
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Anthony Novell | + 5213 word(s) | 5213 | 2020-12-28 10:25:23 | | | |
| 2 | Peter Tang | -108 word(s) | 5105 | 2021-01-30 10:25:27 | | |
Video Upload Options
The blood-brain barrier is the primary obstacle to efficient intracerebral drug delivery. Focused ultrasound, in conjunction with microbubbles, is a targeted and non-invasive way to disrupt the blood-brain barrier. Many commercially available ultrasound contrast agents and agents specifically designed for therapeutic purposes have been investigated in ultrasound-mediated blood-brain barrier opening studies. The new generation of sono-sensitive agents, such as liquid-core droplets, can also potentially disrupt the blood-brain barrier after their ultrasound-induced vaporization.
1. Introduction
The brain homeostasis is maintained by the blood-brain barrier (BBB), composed of tight junctions between endothelial cells on the vessel walls. The BBB, while preventing the entry of potentially harmful compounds, is the primary obstacle to efficient intracerebral delivery of almost all pharmaceuticals developed to treat neurological diseases, especially large molecule compounds [1]. Of the several techniques to deliver drugs across the BBB [1], the use of focused ultrasound (FUS) in conjunction with microbubbles is of great interest as it is targeted, transient, non-invasive, and safe [2]. The effectiveness of this technique was demonstrated for the first time by Hynynen et al. in 2001 [3]. After a few hours, gradual closure of the BBB and normal functioning was observed [1][4][5]. The safety has also been demonstrated in small animals and in non-human primates through histological evaluation and behavioral studies after FUS-mediated BBB disruption at multiple times and locations [6][7]. More recently, phase I and II clinical trials have shown the safety of this technique in humans [8]. This evidence strongly supports that, using suitable parameters, FUS is safe for BBB opening with a great potential to treat many brain diseases.
FUS-induced BBB opening enhances the delivery of drugs in the central nervous system [9][10][11]. Currently, the FUS-induced delivery of several compounds is under investigation for the treatment of diseases such as glioblastoma [12], neurodegenerative diseases like Alzheimer's [13], Parkinson's [14], or genetic diseases [15]. Additionally, FUS combined with microbubbles can achieve therapeutic results alone: it can induce, for example, neurogenesis [16] or reduce the amyloid load in Alzheimer's disease [17]. The approach is usually combined with magnetic resonance imaging (MRI), which enables the treatment guidance, the evaluation of BBB disruption using MR contrast agents, and the monitoring of potential damages during the procedure [3][18][19][20][21][22].
The addition of microbubbles has reduced the amount of ultrasound energy required to open the BBB by 100-fold [2][3]. Upon sonication, microbubbles start oscillating at the frequency of ultrasound. Above a certain acoustic pressure, the previously symmetrical oscillations of the bubble become unstable [23]. Those two different regimens are respectively called stable and inertial cavitation. Inertial cavitation can induce microbubble collapse accompanied by micro-jetting, fragmentation, and shock-wave formation, which may induce vascular endothelium damages [20][22][24][25]. On the other hand, the mechanical stress generated by the stable cavitation can locally and reversibly disrupt the tight junctions present in the vascular endothelial tissue, which increases the BBB permeability [26]. It is generally accepted that stable cavitation is the preferred regime for a safe BBB opening [26]. Figure 1 schematically presents the two oscillation regimens of microbubbles and their potential effects on BBB. The use of low acoustic pressure (few hundreds of kPa) ensures the safety of the technique by limiting any potential damages such as erythrocyte extravasation, hemorrhage, and necrotic damage [27] resulting from local thermal [28] or mechanical effects [24][27].
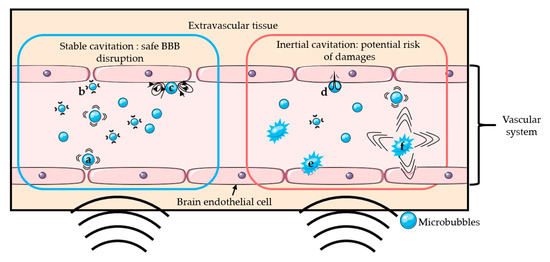
Figure 1. Schematic representation of several mechanisms of BBB disruption: Stable cavitation induces push- (a) and-pull (b) mechanism and microstreaming (c), which can permeabilize the blood-brain barrier safely. Inertial cavitation induces micro-jetting (d), fragmentation (e), and shock-wave (f) that can permeabilize the BBB with risks of damages.
Microbubbles consist of a gas core coated/encapsulated by a stabilizing shell. The coating provides a gas diffusion barrier while the gas core, composed of a heavy molecular weight inert gas, improves the bubble half-life after injection thanks to its low solubility in the surrounding medium [29][30]. Figure 2 shows a typical microscopic picture of polydisperse lipid-shelled microbubbles and their size distribution. The typical size of a microbubble is between 1 and 10 µm in diameter [29]. Sub-micronic bubbles (between 100 nm and 1 µm) are usually named nanobubbles in the literature. In order to be used for BBB opening, bubbles have to be (i) compressible to undergo cavitation, (ii) stable to circulate long enough to fulfill their duty, and (iii) non-toxic. After injection into the bloodstream, microbubbles circulate for only a few minutes before being cleared [31].
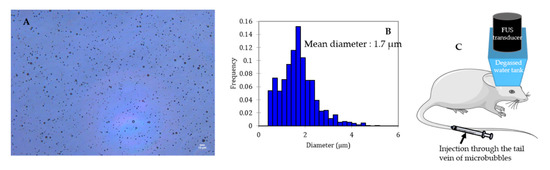
Figure 2. Size distribution characterization of a lipid shelled microbubble solution. Shell composition: 1,2-distearoyl-sn-glycero-3-phosphocholine (DSPC) 9:1 1,2-dimyristoyl-sn-glycero-3-phosphoethanolamine-N-[methoxy(polyethylene glycol)-2000] (DSPE-PEG2000). Core composition: decafluorobutane. (A) Microscopic picture of the solution (dilution: 1:10; scale: 10 µm), (B) size distribution of the solution, (C) schematic representation of BBB opening on mice.
Ultrasound contrast agents (UCAs) initially designed as echogenic contrast agents and clinically approved for diagnostic applications have also been shown capable of disrupting the BBB. However, bubbles can be formulated specifically for BBB disruption. Additionally, UCAs can be loaded with MRI contrast agent molecules, which allows imaging of their biodistribution. Targeting ligands can be added to the UCAs surface to increase their specificity or engineered with embedded drugs for targeted release, which reduces systemic drug effects.
An ultrasound can be used to convert droplets, called emulsions or phase-change contrast agents (PCCA), into microbubbles. For this reason, a droplet can also be designed for BBB opening. In vivo, a sufficient peak rarefactional pressure is necessary to vaporize the droplet's liquid core [32]. Acoustic droplet vaporization (ADV) and the cavitation of the resulting bubble can induce BBB opening [33].
2. Recent Advances on Sono-Sensitive Agents for Ultrasound-Assisted Blood-Brain Barrier Opening
2.1. Commercial Ultrasound Contrast Agents
Commercially available UCAs used for BBB opening are either clinically approved as imaging contrast agents (Food and Drug Administration or European Medicines Agency) or are being developed specifically for therapeutic purposes. The clinically approved microbubbles used to disrupt the BBB with FUS are DefinityTM (Lantheus Medical Imaging, North Billerica, MA, USA), SonoVue®/Lumason® (Bracco, Milan, Italy), OptisonTM (GE Healthcare, Milwaukee, WI, USA), and Sonazoid® (GE Healthcare, Milwaukee, WI, USA). Other commercially available UCAs resulted in successful BBB opening such as USphere® (Trust Bio-sonics, Zhubei City, Taiwan), SIMB® (Advanced Microbubbles Inc, Boulder, CO, USA), Vevo MicroMarker® (Fujifilm, Toronto, ON, Canada), BR-38® and others (Bracco Suisse SA, Geneva, Switzerland), and bubbles ordered from Targeson Inc. (San Diego, CA, USA) These agents consist of lipid or protein shells with gaseous sulfur hexafluoride (SF6) or perfluorocarbon (PFC) cores. Table 1 summarizes the commercial and non-commercial UCAs used to disrupt the BBB with ultrasounds in pre-clinical studies during the past 5 years, their composition, and associated references.
Table 1. Composition and phase of sono-sensitive agents used for BBB opening over the past 5 years.
|
Commercial Name |
Phase |
Core |
Shell |
References |
|
Definity (Lantheus Medical Imaging) |
gas |
C3F8 |
lipid |
[12][13][16][34][35][36][37][38][39][40][41][42][43][44][45][46][47][48][49][50][51] |
|
SonoVue/Lumason (Bracco) |
gas |
SF6 |
lipid |
[15][34][35][52][53][54][55][56][57][58][59][60][61][62][63][64][65][66] |
|
Optison (GE Healthcare) |
gas |
C3F8 |
protein |
|
|
SIMB (Advanced Microbubbles Inc) |
gas |
“gas” |
lipid |
[73] |
|
Vevo MicroMarker (Fujifilm) |
gas |
C4F10 and N2 |
lipid |
[74] |
|
BG8235 similar to BR-38 (Bracco) |
gas |
C4F10 |
lipid |
[75] |
|
Targeson Inc |
gas |
PFC |
lipid |
[76] |
|
USphere (Trust Bio-sonics) |
gas |
C3F8 |
lipid |
[35] |
|
Sonazoid (GE Healthcare) |
gas |
C4F10 |
lipid |
[66] |
|
Sonazoid (GE Healthcare) |
AC: gas and liquid |
Sonazoid bubbles: C4F10 Homemade droplets: C6F12 |
lipid |
[77] |
|
Non-commercial |
liquid |
C3F8 or C4F10 |
lipid |
[33] |
|
Non-commercial |
liquid |
C5F12 |
lipid |
[78] |
|
Non-commercial |
gas |
C3F8 |
protein |
|
|
Non-commercial |
gas |
C3F8 |
self-assembled polymeric nanoparticles |
[82] |
|
Non-commercial |
gas |
C3F8 |
self-assembled polymeric nanoparticles and protein |
[83] |
|
Non-commercial |
gas |
air |
polymer |
|
|
Non-commercial |
gas |
C5F12 |
polymer |
[86] |
|
Non-commercial |
gas |
SF6 |
lipid |
|
|
Non-commercial |
gas |
C4F10 |
lipid |
[6][11][14][37][66][89][90][91][92][93][94][95][96][97][98][99][100][101] |
|
Non-commercial |
gas |
C3F8 |
lipid |
[36][66][102][103][104][105][106][107][108][109][110][111][112][113][114][115][116][117][118][119][120][121][122][123][124] |
AC: Acoustic cluster; PFC: perfluorocarbon; C3F8: octafluoropropane; C4F10: decafluorobutane; C5F12: dodecafluoropentane; C6F12: perfluoromethylcyclopentane; SF6: sulphur hexafluoride; N2: nitrogen.
We came across four studies comparing different commercially available bubbles on their abilities to open the BBB by assessing Evans blue leakage in the brain (commonly used dye for BBB permeability assessment, otherwise blocked by the BBB). The obtained results for these 4 studies are summarized in Table 2, along with the bubble type, injection dose, and ultrasonic parameters used for the BBB disruption. Briefly, Shin et al. compared a dose of SonoVue at 30 µL/kg with two different doses of Definity at 20 µL/kg and 100 µL/kg. Definity microbubbles at a 20 µL/kg dose were more effective for BBB opening and led to fewer damages than SonoVue. For the 100 µL/kg dose of Definity, the BBB opening was more important, and the level of tissue damages (histological evaluation) was similar to SonoVue microbubbles at 30 µL/kg [34]. Wu et al. compared SonoVue, Definity, and USphere at an injected microbubble concentration of 4 × 107 bubbles/kg. For a given set of sonication parameters, the order of Evans blue penetration (from the most important to the weakest) was: SonoVue, Definity, Usphere [35]. Bing et al. compared Optison and Definity performances. The concentration of bubbles was adjusted to inject the same gas volume (1.1–1.2 µL/mL). Evans blue leakage was more important for Optison microbubbles than for Definity [36]. Finally, Omata et al. compared SonoVue and Sonazoid at an injected microbubble concentration of 3 × 109 bubbles/kg and showed a higher Evans blue leakage with the Sonazoid bubbles [66].
