| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Aristidis Anthemidis | + 1898 word(s) | 1898 | 2021-01-11 03:07:34 | | | |
| 2 | Rita Xu | -470 word(s) | 1428 | 2021-01-13 05:13:58 | | |
Video Upload Options
A novel simple and sensitive, time-based flow injection solid phase extraction system was developed for the automated determination of metals at low concentration. The potential of the proposed scheme, coupled with flame atomic absorption spectrometry (FAAS), was demonstrated for trace lead and chromium(VI) determination in environmental water samples. The new sorptive extraction system, consisted of an online microcolumn packed with glass fiber coated with sol–gel poly (diphenylsiloxane) (sol–gel PDPS), which is presented here for the first time.
1. Introduction
Flow injection and related techniques have been proved to be suitable for on-line fluidic manipulation as well as for the successful sample processing in an automated operation mode, meeting the standards of green analytical chemistry [1][2]. Within the last decades, on-line sorbent extraction is by far the most utilized sample preparation technique in flow systems due to its straightforward operation, high preconcentration and separation efficiency, versatility and miniaturization ability, as well as advent of advanced sorptive materials such as the sol–gel-based ones. Most often, on-line microcolumns, packed with an appropriate sorptive material, within a flow network, are employed as an efficient front end to atomic spectrometric techniques such as the flame atomic absorption spectrometry (FAAS) or other detection systems, improving significantly the analytical performance characteristics [3]. A plethora of novel sorbents with desirable characteristics such as fast kinetics, physical chemical and mechanical stability, low cost, and availability have been proposed in on-line sorptive extraction methods for toxic metal determination in various types of samples matrices [4][5][6]. Generally, sorbents can be categorized by the nature of primary interaction mechanism with the target analyte as hydrophobic (reversed phase), polar, and ion-exchange.
Sol–gel technology has been proved to be the vehicle for preparing advanced hybrid inorganic–organic polymer coatings with tunable porosity, selectivity, and noticeably improved chemical and thermal stability. The sol–gel process allows chemical integration of the sol–gel sorbent to the substrate surface in diverse forms, such as particles, fiber, fabric, tube capillary, film, and monoliths that can be used in sample preparation micro-techniques [7][8]. Malik and co-workers reported for the first time the sol–gel process for solid phase microextraction (SPME) using hydroxy-terminated polydimethylsiloxane (PDMS) to create surface-bonded organic inorganic hybrid coating for SPME fiber [9]. The sol–gel PDMS-coated fiber offers better extraction selectivity than the commercially available one and was found to be efficient in extracting both polar and nonpolar analytes from aqueous samples. Due to the chemical bonding and the open porous structure of the sol–gel PDMS coatings, they provide comparable or better extraction efficiency with a shorter equilibrium time and significantly higher thermal stability [7]. The application of SPME fiber coated with sol–gel sorbent for extracting organometals from aqueous samples followed by HPLC analysis was first proposed by Gbatu et al. [10] while, up until today, the sol–gel sorbents for extracting metals and organometallics are used only as in-tube or capillary surface coatings, prior to inductively coupled plasma mass spectrometry (ICP-MS) determination [11]. A comprehensive review of SPME for trace elements speciation was published by Mester et al. [12].
In 2014, Kabir and Furton presented [13][14] a novel green sample pretreatment technique, fabric phase sorptive extraction (FPSE) overcoming the majority of the problems often encountered in modern sample preparation approaches such as low sorbent capacity and long sample preparation time [15]. The inherent porous surface of a natural or synthetic fabric substrate such as cotton, cellulose, polyester, and glass fiber together with the strength of sol–gel derived hybrid sorbents, which are uniformly dispersed in the form of an ultra-thin film into the fabric, result in a plethora of sorbents with significant retention capacity and satisfactorily fast extraction equilibrium to carry out the extraction of the analyte. Recently, Anthemidis et al. reported [16] for the first time the automation of the FPSE technique for on-line extraction and the determination of toxic metals in environmental samples by FAAS. The proposed fabric disk sorptive extraction (FDSE) technique is based on firmly packing an adequate number of fabric disks, obtained from a sol–gel sorbent-coated fabric, into a microcolumn incorporated in an on-line flow injection (FI) system.
