
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Ning Li | + 2099 word(s) | 2099 | 2021-01-07 09:06:25 | | | |
| 2 | Camila Xu | + 2 word(s) | 2101 | 2021-01-12 09:01:59 | | | | |
| 3 | Sirius Huang | -2 word(s) | 2099 | 2023-04-14 04:53:51 | | |
Video Upload Options
Mitochondria are energy-producing structures and the main site for aerobic respiration in cells, and are therefore called the “powerhouse of the cell”.
1. The Role of Mitochondria in Cancer Cells
Mitochondria are associated with many diseases, such as Parkinson’s disease [1], diabetic nephropathy [2], acute kidney injury [3], and Down syndrome [4]. Mitochondria also play an important role for cell signaling, apoptosis regulation, and energy metabolism in drug-induced cancer cells death; therefore, they are considered a significant target in cancer chemotherapy [5]. Some scholars have reviewed the mitochondrion as a target of anticancer therapy over the years [6][7][8][9]. Moreover, modulation of mitochondrial-dependent pathways by natural compounds is diverse (Figure 1). However, few researchers have reviewed natural products that regulate mitochondrial pathway in cancers.
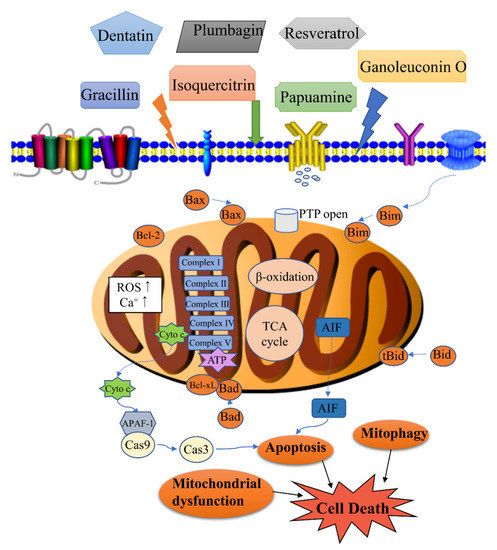
Figure 1. Modulation of mitochondrial-related cell death by natural products. Cell death associated with the activity of natural products includes apoptosis, mitophagy, mitochondrial dysfunction, etc. Apoptosis is regulated by the levels of Bcl-2 (B-cell lymphoma-2) family proteins, release of cytochrome c, and caspase activation. Mitophagy is the targeted phagocytosis and destruction of mitochondria by the autophagy machinery, and it is generally considered as the main mechanism of mitochondrial quality control. A decrease in energy production, an increase of reactive oxygen species (ROS) and permeability transition pore (PTP) opening can lead to mitochondrial dysfunction.
2. Mitochondrial Control of Apoptosis
Mitochondrial involvement is an important pathway in the process of apoptosis. The Bcl-2 protein family regulates apoptosis by controlling mitochondrial permeability. Anti-apoptotic proteins B-cell lymphoma-2 (Bcl-2) and B-cell lymphoma-extra large (Bcl-xL) reside in the outer membrane of mitochondria and inhibit the release of cytochrome c. Pro-apoptotic proteins Bax, Bad, Bid, and Bim can reside in the cytoplasm, translocating to mitochondria after receiving a death signal, and promote cytochrome c release into the cytoplasm. Released cytochrome c binds to apoptotic protease activating factor-1 (Apaf-1) to form apoptosome, amplifying the apoptotic cascade [10][11][12].
Necrotic stimulation leads to increased mitochondrial Ca2+ uptake and ROS production. High levels of Ca2+ and ROS induce the opening of the Cyclophilin-D (Cyp-D) sensitive permeability transition pore (PTP), leading to matrix swelling and Ca2+ release. Swelling damages the outer membrane and releases Ca2+ activating proteases, phosphatases, and nucleases, leading to necrotic degradation [13].
Fission or fusion rates may change under different growth conditions, and result in an increase or decrease in the number of mitochondria. When mitochondria become damaged, their connectivity is reduced, and mitochondria become shorter and rounder. The change from highly branched to fragmented morphologies may be induced by altered fission or fusion rates. At the early stage of apoptosis, the transition from a mitochondrial network to vesicular punctiform mitochondria was detected [14]. Mitochondrial fragmentation occurs in parallel to the formation of apoptotic bodies, increasing the number of the terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) positive nuclei and cleavage of the caspase substrate polymerase (PARP) [15].
3. Mitochondrial Control of Energy Metabolism
Mitochondria provide considerable flexibility for the growth and survival of tumor cells, and play a key role in harsh conditions, such as nutrient depletion and hypoxia. The rapid proliferation of cancer cells requires more mitochondria than normal cells. Therefore, the development of chemotherapeutic drugs for mitochondria is a breakthrough in the fight against cancer. Many scholars have clarified that the mechanical drive of mitochondrial respiration involves the tricarboxylic acid (TCA) cycle, and fatty acid β-oxidation enzymes in the mitochondrial matrix that generate electron donors to fuel respiration and electron transport chain (ETC) complexes, and ATP synthase in the inner mitochondrial membrane (IMM) that carry out oxidative phosphorylation [16]. Some natural products inhibit electron transport chain complexes. Four such complexes are NADH-ubiquinone reductase(complex I), succinate-ubiquinone reductase (complex II), ubiquinol-cytochrome c reductase (complex III), and cytochrome c oxidase (complex IV) [17]. Complex V, which is called ATP synthase, together with the above four complexes, completes oxidative phosphorylation to produce ATP. Inhibition of mitochondrial ETC complex activity can lead to significant mitochondrial dysfunction.
