atoms; thus, it can persist in the environment for years and it accumulates in aquatic habitats [36]. For these reasons, since 2016, the Food and Drug Administration (FDA) has banned its use in consumer products [37]. The mechanism of action against bacteria is still unknown. Unlike other antibacterial compounds, triclocarban does not interfere with the membrane. Therefore, it may act as an inhibitor of lipoprotein synthesis, by mimicking the natural substrate of enoyl-acyl-carrier protein reductase (ENR) enzyme. ENR is a highly conserved enzyme of lipid biosynthetic pathways in bacteria, notably gram-negative, gram-positive, and mycobacterial species [38]. Macsics et al. (2020) recently studied the TCC mechanism of action. Protein demethylmenaquinone methyltranferase (MenG) was proposed as the molecular target of TCC, which inhibits its biosynthesis finally leading to inhibition of the S. aureus menaquinone metabolism [39].
2.1. Diarylureas with Antiparasitic Activity
Diarylureas with antiparasitic activity are summarized in Table 1. TCC and several analogues have been studied in schistosomiasis. This disease, also called bilharzia, is one of the most dramatic parasitic diseases in tropical countries and remains a serious public health problem in the tropics and subtropics, affecting one billion people, with 250 million infected in 74 countries [40]. It is considered one of the most widespread infectious diseases among the WHO-prioritized 17 neglected tropical diseases (NTDs) [41]. Schistosomiasis is caused by the trematode worms of the genus Schistosoma (Platyhelminthes Trematoda). The clinically most relevant species are Schistosoma japonicum, S. haematobium, and S. mansoni [42]. S. japonicum is responsible for intestinal and hepatosplenic schistosomiasis in China, the Philippines, and Indonesia; S. haematobium determines urogenital schistosomiasis in Africa and in some countries of the Arabian Peninsula (it has also recently emerged on the French island of Corsica); S. mansoni causes intestinal and hepatic disease in Africa, the Arabian Peninsula, and Latin America [43]. Praziquantel (PZQ) discovered in the 1970s, is the only drug available for the treatment of schistosomiasis [44]; no schistosomiasis vaccines have been accepted for public use yet [45]. Despite many benefits of PZQ, most notably its high efficacy and excellent tolerability, the drug has important disadvantages, above all its inefficacy against juvenile schistosomes. Furthermore, the increasing administration of PZQ to millions of people annually results in high drug pressure, and thus drug-resistant parasites are likely to evolve. The Medicines for Malaria Venture (MMV) Malaria Box is a collection of over 400 compounds including families of structures identified in phenotypic screens of pharmaceutical and academic libraries against the Plasmodium falciparum malaria parasite [46]. The antischistosomal properties of some of these compounds with confirmed in vitro activity against P. falciparum were studied. Diarylurea MMV665852, a structural analog of TCC, which showed activity against P. falciparum 3D7, was also evaluated against S. mansoni in vitro on newly transformed schistosomula (NTS) and adult S. mansoni worms and showed IC50 values of 4.7 and 0.8 µM, respectively. All the compounds were then tested in vivo using the chronic S. mansoni mouse model. The compound MMV665852 showed the highest in vivo activity, given that treatment of S. mansoni infected mice with a single oral 400 mg/kg dose of MMV665852 reduced worm burden by 52.5% [47]. Then, a search for structures similar to that of MMV665852, using a Tanimoto-Rogers similarity coefficient of 0.85 as the cutoff, identified 46 compounds, 13 of which were diarylureas. All compounds were first tested in vitro on NTS and adult S. mansoni worms. MMV665852, TCC and 1 showed IC50 = 4.7, 0.07, and 1.3 µM, respectively against NTS; TCC and 1 showed IC90 of 0.3 and 2.8 µM, respectively, against NTS. TCC, compounds 1 and 2, tested at 33.3 µM, showed high in vitro activity against adult S. mansoni, showing IC50 values of 0.4, 0.7 and 0.2 µM, and IC90 of 1.4, 2.2 and 0.8 µM, respectively, after a 72-h incubation period. Interestingly, compounds 3 and 4 were found to be schistosomicidal against the adult S. mansoni 72 h post incubation, with calculated IC50 values are of 3.6 and 7.0 µM, respectively [48]. In another work, in order to enlarge the structure-activity relationships for compounds analogues of MMV665852, a series of diarylureas was studied in vitro against the juvenile S. japonicum. Compounds 5 and 6, tested at 32 μM, killed the juvenile worms within 72 h, and exhibited a higher activity than the positive control MMV665852 (IC50 = 2.5 and 2.8 μM versus 4.4 μM of MMV665852). IC50 values against the adult worms were lower than those for juvenile worms (IC50 = 1.5 and 2.2 μM versus 2.2 μM of MMV665852) [49]. However, compound 6 had been described previously in the literature, as an insecticide known to be highly toxic to fish and invertebrates, named flucofuron [50][51]. Recent studies also address it as a toxicant [52][53]. Wu et al. studied twenty diarylureas and found that compound 7 had antischistosomal activity against S. mansoni higher than that of MMV66558, by killing the NTS at a concentration of 10 µM. This compound killed both NTS and adult S. mansoni with IC50 values of 0.15 and 0.19 µM, respectively. In this paper, studies regarding the relationships existing between lipophilicity and metabolic stability were also carried out. The authors found that there were no clear relationships between lipophilicity and metabolic stability. Compound 7 had the best in vivo antischistosomal activity, with worm burden reduction value of 40% following administration of single 100 mg/kg oral doses [19]. In the last years, protein kinase inhibitors were used as starting points for drug discovery in studies on helminth growth and development. Sorafenib and its structurally related analogue regorafenib were tested in vitro against the larval and adult stage of S. mansoni and they were lethal to NTS within 24 h (IC50 = 4.1 and 1.1 µM, respectively, for sorafenib; IC50 = 6.9 and 1.0 µM, respectively for regorafenib). Sorafenib caused the death of all adults, at 33.3 μM, within 24 h [54].