Table 2. Comparison of different commercial bubbles regarding BBB opening.
|
Ref |
Bubble Type |
Injection Dose (µL/kg) |
Number of Bubbles per mL |
Animal |
Acoustic Parameters |
Evans Blue Leakage |
Damages Score |
|
Bing et al. [36] |
Optison |
30 |
7 × 108 |
Sprague Dawley rats (230–300 g) |
PnP = 0.47 MPa |
High |
NA |
|
Definity |
6 |
1 × 1010 |
fc = 0.75 MHz |
Moderate |
NA |
||
|
PRF = 1 Hz |
|||||||
|
duration = 120 s |
|||||||
|
burst = 10 s |
|||||||
|
Shin et al. [34] |
SonoVue |
30 |
2 × 108 |
Sprague Dawley rats (250–300 g) |
PnP = 0.3 MPa |
4.45% |
1 |
|
fc = 0.5 MHz |
|||||||
|
Definity |
20 |
1 × 1010 |
PRF = 2 Hz |
13.72% |
0 |
||
|
Definity |
100 |
1 × 1010 |
duration = 60 s |
16.35% |
1 |
||
|
burst = 10 s |
|||||||
|
Wu et al. [35] |
SonoVue |
200 |
2 × 108 |
Sprague Dawley rats (250–300 g) |
PnP = 0.39 MPa |
0.79 ± 0.24 µM |
0 |
|
fc = 0.4 MHz |
|||||||
|
PRF = 1 Hz |
|||||||
|
Definity |
4 |
1 × 1010 |
duration = 120 s |
0.52 ± 0.25 µM |
0 |
||
|
burst = 10 s |
|||||||
|
USphere |
1.43 |
2.8 × 1010 |
0.2 ± 0.04 µM |
0 |
|||
|
Omata et al. [66] |
Sonazoid |
3333 |
9 × 108 |
ddY mice (6 weeks old) |
Intensity = 0.5 W/cm2 |
18 ± 7 µg/g brain |
0 |
|
SonoVue |
30,000 |
1 × 108 |
fc = 3 MHz |
5 ± 1 µg/g brain |
0 |
||
|
PRF = 10 Hz |
|||||||
|
duration = 180 s |
|||||||
|
burst = 50 s |
PnP = Peak negative Pressure; fc = central frequency; PRF = Pulse repetition frequency; NA: non-assessed; the damage score grades hemorrhage and tissue damages: grade 0—normal tissue, grade 1—scattered or discontinuous erythrocyte extravasation, grade 2—continuous extravasation or microhemorrhage, grade 3—hemorrhage with necrotic damage or gross hemorrhage [27].
In the Wu et al. study, a 200 µL/kg dose of SonoVue microbubbles successfully opened the BBB without damages using a higher peak negative pressure and a more prolonged exposure than the Shin et al. study where damages on the rat brain were observed for a 20 µL/kg dose of SonoVue. This inconsistency between the two studies points out that the BBB opening effectiveness and safety should be studied in-depth, ideally comparing different commercially available bubbles. Omata et al. showed that a 30,000 µL/kg dose of SonoVue successfully opened the mice BBB without damages. However, the comparison with Wu et al. and Shin et al. is trickier as they studied mice for which the skull is thinner than rats. Furthermore, ultrasound parameters such as the frequency used (3 MHz) for BBB opening differs from other studies making the comparison challenging. As another example, the Wu et al. study demonstrates a higher efficacy of SonoVue over Definity while the Shin et al. study tends in the opposite direction. Importantly, injected doses play a critical role. Besides, commercially available agents explicitly made for BBB opening purposes would be desirable.
2.2. Design of Specific Agents for Blood-Brain Barrier Opening
While clinically-approved contrast agents expedite therapeutic clinical trials, their compositions originally developed for diagnostic imaging can be suboptimal for BBB opening [125]. Moreover, designing specific BBB disruption agents makes possible their use as multifunctional agents for imaging and drug delivery purposes in theranostics.
Agents designed for BBB disruption, like UCAs, have to fulfill several criteria such as (i) easily reachable stable cavitation regimen, (ii) long in vivo circulation stability, and (iii) storage stability. Thereby, bubbles must undergo sufficient oscillations for a relatively low peak rarefactional pressure before reaching inertial cavitation. Stable and inertial cavitation thresholds have to be assessed, meaning that the lowest acoustic pressure required for the bubble to reach these regimens has to be estimated. The vaporization threshold for droplets, namely the magnitude of acoustic pressure required to convert a liquid droplet into a gaseous bubble, also has to be considered [126]. Over the 55 studies describing agents designed for BBB opening procedures, only 15 performed at least one test to assess cavitation thresholds or stability performances (Table 3). These tests are crucial for the success and safety of the experiments [89]. Therefore, they should be performed on a routine basis, especially since those parameters can be incredibly different from an agent to another (acoustic stability on echography images ranging from few seconds [66] to days [86] depending on the bubble). Currently, most of the bubble-based therapeutic ultrasound protocols are limited to a few minutes or require a repeatable injection of fresh UCAs to extend the circulation of microbubbles in the body and increase the procedure's efficiency. For commercial microbubbles, the half-life in the bloodstream is typically lower than 10 min [127], and extending the in vivo circulation of these agents is desirable.
Table 3. Stability and cavitation thresholds assessment for sono-sensitive agents for BBB opening; without other specifications, cavitation thresholds are detected with in vitro passive cavitation detection. The storage stability was assessed by taking samples at several times after agent formulation and checking its echographic stability [78], cavitation emissions [106][123], concentration [37], or therapeutic effect [100].
|
Phase |
Core |
Shell |
Storage Stability |
In Vitro Acoustic Stability (Echography) |
In Vivo Half-Life |
Stable Cavitation Threshold |
Inertial Cavitation Threshold |
Ref. |
|
liquid |
C5F12 |
PEG-PLGA |
stable 2 days at 4 °C |
NA |
NA |
VT = 1.0 MPa (fc = 1 MHz) |
[78] |
|
|
liquid |
C3F8 or C4F10 |
DSPC: DSPE-PEG2000 (molar ratio 9:1) |
NA |
NA |
NA |
C3F8 VT = 0.3 MPa |
[33] |
|
|
C4F10 VT = 0.75 MPa |
||||||||
|
(fc = 1.5 MHz) |
||||||||
|
gas |
C3F8 |
DPPC: DSPE-PEG2000: DPTAP (molar ratio 9:2:1) |
NA |
relatively stable 50 min at 37 °C |
NA |
NA |
0.3 MPa (fc = 1MHz) |
[103] |
|
gas |
C3F8 |
DPTAP: DPPC: DSPE-PEG2000 (molar ratio 31,5:3,9:1,8) |
NA |
stable 1 h at 37 °C |
10 min (male C57BL/6J mice 20–25 g) |
0.3 MPa (fc = 1 MHz; BBB opening without damages) |
0.5 MPa |
[104] |
|
(fc = 1 MHz) |
||||||||
|
gas |
C3F8 |
DSPC: DSPE-PEG2000 (molar ratio 9:1) |
NA |
NA |
NA |
NA |
0.175 MPa |
[89] |
|
(fc = 0.25 MHz) |
||||||||
|
0.4 MPa |
||||||||
|
(fc = 1 MHz) |
||||||||
|
gas |
C3F8 |
DBPC: DPPA: DPPE: DSPE-PEG2000 (molar ratio 6,15:2:1:1) |
NA |
NA |
6–8 min (Sprague Dawley rats 230–300 g) |
0.21 MPa in vitro |
0.59 MPa in vitro |
[36] |
|
0.16 MPa in vivo |
0.47 MPa in vivo |
|||||||
|
(fc = 0.75 MHz) |
||||||||
|
gas |
C3F8 |
DBPC: DPPA: DPPE: DSPE-PEG2000 (molar ratio 6:1:2:1) |
stable 2 h at 5×1010 bubbles/mL |
NA |
10 min at 1011 bubbles/mL (Sprague Dawley rats 230–300 g) |
0.31 MPa in vivo |
0.70 MPa in vivo |
[106] |
|
(fc = 0.75 MHz; 1010 bubbles/mL) |
||||||||
|
gas |
C3F8 |
DSPC: DSPE-PEG2000 (molar ratio 9:1) |
NA |
NA |
8 min (nude mice) |
NA |
NA |
[121] |
|
gas |
C3F8 |
DPPC: DPTAP: DSPE-PEG2000 (molar ratio 31,5:3,9:1,8) |
NA |
stable 1 h at 37 °C |
NA |
0.3 MPa |
0.5 MPa |
[105] |
|
(fc = 1 MHz) |
(fc = 1 MHz) |
|||||||
|
gas |
C3F8 |
DSPC: DSPG: DSPE-PEG2000 (molar ratio 21:21:1) |
NA |
stable 1 h at 37 °C |
7.6 min for MB 10,8 min for SPIO-DOX-MB (Sprague Dawley rats 200–250 g) |
0.3 MPa |
0.5 MPa |
[107] |
|
(fc = 1 MHz; BBB opening without damages) |
(BBB opening with damages) |
|||||||
|
gas |
C4F10 |
DSPC: DSPE-PEG2000 (molar ratio 9:1) or DSPC: DSPE-PEG2000-Amine (molar ratio 9:1) or DSPC: DSPE-PEG2000-Amine: DSTAP (molar ratio 7:1:2) |
half-life of 2 h |
NA |
NA |
NA |
NA |
[37] |
|
gas |
C5F12 |
PEGGM-PDSGM |
NA |
stable after 14 days at 37 °C |
10 min (mice 25–35g) |
NA |
NA |
[86] |
|
gas |
C3F8 or C4F10 or SF6 |
DSPC: DSPG: DSPE-PEG2000 (molar ratio 30:60:10) |
NA |
half-life at 37 °C |
C3F8: 130 ± 50 s |
NA |
NA |
[66] |
|
C3F8: 80 ± 5 s |
||||||||
|
C4F10: 190 ± 40 s |
||||||||
|
C4F10: 145 ± 35 s |
||||||||
|
SF6: 20 ± 20 s |
||||||||
|
SF6: 20 ± 5 s |
||||||||
|
gas |
C4F10 |
DSPC: DSPE-PEG2000 (molar ratio 9:1) |
Stable 21 days |
NA |
NA |
NA |
NA |
[100] |
|
gas |
C3F8 |
DSPC: DSPG: DSPE-PEG2000 (molar ratio 10:4:1) |
relatively stable 60 min at 25 °C |
NA |
NA |
0.3 MPa (fc = 1 MHz; BBB opening without damages) |
0.5 MPa (BBB opening with damages) |
[123] |
fc = central frequency; NA = Non-assessed; VT: Vaporization threshold; SPIO-DOX-MB: superparamagnetic iron oxide–doxorubicin–microbubble complex; Core compositions: C3F8: octafluoropropane; C4F10: decafluorobutane; C5F12: dodecafluoropentane; C6F12: perfluoromethylcyclopentane; SF6: sulphur hexafluoride; Shell compositions: DBPC: 1,2-dibehenoyl-sn-glycero-3-phosphocholine; DPPA: 1,2 dipalmitoyl-sn-glycero-3-phosphate; DPPC: 1,2-dipalmitoyl-sn-glycero-3-phosphocholine; DPPE: 1,2-dipalmitoyl-sn-glycero-3-phosphoethanolamine; DPTAP: 1,2-dipalmitoyl-3-trimethylammonium-propane; DSPC: 1,2-distearoyl-sn-glycero-3-phosphocholine; DSPE-PEG2000: 1,2-distearoyl-sn-glycero-3-phosphoethanolamine-N-[methoxy(polyethyleneglycol)-2000]; DSPE-PEG2000-Amine: 1,2-distearoyl-sn-glycero-3-phosphoethanolamine-N-[amino(polyethyleneglycol)2000]; DSPG: 1,2-distearoyl-snglycero-3-phospho-rac-glycerol; DSTAP: 1,2-distearoyl-3-trimethylammonium-propane; PEGGM-PDSGM: poly(ethylene glycol-g-glutamate)-co-poly(distearin-g-glutamate); PEG-PLGA: poly(ethylene glycol)-poly(lactide-co-glycolic acid).