In the frame of evaluation of new sol–gel sorbents in various geometrical shapes, Anthemidis et al. developed a novel green automatic sample preparation approach called fabric fiber sorbent extraction (FFSE) [17]. FFSE is based on the effective packing of sol–gel coated polyester fibers, into a FI on-line microcolumn, which operates as a front end to FAAS or other detectors for metal or organic analytes determination. Taking into consideration the advantages of sol–gel technology in producing the chemically bonded surface of fiber, a new category of sol–gel sorbents is introduced, resulting in a plethora of diverse fiber coatings, which potentially can be packed into an on-line preconcentration mini column for automated flow injection systems.
2. Characterization of Sol–Gel PDPS Coated Glass Fiber
2.1. Fourier Transform Infrared Spectroscopy (FT-IR)
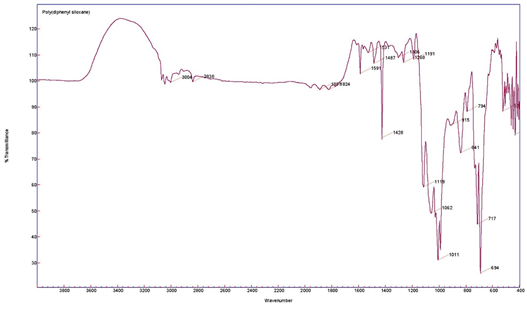
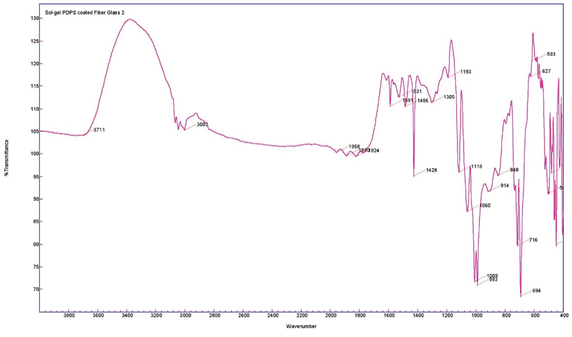
The sol–gel PDPS-coated glass fiber was characterized using Fourier Transform Infrared Spectroscopy (FT-IR). Figure 1 represents the FT-IR spectra of (a) pristine PDPS polymer; and (b) sol–gel PDPS-coated glass fiber.
Figure 1. FT-IR spectra of pristine PDPS polymer (top) and sol–gel PDPS coated glass fiber (bottom).
The characteristic peaks of pristine PDPS polymer appear at 694 cm−1 and 717 cm−1 representing vibration bands for Ph-Si-Ph. The very strong band at 1011 cm−1 represents the vibration band for Si-O-Si [18]. Bands at 1591 cm−1 and 1428 cm−1 can be assigned to skeleton vibration of phenyl groups in PDPS polymer [19]. The shoulder band at 1009 cm−1 originates from SiO2 network formation, and the appearance of a majority of bands from the PDPS spectra in the sol–gel PDPS coated fiberglass spectra is evidence of the successful integration of PDPS polymer into the sol–gel PDPS network coated on the glass fiber substrate.
2.2. Scanning Electron Microscopy (SEM)
The surface morphology of the uncoated glass fiber and sol–gel PDPS glass fibers were investigated using scanning electron microscopy. As can be seen in the SEM images presented in Figure 2, the surface of the sol–gel PDPS coated glass fiber (Figure 2b) is distinctly different than that of the uncoated glass fiber (Figure 2a). The sol–gel PDPS coating on glass fiber looks homogeneous with a characteristic roughened surface. The magnified SEM image of the sol–gel PDPS coated glass fiber (Figure 2c) demonstrated uniform surface coating, thanks to the chemical coating process exploited in sol–gel sorbent coating technology.
Figure 2. Scanning Electron Microscopy images of (a) uncoated glass fiber; (b) sol–gel PDPS coated glass fibers; (c) magnified image of sol–gel PDPS coated individual glass fiber.