Table 1. Diarylureas as antiparasitic agents.
|
Structure |
Compd |
Activity |
Ref |
|
Antiparasitic agents |
|
|
|
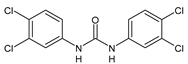 |
MMV665852 |
IC50 = 4.7 µM (S. mansoni NTS) IC50 = 0.8 µM (adult S. mansoni) IC50 = 4.4 µM (juvenile S. japonicum) IC50 = 2.2 µM (adult S. japonicum) |
|
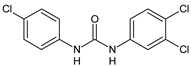 |
Triclocarban (TCC) |
IC50 = 0.07 µM (S. mansoni NTS) IC50 = 0.4 µM (adult S. mansoni) |
[48] |
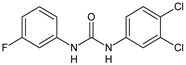 |
1 |
IC50 = 1.3 µM (S. mansoni NTS) IC50 = 0.7 µM (adult S. mansoni) |
[48] |
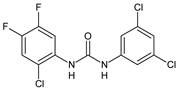 |
2 |
IC50 = 0.2 µM (adult S. mansoni) |
[48] |
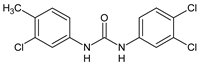 |
3 |
IC50 = 3.6 µM (adult S. mansoni) |
[48] |
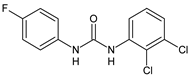 |
4 |
IC50 = 7.0 µM (adult S. mansoni) |
[48] |
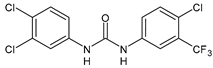 |
5 |
IC50 = 2.5 µM (juvenile S. japonicum) IC50 = 1.5 µM (adult S. japonicum) |
[49] |
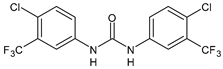 |
6 (Flucofuron) |
IC50 = 2.8 µM (juvenile S. japonicum) IC50 = 1.5 µM (adult S. japonicum) |
[49] |
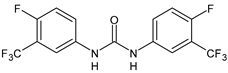 |
7 |
IC50 = 0.15 µM (S. mansoni NTS) IC50 = 0.19 µM (adult S. mansoni) |
[19] |
 |
Sorafenib (BAY-43-9006) |
IC50 = 4.1 µM (S. mansoni NTS) IC50 = 1.1 µM (adult S. mansoni) |
[54] |
 |
Regorafenib (BAY-73-4506) |
IC50 = 6.9 µM (S. mansoni NTS) IC50 = 1.0 µM (adult S. mansoni) |
[54] |
2.2. Diarylureas with Antibacterial and Antifungal Activity
In Table 2, antibacterial and antifungal diarylureas are described. The work by Pujol et al. [55] was devoted to diarylureas designed to overcome the toxicological effect of TCC due to the three chlorine atoms. One or more chlorine atoms of TCC were reduced and/or replaced by pentafluorosulfanyl groups, bioisosteres of the trifluoromethyl groups. Some of the newly synthesized compounds exhibited high potency, broad spectrum of antimicrobial activity against Gram-positive bacteria, and high selectivity index, while displaying a lower spontaneous mutation frequency than TCC. Preliminary experiments suggested a bactericidal mode of action for this family of ureas. Moreover, some of the new molecules removed preexisting S. aureus biofilms, which is important in food industry as well as in hospital settings and displayed a lower spontaneous mutation frequency in S. aureus than TCC. Compound 8 emerged as the most promising compound showing the highest potency against both S. aureus ATCC 12,600 and methicillin-resistant S. aureus MRSA (MIC50 = 0.05 µg/mL versus MIC50 = 0.5 µg/mL of TCC), a broader spectrum of activity, and a higher selectivity index [55]. Le et al. (2020) has recently studied 72 compounds deriving from sorafenib as antibacterials. Compound PK150 showed anti-bacterial activity against several pathogenic strains of S. aureus at sub-micromolar concentrations. MIC value against methicillin sensitive (MSSA) S. aureus NCTC 8325 was 0.3 µM for PK150 and 3 µM for both sorafenib and regorafenib (MIC = 1 and 3 µM for vancomycin and linezolid, respectively). It exhibited a 10-fold enhanced anti-MRSA activity, lack of resistance development under laboratory conditions, killing of persisters, elimination of established biofilms, and in vivo efficacy in a mouse model. Chemical proteomic studies did not reveal a known kinase as target, but interference with menaquinone biosynthesis and dysregulation of protein secretion as putative target mechanisms [56]. Macsics et al. (2020) studied the mechanism of PK150. As TCC, it is an inhibitor of MenG biosynthesis, but unlike TCC, it is not affected by environmentally acquired TCC resistance as it causes over-activation of SpsB, the bacterial signal peptidase I enzyme from S. aureus [39]. The two anticancer agents sorafenib and regorafenib were also investigated as antibacterials and proposed for further studies in this field [57]. Chang et al. (2016) studied antibacterial and antiproliferative activity of sorafenib. The authors compared anti-Staphylococcus activity (MIC, Minimum Inhibitory Concentration) versus antiproliferative activity (IC50) of test agents against HEK-293 human embryonic kidney, K-562 human erythromyeloblastoid leukaemia, HT-29 human colon adenocarcinoma cell lines. It showed antibacterial activity against S. aureus NCTC 8325, with MIC90 (i.e., concentration that inhibits 90% of bacteria strains tested) = 