Several parameters may influence the bubbles/droplets circulation time as well as the cavitation threshold. For this reason, aspects such as size, chemical composition, shell properties (such as surface tension, elasticity, thickness, surface chemistry), and core properties (molecular weight, density, and boiling point for droplets) have to be taken into account when designing a new agent for FUS-induced BBB opening purpose [125][126][127]. Concretely, air, nitrogen, and mostly PFC and SF6 are used as the core gases, while surfactants, lipids, proteins, polymers, or a combination of these materials are used for the shell. For that matter, Figure 3 schematically represents the different possibilities of BBB disruption agent composition.
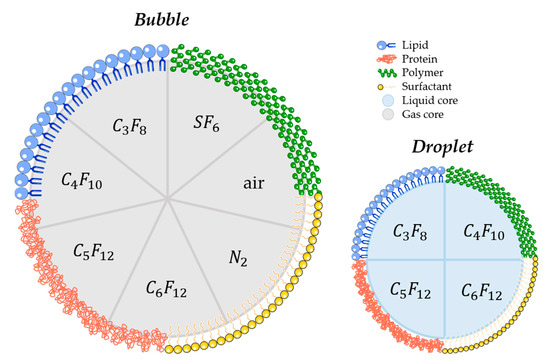
Figure 3. Schematic illustration of most commonly used materials for shell and core composition of agents used for FUS-induced BBB. (C3F8 octafluoropropane; C4F10: decafluorobutane; C5F12: dodecafluoropentane; C6F12: perfluoromethylcyclopentane; SF6: sulphur hexafluoride; N2: nitrogen).
2.3. Bimodal Ultrasound-MRI Contrast Agents for BBB Disruption
MRI is currently used to guide and evaluate the efficiency of therapeutic ultrasound procedures. It allows precise targeting, identification of the lesion, and dynamic feedback on the extent of BBB disruption via MR contrast leakage [1][18]. Two types of agents are used to enhance the contrast on MR images: (i) paramagnetic agents, which consist of a chelate with a paramagnetic core (usually gadolinium) or (ii) superparamagnetic agents composed of iron oxide nanoparticles coated with a hydrophilic organic protective layer such as dextran [128].
Most of the studies reported the use of paramagnetic agents made of gadolinium chelates commercially available such as Gadovist® (Gd-DO3A-butrol), Dotarem® (Gd-DOTA), Magnevist® (Gd-DTPA), Omniscan® (Gd-DTPA-BMA), and Multihance® (Gd-BOPTA) that are co-administered with the UCAs. Aryal et al. incorporated a gadolinium-labeled lipid in the lipid bilayer of liposomes injected as an MRI contrast agent. After BBB opening on mice with Optison® microbubbles, the signal intensity was slightly higher on longitudinal relaxation time (T1)-weighted images for the sonicated hemisphere than the control volume, indicating that the gadolinium-labeled liposomes were effectively delivered to the brain and visible on MR images. Two different sizes of liposomes were compared (77.5 nm and 140 nm), and the relative increase in MRI signal intensity was greater for smaller liposomes than larger ones [68].
Conventional UCAs have also been used for MRI [129] due to the gas-liquid interface producing large local magnetic susceptibility differences visible in transversal relaxation time (T2)-weighted MR images. For that matter, Cheung et al. used two types of bubbles (custom-made air-filled and albumin-coated microbubbles and SonoVue® microbubbles) for in vivo dynamic brain MRI in Sprague-Dawley rats. Transverse relaxation rate enhancements were observed in the brain after bubbles intravenous injection [130].
The microbubble response can be further enhanced by incorporating paramagnetic or superparamagnetic particles into their shells, giving rise to multimodal contrast agents used for BBB disruption and dynamic contrast-enhanced MRI. Liao et al. reported the use of perfluorocarbon-filled albumin-(Gd-DTPA) microbubbles for monitoring FUS-induced BBB opening [131]. T1-weighted MRI confirmed the BBB disruption, and T2-weighted MRI allowed to detect intracerebral hemorrhage. Besides, Fan et al. formulated a multimodal, therapeutic, and active-targeting microbubble encapsulating a superparamagnetic iron oxide-doxorubicin (SPIO-DOX) complex. The lipid microbubble filled with perfluoropropane successfully opened the BBB upon sonication, and the magnetic activation of SPIO nanoparticles triggered the release of chemotherapeutic agent DOX into rat glioma. Figure 4 schematically represents the SPIO-DOX-microbubble complex, the SPIO deposition as a function of 1/T2 values in MR images, and the DOX deposition into the rat brain as a function of the treatment used. The SPIO deposition into the rat brain tumor was correlated with differences in 1/T2 values in MR images (r2 = 0.83) and with DOX deposition (r2 = 0.79), supporting the theranostic capabilities of the SPIO-DOX-microbubble complex [107].
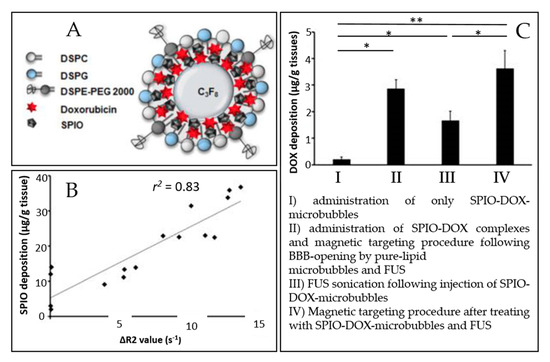
Figure 4. An example of a multimodal agent for BBB disruption, MRI, and drug delivery purposes (extracted from Fan et al. [107], Ivyspring International Publisher, 2016). (A) Illustration of SPIO-DOX-microbubble structure. (B) Correlation between SPIO deposition and ΔR2 value. (C) DOX accumulation measured by high-performance liquid chromatography. *: p < 0.05; **: p < 0.01.
PFC droplets can theoretically be observed on 19-F MRI since it has been reported in vitro [132]. Nevertheless, in vivo 19-F MRI imaging remains challenging due to the low PFC quantities available in the brain after BBB disruption.
2.4. Drug Delivery and Targeting
2.4.1. Targeting
Localized BBB opening can be achieved using physical stimuli (such as FUS or magnetic field), while functionalized agents with a ligand can target overexpressed receptors associated with the treated pathology.
Fan et al. used magnetic targeting as a physical targeting option: particles can be magnetized and become physically sensitive to external magnetic fields. This approach was validated in vivo using the SPIO-DOX-microbubble complex described earlier (schematically represented in Figure 4A). It increased SPIO deposition in the rat brain by 2.8 fold [107]. As well, Wu et al. made lipid-shelled bubbles conjugated with cationic polyethylenimine-coated superparamagnetic iron oxide particles (PSPIO) to open the BBB on mice. The SPIO, here again, was used for magnetic targeting and enhanced the BBB opening by 2.8-fold compared with unconjugated bubbles [123].
Molecular targeting of the circulating microbubbles would allow a BBB opening in the desired area without affecting healthy tissues. Typical ligands used for molecular targeting are antibodies or peptides. Table 4 summarizes the different molecular targeting and drug/gene-complexed bubble options for BBB opening and therapeutic purposes found in the recent literature.
Table 4. Molecular targeting and drug/gene-complexed bubble for BBB opening and therapeutic purpose.
|
Ref |
Core |
Shell |
Molecular Targeting |
Drug/Gene Embedded in the Agent |
|
[84] |
air |
polymer |
NA |
quercetin-modified sulfur nanoparticles-loaded bubble |
|
[86] |
C5F12 |
polymer |
des-octanoyl ghrelin-conjugated bubble |
TGFβ1 inhibitor (LY364947)-loaded bubble |
|
[87] |
SF6 |
lipid |
NA |
GDNFp/BDNFp-loaded liposome bound to bubble |
|
[88] |
SF6 |
lipid |
NA |
ultrasound-sensitizing dye-incorporating nanoparticles bound to bubble |
|
[108] |
C3F8 |
lipid |
NGR-conjugated (targeting)/shBirc5-loaded (gene) liposome bound to bubble |
|
|
[102] |
C3F8 |
lipid |
anti-VEGFR2 antibody-conjugated bubble |
pLUC / pHSV-TK/GCV-loaded bubble |
|
[107] |
C3F8 |
lipid |
NA |
SPIO-DOX-conjugated bubble |
|
[109] |
C3F8 |
lipid |
NA |
GDNFp-loaded cationic bubble |
|
[110] |
C3F8 |
lipid |
NA |
pDC315/Nrf2-loaded bubble |
|
[105] |
C3F8 |
lipid |
folate-conjugated bubble |
pLUC-loaded bubble |
|
[103] |
C3F8 |
lipid |
NA |
pPrestin-loaded bubble |
|
[104] |
C3F8 |
lipid |
NA |
boron-containing polyanion nanoparticles coupled with cationic bubble |
|
[123] |
C3F8 |
lipid |
NA |
PSPIO-GDNFp-loaded bubble |
|
[111] |
C3F8 |
lipid |
phosphatidylserine nanoparticles-microbubble complex |
|
TGFβ1: transforming growth factor; GDNFp: a glial cell line-derived neurotrophic factor plasmid DNA; BDNFp: brain-derived neurotrophic factor plasmid DNA; NGR: Asn-Gly-Arg peptide; shBirc5: short hairpin RNA-Birc5 gene; VEGFR2: vascular endothelial growth factor receptor 2; pLUC: plasmid DNA encoding luciferase gene; pHSV-TK/GCV: plasmid DNA encoding herpes simplex virus type 1 thymidine kinase/ganciclovir gene; SPIO-DOX: superparamagnetic iron oxide-doxorubicin complex; pDC315/Nrf2: plasmid DNA encoding DC315-nuclear factor E2-related factor 2; pPrestin: plasmid DNA encoding Prestin protein; PSPIO-GDNFp: superparamagnetic iron oxide coated with cationic polyethylenimine conjugated with plasmid DNA encoding glial cell line-derived neurotrophic factor; NA: Not applicable).
Chen et al. developed des-octanoyl ghrelin-conjugated microbubbles loaded with TGFβ1 inhibitor to disrupt BBB on glioma-bearing mice. Des-octanoyl ghrelin is a ligand that can bind with BBB. Authors have observed higher BBB disruption with the des-octanoyl ghrelin-conjugated microbubbles than the unconjugated ones (negative contrast intensity of superparamagnetic iron oxide nanoparticles on the T2-weighted MRI images was 0.81-fold higher for the conjugated microbubbles) [86].
Specific biomarkers of the blood-tumor barrier (BTB) could be used to target microbubbles toward these tumors. Indeed, gliomas and brain metastases are tumors known to compromise the integrity of the BBB, resulting in a vasculature known as the BTB, which is highly heterogeneous and characterized by numerous distinct features, including non-uniform permeability and active efflux of molecules [133]. Vascular endothelial growth factor receptor 2 (VEGFR2) is one of the selected targets as it is a specific endothelial molecular marker of angiogenesis, which is exceptionally high in tumor growth and, thus, overexpressed in BTB [134]. Moreover, the inhibition of VEGFR2 with antibodies results in prolonged survival in cancer patients [134]. Functionalized bubbles/droplets with VEGFR2-targeted ligand were formulated for BTB targeting. Chang et al. have used anti-VEGFR2 antibody-conjugated cationic microbubbles to target VEGFR2 in the rat BTB. The microbubble targeting efficiency was evaluated in vitro on C6 glioma cells and was 99.4 ± 0.3%, while it was 6.4 ± 1.2% for unconjugated microbubbles [102]. Most of the targets studied, such as VEGFR2, are directed to endothelial cells. However, it is also possible to target receptors directly expressed on the malignant cells, i.e., folate receptors. Hence, Fan et al. used this receptor as a target for BBB opening and gene delivery in the C6 glioma rat model. They have demonstrated the targeting ability on C6 glioma cells of folate-conjugated DNA-loaded cationic microbubbles in vitro: the folate increased the targeting ability of the complex by 7.6 fold [105]. Besides, Zhao et al. used Asn-Gly-Arg (NGR), a peptide motif that can be used to target CD13 receptors. CD13 is overexpressed in glioma cells and neovascular endothelial cells. In vitro, NGR-linked-shBirc5-loaded liposome complex linked to lipid-shelled microbubble demonstrated a better shBirc5 gene transfection on C6 glioma cells for the targeted microbubble compared to the untargeted ones (36.25% of transfection efficiency vs. 21.26%), thereby demonstrating the targeting profits [108].
