
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Joseph Jankovic | + 2487 word(s) | 2487 | 2021-01-27 07:05:15 | | | |
| 2 | Lily Guo | + 1762 word(s) | 4249 | 2021-01-31 13:48:21 | | |
Video Upload Options
Since its initial approval in 1989 by the US Food and Drug Administration for the treatment of blepharospasm and other facial spasms, botulinum toxin (BoNT) has evolved into a therapeutic modality for a variety of neurological and non-neurological disorders. With respect to neurologic movement disorders, BoNT has been reported to be effective for the treatment of dystonia, bruxism, tremors, tics, myoclonus, restless legs syndrome, tardive dyskinesia, and a variety of symptoms associated with Parkinson’s disease. More recently, research with BoNT has expanded beyond its use as a powerful muscle relaxant and a peripherally active drug to its potential central nervous system applications in the treatment of neurodegenerative disorders. Although BoNT is the most potent biologic toxin, when it is administered by knowledgeable and experienced clinicians, it is one of the safest therapeutic agents in clinical use. The primary aim of this article is to provide an update on recent advances in BoNT research with a focus on novel applications in the treatment of movement disorders.
1. Introduction
Clostridium botulinum, an anaerobic, rod-shaped bacterium, produces a neurotoxin called botulinum toxin (BoNT) during sporulation[1][2][3]. BoNT is the most potent biological toxin, as it causes botulism manifested by paralysis of muscles and eventual fatal respiratory failure[4][5]. When an action potential arrives at the cholinergic presynaptic nerve terminal, there is an influx of calcium into the presynaptic terminal, which then facilitates acetylcholine vesicle fusion with the presynaptic membrane; this fusion is facilitated by a group of proteins referred to as SNARE (soluble N-ethylmaleimide-sensitive factor attachment receptor) proteins, which include SNAP 25 (25 kD synaptosomal-associated protein) and Syntaxin[6][7]. BoNT acts at the cholinergic presynaptic nerve terminal by cleaving and inactivating SNARE proteins, thus inhibiting release of acetylcholine, which in turn prevents muscle contraction and results in local weakness and paralysis [7,8]. BoNT acts at both the extrafusal and intrafusal muscle fibers, thereby preventing contraction of both agonist and antagonist muscles [8][9]. This biologic effect of BoNT has been turned into an advantage in patients troubled by involuntary muscle contractions, excessive secretions, pain, and other conditions [7]. The paralytic effects of BoNT were initially described in 1817 by Justinus Kerner, a German physician, who suggested that the toxin may be potentially useful in the treatment of St. Vitus’ dance, hypersalivation, and hyperhidrosis[10]. The mechanism of action of BoNT injections to account for the typical 3–4 months of duration has not been fully elucidated, and the original proposal that axonal sprouting occurs at the presynaptic nerve terminal after the injection, after which time the neuromuscular junction integrity is restored when the original nerve terminals regain their exocytic function, hence necessitating repeat injections[11], has been challenged[12].
In 1981, Dr. Jankovic initially injected BoNT into a patient for treatment of blepharospasm (BSP)[10] and subsequently published the results of the first double-blind, placebo-controlled trial of BoNT in cranial–cervical dystonia[13]. The results of this trial, along with additional data, were used by the United States Food and Drug Administration (FDA) to approve BoNT in 1989 for the treatment of BSP and facial nerve disorders such as hemifacial spasm (HFS) [14]. Although only BoNT types A and B have been approved for clinical use by the FDA, there are a total of eight different subtypes: BoNT A to H[6].
There are currently four FDA approved BoNT formulations: the three types of botulinum toxin type A (BoNTA) available are onabotulinumtoxinA (Botox; Allergan, CA, USA), abobotulinumtoxinA (Dysport; Ipsen-Pharma, UK), and incobotulinumtoxinA (Xeomin; Merz Pharma, Germany); rimabotulinumtoxinB (Myobloc in the USA; Supernus Pharmaceuticals, Inc, Rockville, MD; Neurobloc in Europe, Sloan Pharma, Switzerland) is a BoNTB preparation[6][10].
There are several BoNT preparations that are currently in development but have not yet been approved by the FDA. DaxibotulinumtoxinA is a novel BoNTA preparation that was recently evaluated in a phase 3 trial (ASPEN-1) in cervical dystonia (https://www.businesswire.com/news/home/20201014005360/en/). This study enrolled 301 patients from 60 sites in the U.S., Canada, and Europe and confirmed the findings of an earlier phase 2 study[15] in that it found that daxibotulinumtoxinA is safe and effective. Interestingly, at doses of 125 U, it has a median duration of effect (based on the median time to loss of 80% of the peak treatment effect) of 24 weeks. This relatively long duration of action offers potential advantage over other formulations in that it may allow increasing the intervisit interval beyond the conventional 3–4 months. LanbotulinumtoxinA (Prosigne; Shanghai, China) is a new preparation of BoNTA marketed chiefly in Asia[16][17].
The doses of different formulations are not interchangeable, but based on prior studies, the following ratios are often used in clinical practice when switching from one to another BoNT product to achieve similar results: onabotulinumtoxinA:incobotulinumtoxinA = 1:1; onabotulinumtoxinA:abobotulinumtoxinA = 1:2.5, and onabotulinumtoxinA:rimabotulinumtoxinB = 1:50[10].
With long term use of BoNT, there is a risk of developing neutralizing antibodies (NAbs)[18], and patients may stop responding to BoNT. Factors that increase the risk of developing resistance to BoNT include a high protein load in some formulations, large individual and cumulative doses of BoNT, and short intervisit intervals, especially booster injections[18][19][20][21][22][23]. Immunogenicity varies among the different products and has been reported as low as 0% for incobotulinumtoxinA and as high as 42.4% for rimabotulinumtoxinB [20]. Brin et al. [23] noted a 1.2% frequency of NAbs based on mouse protection assay (MPA) in patients treated for cervical dystonia with onabotulinumtoxinA. In contrast, Albrecht et al.[18] reported a prevalence of 14% of NAbs in 596 treated with BoNTA, mostly abobotulinumtoxinA, for a mean of 5.3 years based on mouse hemidiaphragm assay (MHDA). Since biological assays such as MPA and MHDA are difficult to perform and they involve sacrificing animals, there is a huge unmet need to develop a simple, inexpensive, sensitive, and specific test for BoNT-blocking antibodies. If a patient reports lack of improvement (less than 25%) after at least two or three consecutive treatment visits, this raises a high level of suspicion of immunoresistance[20]. In this case, unilateral brow injection may be performed as a clinically useful test[20]. This involves injection of BoNT in the right medial eyebrow and reassessing in 1–2 weeks for paralysis of the right procerus/corrugator as manifested by asymmetric frowning, which would disprove immunoresistance [20]. We have not included spasticity in this article, as we believe it is beyond the scope of this review. The reader is referred to some recent reviews on this topic[24][25][26].