4 mg/L. However, the authors concluded that it lacked selectivity, having a low selectivity ratio (IC50 for human cells/MIC for S. aureus) of 0.7–0.8, thus assessing it is not a feasible candidate for treatment of MRSA infection. Then, the authors studied other diarylureas and found that compounds 6 was highly potent against S. aureus (NCTC 8325 and ATCC 12598) and S. epidermidis (ATCC 12,228 and ATCC 35,984) strains, with MIC = 0.25 mg/L. It was classified as bactericidal. The selectivity ratio for this compound was higher than sorafenib, ranging between 4 and 19 [58]. In 2004, Francisco et al. [59] synthesized and studied a series of phenyl thiazolyl urea as new inhibitors of the bacterial cell-wall biosynthesis. Indeed, the peptidoglycan biosynthesis requires 10 synthetic transformations and, correspondingly, 10 specific enzymes, including MurA and MurB. Phenyl thiazolyl urea 9 demonstrated a good activity against MurA and MurB and gram-positive bacteria including MRSA, vancomycin resistant Enterococcus (VRE), and penicillin-resistant Streptococcus pneumoniae (PRSP), with MIC values ranging between 0.5 and 8.0 µg/mL. However, when tested in the presence of 4% bovine serum albumin, their MICs increased significantly [59]. The importance of compound 9 is underlined by its use as a positive control for studies on MurB [60]. Hassan et al. (2014) reported a study on diarylureas with activity against bacteria and fungi. Compound 10 possessed antimicrobial activity against the tested Gram-positive bacterium Bacillus subtilis (NCTC-10400) and Gram negative bacteria Pseudomonas aeruginosa (ATCC 10145) and Escherichia coli (ATCC 23,282) showing mean values of inhibition zones (in mm) of 14.0 versus 34.0, 32.0 and 30.0 of erythromycin, respectively. The compound showed weak antifungal activity [61]. Diarylurea PQ401 is a small molecule previously described as inhibitor of the insulin like growth factor I receptor (IGF-1R) signaling and then studied in breast cancer and osteosarcoma [15]. Recently, it has been proposed as a lead candidate for repurposing as a membrane-active antimicrobial agent because it is able to kill both growing and non-growing antibiotic-tolerant MRSA by lipid bilayer disruption. When tested against a panel of antibiotic-resistant S. aureus strains (MW2, ATCC 33591, JE2, BF1‒5, BF7,8, BF10,11), including MRSA clinical isolates and a vancomycin-resistant S. aureus (VRSA; strain VRS1), it showed a MIC value of 4 µg/mL. PQ401 was demonstrated to be active against MRSA (MW2, ATCC 33591, JE2) and VRS1 strains showing a minimum bactericidal concentration (MBC) of 4 g/mL. Unlike other well-studied membrane-disrupting cationic antimicrobial low-molecular-weight antimicrobials, maximum membrane activity was shown by PQ401 in its neutral form rather than its cationic form. PQ401 also showed efficacy in both the Caenorhabditis elegans and Galleria mellonella models of MRSA infection [62]. A series of the diarylureas was synthesized and screened for antimicrobial activity against Gram positive and negative bacteria and fungi. Compounds 11 and 12 showed activity against Proteus mirabilis ATCC 19,181 comparable to that of standard ciprofloxacin (zone of inhibition in 23 and 24 mm, respectively, at a concentration of 200 µg/mL compared to 30 mm of ciprofloxacin) [63]. In the work by Gezegen et al. (2017), a series of diarylureas was studied as antimicrobial and antiproliferative agents. Antimicrobial activity studies were carried out against Gram-positive and Gram-negative bacteria and yeasts, by using piperacillin/tazobactam (P/T = 8/1) and fluconazole as positive controls. The most interesting compounds were 13 and 14. They were both more active than reference against Shigella boydii ATCC 9905 and Enterococcus faecalis ATCC 29,212 (MIC = 31.3 µg/mL versus 62.5 µg/mL), a clinically significant pathogen implicated in different types of infections [64], and against Bacillus cereus ATCC 10,987 (MIC = 31.3 µg/mL versus 125 µg/mL), while they showed the same activity of the reference against Klebsiella pneumoniae ATCC 10,031 (MIC = 31.3 µg/mL) [65]. Diarylureas 15–17, bearing an aminoguanidine group that is a common moiety in medicinal chemistry [66], were active against MRSA NRS123 showing MIC values of 10 µg/mL (for compounds 15 and 17) and 8 µg/mL (for compound 16) [67]. Compound 16 was chosen as the lead compound for further