Molecular targeting was always associated with drug/gene loaded into the bubble in the browsed literature. Indeed, molecular targeting may improve drug delivery: Fan et al. have observed better gene transfection efficiency in vivo for their folate-conjugated microbubbles than those without conjugation (luciferase gene expression 4.7 fold higher after 24 h) [105].
2.4.2. Drug and Gene Delivery
Drug delivery through the BBB with FUS can be reached by (i) co-injection of microbubbles and free drugs or drug carriers or (ii) encapsulating or covalently linking the therapeutics to the agent shell [32][127]. Loading drugs into microbubbles' shells enables better spatial control to deliver the drug to the treated site. The drug's encapsulation would also decrease the side effects induced by the circulation of a high drug dose in the vasculature [30][32].
Non-viral gene delivery by plasmid DNA complexation with bubbles was studied in numerous studies [87][102][103][105][108][109][110]. Thereby, glial cell line-derived neurotrophic factor (GDNF) [87][109][123], brain-derived neurotrophic factor (BDNF) [87] and nuclear factor E2-related factor 2 (Nrf2) [110] have been studied for Parkinson's disease treatment. Gene therapy with neurotrophic factors is a promising approach to improving current Parkinson's disease therapy. It has been found to reduce progressive neuronal loss and play a crucial role in the development, survival, and maintenance of the central and peripheral nervous system [87]. Nrf2 is a nuclear factor that activates the antioxidant response element pathway and protects the brain by regulating redox status. Nrf2 might be useful in Parkinson's disease therapy as reactive oxygen species play an important role in disease development [110]. Thus, these factors have been encoded in plasmid DNA and complexed with microbubble for BBB disruption and drug delivery on Parkinson's disease rodent model [87][109][110][123]. Neurotrophic factor delivery provided a neuroprotective effect by showing evidence of improvement of behavioral deficits [87][109][123], while Nrf2 gene transfection enabled the reduction of reactive oxygen species levels [110].
Interestingly, Wu et al. have used pPrestin-microbubble to disrupt the BBB, modify, and activate neurons within mice brain for spatiotemporal neuromodulation [103]. Prestin is a transmembrane protein that exists in the mammalian auditory system and functions as an electromechanical transducer. The cellular transfection rate with pPrestin-microbubble was 1.5-fold higher than with commercial transfection agents (LT-1) [103].
Chang et al. used anti-tumor suicide gene therapy for glioblastoma therapy on Sprague-Dawley rats using cationic VEGFR2-targeted microbubbles, complexed with luciferase gene and herpes simplex virus type 1 thymidine kinase/ganciclovir (gene suicide system) encoding plasmid DNA (pLUC and pHSV-TK/GCV). Anti-tumor suicide gene therapy involves tumor-targeted transfection of a suicide gene that encodes an enzyme for converting non-toxic prodrugs into toxic products to kill tumor cells. Both VEGFR2-targeting and pHSV-TK contributed to improving the anti-tumor efficiency: 25 days after tumor implantation, the tumor volume was 9.7 ± 5.2 mm3 for the pHSV-TK/GCV-loaded VEGFR2-targeted microbubbles-treated group, 40.1 ± 4.3 mm3 for the untargeted pHSV-TK/GCV-loaded microbubbles-treated group, and approximately 68 ± 8 mm3 for the untreated group [102].
Another technique of gene delivery therapy is the RNA interference technique, which consists of protein expression inhibition. The RNA interference technique includes small interfering RNA (siRNA) and short hairpin RNA (shRNA). Zhao et al. used this technique on an orthotopic C6 glioma rat model with NGR-conjugated shBirc5-loaded liposome attached to microbubbles. Birc5 is a protein in the family of apoptosis inhibitors. The Birc5 gene is only expressed in malignant tumors and not in normal tissue. Thus, a plasmid containing shRNA for the Birc5 gene could enter the cell, decrease Birc5 gene transcription in a targeted manner, promote tumor cell apoptosis, and reduce angiogenesis without affecting normal cells. The triple function agent (tumor cell targeting, delivering gene, and BBB opening) in conjunction with FUS exhibited a significant therapeutic effect, higher than the control group: median survival times were 38 and 21 days, respectively [108].
Besides, Chen et al. have proposed another cancer treatment possibility combined with molecular targeting: des-octanoyl ghrelin-conjugated microbubbles were loaded with TGFβ1 inhibitor (LY364947). Transforming growth factor TGFβ plays an essential role in the functional regulation of tumor interstitium; it also controls the permeability of the BBB and reduces the permeability of the brain's endothelial cell. Therefore, inhibition of TGFβ in cancer cells is expected to improve the therapeutic effects of chemotherapy. Thus, the deposition of doxorubicin in mice brain tissues was higher for the mice treated with TGFβ1 inhibitor-loaded conjugated bubbles (45 ± 5 µg/g of tissue) than for the group treated with unloaded conjugated bubbles (35 ± 5 µg/g of tissue). The median survival time was also increased (44 days for loaded microbubbles, 38 days for unloaded microbubbles) [86].
As mentioned earlier, anti-cancer drug-loaded bubbles have also been explored for cancer treatment (Figure 6) [107]. Interestingly, Fan et al. [104] proposed an alternative to boron neutron capture therapy consisting of radiotherapy based on boron agent delivery to the brain [135]. For a more efficient tumor-targeted boron delivery, the authors fabricated boron-containing nanoparticles-loaded microbubbles for the treatment of the glioma-bearing mice model. The complex successfully disrupted the BTB and delivered boron into the brain tumor (76.6 ± 3.6% of boron uptake in tumor 4 min after drug delivery) [104].
Several studies came up with other innovative drug delivery. Briefly, Zhao et al. used phosphatidylserine nanoparticles-microbubbles complexes to monitor inflammatory reaction [111]. The complex successfully and safely opened the BBB and activated the microglia/macrophage in the rat brain. In addition, Liu et al. used sulfur nanoparticles-quercetin complex embedded in the microbubble shell for Alzheimer's treatment [84]. Those microbubbles successfully opened the BBB on mice and allowed for a rapid accumulation of nanoparticles-quercetin complex in the brain, leading to improved Alzheimer's disease outcome (significant increase of success on Morris water maze experiment after treatment). Finally, Ha et al. proposed a drug delivery system by binding microbubble with ultrasound-sensitizing dye-incorporating nanoparticles. They successfully delivered those nanoparticles to the U87MG (human glioblastoma cell line) located in the mouse brain (fluorescence intensity 1.5 times higher than the control group) [88].
Among the multifunctional bubbles for drug delivery strategies found in the recent literature, most of them have to be destroyed to release the active principle embedded in their shell. However, bubble destruction results in a significant risk of brain damage (hemorrhage), which is not acceptable. Although safe BBB disruption with no evidence of acute or chronic inflammation was pointed out after bubble destruction by some studies [87][103], DNA delivery has been reported without destroying the DNA-loaded bubble [102]. Indeed, DNA loaded in bubbles might penetrate cells through endocytosis when the bubble undergoes stable cavitation [102].
References
- Burgess, A.; Shah, K.; Hough, O.; Hynynen, K. Focused ultrasound-mediated drug delivery through the blood–brain barrier. Expert Rev. Neurother. 2015, 15, 477–491, doi:10.1586/14737175.2015.1028369.
- Burgess, A.; Hynynen, K. Microbubble-Assisted Ultrasound for Drug Delivery in the Brain and Central Nervous System. In Therapeutic Ultrasound; Advances in Experimental Medicine and Biology; Escoffre, J.-M., Bouakaz, A., Eds; Springer International Publishing: Cham, Switzerland, 2016; Volume 880, pp. 293–308 ISBN 978-3-319-22535-7.
- Hynynen, K.; McDannold, N.; Vykhodtseva, N.; Jolesz, F.A. Noninvasive MR Imaging–guided Focal Opening of the Blood-Brain Barrier in Rabbits. Radiology 2001, 220, 640–646, doi:10.1148/radiol.2202001804.
- Marty, B.; Larrat, B.; Van Landeghem, M.; Robic, C.; Robert, P.; Port, M.; Le Bihan, D.; Pernot, M.; Tanter, M.; Lethimonnier, F.; et al. Dynamic Study of Blood–Brain Barrier Closure after its Disruption using Ultrasound: A Quantitative Analysis. J. Cereb. Blood Flow Metab. 2012, 32, 1948–1958, doi:10.1038/jcbfm.2012.100.
- Conti, A.; Mériaux, S.; Larrat, B. About the Marty model of blood-brain barrier closure after its disruption using focused ultrasound. Phys. Med. Biol. 2019, 64, 14NT02, doi:10.1088/1361-6560/ab259d.
- Downs, M.E.; Buch, A.; Karakatsani, M.E.; Konofagou, E.E.; Ferrera, V.P. Blood-Brain Barrier Opening in Behaving Non-Human Primates via Focused Ultrasound with Systemically Administered Microbubbles. Sci. Rep. 2015, 5, 15076, doi:10.1038/srep15076.
- Downs, M.E.; Buch, A.; Sierra, C.; Karakatsani, M.E.; Chen, S.; Konofagou, E.E.; Ferrera, V.P. Long-Term Safety of Repeated Blood-Brain Barrier Opening via Focused Ultrasound with Microbubbles in Non-Human Primates Performing a Cognitive Task. PLoS ONE 2015, 10, e0125911, doi:10.1371/journal.pone.0125911.
- FUS Foundation. Available online: https://www.fusfoundation.org/ (accessed on 1 November 2020).
- Conti, A.; Magnin, R.; Gerstenmayer, M.; Tsapis, N.; Dumont, E.; Tillement, O.; Lux, F.; Le Bihan, D.; Mériaux, S.; Della Penna, S.; et al. Empirical and Theoretical Characterization of the Diffusion Process of Different Gadolinium-Based Nanoparticles within the Brain Tissue after Ultrasound-Induced Permeabilization of the Blood-Brain Barrier. Contrast Media Mol. Imaging 2019, 2019, 1–13, doi:10.1155/2019/6341545.
- Mériaux, S.; Conti, A.; Larrat, B. Assessing Diffusion in the Extra-Cellular Space of Brain Tissue by Dynamic MRI Mapping of Contrast Agent Concentrations. Front. Phys. 2018, 6, 38, doi:10.3389/fphy.2018.00038.
- Valdez, M.A.; Fernandez, E.; Matsunaga, T.; Erickson, R.P.; Trouard, T.P. Distribution and Diffusion of Macromolecule Delivery to the Brain via Focused Ultrasound using Magnetic Resonance and Multispectral Fluorescence Imaging. Ultrasound Med. Biol. 2020, 46, 122–136, doi:10.1016/j.ultrasmedbio.2019.08.024.
- Alli, S.; Figueiredo, C.A.; Golbourn, B.; Sabha, N.; Wu, M.Y.; Bondoc, A.; Luck, A.; Coluccia, D.; Maslink, C.; Smith, C.; et al. Brainstem blood brain barrier disruption using focused ultrasound: A demonstration of feasibility and enhanced doxorubicin delivery. J. Control. Release 2018, 281, 29–41, doi:10.1016/j.jconrel.2018.05.005.