Our manuscript provides a comprehensive review of botulinum toxin in movement disorders. Figure 1 depicts the variety of movement disorders where botulinum toxin is used for therapeutic purposes.
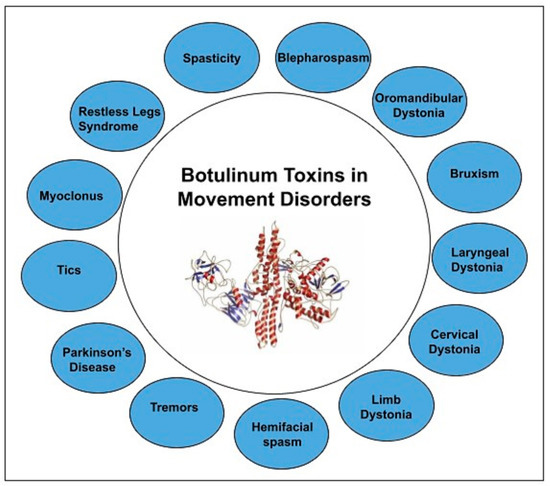
Figure 1. BoNT in Movement disorders.
2. Discussion of BoNT Use in Different Indications
2.1. Dystonia
Dystonia is defined as a movement disorder characterized by sustained or intermittent muscle contractions causing abnormal, often repetitive, patterned movements, postures, or both[27][28].
It is frequently associated with activity but may also be present at rest and worsens with stress, anxiety, and fatigue [29]. The prevalence has been reported to range between 15 and 225 per 100,000 individuals[30].
We conducted a PubMed search on 28 May 2020; using the title words botulinum and dystonia, a total of 438 articles were identified. 340 of these were in English and were human studies. Of the 340 articles, 71 were review articles, 154 were either prospective or retrospective trials, 32 were randomized controlled trials (RCTs), 58 were case reports, 6 were commentaries, 8 were unavailable for review, and 11 articles were irrelevant. The clinical composition of 340 articles were different types of dystonia: 2 axial, 1 blepharospasm (BSP), 49 multiple types, 186 cervical dystonia (CD), 1 cranial, 15 unspecified, 17 laryngeal, 28 limb, 5 lingual, 30 oromandibular dystonia (OMD), 1 torsion, and 5 tardive.
2.2. Hemifacial Spasm
Hemifacial spasm (HFS) is a peripherally induced unilateral facial movement disorder characterized by irregular, clonic, or tonic contractions of muscles innervated by the ipsilateral facial nerve. Its estimated prevalence is around 10 in 100,000 [31][32]. The condition usually begins as spasms of lower eyelid on one side of the face, which eventually spreads to upper eyelid and other muscles in ipsilateral face, often associated with elevation of ipsilateral eyebrow referred to as the “other Babinski sign”[33]. The estimated prevalence is 14.5 and 7.4 per 100,000 in women and men, respectively[31]. Primary HFS is thought to be related to compression of the facial nerve at the exit zone by an aberrant blood vessel loop. Secondary HFS is related to prior facial nerve injury or Bell’s palsy or brain stem damage[34]; 76% and 21% of HFS are primary and secondary respectively[35]. There is some evidence that facial motor nucleus excitability is reduced after BoNT injections [36].
We conducted a PubMed search on 21 April 2020. Using botulinum and hemifacial as title words, we identified 157 articles; of these, 118 were in English and were human studies. Of the 118 articles, 9 were review articles, 74 were either prospective or retrospective trials, 13 were RCTs, 5 were case reports, 6 were commentaries, and 11 articles were irrelevant.
In 1985, Savino et al. published one of the earliest case series in 15 patients who experienced relief of HFS after BoNT injections[37]. In a series of patients with BSP (n = 70), HFS (n = 13), CD (n = 195), hand dystonia (n = 22), and oromandibular dystonia (n = 45) who underwent BoNT injections, 94%, 92%, 90%, 77%, and 73% experienced relief of their symptoms, respectively [38]. In another series, 98% of 130 patients with HFS patients experienced relief of symptoms after BoNT injection [39]. In a retrospective review of 100 HFS patients who were treated with a mean dose of 28 U of onabotulinumtoxinA and were followed for 4 years, showed a mean duration of effect of around 3.1 months and latency to onset of effect of 7.1 days[40]. There are numerous prospective and retrospective trial which evaluated the use of BoNTA injections that showed safety and benefit in patients with HFS[41][42][43][44][45][46]. Cakmur et al. evaluated pretarsal versus preseptal injections in 28 and 25 patients with HFS and BSP, respectively, and found that pretarsal BoNTA had better relief of symptoms, longer duration of effect, and lower incidence of ptosis [47]. Results from another study of 72 HFS and 38 BSP patients with a crossover design concluded that pretarsal and preseptal injections provided similar beneficial effects; however, the pretarsal group had longer duration of benefit[48]. A systematic review that was published recently stated that they did not identify RCTs of BoNTA in HFS [49]. In our practice, we inject mainly in the pretarsal portion of the orbicularis oculi in patients with BSP and HFS. Cochrane review (based on a single study with study size = 11) concluded that the benefit rate of BoNT in HFS was between 76–100% and that due to this effect size, it would be extremely hard and unethical to conduct new placebo-controlled trials with a large sample size[50].
Side effects of BoNT for HFS include ptosis (7.8–36%), double vision (1.6%), blurred vision (2.5%), dry eyes/exposure keratitis (2.5%), dysphagia (5.5%), facial droop (3.5–5.5%), eye lid swelling/ecchymosis (3.8%), nausea (2.5%), and conjunctival redness [47][51][52].
Tunc et al.[53] assessed BoNT injections efficacy in 69 patients with idiopathic HFS (n = 46) and those with HFS due to definite neurovascular compression (n = 23) and found that those with idiopathic HFS had more robust improvement with BoNT. Although some favor surgical vascular decompression as a treatment of HFS, most neurologists prefer BoNT, as there is a lower risk of permanent adverse effects such as facial paralysis and deafness [54]. Table 5 discusses the various randomized trials involving BoNT and HFS.