studies on a series of diarylureas bearing an alkoxy side chain in lieu of the n-butyl moiety. The substitution with an isopentyloxy or cycloheptyloxy group gave compounds 18 and 19, which showed activity in vitro against MRSA (MIC values between 2 and 4 µg/mL versus vancomycin ranging from 0.5 to 1 µg/mL). Compound 19 was chosen for further in vivo studies using a C. elegans animal model. The antibacterial activity was confirmed as 19, at 10 µg/mL, reduced the burden of MRSA USA400 by more than 50% in infected worms, which is a result better than that observed for vancomycin (at the same concentration it reduces the bacterial burden by 25%). Compound 19 also showed better pharmacokinetics, showing enhanced stability to hepatic metabolism, it was also demonstrated to be suitable for intravenous or topic administration (for treatment MRSA skin infections) [67]. In a sequent work, the investigation of action mechanism of a series diphenylureas revealed that they exert their antibacterial effect by interfering with bacterial cell wall synthesis. Interestingly, both compounds 18 and 19 are able of re-sensitizing VRSA to the effect of vancomycin. Furthermore, compound 18 can penetrate staphylococcal biofilms (S. aureus and S. epidermidis) to reduce the burden of bacteria present within the biofilm, although at high concentrations [68]. Upadhayaya et al. (2009) studied several quinoline derivatives as antimycobacterials. Tuberculosis (TB) is an old human disease and represent a major threat for mankind especially because of the emergence of resistance strain of Mycobacterium tuberculosis against antibiotics. Compounds 20 and 21 inhibited M. tuberculosis H37Rv up to 98% and 94%, respectively, at a concentration of 6.25 µg/mL. They showed MIC values of 6.25 µg/mL and 3.125 µg/mL, respectively [69]. Compounds 22 and 23 showed MIC values of 6.0 and 5.2 µg/mL, respectively, against M. tuberculosis pathogenic strain H37Rv and 2.0 and 1.0, respectively, against M. tuberculosis nonpathogenic strain mc26030. They also showed selective inhibition of mycolic acid biosynthesis. At the same time, these molecules also executed their potent immunomodulatory activity by up-regulation of the pro-inflammatory cytokines IFN-g and IL-12 and down-regulation of IL-10 [70]. Compound 24 was studied for antifungal activity, containing 1,2,4-triazole, showed good antifungal activity against Phomopsis species. These are known as Phomopsis cane and leaf spot (P. viticola) causes economic losses to the vine grape production in the USA and Europe, while P. obscurans is responsible of Phomopsis leaf blight and fruit rot of strawberry. At 30 μM, compound 24 inhibited the growth of P. obscurans and P. viticola by 80% and 100%, respectively, after 120 h exposure, showing an activity similar to that of the positive control Captan, a well-known multisite inhibitor fungicide with no systemic activity, used as a commercial protectant fungicide to prevent anthracnose diseases in fruits and ornamentals [71].
Table 2. Diarylureas as antibacterials and antifungals.
|
Structure |
Compd |
Activity |
Ref |
|
Antibacterial activity |
|
||
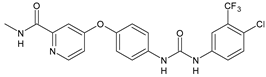
 |
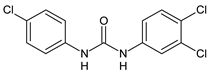
8 |
MIC50 = 0.05 µg/mL (S. aureus ATCC 12600 and MRSA) |
[55] |
 |
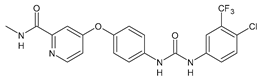
PK150 |
MIC = 0.3 µM (S. aureus NCTC 8325) |
[56] |
 |
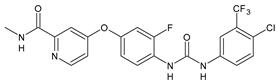
Sorafenib (BAY-43-9006) |
MIC = 3 µM (S. aureus NCTC 8325) |
[56] |
 |
Regorafenib (BAY-73-4506) |
MIC = 3 µM (S. aureus NCTC 8325) |
[56] |
 |
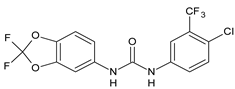
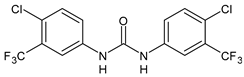
Triclocarban (TCC) |
MIC50 = 0.5 µg/mL (S. aureus) |
[55] |
 |
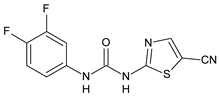
6 (Flucofuron) |
MIC = 0.25 mg/L (S. aureus NCTC 8325; ATCC 12598 and S. epidermidis ATCC 12228; 35984) |
[58] |
 |
9 |
MIC = 0.5‒8.0 µg/mL (different gram-positive bacteria) |
[59] |
 |
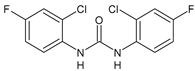
10 |
Inhibition zones = 14.0 mm Gram(+) B. subtilis NCTC-10400 and Gram(‒) P. aeruginosa ATCC 10145 and E. coli ATCC 23282 |
[61] |
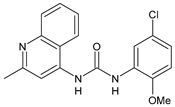 |
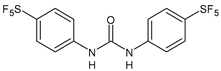
PQ401 |
MIC = 4 µg/mL (different S. aureus) MBC = 4 g/mL (S. aureus VRS1 strains) |
[62] |
 |
11 |
Inhibition zone = 23 mm, at a concentration of 200 µg/mL (P. mirabilis ATCC 19181) |
[63] |