3. Materials and Methods
3.1. Sol–Gel PDPS Coated Glass Fiber and Chelating Agent
Although the chelating agent ammonium pyrrolidine dithiocarbamate (APDC) is a highly polar chelating agent with logKow < 1.10, after complexing with metal ions, the electron-rich center of APDC becomes overall electronically charge-neutral and therefore, the complex turns relatively hydrophobic. Taking this observation into consideration, a new fiber substrate, glass-fiber was selected along with a non-polar organic polymer, poly-diphenylsiloxane (PDPS) and a methyl terminated trimethoxysilane (MTMS) inorganic precursor in order to create the robust hydrophobic extraction media. Due to the strong covalent bond between the glass fiber substrate and the sol–gel PDPS sorbent network, various organic solvents as well as acidic or alkaline solutions can be used for the effective elution of the extracted analytes without risking the potential loss of the extracting sorbent.
3.2. Construction of the Microcolumn
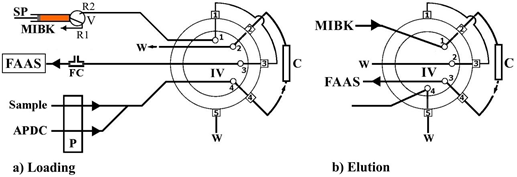
The microcolumns were constructed from a plastic (polypropylene) 1.0 mL disposable syringe (length ≈ 12 cm; i.d. 5.0 mm) with a luer slip centered tip. The main body of syringe was cut properly at a length ca. 40 mm., and the obtained packed column had an effective length of 30 mm and i.d. of 5.0 mm. At first, the glass fibers were cut in small pieces (ca. 1–2 mm) using scissors, so that they could be packed in the column. An amount of 200 mg of glass fiber coated with sol–gel PDPS was firmly packed into the column. It was not necessary to establish frits at the two ends of the column for glass fiber immobilization. Two push-fit connections made of silicon plug and short PTFE tubing (i.d. ≈ 0.5 mm) were used in order be installed in the FI manifold, at ports 2 and 4 (Figure 3).
Figure 3. Schematic diagram of the FI-FCSE-FAAS method for Pb(II) and Cr(VI) determination.
3.3. Automatic operation procedure
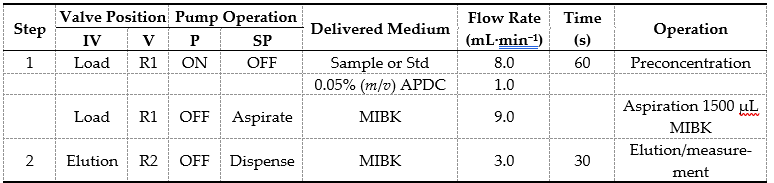
The automatic on-line FI-FCSE-FAAS analytical procedure is operated in two main steps: sample loading and the elution step, which are presented schematically in Figure 3, while the operational sequences of the developed method are summarized in Table 1.
Table 1. Operational sequences of the FI-FCSE-FAAS system for metal determination.
References
- Miró, M.; Hansen, E.H. On-line sample processing involving microextraction techniques as a front-end to atomic spectrometric detection for trace metal assays: A review. Chim. Acta 2013, 782, 1–11, doi:10.1016/j.aca.2013.03.019.
- Trojanowicz, M.; Kołacińska, K. Recent advances in flow injection analysis. Analyst 2016, 141, 2085–2139, doi:10.1039/c5an02522b.
- Yu, Y.; Wang, J. Recent advances in flow-based sample pretreatment for the determination of metal species by atomic spectrometry. Sci. Bull. 2013, 58, 1992–2002, doi:10.1007/s11434-013-5666-9.
- Lemos, V.A.; Teixeira, L.S.G.; Bezzera, M.A.; Costa, A.C.S.; Castro, J.T.; Cardoso, L.A.M.; de Jesus, D.S.; Santos, E.S.; Baliza, P.X.; Santos, L.N. New materials for solid-phase extraction of trace elements. Spectrosc. Rev. 2008, 43, 303–334, doi:10.1080/05704920802031341.