2.3. Tremors
Tremor, an involuntary, rhythmic, oscillatory movement of a body part, is the most common movement disorder in a movement disorder clinic[55][56]. When oral medications do not adequately control the tremors, as is the case in 30% of patients with essential tremor (ET), BoNT should be considered as a therapeutic option[57]. We conducted a PubMed search on 9 July 2020; using botulinum and tremor as title words, a total of 49 articles were identified. Of those, 43 of these were in English and were human studies. Of the 43 articles, 4 were review articles, 18 were either prospective or retrospective trials, 8 were RCTs, 12 were case reports, and 1 was commentary. The clinical subsets of the 43 articles were 8 ET, 2 ET PD, 3 jaw tremor, 6 palatal, 5 multiple tremor types, 9 vocal, 1 each for PD, tremor/tic, orthostatic, head tremor with CD, neuropathy-associated, head tremor, and multiple sclerosis associated with Holmes tremor.
In 1981, Jankovic and colleagues reported the earliest series of 51 patients with different tremor types who benefited from BoNT [58]. Trosh and Pullman published a prospective study with 26 patients (12 and 14 of PD and ET, respectively) who also benefited from BoNT [59]. Fixed doses, limited muscles being injected, and complicating weakness postinjections initially made BoNT use for tremors unsatisfactory. In 2015, a series of 28 PD patients with tremors underwent muscle selection of incobotulinumtoxinA and patients improved at 16 weeks [60]. In an open-label prospective trial, 31 ET patients received 3 cycles of BoNTA based on kinematic analysis guided muscle selection and dose administered; it showed that BoNTA reduced tremor by 47.7% at 6 weeks and the improvement lasted 18–30 weeks. In a series of 10 patients with ET who received BoNTA using kinematics every 16 weeks, a 33.8% functional improvement was noted when selected muscles were injected[61]. The series was later expanded to include 28 PD and 24 ET patients who were injected with BoNTA using computer-based kinematics[61].
Mittal and Jankovic (2019) had provided a systematic review of BoNT in tremors and concluded that most studies were open-label and that there was a need for well-designed controlled trials of BoNT in the treatment of ET and PD tremors. In a retrospective analysis by Niemann and Jankovic[62] of 91 patients (53 ET, 31 dystonic, 9 PD, 1 cerebellar), 81.3% of whom received injections into flexor carpi radialis or ulnaris (mean dose per limb 71.8 units of onabotulinumtoxinA), only 12.2% had transient weakness. This is in contrast to earlier double-blind, placebo-controlled studies by Jankovic et al. [63] and Brin et al.[64], during which the wrist extensors were also injected and, as a result, many patients experienced finger extensor weakness. Therefore, we no longer inject the extensor hand muscles[62]. In a series of 19 patients with proximal tremors, injections in muscles such as supra/infraspinatus, teres major/minor, biceps, triceps, deltoid, and pectoralis major resulted in at least moderate benefit in 63%, but 15% had no benefit [65]. In 20 patients with severe ET, BoNT (mean total dose 95.5 ± 40.58 per patient) improvement was noted in activities of daily living and in severity tremor scale [66]. The investigators also concluded that excluding extensor carpi muscle did not affect efficacy of BoNT. Table 1 lists RCTs associated with tremors and BoNT.
Table 1. RCTs in tremors and BoNT.
|
Study |
Study Design and Goal |
Method |
Results |
|
Mittal et al., 2018 [216] |
Randomized, double-blind placebo-controlled, prospective crossover trial
Assessed safety and usefulness of incobotulinumtoxinA for management of essential tremor (ET)
|
n = 33 Either placebo (normal saline) or 80–120 units of incobotulinumtoxinA with EMG guidance was injected in hand and forearm of patients with moderate to severe ET
|
Fahn Tolosa Marin score median comparison between incobotulinumtoxinA /placebo was 2 and placebo/incobotulinumtoxinA was 3 at week 8
Two patients in incobotulinumtoxinA group at hand weakness
IncobotulinumtoxinA was found useful in improving tremor scores in patients with ET
|
|
Mittal et al., 2017 [217] |
Randomized, double-blind placebo-controlled, prospective crossover trial
Assessed safety and usefulness of incobotulinumtoxinA for management of PD tremor
|
n = 30 Patients either received placebo or 7–12 injections of incobotulinumtoxinA (total dose 85–110units, using EMG guidance). The lumbricals (97%), FCR (90%), FDS (87%), FCU, pronator, and biceps (83%) were the most commonly injected muscles. |
UPDRS rest tremor (p < 0.001) and NIHCGC improved (p < 0.001) significantly at weeks 4 and 8.
IncobotulinumtoxinA was found useful in improving PD tremor scores and patient symptoms.
|
|
Bertram et al., 2013 [218] |
Randomized, double-blind placebo-controlled, prospective crossover trial
Studied safety and efficacy of abobotulinumtoxinA for postural orthostatic tremor (POT)
|
n = 8 POT diagnosed with electrophysiology were randomized to receive either placebo or 200 units of abobotulinumtoxinA in tibialis anterior. |
The tremor frequency remained unchanged. 200 units of abobotulinumtoxinA did not affect patient symptoms of unsteadiness and falls in POT. |
|
Walt et al., 2012 [219] |
Randomized double-blind crossover study
|
n = 23 Each limb was randomly assigned to either 100 units BoNTA (under EMG guidance) or placebo (0.9%) and the other treatment at 12 weeks.
|
Bain score improved after BoNT at 6 (p = 0.0005) and 12 weeks (p = 0.0001). Hand weakness was more common in BoNT group (42.2%) compared to placebo (6.1%). BoNTA can improve arm tremor in MS. |
|
Adler et al., 2004 [220] |
Randomized prospective study
Assessed BoNTA for voice tremor management |
n = 13 13 patients with voice tremor were randomized to receive either 1.25 or 2.5 or 3.75 U of BoNTA. |
Mean time of onset of efficacy was 2.3 days; mean tremor severity score improved by 1.4 points at week 2. Dysphagia was a noted adverse event. |
|
Brin et al., 2001 [213] |
Randomized double-blind trial
OnabotulinumtoxinA
Evaluates BoNTA for ET of hand |
n = 133 133 ET patients received 50U (n = 43) or 100 U (n = 45) or placebo (n = 45) under EMG guidance into FCR, FCU, ECR, and ECU.