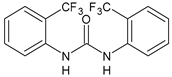 |
12 |
Inhibition zone = 24 mm, at a concentration of 200 µg/mL (P. mirabilis ATCC 19181) |
[63] |
 |
13 |
MIC = 31.3 µg/mL (S. boydii ATCC 9905; E. faecalis ATCC 29212; B. cereus ATCC 10987; K. pneumoniae ATCC 10031) |
[65] |
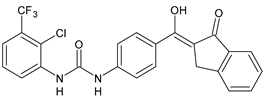
 |
14 |
MIC = 31.3 µg/mL (S. boydii ATCC 9905; E. faecalis ATCC 29212; B. cereus ATCC 10987; K. pneumoniae ATCC 10031) |
[65] |
 |
15 |
MIC = 10 µg/mL (MRSA NRS123) |
[67] |
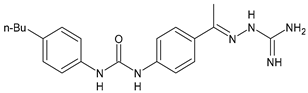 |
16 |
MIC = 8 µg/mL (MRSA NRS123) |
[67] |
 |
17 |
MIC = 10 µg/mL (MRSA NRS123) |
[67] |
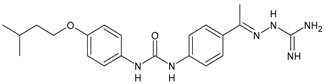 |
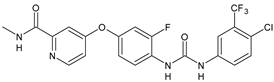
18 |
MIC = 2‒4 µg/mL (various MRSA) |
|
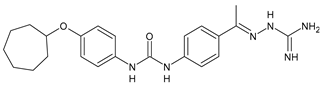 |
19 |
MIC = 2‒4 µg/mL (various MRSA) |
[68] |
 |
20 |
MIC = 6.25 µg/mL (M. tuberculosis H37Rv) |
[69] |
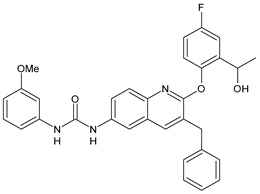 |
21 |
MIC = 3.125 µg/mL (M. tuberculosis H37Rv) |
[69] |
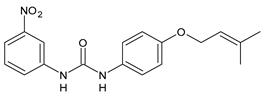 |
22 |
MIC = 6.0 µg/mL (M. tuberculosis pathogenic strain H37Rv) MIC = 2.0 µg/mL (M. tuberculosis nonpathogenic strain mc26030) |
[70] |
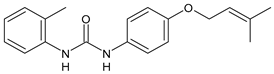 |
23 |
MIC = 5.2 µg/mL (M. tuberculosis pathogenic strain H37Rv) MIC = 1.0 µg/mL (M. tuberculosis nonpathogenic strain mc26030) |
[70] |
|
Antifungal activity |
|
|
|
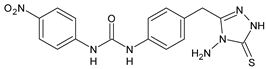 |
24 |
Inhibition, at 30 μM, of the growth of P. obscurans by 80% and P. viticola and 100%, after 120 h exposure |
[71] |
References
- World Health Organization. Tackling antimicrobial resistance. Eurohealth 2020, 26.
- Pushpakom, S.; Iorio, F.; Eyers, P.A.; Escott, K.J.; Hopper, S.; Wells, A.; Doig, A.; Guilliams, T.; Latimer, J.; McNamee, C.; et al. Drug repurposing: Progress, challenges and recommendations. Rev. Drug Discov. 2019, 18, 41‒48.
- Nascimento, I.J.D.S.; de Aquino, T.M.; da Silva-Júnior, E.F. Drug repurposing: A strategy for discovering inhibitors against emerging viral infections. Med. Chem. 2020, in press.
- Reperant, L.A.; Osterhaus, A. Aids, Avian flu, SARS, MERS, Ebola, Zika… What next? Vaccine 2017, 35, 4470–4474.
- Velavan, T.P.; Meyer, C.G. The COVID-19 epidemic. Med. Int. Health 2020, 25, 278–280.
- Pascarella, G.; Strumia, A.; Piliego, C.; Bruno, F.; Del Buono, R.; Costa, F.; Scarlata, S.; Agrò, F.E. COVID-19 diagnosis and management: A comprehensive review. Intern. Med. 2020, 288, 192–206.
- Fadaka, A.O.; Sibuyi, N.R.S.; Adewale, O.B.; Bakare, O.O.; Akanbi, M.O.; Klein, A.; Madiehe, A.M.; Meyer, M. Understanding the epidemiology, pathophysiology, diagnosis and management of SARS-CoV-2. Intern. Med. Res. 2020, 48, 0300060520949077.
- Wise, J. Covid-19: New coronavirus variant is identified in UK. BMJ 2020, 371, m4857, doi:10.1136/bmj.m4857.
- Mokhtari, T.; Hassani, F.; Ghaffari, N.; Ebrahimi, B.; Yarahmadi, A.; Hassanzadeh, G. COVID-19 and multiorgan failure: A narrative review on potential mechanisms. J. Mol. Histol. 2020, 51, 613–628.
- Zaim, S.; Chong, J.H.; Sankaranarayanan, V.; Harky, A. COVID-19 and multiorgan response. Probl. Cardiol. 2020, 45, 100618.
- Gautret, P.; Lagier, J.-C.; Parola, P.; Hoang, V.T.; Meddeb, L.; Mailhe, M.; Doudier, B.; Courjon, J.; Giordanengo, V.; Vieira, V.E.; et al. Hydroxychloroquine and azithromycin as a treatment of COVID-19: Results of an open-label non-randomized clinical trial. J. Antimicrob. Agents 2020, 56, 105949.
- Ginsburg, A.S.; Klugman, K.P. COVID-19 pneumonia and the appropriate use of antibiotics. Lancet Glob. Health 2020, 8, e1453–
- Farha, M.A.; Brown, E.D. Drug repurposing for antimicrobial discovery. Microbiol. 2019, 4, 565‒577.
- Garuti, L.; Roberti, M.; Bottegoni, G.; Ferraro, M. Diaryl urea: A privileged structure in anticancer agents. Med. Chem. 2016, 23, 1528–1548.
- Catalano, A.; Iacopetta, D.; Sinicropi, M.S.; Franchini, C. Diarylureas as antitumor agents. Sci. 2020, 11, 374.
- Sikka, P.; Sahu, J.K.; Mishra, A.K.; Hashim, S.R. Role of Aryl Urea Containing Compounds in Medicinal Chemistry. Chem. 2015, 5, 479–483.
- Asif, M. Short notes on diaryl ureas J. Adv. Res. Biochem. Pharmacol. 2018, 1, 38‒41.
- Gemma, S.; Federico, S.; Brogi, S.; Brindisi, M.; Butini, S.; Campiani, G. Dealing with schistosomiasis: Current drug discovery strategies. In Annual Reports in Medicinal Chemistry; Elsevier: Amsterdam, The Netherland, 2019; Volume 53, pp. 107–138, ISBN 9780128198667.