- Weber-Adrian, D.; Kofoed, R.H.; Chan, J.W.Y.; Silburt, J.; Noroozian, Z.; Kügler, S.; Hynynen, K.; Aubert, I. Strategy to enhance transgene expression in proximity of amyloid plaques in a mouse model of Alzheimer’s disease. Theranostics 2019, 9, 8127–8137, doi:10.7150/thno.36718.
- Karakatsani, M.E.; Wang, S.; Samiotaki, G.; Kugelman, T.; Olumolade, O.O.; Acosta, C.; Sun, T.; Han, Y.; Kamimura, H.A.S.; Jackson-Lewis, V.; et al. Amelioration of the nigrostriatal pathway facilitated by ultrasound-mediated neurotrophic delivery in early Parkinson’s disease. J. Control. Release 2019, 303, 289–301, doi:10.1016/j.jconrel.2019.03.030.
- Hsu, Y.-H.; Liu, R.-S.; Lin, W.-L.; Yuh, Y.-S.; Lin, S.-P.; Wong, T.-T. Transcranial pulsed ultrasound facilitates brain uptake of laronidase in enzyme replacement therapy for Mucopolysaccharidosis type I disease. Orphanet J. Rare Dis. 2017, 12, 109, doi:10.1186/s13023-017-0649-6.
- Mooney, S.J.; Shah, K.; Yeung, S.; Burgess, A.; Aubert, I.; Hynynen, K. Focused Ultrasound-Induced Neurogenesis Requires an Increase in Blood-Brain Barrier Permeability. PLoS ONE 2016, 11, e0159892, doi:10.1371/journal.pone.0159892.
- Burgess, A.; Dubey, S.; Yeung, S.; Hough, O.; Eterman, N.; Aubert, I.; Hynynen, K. Alzheimer Disease in a Mouse Model: MR Imaging–guided Focused Ultrasound Targeted to the Hippocampus Opens the Blood-Brain Barrier and Improves Pathologic Abnormalities and Behavior. Radiology 2014, 273, 736–745, doi:10.1148/radiol.14140245.
- Lamsam, L.; Johnson, E.; Connolly, I.D.; Wintermark, M.; Hayden Gephart, M. A review of potential applications of MR-guided focused ultrasound for targeting brain tumor therapy. Neurosurg. Focus 2018, 44, E10, doi:10.3171/2017.11.FOCUS17620.
- Conti, A.; Kamimura, H.A.; Novell, A.; Duggento, A.; Toschi, N. Magnetic Resonance Methods for Focused ultrasound-induced Blood-Brain Barrier Opening. Front. Phys. 2020, doi:10.3389/fphy.2020.547674.
- Kamimura, H.A.; Flament, J.; Valette, J.; Cafarelli, A.; Aron Badin, R.; Hantraye, P.; Larrat, B. Feedback control of microbubble cavitation for ultrasound-mediated blood–brain barrier disruption in non-human primates under magnetic resonance guidance. J. Cereb. Blood Flow Metab. 2019, 39, 1191–1203, doi:10.1177/0271678X17753514.
- Kamimura, H.A.S.; Wang, S.; Wu, S.-Y.; Karakatsani, M.E.; Acosta, C.; Carneiro, A.A.O.; Konofagou, E.E. Chirp- and random-based coded ultrasonic excitation for localized blood-brain barrier opening. Phys. Med. Biol. 2015, 60, 7695–7712, doi:10.1088/0031-9155/60/19/7695.
- Novell, A.; Kamimura, H.A.S.; Cafarelli, A.; Gerstenmayer, M.; Flament, J.; Valette, J.; Agou, P.; Conti, A.; Selingue, E.; Aron Badin, R.; et al. A new safety index based on intrapulse monitoring of ultra-harmonic cavitation during ultrasound-induced blood-brain barrier opening procedures. Sci. Rep. 2020, 10, 10088, doi:10.1038/s41598-020-66994-8.
- Qin, S.; Caskey, C.F.; Ferrara, K.W. Ultrasound contrast microbubbles in imaging and therapy: Physical principles and engineering. Phys. Med. Biol. 2009, 54, R27–R57, doi:10.1088/0031-9155/54/6/R01.
- Lentacker, I.; De Cock, I.; Deckers, R.; De Smedt, S.C.; Moonen, C.T.W. Understanding ultrasound induced sonoporation: Definitions and underlying mechanisms. Adv. Drug Deliv. Rev. 2014, 72, 49–64, doi:10.1016/j.addr.2013.11.008.
- Hwang, J.H.; Tu, J.; Brayman, A.A.; Matula, T.J.; Crum, L.A. Correlation between inertial cavitation dose and endothelial cell damage in vivo. Ultrasound Med. Biol. 2006, 32, 1611–1619, doi:10.1016/j.ultrasmedbio.2006.07.016.
- Lee, H.; Kim, H.; Han, H.; Lee, M.; Lee, S.; Yoo, H.; Chang, J.H.; Kim, H. Microbubbles used for contrast enhanced ultrasound and theragnosis: A review of principles to applications. Biomed. Eng. Lett. 2017, 7, 59–69, doi:10.1007/s13534-017-0016-5.
- Meng, Y.; Pople, C.B.; Lea-Banks, H.; Abrahao, A.; Davidson, B.; Suppiah, S.; Vecchio, L.M.; Samuel, N.; Mahmud, F.; Hynynen, K.; et al. Safety and efficacy of focused ultrasound induced blood-brain barrier opening, an integrative review of animal and human studies. J. Control. Release 2019, 309, 25–36, doi:10.1016/j.jconrel.2019.07.023.
- Kamimura, H.A.S.; Aurup, C.; Bendau, E.V.; Saharkhiz, N.; Kim, M.G.; Konofagou, E.E. Iterative Curve Fitting of the Bioheat Transfer Equation for Thermocouple-Based Temperature Estimation In Vitro and In~ Vivo. Ieee Trans. Ultrason. Ferroelectr. Freq. Control. 2020, 67, 70–80, doi:10.1109/TUFFC.2019.2940375.
- Kooiman, K.; Vos, H.J.; Versluis, M.; de Jong, N. Acoustic behavior of microbubbles and implications for drug delivery. Adv. Drug Deliv. Rev. 2014, 72, 28–48, doi:10.1016/j.addr.2014.03.003.
- Delalande, A.; Bastié, C.; Pigeon, L.; Manta, S.; Lebertre, M.; Mignet, N.; Midoux, P.; Pichon, C. Cationic gas-filled microbubbles for ultrasound-based nucleic acids delivery. Biosci. Rep. 2017, 37, BSR20160619, doi:10.1042/BSR20160619.
- Stride, E.; Segers, T.; Lajoinie, G.; Cherkaoui, S.; Bettinger, T.; Versluis, M.; Borden, M. Microbubble Agents: New Directions. Ultrasound Med. Biol. 2020, 46, 1326–1343, doi:10.1016/j.ultrasmedbio.2020.01.027.
- Yildirim, A.; Blum, N.T.; Goodwin, A.P. Colloids, nanoparticles, and materials for imaging, delivery, ablation, and theranostics by focused ultrasound (FUS). Theranostics 2019, 9, 2572–2594, doi:10.7150/thno.32424.
- Wu, S.-Y.; Fix, S.M.; Arena, C.B.; Chen, C.C.; Zheng, W.; Olumolade, O.O.; Papadopoulou, V.; Novell, A.; Dayton, P.A.; Konofagou, E.E. Focused ultrasound-facilitated brain drug delivery using optimized nanodroplets: Vaporization efficiency dictates large molecular delivery. Phys. Med. Biol. 2018, 63, 035002, doi:10.1088/1361-6560/aaa30d.
- Shin, J.; Kong, C.; Cho, J.S.; Lee, J.; Koh, C.S.; Yoon, M.-S.; Na, Y.C.; Chang, W.S.; Chang, J.W. Focused ultrasound–mediated noninvasive blood-brain barrier modulation: Preclinical examination of efficacy and safety in various sonication parameters. Neurosurg. Focus 2018, 44, E15, doi:10.3171/2017.11.FOCUS17627.
- Wu, S.-K.; Chu, P.-C.; Chai, W.-Y.; Kang, S.-T.; Tsai, C.-H.; Fan, C.-H.; Yeh, C.-K.; Liu, H.-L. Characterization of Different Microbubbles in Assisting Focused Ultrasound-Induced Blood-Brain Barrier Opening. Sci. Rep. 2017, 7, 46689, doi:10.1038/srep46689.
- Bing, C.; Hong, Y.; Hernandez, C.; Rich, M.; Cheng, B.; Munaweera, I.; Szczepanski, D.; Xi, Y.; Bolding, M.; Exner, A.; et al. Characterization of different bubble formulations for blood-brain barrier opening using a focused ultrasound system with acoustic feedback control. Sci. Rep. 2018, 8, 7986, doi:10.1038/s41598-018-26330-7.
- Tan, J.-K.Y.; Pham, B.; Zong, Y.; Perez, C.; Maris, D.O.; Hemphill, A.; Miao, C.H.; Matula, T.J.; Mourad, P.D.; Wei, H.; et al. Microbubbles and ultrasound increase intraventricular polyplex gene transfer to the brain. J. Control. Release 2016, 231, 86–93, doi:10.1016/j.jconrel.2016.02.003.
- McDannold, N.; Zhang, Y.; Supko, J.G.; Power, C.; Sun, T.; Peng, C.; Vykhodtseva, N.; Golby, A.J.; Reardon, D.A. Acoustic feedback enables safe and reliable carboplatin delivery across the blood-brain barrier with a clinical focused ultrasound system and improves survival in a rat glioma model. Theranostics 2019, 9, 6284–6299, doi:10.7150/thno.35892.
- Yoon, K.; Lee, W.; Chen, E.; Lee, J.E.; Croce, P.; Cammalleri, A.; Foley, L.; Tsao, A.L.; Yoo, S.-S. Localized Blood–Brain Barrier Opening in Ovine Model Using Image-Guided Transcranial Focused Ultrasound. Ultrasound Med. Biol. 2019, 45, 2391–2404, doi:10.1016/j.ultrasmedbio.2019.05.023.
- Xhima, K.; Nabbouh, F.; Hynynen, K.; Aubert, I.; Tandon, A. Noninvasive delivery of an α-synuclein gene silencing vector with magnetic resonance-guided focused ultrasound: Noninvasive Knockdown of Brain α-Syn. Mov. Disord. 2018, 33, 1567–1579, doi:10.1002/mds.101.
- Arvanitis, C.D.; Askoxylakis, V.; Guo, Y.; Datta, M.; Kloepper, J.; Ferraro, G.B.; Bernabeu, M.O.; Fukumura, D.; McDannold, N.; Jain, R.K. Mechanisms of enhanced drug delivery in brain metastases with focused ultrasound-induced blood–tumor barrier disruption. Proc. Natl. Acad. Sci. USA 2018, 115, E8717–E8726, doi:10.1073/pnas.1807105115.
- Pelekanos, M.; Leinenga, G.; Odabaee, M.; Odabaee, M.; Saifzadeh, S.; Steck, R.; Götz, J. Establishing sheep as an experimental species to validate ultrasound-mediated blood-brain barrier opening for potential therapeutic interventions. Theranostics 2018, 8, 2583–2602, doi:10.7150/thno.22852.
- Park, J.; Aryal, M.; Vykhodtseva, N.; Zhang, Y.-Z.; McDannold, N. Evaluation of permeability, doxorubicin delivery, and drug retention in a rat brain tumor model after ultrasound-induced blood-tumor barrier disruption. J. Control. Release 2017, 250, 77–85, doi:10.1016/j.jconrel.2016.10.011.
- Alkins, R.; Burgess, A.; Kerbel, R.; Wels, W.S.; Hynynen, K. Early treatment of HER2-amplified brain tumors with targeted NK-92 cells and focused ultrasound improves survival. Neuro-Oncol. 2016, 18, 974–981, doi:10.1093/neuonc/nov318.