- Arkhipova, A.A.; Statkus, M.A.; Tsizin, G.I.; Zolotov, Y.A. Preconcentration of elements as hydrophobic complexes with low-polar adsorbents. Anal. Chem. 2015, 70, 1413–1431, doi:10.1134/S1061934815120047.
- Kanao, E.; Kubo, T.; Otsuka, K. Carbon_Based Nanomaterials for Separation Media. Chem. Soc. Jpn. 2020, 93, 482–489, doi:10.1246/bscj.20190372.
- Kabir, A.; Furton, K.G.; Malik, A. Innovation in sol-gel microextraction phases for solvent-free sample preparation in analytical chemistry. Trends Anal. Chem. 2013, 45, 197–218, doi:10.1016/j.trac.2012.11.014.
- Fumes, B.H.; Silva, M.R.; Andrade, F.N.; Nazario, C.E.D.; Lanças, F.M. Recent advances and future trends in new materials for sample preparation. Trends Anal. Chem. 2015, 71, 9–25, doi:10.1016/j.trac.2015.04.011.
- Chong, S.L.; Wang, D.; Hayes, J.D.; Wilhite, B.W.; Malik, A. Sol-gel coating technology for the preparation of solid-phase microextraction fibers of enhanced thermal stability. Chem. 1997, 69, 3889–3898, doi:10.1021/ac9703360.
- Gbatu, T.P.; Sutton, K.L.; Caruso, J.A. Development of new SPME fibers by sol-gel technology for SPME-HPLC determination of organometals. Chim. Acta 1999, 402, 67–79, doi:10.1016/S0003-2670(99)00532-2.
- Zheng, F.; Hu, B. Preparation of a high pH-resistant AAPTS-silica coating and its application to capillary microextraction (CME) of Cu, Zn, Ni, Hg and Cd from biological samples followed by on-line ICP-MS detection. Anal. Chim. Acta 2007, 605, 1–10, doi:10.1016/j.aca.2007.10.018.
- Mester, Z.; Sturgeon, R.E. Trace element speciation using solid phase microextraction. Acta Part B 2005, 60, 1243–1269, doi:10.1016/j.sab.2005.06.013.
- Kabir, A.; Furton, K.G. Fabric Phase Sorptive Extraction (FPSE). U.S. Patent and Trademark Office 14,216,121, 17 March 2014.
- Kumar, R.; Gaurav; Heena; Malik, A.K.; Kabir, A.; Furton, K.G. Efficient analysis of selected estrogens using fabric phase sorptive extraction and high performance liquid chromatography-fluorescence detection. Chromatogr. A 2014, 1359, 16–25, doi:10.1016/j.chroma.2014.07.013.
- Kazantzi, V.; Anthemidis, A. Fabric sol-gel Phase Sorptive Extraction Technique: A Review. Separations 2017, 4, 20, doi:10.390/separations4020020.
- Anthemidis, A.; Kazantzi, V.; Samanidou, V.; Kabir, A.; Furton, K.G. An automated flow injection system for metal determination by flame atomic absorption spectrometry involving on-line fabric disk sorptive extraction technique. Talanta 2016, 156, 64–70, doi:10.1016/j.talanta.2016.05.012.
- Kazantzi, V.; Kabir, A.; Furton, K.G.; Anthemidis, A. Fabric fiber sorbent extraction for on-line toxic metal determination by atomic absorption spectrometry: Determination of lead and cadmium in energy and soft drinks. J. 2018, 137, 285–291, doi:10.1016/j.micro.2017.11.006.
- Montesdeoca-Esponda, S.; Sosa-Ferrera, Z.; Kabir, A.; Furton, K.G.; Santana-Rodriguez, J.J. Fabric phase sorptive extraction followed by UHPLC-MS/MS for the analysis of benzotriazole UV stabilizers in sewage samples. Anal. Bioanal. Chem. 2015, 407, 8137–8150.
- Shang, X.; Cao, X.; Ma, Y.; Kumaravel, J.J.; Zheng, K.; Zhang, J.; Zhang, R. Diphenylsiloxane-bridged ladder-like hydrido-polysiloxane and the derivatisation by triphenylsiloxy substitution. Eur. Polym. J. 2018, 106, 53–62.