- Wu, J.; Wang, C.; Leas, D.; Vargas, M.; White, K.L.; Shackleford, D.M.; Chen, G.; Sanford, A.G.; Hemsley, R.M.; Davis, P.H.; et al. Progress in antischistosomal N,N′-diaryl urea SAR. Med. Chem. Lett. 2018, 28, 244–248.
- Zhang, Y.; Anderson, M.; Weisman, J.L.; Lu, M.; Choy, C.J.; Boyd, V.A.; Price, J.; Siga, M.; Clark, J.; Connelly, M.; et al. Evaluation of diarylureas for activity against Plasmodium falciparum. ACS Med. Chem. Lett. 2010, 1, 460–465.
- Yang, W.; Wang, Y.; Lai, A.; Qiao, J.X.; Wang, T.C.; Hua, J.; Price, L.A.; Shen, H.; Chen, X.; Wong, P.; et al. Discovery of 4-aryl-7-hydroxyindoline-based P2Y1 antagonists as novel antiplatelet agents. Med. Chem. 2014, 57, 6150–6164.
- Dighe, S.N.; Dua, K.; Chellappan, D.K.; Katavic, P.L.; Collet, T.A. Recent update on anti-dengue drug discovery. J. Med. Chem. 2019, 176, 431–455.
- Nguyen, T.; German, N.; Decker, A.M.; Langston, T.L.; Gamage, T.F.; Farquhar, C.E.; Li, J.-X.; Wiley, J.L.; Thomas, B.F.; Zhang, Y. Novel diarylurea based allosteric modulators of the cannabinoid CB1 receptor: Evaluation of importance of 6-pyrrolidinylpyridinyl substitution. Med. Chem. 2017, 60, 7410–7424.
- Witherington, J.; Blaney, E.L.; Bordas, V.; Elliott, R.L.; Gaiba, A.; Garton, N.; Green, P.M.; Naylor, A.; Smith, D.G.; Spalding, D.J.; et al. Pyridone derivatives as potent, orally bioavailable VLA-4 integrin antagonists. Med. Chem. Lett. 2006, 16, 5538–5541.
- Jiang, N.; Bu, Y.; Wang, Y.; Nie, M.; Zhang, D.; Zhai, X. Design, Synthesis and Structure–Activity Relationships of Novel Diaryl Urea Derivatives as Potential EGFR Inhibitors. Molecules 2016, 21, 1572.
- Catalano, A. COVID-19: Could Irisin Become the Handyman Myokine of the 21st Century? Coronaviruses 2020, 1, 32–41.
- Rapkiewicz, A.V.; Mai, X.; Carsons, S.E.; Pittaluga, S.; Kleiner, D.E.; Berger, J.S.; Thomas, S.; Adler, N.M.; Charytan, D.M.; Gasmi, B.; et al. Megakaryocytes and platelet-fibrin thrombi characterize multi-organ thrombosis at autopsy in COVID-19: A case series. E Clin. Med. 2020, 24, 100434.
- Mazza, M.G.; Lorenzo, R.D.; Conte, C.; Poletti, S.; Vai, B.; Bollettini, I.; Melloni, E.M.T.; Furlan, R.; Ciceri, F.; Rovere-Querini, P.; et al. Anxiety and depression in COVID-19 survivors: Role of inflammatory and clinical predictors. Brain Behav. Immun. 2020, 89, 594–600.
- Phillips, N.; Park, I.-W.; Robinson, J.R.; Jones, H.P. The perfect storm: COVID-19 health disparities in US blacks. Racial Ethn. Health Disparities 2020, 1–8, doi:10.1007/s40615-020-00871-y.
- Patini, R.; Mangino, G.; Martellacci, L.; Quaranta, G.; Masucci, L.; Gallenzi, P. The effect of different antibiotic regimens on bacterial resistance: A systematic review. Antibiotics 2020, 9, 22.
- Balakrishna, K.; Rath, A.; Praveenkumarreddy, Y.; Guruge, K.S.; Subedi, B. A review of the occurrence of pharmaceuticals and personal care products in Indian water bodies. Environ. Saf. 2017, 137, 113–120.
- Bergman, Å.; Heindel, J.J.; Jobling, S.; Kidd, K.A.; Zoeller, R.T. The State-of-the-Science of Endocrine Disrupting Chemicals, World Health Organization and United Nations Environment Programme Report–2012; UNEP/WHO: Geneva, Switzerland, 2013. Available online: www.who.int/ceh/publications/endocrine/en/index.html (accessed on).
- Liu, J.; Qiu, H.; Morisseau, C.; Hwang, S.H.; Tsai, H.; Ulu, A.; Chiamvimonvat, N.; Hammock, B.D. Inhibition of soluble epoxide hydrolase contributes to the anti-inflammatory effect of antimicrobial triclocarban in a murine model. Appl. Pharmacol. 2011, 255, 200–206.
- Xie, W.; Zhang, W.; Ren, J.; Li, W.; Zhou, L.; Cui, Y.; Chen, H.; Yu, W.; Zhuang, X.; Zhang, Z.; et al. Metabonomics indicates inhibition of fatty acid synthesis, beta-oxidation, and tricarboxylic acid cycle in triclocarban-induced cardiac metabolic alterations in male mice. Agric. Food Chem. 2018, 66, 1533–1542.
- Geer, L.A.; Pycke, B.F.G.; Waxenbaum, J.; Sherer, D.M.; Abulafia, O.; Halden, R.U. Association of birth outcomes with fetal exposure to parabens, triclosan and triclocarban in an immigrant population in Brooklyn, New York. Hazard. Mater. 2017, 323, 177–183.