In 100 U = 30U FCR, 30U FCU, 20U ECR, 20U ECU
In 50U = 15U FCR, 15U FCU, 10U ECR, 10U ECU
|
Postural component was lower after weeks 4 and 16, while kinetic component was lower at week 6. Grip strength was lower in high- and low-dose BoNT.
|
|
Jankovic et al., 1996 [212] |
Randomized double-blind placebo-controlled trial
Assesses BoNTA for essential hand tremor |
n = 25 BoNTA or Placebo injected into wrist flexors and extensors |
Tremors improved at 4 weeks (p < 0.05) compared to placebo. All BoNTA treated patients had finger weakness. |
|
Pahwa et al., 1995 [221] |
Randomized double-blind placebo-controlled trial
Assess BoNT for essential head tremor
|
n = 10 10 patients with head tremor got either normal saline or BoNT under EMG guidance and had the other treatment after 3 months
40 U in each SCM and 60 U in each splenius capitis |
Examiner 50% and 10% improvement in BoNT and placebo group
They inferred that BoNT may be helpful if patients did not respond to oral medications.
|
|
Rajan et al., 2020 [222] |
Randomized placebo-controlled trial
Assessed BoNT in upper extremity dystonic hand tremor |
n = 30 15 received placebo and 15 received onabotulinumtoxin A |
Fahn–Tolosa–Marin tremor rating scale total score was lower in BoNT group at weeks 6 (p < 0.001) and 12 (p = 0.03). |
|
Abbreviations |
Blepharospasm—BSP Botulinum toxin—BoNT Botulinum toxin A—BoNTA Botulinum toxin B—BoNTB Cervical dystonia—CD Electromyography—EMG Essential tremor—ET Extensor carpi radialis—ECR Extensor carpi ulnaris—ECU Flexor carpi radialis—FCR Flexor carpi ulnaris—FCU Parkinson’s disease—PD Postural orthostatic tremor—POT Sternocleidomastoid—SCM Unified Parkinson’s disease rating scale—UPDRS National Institutes of Health Collaborative Genetic Criteria—NIHCGC
|
|
|
2.4. Parkinson’s Disease
Parkinson’s Disease
PD is a neurodegenerative disease with incidence around 118 per 100,000 person years[67]. There are a variety of symptoms in PD that have been amenable to the treatment with BoNT including hand tremors, jaw tremors, axial dystonia, rectal dystonia, freezing of gait, sialorrhea, and levodopa-induced dyskinesias[55][68]. We conducted a PubMed search on 11 July 2020; using botulinum and Parkinson as title words, a total of 58 articles were identified. Of these, 49 of these were in English and were human studies. Of the 49 articles, 7 were review articles, 19 were either prospective or retrospective trials, 14 were RCTs, 7 were case reports, 1 was unavailable for review, and 1 was a commentary. Table 2 lists PD-related conditions amenable to BoNT treatment. Table 3 lists RCTs associated with PD and BoNT.
Dystonia
|
Numerous studies have tried treating various dystonic symptoms in patients with Parkinson’s disease [224,226,227]. |
| Jaw tremors | In three patients with PD jaw tremor who underwent Dysport injection, mean dose of 53 units into each masseter and improvement was noted in jaw tremor in all three patients without side effects [228]. |
| Freezing of gait | Freezing of gait (FOG) is thought to be due to activation of both agonist and antagonist muscle in the legs, which is similar to pathophysiology of dystonia, hence studies have looked into botulinum for freezing of gait [229,230]. |
| Sialorrhea | Increased drooling is seen in about 10% of PD [231] and multiple studies have looked at used of botulinum injection for sialorrhea [231,232]. |
| Overactive bladder | In four PD and two MSA patients with overactive bladder (OAB) complaints, 200 U BoNTA was injected into detrusor, and all patients experienced relief of symptoms without systemic adverse effects [233]. Similar results were seen in eight PD patients with OAB post-BoNTA [234]. |
| Constipation | In a study with PD patients with constipation (after excluding those related to slow movement in colon), in an open-label study, Botox was injected into puborectalis muscles and noted improvement in symptoms in 10 patients at 2 months [235]. |
Table 3. RCT associated with PD and BoNT.
| Study | Study Design and Goal | Method | Results |
|---|---|---|---|
| Rieu et al., 2018 [244] | Double-blind randomized trial Assessed incobotulinumtoxinA for foot dystonia related to Parkinson’s disease |
45 PD patients were injected with either 100UI incobotulinumtoxinA or placebo in flexor digitorum longus and brevis | Mean clinical global impression was better in the treatment group as compared to the placebo Pain and dystonia severity were reduced in the treatment group |
| Bruno et al., 2018 [245] | Randomized placebo-controlled double-blind crossover prospective trial Assessed BoNT for limb pain in PD |
n = 12 BoNTA under EMG was used at average dose of 241.6 U |
Temporary muscle weakness was seen in two patients (one in each group) BoNTA led to NRS score to drop significantly at week 4 (−1.75 points lower), whereas there was not a significant change in the placebo group |
| Narayanaswami et al., 2016 [232] | Randomized placebo-controlled double-blind crossover prospective trial Assessed incobotulinumtoxinA for treatment of drooling in PD |
n = 9 Subjects were randomized to receive either 100 U of incobotulinumtoxinA or saline was injected into each submandibular (30 U) and parotid glands (20 U). |
Saliva weight was similar between both groups pre- and postinjections One patient had difficulty chewing and swallowing while another had thicker saliva during the incobotulinumtoxinA injections In this study, incobotulinumtoxinA was not helpful for drooling in PD |
| Bonanni L et al., 2007 [246] | Randomized blinded crossover trial AbobotulinumtoxinA was used Assesses BoNT for lateral axial dystonia due to Parkinson’s disease |
n = 9 with lateral axial dystonia due to Parkinson’s disease Four patients received BoNT and five got placebo, and five got placebo and then switched over after 3 months 500 units were injected in four paraspinal muscle sites |
Six patients found BoNT to be effective, two had no change, and one had subjective improvement without change in lateral bending |