- Coluccia, D.; Figueiredo, C.A.; Wu, M.Y.; Riemenschneider, A.N.; Diaz, R.; Luck, A.; Smith, C.; Das, S.; Ackerley, C.; O’Reilly, M.; et al. Enhancing glioblastoma treatment using cisplatin-gold-nanoparticle conjugates and targeted delivery with magnetic resonance-guided focused ultrasound. Nanomed. Nanotechnol. Biol. Med. 2018, 14, 1137–1148, doi:10.1016/j.nano.2018.01.021.
- Xhima, K.; Markham-Coultes, K.; Nedev, H.; Heinen, S.; Saragovi, H.U.; Hynynen, K.; Aubert, I. Focused ultrasound delivery of a selective TrkA agonist rescues cholinergic function in a mouse model of Alzheimer’s disease. Sci. Adv. 2020, 6, eaax6646, doi:10.1126/sciadv.aax6646.
- Noroozian, Z.; Xhima, K.; Huang, Y.; Kaspar, B.K.; Kügler, S.; Hynynen, K.; Aubert, I. MRI-Guided Focused Ultrasound for Targeted Delivery of rAAV to the Brain. In Adeno-Associated Virus Vectors; Castle, M.J., Ed.; Methods in Molecular Biology; Springer New York: New York, NY, USA, 2019; Volume 1950, pp. 177–197 ISBN 978-1-4939-9138-9.
- Huang, Y.; Alkins, R.; Schwartz, M.L.; Hynynen, K. Opening the Blood-Brain Barrier with MR Imaging–guided Focused Ultrasound: Preclinical Testing on a Trans–Human Skull Porcine Model. Radiology 2017, 282, 123–130, doi:10.1148/radiol.2016152154.
- Shin, J.; Kong, C.; Lee, J.; Choi, B.Y.; Sim, J.; Koh, C.S.; Park, M.; Na, Y.C.; Suh, S.W.; Chang, W.S.; et al. Focused ultrasound-induced blood-brain barrier opening improves adult hippocampal neurogenesis and cognitive function in a cholinergic degeneration dementia rat model. Alzheimers Res. 2019, 11, 110, doi:10.1186/s13195-019-0569-x.
- Jung, B.; Huh, H.; Lee, E.; Han, M.; Park, J. An advanced focused ultrasound protocol improves the blood-brain barrier permeability and doxorubicin delivery into the rat brain. J. Control. Release 2019, 315, 55–64, doi:10.1016/j.jconrel.2019.10.044.
- Rich, M.C.; Sherwood, J.; Bartley, A.F.; Whitsitt, Q.A.; Lee, M.; Willoughby, W.R.; Dobrunz, L.E.; Bao, Y.; Lubin, F.D.; Bolding, M. Focused ultrasound blood brain barrier opening mediated delivery of MRI-visible albumin nanoclusters to the rat brain for localized drug delivery with temporal control. J. Control. Release 2020, 324, 172–180, doi:10.1016/j.jconrel.2020.04.054.
- Alecou, T.; Giannakou, M.; Damianou, C. Amyloid β Plaque Reduction With Antibodies Crossing the Blood-Brain Barrier, Which Was Opened in 3 Sessions of Focused Ultrasound in a Rabbit Model: Amyloid β Plaque Reduction With Focused Ultrasound. J. Ultrasound Med. 2017, 36, 2257–2270, doi:10.1002/jum.14256.
- Chu, P.-C.; Chai, W.-Y.; Tsai, C.-H.; Kang, S.-T.; Yeh, C.-K.; Liu, H.-L. Focused Ultrasound-Induced Blood-Brain Barrier Opening: Association with Mechanical Index and Cavitation Index Analyzed by Dynamic Contrast-Enhanced Magnetic-Resonance Imaging. Sci. Rep. 2016, 6, 33264, doi:10.1038/srep33264.
- Yang, F.-Y.; Chang, W.-Y.; Lin, W.-T.; Hwang, J.-J.; Chien, Y.-C.; Wang, H.-E.; Tsai, M.-L. Focused ultrasound enhanced molecular imaging and gene therapy for multifusion reporter gene in glioma-bearing rat model. Oncotarget 2015, 6, 36260–36268, doi:10.18632/oncotarget.5389.
- Lin, C.-Y.; Hsieh, H.-Y.; Pitt, W.G.; Huang, C.-Y.; Tseng, I.-C.; Yeh, C.-K.; Wei, K.-C.; Liu, H.-L. Focused ultrasound-induced blood-brain barrier opening for non-viral, non-invasive, and targeted gene delivery. J. Control. Release 2015, 212, 1–9, doi:10.1016/j.jconrel.2015.06.010.
- Horodyckid, C.; Canney, M.; Vignot, A.; Boisgard, R.; Drier, A.; Huberfeld, G.; François, C.; Prigent, A.; Santin, M.D.; Adam, C.; et al. Safe long-term repeated disruption of the blood-brain barrier using an implantable ultrasound device: A multiparametric study in a primate model. J. Neurosurg. 2017, 126, 1351–1361, doi:10.3171/2016.3.JNS151635.
- Beccaria, K.; Canney, M.; Goldwirt, L.; Fernandez, C.; Piquet, J.; Perier, M.-C.; Lafon, C.; Chapelon, J.-Y.; Carpentier, A. Ultrasound-induced opening of the blood-brain barrier to enhance temozolomide and irinotecan delivery: An experimental study in rabbits. J. Neurosurg. 2016, 124, 1602–1610, doi:10.3171/2015.4.JNS142893.
- Morse, S.V.; Pouliopoulos, A.N.; Chan, T.G.; Copping, M.J.; Lin, J.; Long, N.J.; Choi, J.J. Rapid Short-pulse Ultrasound Delivers Drugs Uniformly across the Murine Blood-Brain Barrier with Negligible Disruption. Radiology 2019, 291, 459–466, doi:10.1148/radiol.2019181625.
- Zhang, D.Y.; Dmello, C.; Chen, L.; Arrieta, V.A.; Gonzalez-Buendia, E.; Kane, J.R.; Magnusson, L.P.; Baran, A.; James, C.D.; Horbinski, C.; et al. Ultrasound-mediated Delivery of Paclitaxel for Glioma: A Comparative Study of Distribution, Toxicity, and Efficacy of Albumin-bound Versus Cremophor Formulations. Clin. Cancer Res. 2020, 26, 477–486, doi:10.1158/1078-0432.CCR-19-2182.
- Chai, W.-Y.; Chu, P.-C.; Tsai, C.-H.; Lin, C.-Y.; Yang, H.-W.; Lai, H.-Y.; Liu, H.-L. Image-Guided Focused-Ultrasound CNS Molecular Delivery: An Implementation via Dynamic Contrast-Enhanced Magnetic-Resonance Imaging. Sci. Rep. 2018, 8, 4151, doi:10.1038/s41598-018-22571-8.
- Xi, X.-P.; Zong, Y.-J.; Ji, Y.-H.; Wang, B.; Liu, H.-S. Experiment research of focused ultrasound combined with drug and microbubble for treatment of central nervous system leukemia. Oncotarget 2018, 9, 5424–5434, doi:10.18632/oncotarget.23521.
- Morse, S.V.; Boltersdorf, T.; Harriss, B.I.; Chan, T.G.; Baxan, N.; Jung, H.S.; Pouliopoulos, A.N.; Choi, J.J.; Long, N.J. Neuron labeling with rhodamine-conjugated Gd-based MRI contrast agents delivered to the brain via focused ultrasound. Theranostics 2020, 10, 2659–2674, doi:10.7150/thno.42665.
- Kung, Y.; Huang, H.-Y.; Liao, W.-H.; Huang, A.P.-H.; Hsiao, M.-Y.; Wu, C.-H.; Liu, H.-L.; Inserra, C.; Chen, W.-S. A Single High-Intensity Shock Wave Pulse With Microbubbles Opens the Blood-Brain Barrier in Rats. Front. Bioeng. Biotechnol. 2020, 8, 402, doi:10.3389/fbioe.2020.00402.
- Tran, V.L.; Novell, A.; Tournier, N.; Gerstenmayer, M.; Schweitzer-Chaput, A.; Mateos, C.; Jego, B.; Bouleau, A.; Nozach, H.; Winkeler, A.; et al. Impact of blood-brain barrier permeabilization induced by ultrasound associated to microbubbles on the brain delivery and kinetics of cetuximab: An immunoPET study using 89Zr-cetuximab. J. Control. Release 2020, 328, 304–312, doi:10.1016/j.jconrel.2020.08.047.
- Luo, J.; Xie, C.; Zhang, W.; Cai, Y.; Ding, J.; Wang, Y.; Hao, Y.; Zhang, Y.; Guan, Y. Experimental mouse model of NMOSD produced by facilitated brain delivery of NMO-IgG by microbubble-enhanced low-frequency ultrasound in experimental allergic encephalomyelitis mice. Mult. Scler. Relat. Disord. 2020, 46, 102473, doi:10.1016/j.msard.2020.102473.
- Omata, D.; Maruyama, T.; Unga, J.; Hagiwara, F.; Munakata, L.; Kageyama, S.; Shima, T.; Suzuki, Y.; Maruyama, K.; Suzuki, R. Effects of encapsulated gas on stability of lipid-based microbubbles and ultrasound-triggered drug delivery. J. Control. Release 2019, 311–312, 65–73, doi:10.1016/j.jconrel.2019.08.023.
- Sun, T.; Zhang, Y.; Power, C.; Alexander, P.M.; Sutton, J.T.; Aryal, M.; Vykhodtseva, N.; Miller, E.L.; McDannold, N.J. Closed-loop control of targeted ultrasound drug delivery across the blood–brain/tumor barriers in a rat glioma model. Proc. Natl. Acad. Sci. USA 2017, 114, E10281–E10290, doi:10.1073/pnas.1713328114.
- Aryal, M.; Papademetriou, I.; Zhang, Y.-Z.; Power, C.; McDannold, N.; Porter, T. MRI Monitoring and Quantification of Ultrasound-Mediated Delivery of Liposomes Dually Labeled with Gadolinium and Fluorophore through the Blood-Brain Barrier. Ultrasound Med. Biol. 2019, 45, 1733–1742, doi:10.1016/j.ultrasmedbio.2019.02.024.
- Yao, X.; Adams, M.S.; Jones, P.D.; Diederich, C.J.; Verkman, A.S. Noninvasive, Targeted Creation of Neuromyelitis Optica Pathology in AQP4-IgG Seropositive Rats by Pulsed Focused Ultrasound. J. Neuropathol. Exp. Neurol. 2019, 78, 47–56, doi:10.1093/jnen/nly107.
- Stavarache, M.A.; Petersen, N.; Jurgens, E.M.; Milstein, E.R.; Rosenfeld, Z.B.; Ballon, D.J.; Kaplitt, M.G. Safe and stable noninvasive focal gene delivery to the mammalian brain following focused ultrasound. J. Neurosurg. 2019, 130, 989–998, doi:10.3171/2017.8.JNS17790.
- Kobus, T.; Zervantonakis, I.K.; Zhang, Y.; McDannold, N.J. Growth inhibition in a brain metastasis model by antibody delivery using focused ultrasound-mediated blood-brain barrier disruption. J. Control. Release 2016, 238, 281–288, doi:10.1016/j.jconrel.2016.08.001.
- Mulik, R.S.; Bing, C.; Ladouceur-Wodzak, M.; Munaweera, I.; Chopra, R.; Corbin, I.R. Localized delivery of low-density lipoprotein docosahexaenoic acid nanoparticles to the rat brain using focused ultrasound. Biomaterials 2016, 83, 257–268, doi:10.1016/j.biomaterials.2016.01.021.
- Song, K.-H.; Fan, A.C.; Hinkle, J.J.; Newman, J.; Borden, M.A.; Harvey, B.K. Microbubble gas volume: A unifying dose parameter in blood-brain barrier opening by focused ultrasound. Theranostics 2017, 7, 144–152, doi:10.7150/thno.15987.