- Wang, Y.; Teng, Y.; Wang, D.; Han, K.; Wang, H.; Kang, L. The fate of triclocarban in activated sludge and its influence on biological wastewater treatment system. Environ. Manag. 2020, 276, 111237.
- Yun, H.; Liang, B.; Kong, D.; Li, X.; Wang, A. Fate, risk and removal of triclocarban: A critical review. Hazard. Mater. 2020, 387, 121944.
- Batra, S.; Tusi, Z.; Madapa, S. Medicinal chemistry of ureido derivatives as anti-infectives. Anti-Infect. Agents Med. Chem. (Former. Curr. Med. Chem. Anti-Infect. Agents) 2006, 5, 135–160.
- Macsics, R.; Hackl, M.W.; Fetzer, C.; Mostert, D.; Bender, J.; Layer, F.; Sieber, S.A. Comparative Target Analysis of Chlorinated Biphenyl Antimicrobials Highlights MenG as Molecular Target of Triclocarban. Environ. Microbiol. 2020, doi:10.1128/AEM.00933-20.
- McManus, D.P.; Dunne, D.W.; Sacko, M.; Utzinger, J.; Vennervald, B.J.; Zhou, X.N. Schistosomiasis. Rev. Dis. Primers 2018, 4, 13.
- Herricks, J.R.; Hotez, P.J.; Wanga, V.; Coffeng, L.E.; Haagsma, J.A.; Basanez, M.G.; Buckle, G.; Budke, C.M.; Carabin, H.; Fevre, E.M.; et al. The global burden of disease study 2013: What does it mean for the NTDs? PLoS Negl. Trop. Dis. 2017, 11, e0005424.
- Campbell, C.H., Jr.; Binder, S.; King, C.H.; Knopp, S.; Rollinson, D.; Person, B.; Webster, B.; Allan, F.; Utzinger, J.; Ame, S.M.; et al. SCORE operational research on moving toward interruption of schistosomiasis transmission. J. Trop. Med. Hyg. 2020, 103 (Suppl. 1), 58–65.
- McManus, D.P.; Bergquist, R.; Cai, P.; Ranasinghe, S.; Tebeje, B.M.; You, H. Schistosomiasis-from immunopathology to vaccines. Immunopathol. 2020, 42, 355–371.
- Mäder, P.; Rennar, G.A.; Ventura, A.M.P.; Grevelding, C.G.; Schlitzer, M. Chemotherapy for fighting schistosomiasis: Past, present and future. ChemMedChem 2018, 13, 2374–2389.
- McManus, D.P. Recent progress in the development of liver fluke and blood fluke vaccines. Vaccines 2020, 8, 553.
- Dang, N.L.; Hughes, T.B.; Taramelli, D. Open source drug discovery with the Malaria Box compound collection for neglected diseases and beyond. PLoS Pathog. 2016, 12, e1005763.
- Ingram-Sieber, K.; Cowan, N.; Panic, G.; Vargas, M.; Mansour, N.R.; Bickle, Q.D.; Wells, T.N.C.; Spangenberg, T.; Keiser, J. Orally active antischistosomal early leads identified from the open access Malaria Box. PLoS Negl. Trop. Dis. 2014, 8, 30.
- Cowan, N.; Dätwyler, P.; Ernst, B.; Wang, C.; Vennerstro, J.L.; Spangenberg, T.; Keiser, J. Activities of N,N’-diarylurea MMV665852 analogs against Schistosoma mansoni. Agents Chemother. 2015, 59, 1935–1941.
- Yao, H.; Liu, F.; Chen, J.; Li, Y.; Cui, J.; Qiao, C. Antischistosomal activity of N,N′-arylurea analogs against Schistosoma japonicum. Med. Chem. Lett. 2016, 26, 1386–1390.
- Cole, S.; Codling, I.D.; Parr, W.; Zabel, T. Guidelines for managing water quality impacts within UK European marine sites. In UK Marine SACs Project; 1999; Volume 88.
- Tomlin CDS. The Pesticide Manua, 10th ed.; Royal Society of Chemistry: Cambridge, UK; British Crop Protection Council: London, UK, 1995.
- Lee, J.; Hong, S.; Kim, T.; Lee, C.; An, S.A.; Kwon, B.O.; Lee, S.; Moon, H.-B.; Giesy, J.-P.; Khim, J.S. Multiple bioassays and targeted and nontargeted analyses to characterize potential toxicological effects associated with sediments of masan bay: Focusing on AhR-mediated potency. Sci. Technol. 2020, 54, 4443–4454.
- Chiaia-Hernández, A.C.; Scheringer, M.; Müller, A.; Stieger, G.; Wächter, D.; Keller, A.; Pintado-Herrera, M.G.; Lara-Martin, P.A.; Bucheli, T.D.; Hollender, J. Target and suspect screening analysis reveals persistent emerging organic contaminants in soils and sediments. Total Environ. 2020, 740, 140181.
- Cowan, N.; Keiser, J. Repurposing of anticancer drugs: In vitro and in vivo activities against Schistosoma mansoni. Parasite Vect. 2015, 8, 417–426.
- Pujol, E.; Blanco-Cabra, N.; Julián, E.; Leiva, R.; Torrents, E.; Vázquez, S. Pentafluorosulfanyl-containing triclocarban analogs with potent antimicrobial activity. Molecules 2018, 23, 2853.