| Tassorelli et al., 2014 [247] | Randomized placebo-controlled double-blind prospective trial Assessed if BoNTA helped increase rehabilitation effects in PD patients with Pisa syndrome |
n = 26 They were randomized to receive rehabilitation therapy with or without BoNTA (total dose 50–200 UI) |
Patients who received rehabilitation therapy had better posture, but those who also received BoNTA had more pain reduction and longer improvement in clinical variables |
| Chinnapongse et al., 2012 [248] | Randomized placebo-controlled double-blind with sequential dose escalation Assessed BoNTB for sialorrhea in PD |
n = 54 They were randomized and given either placebo or 1500U/2500U/3500U of BoNTB into submandibular (250 units for each side) and parotid glands |
Dry mouth was seen in 15% of BoNTB patients. Drooling frequency and severity scale was better in BoNTB arm than placebo at four weeks (p < 0.05), and this was dose-dependent BoNTB is safe and effective for treatment of sialorrhea in PD |
| Espay et al., 2011 [225] | Double-blind crossover trial Assessed cervical BoNTA for treatment of levodopa-induced dyskinesia |
n = 12 EMG-guided BoNTA or placebo was injected in neck muscles. SCM 25U, Splenius capitis 50U divided into each side, trapezius 25 U bilaterally |
Four patients finished the 6-month trial There was a lack of positive effect. There was neck weakness |
| Guidubaldi et al., 2011 [249] | Randomized double-blind crossover trial Assessed BoNTA versus BoNTB for drooling in PD or ALS |
n = 27 (15 ALS and 12PD) Either got BoNTA or BoNTB ultrasound-guided into parotid and submandibular glands Either 250 U of abobotulinumtoxinA (BoNTA) or Neurobloc 2500 U (BoNTB) |
Latency to benefit was shorter for BoNTA (6.6 ± 4.1days) and BoNTB (3.2 ± 3.7days) Duration of effect was similar between both groups |
| Lagalla et al., 2009 [250] | Randomized double-blind placebo-controlled trial Assessed BoNTB for drooling in PD |
n = 36 Patients either got 4000 U of BoNTB or placebo |
Patients who received BoNTB noted 44.4% and 33.3% (moderate and dramatic) reduction in sialorrhea Useful effects lasted 19.2 ± 6.3 weeks in BoNTB-treated patients (p < 0.0001) |
| Kalf et al., 2007 [251] | Randomized prospective trial Compares BoNTA in submandibular versus parotid injections |
n = 17 These patients either received 150 MU abobotulinumtoxinA divided between each gland, either submandibular or parotid |
Two patients developed transient dysphagia (one in each group) Dry mouth was noted in three and one time after submandibular and parotid groups, respectively Within the submandibular group, DSFS and social consequences were improved. This was not seen in the parotid group 50% and 22% of patients in the submandibular and parotid groups were noted as responders |
| Lagalla et al., 2006 [252] | Double-blind randomized placebo-controlled study Assessed BoNTA for drooling in PD |
n = 32 They received 50 U of onabotulinumtoxinA in each parotid or placebo |
Patient that received BoNT had improved frequency of drooling and reduced social disability No adverse effects were reported |
| Wieler et al., 2005 [253] | Double-blind randomized placebo-controlled crossover study Assessed BoNTA for freezing of gait (FOG) |
n = 12 Patients got either BoNTA or placebo and had crossover for five visits 200–300 U was given in the gastrocnemius and soleus under EMG guidance (up to 150 U per limb) |
FOG did not improve after BoNT |
| Fernandez et al., 2004 [229] | Double-blind randomized placebo-controlled study Assessed BoNTB for FOG |
n = 14 14 were randomly given either 5000 U of BoNTB (n = 9) or placebo (n = 5) Injections were in soleus and gastrocnemius |
No difference noted in FOG between two groups |
| Dogu et al., 2004 [254] | Randomized prospective trial Assessed US-guided versus anatomically injected intraparotid BoNTA for drooling in PD |
n = 15 Patients were randomly given either US-guided (n = 8) or blind (n = 7) onabotulinumtoxinA injections into parotid (15 U in each parotid) |
Two patients in US-guided group had dry mouth Mean time to have lower saliva production was 4.1 days and duration of effect was about 4.4 months US guidance may be safe and easy to use |
| Ondo et al., 2004 [255] | Double-blind randomized placebo-controlled study Looks at BoNTB (rimabotulinumtoxinB) for drooling in PD |
n = 16 They either received BoNTB (1000 U in each parotid or 250 U in each submandibular) or placebo. |
Patients who got BoNT did improve on visual analogue scale (p < 0.001) and drooling scale (p < 0.05) BoNTB is effective for drooling in PD |
Abbreviations: ALS—Amyotrophic lateral sclerosis; oNT—Botulinum toxin; BoNTA—Botulinum toxin A—BoNTB—Botulinum toxin B; DSFS—Drooling severity and frequency score; EMG—Electromyography; FOG—Freezing of gait; MSA—Multiple system atrophy; PD—Parkinson’s disease; Ultrasound—US.
3. Conclusions
BoNT is a safe and powerful treatment strategy for a variety of hyperkinetic movement disorders. The indications have gradually expanded over the last four decades, making BoNT one the most versatile drugs in the world. With advancing research into mechanisms of action, improved methods of administration, and novel formulations, the field of therapeutic BoNT will continue to grow[14][69].
References
- Padda, I.S.; Tadi, P. Botulinum Toxin. In StatPearls [Internet]; StatPearls Publishing: Treasure Island, FL, USA , 2020.
- Pirazzini, M.; Rossetto, O.; Eleopra, R.; Montecucco, C. Botulinum neurotoxins: Biology, pharmacology, and toxicology. Pharmacol. Rev. 2017, 69, 200–235.
- Rossetto, O.; Pirazzini, M.; Fabris, F.; Montecucco, C. Botulinum Neurotoxins: Mechanism of Action. In Handbook of Experimental Pharmacology; Springer: Berlin/Heidelberg, Germany, 2020; doi:10.1007/164_2020_355.
- Lonati, D.; Schicchi, A.; Crevani, M.; Buscaglia, E.; Scaravaggi, G.; Maida, F.; Cirronis, M.; Petrolini, V.M.; Locatelli, C.A. Foodborne botulism: Clinical diagnosis and medical treatment. Toxins 2020, 12, 509 .