- Hartman, R.K.; Hallam, K.A.; Donnelly, E.M.; Emelianov, S.Y. Photoacoustic imaging of gold nanorods in the brain delivered via microbubble-assisted focused ultrasound: A tool for in vivo molecular neuroimaging. Laser Phys. Lett. 2019, 16, 025603, doi:10.1088/1612-202X/aaf89e.
- Papachristodoulou, A.; Signorell, R.D.; Werner, B.; Brambilla, D.; Luciani, P.; Cavusoglu, M.; Grandjean, J.; Silginer, M.; Rudin, M.; Martin, E.; et al. Chemotherapy sensitization of glioblastoma by focused ultrasound-mediated delivery of therapeutic liposomes. J. Control. Release 2019, 295, 130–139, doi:10.1016/j.jconrel.2018.12.009.
- Lin, C.-Y.; Hsieh, H.-Y.; Chen, C.-M.; Wu, S.-R.; Tsai, C.-H.; Huang, C.-Y.; Hua, M.-Y.; Wei, K.-C.; Yeh, C.-K.; Liu, H.-L. Non-invasive, neuron-specific gene therapy by focused ultrasound-induced blood-brain barrier opening in Parkinson’s disease mouse model. J. Control. Release 2016, 235, 72–81, doi:10.1016/j.jconrel.2016.05.052.
- Åslund, A.K.O.; Snipstad, S.; Healey, A.; Kvåle, S.; Torp, S.H.; Sontum, P.C.; Davies, C. de L.; van Wamel, A. Efficient Enhancement of Blood-Brain Barrier Permeability Using Acoustic Cluster Therapy (ACT). Theranostics 2017, 7, 23–30, doi:10.7150/thno.16577.
- Zhang, X.; Hu, J.; Zhao, G.; Huang, N.; Tan, Y.; Pi, L.; Huang, Q.; Wang, F.; Wang, Z.; Wang, Z.; et al. PEGylated PLGA-based phase shift nanodroplets combined with focused ultrasound for blood brain barrier opening in rats. Oncotarget 2017, 8, 38927–38936, doi:10.18632/oncotarget.17155.
- Mead, B.P.; Mastorakos, P.; Suk, J.S.; Klibanov, A.L.; Hanes, J.; Price, R.J. Targeted gene transfer to the brain via the delivery of brain-penetrating DNA nanoparticles with focused ultrasound. J. Control. Release 2016, 223, 109–117, doi:10.1016/j.jconrel.2015.12.034.
- Timbie, K.F.; Afzal, U.; Date, A.; Zhang, C.; Song, J.; Wilson Miller, G.; Suk, J.S.; Hanes, J.; Price, R.J. MR image-guided delivery of cisplatin-loaded brain-penetrating nanoparticles to invasive glioma with focused ultrasound. J. Control. Release 2017, 263, 120–131, doi:10.1016/j.jconrel.2017.03.017.
- Curley, C.T.; Mead, B.P.; Negron, K.; Kim, N.; Garrison, W.J.; Miller, G.W.; Kingsmore, K.M.; Thim, E.A.; Song, J.; Munson, J.M.; et al. Augmentation of brain tumor interstitial flow via focused ultrasound promotes brain-penetrating nanoparticle dispersion and transfection. Sci. Adv. 2020, 6, eaay1344, doi:10.1126/sciadv.aay1344.
- Baghirov, H.; Snipstad, S.; Sulheim, E.; Berg, S.; Hansen, R.; Thorsen, F.; Mørch, Y.; Davies, C. de L.; Åslund, A.K.O. Ultrasound-mediated delivery and distribution of polymeric nanoparticles in the normal brain parenchyma of a metastatic brain tumour model. PLoS ONE 2018, 13, e0191102, doi:10.1371/journal.pone.0191102.
- Åslund, A.K.O.; Berg, S.; Hak, S.; Mørch, Ý.; Torp, S.H.; Sandvig, A.; Widerøe, M.; Hansen, R.; de Lange Davies, C. Nanoparticle delivery to the brain—By focused ultrasound and self-assembled nanoparticle-stabilized microbubbles. J. Control. Release 2015, 220, 287–294, doi:10.1016/j.jconrel.2015.10.047.
- Liu, Y.; Gong, Y.; Xie, W.; Huang, A.; Yuan, X.; Zhou, H.; Zhu, X.; Chen, X.; Liu, J.; Liu, J.; et al. Microbubbles in combination with focused ultrasound for the delivery of quercetin-modified sulfur nanoparticles through the blood brain barrier into the brain parenchyma and relief of endoplasmic reticulum stress to treat Alzheimer’s disease. Nanoscale 2020, 12, 6498–6511, doi:10.1039/C9NR09713A.
- May, J.-N.; Golombek, S.K.; Baues, M.; Dasgupta, A.; Drude, N.; Rix, A.; Rommel, D.; von Stillfried, S.; Appold, L.; Pola, R.; et al. Multimodal and multiscale optical imaging of nanomedicine delivery across the blood-brain barrier upon sonopermeation. Theranostics 2020, 10, 1948–1959, doi:10.7150/thno.41161.
- Chen, Y.-C.; Chiang, C.-F.; Wu, S.-K.; Chen, L.-F.; Hsieh, W.-Y.; Lin, W.-L. Targeting microbubbles-carrying TGFβ1 inhibitor combined with ultrasound sonication induce BBB/BTB disruption to enhance nanomedicine treatment for brain tumors. J. Control. Release 2015, 211, 53–62, doi:10.1016/j.jconrel.2015.05.288.
- Lin, C.-Y.; Lin, Y.-C.; Huang, C.-Y.; Wu, S.-R.; Chen, C.-M.; Liu, H.-L. Ultrasound-responsive neurotrophic factor-loaded microbubble- liposome complex: Preclinical investigation for Parkinson’s disease treatment. J. Control. Release 2020, 321, 519–528, doi:10.1016/j.jconrel.2020.02.044.
- Ha, S.-W.; Hwang, K.; Jin, J.; Cho, A.-S.; Kim, T.Y.; Hwang, S.I.; Lee, H.J.; Kim, C.-Y. Ultrasound-sensitizing nanoparticle complex for overcoming the blood-brain barrier: An effective drug delivery system. Int. J. Nanomed. 2019, 14, 3743–3752, doi:10.2147/IJN.S193258.
- Ilovitsh, T.; Ilovitsh, A.; Foiret, J.; Caskey, C.F.; Kusunose, J.; Fite, B.Z.; Zhang, H.; Mahakian, L.M.; Tam, S.; Butts-Pauly, K.; et al. Enhanced microbubble contrast agent oscillation following 250 kHz insonation. Sci. Rep. 2018, 8, 16347, doi:10.1038/s41598-018-34494-5.
- Wu, S.-Y.; Chen, C.C.; Tung, Y.-S.; Olumolade, O.O.; Konofagou, E.E. Effects of the microbubble shell physicochemical properties on ultrasound-mediated drug delivery to the brain. J. Control. Release 2015, 212, 30–40, doi:10.1016/j.jconrel.2015.06.007.
- Wu, S.-Y.; Aurup, C.; Sanchez, C.S.; Grondin, J.; Zheng, W.; Kamimura, H.; Ferrera, V.P.; Konofagou, E.E. Efficient Blood-Brain Barrier Opening in Primates with Neuronavigation-Guided Ultrasound and Real-Time Acoustic Mapping. Sci. Rep. 2018, 8, 7978, doi:10.1038/s41598-018-25904-9.
- Karakatsani, M.E.; Samiotaki, G.; Downs, M.E.; Ferrera, V.P.; Konofagou, E.E. Targeting Effects on the Volume of the Focused Ultrasound Induced Blood-Brain Barrier Opening in Non-Human Primates in vivo. 2018, 28.
- Samiotaki, G.; Karakatsani, M.E.; Buch, A.; Papadopoulos, S.; Wu, S.Y.; Jambawalikar, S.; Konofagou, E.E. Pharmacokinetic analysis and drug delivery efficiency of the focused ultrasound-induced blood-brain barrier opening in non-human primates. Magn. Reson. Imaging 2017, 37, 273–281, doi:10.1016/j.mri.2016.11.023.
- Sierra, C.; Acosta, C.; Chen, C.; Wu, S.-Y.; Karakatsani, M.E.; Bernal, M.; Konofagou, E.E. Lipid microbubbles as a vehicle for targeted drug delivery using focused ultrasound-induced blood–brain barrier opening. J. Cereb. Blood Flow Metab. 2017, 37, 1236–1250, doi:10.1177/0271678X16652630.
- Wang, S.; Karakatsani, M.E.; Fung, C.; Sun, T.; Acosta, C.; Konofagou, E. Direct brain infusion can be enhanced with focused ultrasound and microbubbles. J. Cereb. Blood Flow Metab. 2017, 37, 706–714, doi:10.1177/0271678X16637881.
- Zhang, Y.; Tan, H.; Bertram, E.H.; Aubry, J.-F.; Lopes, M.-B.; Roy, J.; Dumont, E.; Xie, M.; Zuo, Z.; Klibanov, A.L.; et al. Non-Invasive, Focal Disconnection of Brain Circuitry Using Magnetic Resonance-Guided Low-Intensity Focused Ultrasound to Deliver a Neurotoxin. Ultrasound Med. Biol. 2016, 42, 2261–2269, doi:10.1016/j.ultrasmedbio.2016.04.019.
- Karakatsani, M.E.; Kugelman, T.; Ji, R.; Murillo, M.; Wang, S.; Niimi, Y.; Small, S.A.; Duff, K.E.; Konofagou, E.E. Unilateral Focused Ultrasound-Induced Blood-Brain Barrier Opening Reduces Phosphorylated Tau from The rTg4510 Mouse Model. Theranostics 2019, 9, 5396–5411, doi:10.7150/thno.28717.
- Yang, Y.; Zhang, X.; Ye, D.; Laforest, R.; Williamson, J.; Liu, Y.; Chen, H. Cavitation dose painting for focused ultrasound-induced blood-brain barrier disruption. Sci. Rep. 2019, 9, 2840, doi:10.1038/s41598-019-39090-9.
- Ye, D.; Sultan, D.; Zhang, X.; Yue, Y.; Heo, G.S.; Kothapalli, S.V.V.N.; Luehmann, H.; Tai, Y.; Rubin, J.B.; Liu, Y.; et al. Focused ultrasound-enabled delivery of radiolabeled nanoclusters to the pons. J. Control. Release 2018, 283, 143–150, doi:10.1016/j.jconrel.2018.05.039.
- Pouliopoulos, A.N.; Jimenez, D.A.; Frank, A.; Robertson, A.; Zhang, L.; Kline-Schoder, A.R.; Bhaskar, V.; Harpale, M.; Caso, E.; Papapanou, N.; et al. Temporal Stability of Lipid-Shelled Microbubbles During Acoustically-Mediated Blood-Brain Barrier Opening. Front. Phys. 2020, 8, 137, doi:10.3389/fphy.2020.00137.
- Galan-Acosta, L.; Sierra, C.; Leppert, A.; Pouliopoulos, A.N.; Kwon, N.; Noel, R.L.; Tambaro, S.; Presto, J.; Nilsson, P.; Konofagou, E.E.; et al. Recombinant BRICHOS chaperone domains delivered to mouse brain parenchyma by focused ultrasound and microbubbles are internalized by hippocampal and cortical neurons. Mol. Cell. Neurosci. 2020, 105, 103498, doi:10.1016/j.mcn.2020.103498.
- Chang, E.-L.; Ting, C.-Y.; Hsu, P.-H.; Lin, Y.-C.; Liao, E.-C.; Huang, C.-Y.; Chang, Y.-C.; Chan, H.-L.; Chiang, C.-S.; Liu, H.-L.; et al. Angiogenesis-targeting microbubbles combined with ultrasound-mediated gene therapy in brain tumors. J. Control. Release 2017, 255, 164–175, doi:10.1016/j.jconrel.2017.04.010.
- Wu, C.-Y.; Fan, C.-H.; Chiu, N.-H.; Ho, Y.-J.; Lin, Y.-C.; Yeh, C.-K. Targeted delivery of engineered auditory sensing protein for ultrasound neuromodulation in the brain. Theranostics 2020, 10, 3546–3561, doi:10.7150/thno.39786.