- Le, P.; Kunold, E.; Macsics, R.; Rox, K.; Jennings, M.C.; Ugur, I.; Reinecke, M.; Chaves-Moreno, D.; Hackl, M.W.; Fetzer, C.; et al. Repurposing human kinase inhibitors to create an antibiotic active against drug-resistant Staphylococcus aureus, persisters and biofilms. Chem. 2020, 12, 145–158.
- Roberts, J.L.; Tavallai, M.; Nourbakhsh, A.; Fidanza, A.; Cruz-Luna, T.; Smith, E.; Siembida, P.; Plamondon, P.; Cycon, K.A.; Doern, C.D.; et al. GRP78/Dna K is a target for Nexavar/Stivarga/Votrient in the treatment of human malignancies, viral infections and bacterial diseases. Cell. Physiol. 2015, 230, 2552–2578.
- Chang, H.C.; Huang, Y.T.; Chen, C.S.; Chen, Y.W.; Huang, Y.T.; Su, J.C.; Teng, L.J.; Shiau, C.W.; Chiu, H.C. In vitro and in vivo activity of a novel sorafenib derivative SC5005 against MRSA. Antimicrob. Chemother. 2016, 71, 449–459.
- Francisco, G.D.; Li, Z.; Albright, J.D.; Eudy, N.H.; Katz, A.H.; Petersen, P.J.; Labthavikul, P.; Singh, G.; Yang, Y.; Rasmussen, B.A.; et al. Phenyl thiazolyl urea and carbamate derivatives as new inhibitors of bacterial cell-wall biosynthesis. Med. Chem. Lett. 2004, 14, 235–238.
- Jukič, M.; Hrast, M.; Patin, D.; Ogorevc, E.; Barreteau, H.; Gobec, S. Virtual screening approach and biochemical evaluation on MurB. Data Collect. 2019, 24, 100276.
- Hassan, M.A.; Sayed, G.H.; El-Nagar, A.M.; Hussien, A.M. A convenient synthesis of some diarylurea and thiourea derivatives as antimicrobial compounds. Process Eng. Res. 2014, 25, 1–11.
- Kim, W.; Zou, G.; Pan, W.; Fricke, N.; Faizi, H.A.; Kim, S.M.; Khader, R.; Li, S.; Lee, K.; Escorba, L.; et al. The neutrally charged diarylurea compound PQ401 kills antibiotic-resistant and antibiotic-tolerant Staphylococcus aureus. mBio 2020, 11, 603.
- Sarveswari, S.; Vijayakumar, V. A Facile Synthesis of diaylureas and their antimicrobial evaluation. Chiang Mai J. Sci. 2018, 45, 997–1007.
- Pozzi, C.; Ferrari, S.; Cortesi, D.; Luciani, R.; Stroud, R.M.; Catalano, A.; Costi, M.P.; Mangani, S. The structure of Enterococcus faecalis thymidylate synthase provides clues about folate bacterial metabolism. Acta Crystallogr. D Biol. Crystallogr. 2012, 68, 1232–1241.
- Gezegen, H.; Hepokur, C.; Tutar, U.; Ceylan, M. Synthesis and Biological Evaluation of Novel 1‐(4‐(Hydroxy(1‐oxo‐1,3‐dihydro‐2H‐inden‐2‐ylidene)methyl)phenyl)‐3‐phenylurea Derivatives. Biodivers. 2017, 14, e1700223.
- Ceramella, J.; Mariconda, A.; Rosano, C.; Iacopetta, D.; Caruso, A.; Longo, P.; Sinicropi, M.S.; Saturnino, C. α–ω Alkenyl-bis-S-guanidine thiourea dihydrobromide affects HeLa cell growth hampering tubulin polymerization. ChemMedChem 2020, in press.
- Eissa, I.H.; Mohammad, H.; Qassem, O.A.; Younis, W.; Abdelghany, T.M.; Elshafeey, A.; Moustafa, M.M.A.R.; Seleem, M.N.; Mayhoub, A.S. Diphenylurea derivatives for combating methicillin- and vancomycin-resistant Staphylococcus aureus. J. Med. Chem. 2017, 130, 73–85.
- Mohammad, H.; Younis, W.; Ezzat, H.G.; Peters, C.E.; AbdelKhalek, A.; Cooper, B.; Pogliano, K.; Pogliano, J.; Mayhoub, A.S.; Seleem, M.N. Bacteriological profiling of diphenylureas as a novel class of antibiotics against methicillin-resistant Staphylococcus aureus. PLoS ONE 2017, 12, e0182821.
- Upadhayaya, R.S.; Kulkarni, G.M.; Vasireddy, N.R.; Vandavasi, J.K.; Dixit, S.S.; Sharma, V. Chattapadhayaya, Design, synthesis and biological evaluation of novel triazole, urea and thiourea derivatives of quinoline against Mycobacterium tuberculosis. Med. Chem. 2009, 17, 4681–4692.
- Velappan, A.B.; Raja, M.R.C.; Datta, D.; Tsai, Y.T.; Halloum, I.; Wan, B.; Kremer, L.; Gramajo, H.; Franzblau, S.G.; Mahapatra, S.K.; et al. Attenuation of Mycobacterium species through direct and macrophage mediated pathway by unsymmetrical diaryl urea. J. Med. Chem. 2017, 125, 825–841.
- Kocyigit-Kaymakcioglu, B.; Celen, A.O.; Tabanca, N.; Ali, A.; Khan, S.I.; Khan, I.A.; Wedge, D.E. Synthesis and biological activity of substituted urea and thiourea derivatives containing 1,2,4-triazole moieties. Molecules 2013, 18, 3562–3576.