- Wang, Y.; Fry, H.C.; Skinner, G.E.; Schill, K.M.; Duncan, T.V. Detection and Quantification of Biologically Active Botulinum Neurotoxin Serotypes A and B Using a Förster Resonance Energy Transfer-Based Quantum Dot Nanobiosensor. ACS Appl. Mater. Interfaces 2017, 9, 18, doi:10.1021/acsami.7b08736.
- Camargo, C.H.F.; Teive, H.A.G. Use of botulinum toxin for movement disorders. Drugs Context 2019, 8, 1–14, doi:10.7573/dic.212586.
- Rossetto, O.; Pirazzini, M.; Montecucco, C. Botulinum neurotoxins: Genetic, structural and mechanistic insights. Nat. Rev. Genet. 2014, 12, 535–549, doi:10.1038/nrmicro3295.
- Rosales, R.L.; Dressler, D. On muscle spindles, dystonia and botulinum toxin. Eur. J. Neurol. 2010, 17, 71–80, doi:10.1111/j.1468-1331.2010.03056.x.
- Hallett, M.; Albanese, A.; Dressler, D.; Segal, K.R.; Simpson, D.M.; Truong, D.; Jankovic, J. Evidence-based review and assessment of botulinum neurotoxin for the treatment of movement disorders. Toxicon 2013, 67, 94–114, doi:10.1016/j.toxicon.2012.12.004.
- Jankovic, J. Botulinum toxin: State of the art. Mov. Disord. 2017, 32, 1131–1138, doi:10.1002/mds.27072.
- De Paiva, A.; Meunier, F.A.; Molgó, J.; Aoki, K.R.; Dolly, J.O. Functional repair of motor endplates after botulinum neurotoxin type a poisoning: Biphasic switch of synaptic activity between nerve sprouts and their parent terminals. Proc. Natl. Acad. Sci. USA. 1999, 96, 3200–3205, doi:10.1073/pnas.96.6.3200.
- Rogozhin, A.A.; Pang, K.K.; Bukharaeva, E.; Young, C.; Slater, C.R. Recovery of mouse neuromuscular junctions from single and repeated injections of botulinum neurotoxin A. J. Physiol. 2008, 586, 3163–3182, doi:10.1113/jphysiol.2008.153569.
- Jankovic, J.; Orman, J. Botulinum a toxin for cranial-cervicaldystonia: A double-blind, placebo-controlled study. Neurology 1987, 37, 616–623, doi:10.1212/wnl.37.4.616.
- Jankovic, J. An update on new and unique uses of botulinum toxin in movement disorders. Toxicon 2018, 147, 84–88, doi:10.1016/j.toxicon.2017.09.003.
- Jankovic, J.; Truong, D.; Patel, A.T.; Brashear, A.; Evatt, M.; Rubio, R.G.; Oh, C.K.; Snyder, D.; Shears, G.; Comella, C. Injectable DaxibotulinumtoxinA in Cervical Dystonia: A Phase 2 Dose-Escalation Multicenter Study. Mov. Disord. Clin. Pract. 2018, 5, 273–282, doi:10.1002/mdc3.12613.
- Popescu, M.N.; Popescu, S.I.; Cernat, C.C.; Boariu, A.M.; Călin, E.; Vieru, A.; Muşat, O. Injecting botulinum toxin into the treatment of blepharospasm. Rom. J. Ophthalmol. 2018, 61, 162–165, doi:10.22336/rjo.2018.24.
- Dressler, D. Clinical Pharmacology of Botulinum Toxin Drugs. In Handbook of Experimental Pharmacology; Springer: Berlin/Heidelberg, Germany, 2019.
- Albrecht, P.; Jansen, A.; Lee, J.I.; Moll, M.; Ringelstein, M.; Rosenthal, D.; Bigalke, H.; Aktas, O.; Hartung, H.P.; Hefter, H. High prevalence of neutralizing antibodies after long-term botulinum neurotoxin therapy. Neurology 2019, 92, E48–E54, doi:10.1212/WNL.0000000000006688.
- Atassi, M.Z.; Jankovic, J.; Dolimbek, B.Z. Neutralizing antibodies in dystonic patients who still respond well to botulinum toxin type A. Neurology 2008, 71, 1040–1041, doi:10.1212/01.wnl.0000327865.05877.17.
- Bellows, S.; Jankovic, J. Immunogenicity associated with botulinum toxin treatment. Toxins 2019, 11, 491, doi:10.3390/toxins11090491.
- Naumann, M.; Boo, L.M.; Ackerman, A.H.; Gallagher, C.J. Immunogenicity of botulinum toxins. J. Neural Transm. 2013, 120, 275–290.
- Jankovic, J.; Schwartz, K. Response and immunoresistance to botulinum toxin injections. Neurology 1995, 45, 1743–1746, doi:10.1212/WNL.45.9.1743.
- Brin, M.F.; Comella, C.L.; Jankovic, J.; Lai, F.; Naumann, M.; Ahmed, F.; Brashear, A.; Chehrenama, M.; Erjanti, H.; Evatt, M.; et al. Long-term treatment with botulinum toxin type A in cervical dystonia has low immunogenicity by mouse protection assay. Mov. Disord. 2008, 23, 1353–1360, doi:10.1002/mds.22157.
- Andraweera, N.D.; Andraweera, P.H.; Lassi, Z.S.; Kochiyil, V. Effectiveness of Botulinum Toxin A injection in managing mobility related outcomes in adult patients with cerebral palsy—Systematic review. Am. J. Phys. Med. Rehabil. 2020 , doi:10.1097/phm.0000000000001653.(Article published ahead of print)
- Hareb, F.; Bertoncelli, C.M.; Rosello, O.; Rampal, V.; Solla, F. Botulinum Toxin in Children with Cerebral Palsy: An Update. Neuropediatrics 2020, 51, 1–5.
- Jia, S.; Liu, Y.; Shen, L.; Liang, X.; Xu, X.; Wei, Y. Botulinum Toxin Type A for Upper Limb Spasticity in Poststroke Patients: A Meta-analysis of Randomized Controlled Trials. J. Stroke Cerebrovasc. Dis. 2020, 29, 104682, doi:10.1016/j.jstrokecerebrovasdis.2020.104682.
- Albanese, A.; Bhatia, K.; Bressman, S.B.; Delong, M.R.; Fahn, S.; Fung, V.S.C.; Hallett, M.; Jankovic, J.; Jinnah, H.A.; Klein, C.; et al. Phenomenology and classification of dystonia: A consensus update. Mov. Disord. 2013, 28, 863–873.