- Fan, C.-H.; Wang, T.-W.; Hsieh, Y.-K.; Wang, C.-F.; Gao, Z.; Kim, A.; Nagasaki, Y.; Yeh, C.-K. Enhancing Boron Uptake in Brain Glioma by a Boron-Polymer/Microbubble Complex with Focused Ultrasound. Acs Appl. Mater. Interfaces 2019, 11, 11144–11156, doi:10.1021/acsami.8b22468.
- Fan, C.-H.; Chang, E.-L.; Ting, C.-Y.; Lin, Y.-C.; Liao, E.-C.; Huang, C.-Y.; Chang, Y.-C.; Chan, H.-L.; Wei, K.-C.; Yeh, C.-K. Folate-conjugated gene-carrying microbubbles with focused ultrasound for concurrent blood-brain barrier opening and local gene delivery. Biomaterials 2016, 106, 46–57, doi:10.1016/j.biomaterials.2016.08.017.
- Cheng, B.; Bing, C.; Xi, Y.; Shah, B.; Exner, A.A.; Chopra, R. Influence of Nanobubble Concentration on Blood–Brain Barrier Opening Using Focused Ultrasound Under Real-Time Acoustic Feedback Control. Ultrasound Med. Biol. 2019, 45, 2174–2187, doi:10.1016/j.ultrasmedbio.2019.03.016.
- Fan, C.-H.; Cheng, Y.-H.; Ting, C.-Y.; Ho, Y.-J.; Hsu, P.-H.; Liu, H.-L.; Yeh, C.-K. Ultrasound/Magnetic Targeting with SPIO-DOX-Microbubble Complex for Image-Guided Drug Delivery in Brain Tumors. Theranostics 2016, 6, 1542–1556, doi:10.7150/thno.15297.
- Zhao, G.; Huang, Q.; Wang, F.; Zhang, X.; Hu, J.; Tan, Y.; Huang, N.; Wang, Z.; Wang, Z.; Cheng, Y. Targeted shRNA-loaded liposome complex combined with focused ultrasound for blood brain barrier disruption and suppressing glioma growth. Cancer Lett. 2018, 418, 147–158, doi:10.1016/j.canlet.2018.01.035.
- Fan, C.-H.; Ting, C.-Y.; Lin, C.; Chan, H.-L.; Chang, Y.-C.; Chen, Y.-Y.; Liu, H.-L.; Yeh, C.-K. Noninvasive, Targeted and Non-Viral Ultrasound-Mediated GDNF-Plasmid Delivery for Treatment of Parkinson’s Disease. Sci. Rep. 2016, 6, 19579, doi:10.1038/srep19579.
- Long, L.; Cai, X.; Guo, R.; Wang, P.; Wu, L.; Yin, T.; Liao, S.; Lu, Z. Treatment of Parkinson’s disease in rats by Nrf2 transfection using MRI-guided focused ultrasound delivery of nanomicrobubbles. Biochem. Biophys. Res. Commun. 2017, 482, 75–80, doi:10.1016/j.bbrc.2016.10.141.
- Zhao, R.; Jiang, J.; Li, H.; Chen, M.; Liu, R.; Sun, S.; Ma, D.; Liang, X.; Wang, S. Phosphatidylserine-microbubble targeting-activated microglia/macrophage in inflammation combined with ultrasound for breaking through the blood–brain barrier. J. Neuroinflammation 2018, 15, 334, doi:10.1186/s12974-018-1368-1.
- Zhang, N.; Yan, F.; Liang, X.; Wu, M.; Shen, Y.; Chen, M.; Xu, Y.; Zou, G.; Jiang, P.; Tang, C.; et al. Localized delivery of curcumin into brain with polysorbate 80-modified cerasomes by ultrasound-targeted microbubble destruction for improved Parkinson’s disease therapy. Theranostics 2018, 8, 2264–2277, doi:10.7150/thno.23734.
- Pan, M.; Zhang, Y.; Deng, Z.; Yan, F.; Hong, G. Noninvasive and Local Delivery of Adenoviral-Mediated Herpes Simplex Virus Thymidine Kinase to Treat Glioma Through Focused Ultrasound-Induced Blood-Brain Barrier Opening in Rats. J. Biomed. Nanotechnol. 2018, 14, 2031–2041, doi:10.1166/jbn.2018.2642.
- Zhao, B.; Chen, Y.; Liu, J.; Zhang, L.; Wang, J.; Yang, Y.; Lv, Q.; Xie, M. Blood-brain barrier disruption induced by diagnostic ultrasound combined with microbubbles in mice. Oncotarget 2018, 9, 4897–4914, doi:10.18632/oncotarget.23527.
- Shen, Y.; Pi, Z.; Yan, F.; Yeh, C.-K.; Zeng, X.; Diao, X.; Hu, Y.; Chen, S.; Chen, X.; Zheng, H. Enhanced delivery of paclitaxel liposomes using focused ultrasound with microbubbles for treating nude mice bearing intracranial glioblastoma xenografts. Int. J. Nanomed. 2017, 12, 5613–5629, doi:10.2147/IJN.S136401.
- Xu, Y.; Cui, H.; Zhu, Q.; Hua, X.; Xia, H.; Tan, K.; Gao, Y.; Zhao, J.; Liu, Z. Unilateral Opening of Rat Blood-Brain Barrier Assisted by Diagnostic Ultrasound Targeted Microbubbles Destruction. Biomed. Res. Int. 2016, 2016, 1–10, doi:10.1155/2016/4759750.
- Shen, Y.; Guo, J.; Chen, G.; Chin, C.T.; Chen, X.; Chen, J.; Wang, F.; Chen, S.; Dan, G. Delivery of Liposomes with Different Sizes to Mice Brain after Sonication by Focused Ultrasound in the Presence of Microbubbles. Ultrasound Med. Biol. 2016, 42, 1499–1511, doi:10.1016/j.ultrasmedbio.2016.01.019.
- Cui, H.; Zhu, Q.; Xie, Q.; Liu, Z.; Gao, Y.; He, Y.; Tan, X.; Xu, Y. Low intensity ultrasound targeted microbubble destruction assists MSCs delivery and improves neural function in brain ischaemic rats. J. Drug Target. 2020, 28, 320–329, doi:10.1080/1061186X.2019.1656724.
- Blackmore, D.G.; Turpin, F.; Mohamed, A.Z.; Zong, F.; Pandit, R.; Pelekanos, M.; Nasrallah, F.; Sah, P.; Bartlett, P.F.; Götz, J. Multimodal analysis of aged wild-type mice exposed to repeated scanning ultrasound treatments demonstrates long-term safety. Theranostics 2018, 8, 6233–6247, doi:10.7150/thno.27941.
- Sun, Y.; Wang, H.; Wang, P.; Zhang, K.; Geng, X.; Liu, Q.; Wang, X. Tumor targeting DVDMS-nanoliposomes for an enhanced sonodynamic therapy of gliomas. Biomater. Sci. 2019, 7, 985–994, doi:10.1039/C8BM01187G.
- Pi, Z.; Huang, Y.; Shen, Y.; Zeng, X.; Hu, Y.; Chen, T.; Li, C.; Yu, H.; Chen, S.; Chen, X. Sonodynamic Therapy on Intracranial Glioblastoma Xenografts Using Sinoporphyrin Sodium Delivered by Ultrasound with Microbubbles. Ann. Biomed. Eng. 2019, 47, 549–562, doi:10.1007/s10439-018-02141-9.
- Omata, D.; Hagiwara, F.; Munakata, L.; Shima, T.; Kageyama, S.; Suzuki, Y.; Azuma, T.; Takagi, S.; Seki, K.; Maruyama, K.; et al. Characterization of Brain-Targeted Drug Delivery Enhanced by a Combination of Lipid-Based Microbubbles and Non-Focused Ultrasound. J. Pharm. Sci. 2020, 109, 2827–2835, doi:10.1016/j.xphs.2020.06.008.
- Wu, C.-Y.; Huang, R.-Y.; Liao, E.-C.; Lin, Y.-C.; Ho, Y.-J.; Chang, C.-W.; Chan, H.-L.; Huang, Y.-Z.; Hsieh, T.-H.; Fan, C.-H.; et al. A preliminary study of Parkinson’s gene therapy via sono-magnetic sensing gene vector for conquering extra/intracellular barriers in mice. Brain Stimul. 2020, 13, 786–799, doi:10.1016/j.brs.2020.02.024.
- Qu, F.; Wang, P.; Zhang, K.; Shi, Y.; Li, Y.; Li, C.; Lu, J.; Liu, Q.; Wang, X. Manipulation of Mitophagy by “All-in-One” nanosensitizer augments sonodynamic glioma therapy. Autophagy 2020, 16, 1413–1435, doi:10.1080/15548627.2019.1687210.
- Song, K.-H.; Harvey, B.K.; Borden, M.A. State-of-the-art of microbubble-assisted blood-brain barrier disruption. Theranostics 2018, 8, 4393–4408, doi:10.7150/thno.26869.
- Lea-Banks, H.; O’Reilly, M.A.; Hynynen, K. Ultrasound-responsive droplets for therapy: A review. J. Control. Release 2019, 293, 144–154, doi:10.1016/j.jconrel.2018.11.028.
- Boissenot, T.; Bordat, A.; Fattal, E.; Tsapis, N. Ultrasound-triggered drug delivery for cancer treatment using drug delivery systems: From theoretical considerations to practical applications. J. Control. Release 2016, 241, 144–163, doi:10.1016/j.jconrel.2016.09.026.
- Morfin, J.-F.; Beloeil, J.-C.; Tóth, É. Agents de Contraste pour l’IRM. 2014, 23.
- Kogan, P.; Gessner, R.C.; Dayton, P.A. Microbubbles in imaging: Applications beyond ultrasound. Bubble Sci. Eng. Technol. 2010, 2, 3–8, doi:10.1179/175889610X12730566149100.
- Cheung, J.S.; Chow, A.M.; Guo, H.; Wu, E.X. Microbubbles as a novel contrast agent for brain MRI. NeuroImage 2009, 46, 658–664, doi:10.1016/j.neuroimage.2009.02.037.
- Liao, A.-H.; Liu, H.-L.; Su, C.-H.; Hua, M.-Y.; Yang, H.-W.; Weng, Y.-T.; Hsu, P.-H.; Huang, S.-M.; Wu, S.-Y.; Wang, H.-E.; et al. Paramagnetic perfluorocarbon-filled albumin-(Gd-DTPA) microbubbles for the induction of focused-ultrasound-induced blood–brain barrier opening and concurrent MR and ultrasound imaging. Phys. Med. Biol. 2012, 57, 2787–2802, doi:10.1088/0031-9155/57/9/2787.
- Rapoport, N.; Nam, K.-H.; Gupta, R.; Gao, Z.; Mohan, P.; Payne, A.; Todd, N.; Liu, X.; Kim, T.; Shea, J.; et al. Ultrasound-mediated tumor imaging and nanotherapy using drug loaded, block copolymer stabilized perfluorocarbon nanoemulsions. J. Control. Release 2011, 153, 4–15, doi:10.1016/j.jconrel.2011.01.022.
- Arvanitis, C.D.; Ferraro, G.B.; Jain, R.K. The blood–brain barrier and blood–tumour barrier in brain tumours and metastases. Nat. Rev. Cancer 2020, 20, 26–41, doi:10.1038/s41568-019-0205-x.
- Fontanella, C.; Ongaro, E.; Bolzonello, S.; Guardascione, M.; Fasola, G.; Aprile, G. Clinical advances in the development of novel VEGFR2 inhibitors. Ann. Transl. Med. 2014, 2, 10.
- Deng, Z.; Sheng, Z.; Yan, F. Ultrasound-Induced Blood-Brain-Barrier Opening Enhances Anticancer Efficacy in the Treatment of Glioblastoma: Current Status and Future Prospects. J. Oncol. 2019, 2019, 1–9, doi:10.1155/2019/2345203.