- Balint, B.; Mencacci, N.E.; Valente, E.M.; Pisani, A.; Rothwell, J.; Jankovic, J.; Vidailhet, M.; Bhatia, K.P. Dystonia. Nat. Rev. Dis. Prim. 2018, 4, doi:10.1038/s41572-018-0023-6.
- Bakke, M.; Baram, S.; Dalager, T.; Biernat, H.B.; Møller, E. Oromandibular dystonia, mental distress and oro-facial dysfunction—A follow-up 8–10 years after start of treatment with botulinum toxin. J. Oral Rehabil. 2019, 46, 441–449, doi:10.1111/joor.12768.
- Steeves, T.D.; Day, L.; Dykeman, J.; Jette, N.; Pringsheim, T. The prevalence of primary dystonia: A systematic review and meta-analysis. Mov. Disord. 2012, 27, 1789–1796, doi:10.1002/mds.25244.
- Rudzińska, M.; Wójcik, M.; Szczudlik, A. Hemifacial spasm non-motor and motor-related symptoms and their response to botulinum toxin therapy. J. Neural Transm. 2010, 117, 765–772, doi:10.1007/s00702-010-0416-5.
- Ozzello, D.J.; Giacometti, J.N. Botulinum toxins for treating essential blepharospasm and hemifacial spasm. Int. Ophthalmol. Clin. 2018, 58, 49–61, doi:10.1097/IIO.0000000000000203.
- Stamey, W.; Jankovic, J. The other Babinski sign in hemifacial spasm. Neurology 2007, 69, 402–404, doi:10.1212/01.wnl.0000266389.52843.3b.
- Butera, C.; Guerriero, R.; Amadio, S.; Ungaro, D.; Tesfaghebriel, H.; Bianchi, F.; Comi, G.; Del Carro, U. Functional end-plate recovery in long-term botulinum toxin therapy of hemifacial spasm: A nerve conduction study. Neurol. Sci. 2013, 34, 209–215, doi:10.1007/s10072-012-0987-z.
- Karp, B.I.; Alter, K. Botulinum Toxin Treatment of Blepharospasm, Orofacial/Oromandibular Dystonia, and Hemifacial Spasm. Semin. Neurol. 2016, 36, 84–91, doi:10.1055/s-0036-1571952.
- Ishikawa, M.; Takashima, K.; Kamochi, H.; Kusaka, G.; Shinoda, S.; Watanabe, E. Treatment with botulinum toxin improves the hyperexcitability of the facial motoneuron in patients with hemifacial spasm. Neurol. Res. 2010, 32, 656–660, doi:10.1179/174313209X431129.
- Savino, P.J.; Sergott, R.C.; Bosley, T.M.; Schatz, N.J. Hemifacial Spasm Treated With Botulinum A Toxin Injection. Arch. Ophthalmol. 1985, 103, 1305–1306, doi:10.1001/archopht.1985.01050090057031.
- Jankovic, J.; Schwartz, K.; Donovan, D.T. Botulinum toxin treatment of cranial-cervical dystonia, spasmodic dysphonia, other focal dystonias and hemifacial spasm. J. Neurol. Neurosurg. Psychiatry 1990, 53, 633–639, doi:10.1136/jnnp.53.8.633.
- Taylor, J.D.N.; Kraft, S.P.; Kazdan, M.S.; Flanders, M.; Cadera, W.; Orton, R.B. Treatment of blepharospasm and hemifacial spasm with botulinum A toxin: A Canadian multicentre study. Can. J. Ophthalmol. 1991, 26, 133–138.
- Batisti, J.P.M.; Kleinfelder, A.D.F.; Galli, N.B.; Moro, A.; Munhoz, R.P.; Teive, H.A.G. Treatment of hemifacial spasm with botulinum toxin type a: Effective, long lasting and well tolerated. Arq. Neuro-Psiquiatr. 2017, 75, 87–91, doi:10.1590/0004-282x20160191.
- Price, J.; O’Day, J. Efficacy and side effects of botulinum toxin treatment for blepharospasm and hemifacial spasm. Aust. N. Z. J. Ophthalmol. 1994, 22, 255–260, doi:10.1111/j.1442-9071.1994.tb00793.x.
- Snir, M.; Weinberger, D.; Bourla, D.; Kristal-Shalit, O.; Dotan, G.; Axer-Siegel, R. Quantitative changes in botulinum toxin a treatment over time in patients with essential blepharospasm and idiopathic hemifacial spasm. Am. J. Ophthalmol. 2003, 136, 99–105, doi:10.1016/S0002-9394(03)00075-8.
- Cillino, S.; Raimondi, G.; Guépratte, N.; Damiani, S.; Cillino, M.; Di Pace, F.; Casuccio, A. Long-term efficacy of botulinum toxin A for treatment of blepharospasm, hemifacial spasm, and spastic entropion: A multicentre study using two drug-dose escalation indexes. Eye 2010, 24, 600–607, doi:10.1038/eye.2009.192.
- Cannon, P.S.; MacKenzie, K.R.; Cook, A.E.; Leatherbarrow, B. Difference in response to botulinum toxin type A treatment between patients with benign essential blepharospasm and hemifacial spasm. Clin. Exp. Ophthalmol. 2010, 38, 688–691, doi:10.1111/j.1442-9071.2010.02303.x.
- Bastola, P.; Chaudhary, M.; Agrawal, J.P.; Shah, D.N. The role of the injection botulinum toxin A in cases of essential blepharospasm syndrome, hemifacial spasm and meige’s syndrome. Kathmandu Univ. Med. J. 2010, 8, 305–310, doi:10.3126/kumj.v8i3.6217.
- Colosimo, C.; Chianese, M.; Giovannelli, M.; Contarino, M.F.; Bentivoglio, A.R. Botulinum toxin type B in blepharospasm and hemifacial spasm. J. Neurol. Neurosurg. Psychiatry 2003, 74, 687, doi:10.1136/jnnp.74.5.687.
- Çakmur, R.; Ozturk, V.; Uzunel, F.; Donmez, B.; Idiman, F. Comparison of preseptal and pretarsal injections of botulinum toxin in the treatment of blepharospasm and hemifacial spasm. J. Neurol. 2002, 249, 64–68, doi:10.1007/PL00007849.
- Sacramento, D.R.C.; Lima, A.; Maia, D.P.; Cunningham, M.; Maciel, R.H.; Camargos, S.T.; Cardoso, F. Comparison of techniques of botulinum toxin injections for blepharospasm and hemifacial spasm. Mov. Disord. 2019, 34, 1401–1403, doi:10.1002/mds.27789.
- Duarte, G.S.; Rodrigues, F.B.; Castelão, M.; Marques, R.E.; Ferreira, J.; Sampaio, C.; Moore, A.P.; Costa, J. Botulinum toxin type A therapy for hemifacial spasm. Cochrane database Syst. Rev. 2020, 11, doi:10.1002/14651858.CD004899.pub3.
- Costa, J.; Espírito-Santo, C.C.; Borges, A.A.; Ferreira, J.; Coelho, M.M.; Moore, P.; Sampaio, C. Botulinum toxin type A therapy for hemifacial spasm. Cochrane Database Syst. Rev. 2005, doi:10.1002/14651858.cd004899.pub2.
- Kalra, H.K.; Magoon, E.H. Side effects of the use of botulinum toxin for treatment of benign essential blepharospasm and hemifacial spasm. Ophthalmic Surg. 1990, 21, 335–338, doi:10.3928/1542-8877-19900501-08.
- Brin, M.F.; Fahn, S.; Moskowitz, C.; Friedman, A.; Shale, H.M.; Greene, P.E.; Blitzer, A.; List, T.; Lange, D.; Lovelace, R.E.; et al. Localized injections of botulinum toxin for the treatment of focal dystonia and hemifacial spasm. Mov. Disord. 1987, 2, 237–254, doi:10.1002/mds.870020402.
- Tunç, T.; Çavdar, L.; Karadaǧ, Y.S.; Okuyucu, E.; Coşkun, Ö.; Inan, L.E. Differences in improvement between patients with idiopathic versus neurovascular hemifacial spasm after botulinum toxin treatment. J. Clin. Neurosci. 2008, 15, 253–256, doi:10.1016/j.jocn.2007.02.002.
- Lu, A.Y.; Yeung, J.T.; Gerrard, J.L.; Michaelides, E.M.; Sekula, R.F.; Bulsara, K.R. Hemifacial spasm and neurovascular compression. Sci. World J. 2014, 2014, doi:10.1155/2014/349319.
- Mittal, S.O.; Lenka, A.; Jankovic, J. Botulinum toxin for the treatment of tremor. Park. Relat. Disord. 2019, 63, 31–41, doi:10.1016/j.parkreldis.2019.01.023.
- Kreisler, A.; Bouchain, B.; Defebvre, L.; Krystkowiak, P. Treatment with Botulinum Neurotoxin Improves Activities of Daily Living and Quality of Life in Patients with Upper Limb Tremor. Tremor Other Hyperkinet. Mov. 2019, 9, 1–7, doi:10.7916/tohm.v0.640.
- Samotus, O.; Lee, J.; Jog, M. Personalized bilateral upper limb essential tremor therapy with botulinum toxin using kinematics. Toxins 2019, 11, 1–12, doi:10.3390/toxins11020125.
- Jankovic, J.; Schwartz, K. Botulinum toxin treatment of tremors. Neurology 1991, 41, 1185–1188, doi:10.1212/wnl.41.8.1185.
- Trosch, R.M.; Pullman, S.L. Botulinum toxin a injections for the treatment of hand tremors. Mov. Disord. 1994, 9, 601–609, doi:10.1002/mds.870090604.
- Rahimi, F.; Samotus, O.; Lee, J.; Jog, M. Effective management of upper limb parkinsonian tremor by incobotulinumtoxina injections using sensor-based biomechanical patterns. Tremor Other Hyperkinetic Mov. 2015, 2015, 1–13, doi:10.7916/D8BP0270.
- Samotus, O.; Kum, N.; Rizek, P.; Jog, M. Botulinum Toxin Type A Injections as Monotherapy for Upper Limb Essential Tremor Using Kinematics. Can. J. Neurol. Sci. 2018, 45, 11–22, doi:10.1017/cjn.2017.260.
- Niemann, N.; Jankovic, J. Botulinum toxin for the treatment of hand tremor. Toxins 2018, 10, 299, doi:10.3390/toxins10070299.
- Jankovic, J.; Schwartz, K.; Clemence, W.; Aswad, A.; Mordaunt, J. A randomized, double-blind, placebo-controlled study to evaluate botulinum toxin type A in essential hand tremor. Mov. Disord. 1996, 11, 250–256, doi:10.1002/mds.870110306.
- Brin, M.F.; Lyons, K.E.; Doucette, J.; Adler, C.H.; Caviness, J.N.; Comella, C.L.; Dubinsky, R.M.; Friedman, J.H.; Manyam, B.V.; Matsumoto, J.Y.; et al. A randomized, double masked, controlled trial of botulinum toxin type A in essential hand tremor. Neurology 2001, 56, 1523–1528, doi:10.1212/WNL.56.11.1523.
- Kim, S.D.; Yiannikas, C.; Mahant, N.; Vucic, S.; Fung, V.S.C. Treatment of proximal upper limb tremor with botulinum toxin therapy. Mov. Disord. 2014, 29, 835–838, doi:10.1002/mds.25739.
- Pacchetti, C.; Mancini, F.; Bulgheroni, M.; Zangaglia, R.; Cristina, S.; Sandrini, G.; Nappi, G. Botulinum toxin treatment for functional disability induced by essential tremor. Neurol. Sci. 2000, 21, 349–353, doi:10.1007/s100720070049.
- Egevad, G.; Petkova, V.Y.; Vilholm, O.J. Sialorrhea in patients with Parkinson’s disease: Safety and administration of botulinum neurotoxin. J. Parkinsons. Dis. 2014, 4, 321–326, doi:10.3233/JPD-140379.
- Jocson, A.; Lew, M. Use of botulinum toxin in Parkinson’s disease. Park. Relat. Disord. 2019, 59, 57–64, doi:10.1016/j.parkreldis.2018.12.002.
- Schneider, S.A.; Edwards, M.J.; Cordivari, C.; Macleod, W.N.; Bhatia, K.P. Botulinum toxin A may be efficacious as treatment for jaw tremor in Parkinson’s disease. Mov. Disord. 2006, 21, 1722–1724, doi:10.1002/mds.21019.