Cardiolipin, which consists of two phosphatidyl residues linked by a glycerol bridge, is a unique phospholipid dimer in the inner mitochondrial membrane in all eukaryotes. Cardiolipins play an important role in preserving mitochondrial structure and function. They support membrane dynamics and stabilize the lateral organization of protein-rich membranes in mitochondria [18]. Cardiolipins are involved in mitochondrial cristae morphology and stability [19], mitochondrial quality control, and dynamics by fission and fusion [20][21] and mitophagy [22]. They can also serve as a binding platform to recruit apoptotic factors in the apoptotic process [23][24]. However, it is still not clear how these events are interconnected and cooperate. In addition, cardiolipins are very susceptible to damage from ROS because of their high content of unsaturated acyl chains. Thus, the stability and function of mitochondria can be impaired by the biophysical properties of the membranes that are altered [25].
In this paper, we attempt to summarize the mechanisms through which natural products exert anticancer effects, as published in the past five years, by using a structural classification, with emphasis on the molecular mechanisms of mitochondrial involvement. Through all the reports, we found that most natural products regulate a series of proteins, such as Bax, Bcl-2, and caspases-3 and -9. Moreover, inhibitors of electron transport chain complexes can also exert anticancer activity. Details can be found in Table 1.
Table 1. Natural products (1–81) regulated mitochondria by different mechanisms in cancer cells.
| No. | Isolated Compound | Origin | Cell Line | Mechanism | Reference |
|---|---|---|---|---|---|
| Terpenoids | |||||
| 1 | Ganoleuconin O | Ganoderma leucocontextum | Huh7.5 | Fatty acid immobilization, loss of the mitochondrial lipid cardiolipin | [19] |
| 2 | Lupeol | Bombax ceiba | SK-RC-45 | Mitochondrial hyper fission | [20] |
| 3 | Betulinic acid | Betula alba | HeLa | Cardiolipin modification, ROS generation, Bad, caspase 9 | [21][22] |
| 4 | Alisol B-23-acetate | Alisma orientale | A549, NCI-H292 | ROS generation, Bcl-2↓, Bax↑, activation of caspase-3, -9, release of cytochrome c/AIF | [23] |
| 5 | Genipin | Gardenia jasminoides | N18TG2 | Activation of dicarboxylate carrier, decreased activity of UCP1, UCP3, and complex III of the respiratory chain, UCP2 inhibition | [24] |
| 6 | Alternol | Yew tree | PC-3 | Decrease of mitochondrial respiration, isocitric acid, fumaric acid and malic acid, ATP production | [25][26] |
| 7 | Cyathin Q | Cyathus africanus | HCT116 | Bcl-2↓, Bax↑, Bcl-xL↓, ROS generation, release of cytochrome c | [27] |
| 8 | 3α-hydroxy-19α-hydrogen-29-aldehyde-27-lupanoic acid | Potentilla discolor | HepG2 | Bcl-2↓, Bax↑, release of cytochrome c | [28] |
| 9 | Uvedafolin | Smallanthus sonchifolius | HeLa | MMP loss, release of cytochrome c | [29] |
| 10 | Heteronemin | Hippospongia sp. | Molt4 | ROS generation | [30] |
| 11 | Jatrogossone A | Jatropha gossypiifolia | KOPN-8 | MMP loss, ROS generation | [31] |
| 12 | Walsuronoid B | Walsura robusta | Bel-7402, HepG2 | ROS generation, mitochondrial and lysosomal dysfunction | [32] |
| 13 | Ferruginol | Podocarpus ferruginea | MDA-T32 | ROS generation, MMP loss, Bcl-2↓ | [33][34] |
| 14 | Lobocrassin B | Lobophytum crassum | CL1-5, H520, BEAS-2B |
Bcl-2↓, Bax↑, ROS generation, MMP loss, release of cytochrome c, activation of caspase-3 | [35] |
| 15 | Aellinane | Euphorbia aellenii | Caov-4 | Bcl-2↓, Bax↑, ROS generation, MMP loss | [36] |
| 16 | Tingenin B | Maytenus sp. | MCF-7s | Bcl-2↓, Bax↑, MMP loss | [37] |
| 17 | 3-O-trans-p-coumaroyl alphitolic acid | Ziziphus jujuba | PC-3 | ROS generation | [38] |
| 18 | Zerumbone | Zingiber zerumbet | PC-3, DU-145 | Tubulin binding and crosstalk between endoplasmic reticulum stress and mitochondrial insult | [39][40] |
| Flavonoids | |||||
| 19 | Isoquercitrin | Hibiscus cannabinus | MDA-MB-231 | LSD1-induced mitochondrial-mediated apoptosis pathway | [41][42] |
| 20 | Luteolin | Cauliflower, peanut, and carrot | SW1990 | Inhibitor of Bcl-2, mitochondrial permeabilization | [43] |
| 21 | Dihydromyricetin | Ampelopsis grossedentata | HepG2 | Akt/Bad signal pathway, mitochondrial apoptotic pathway, Bax↑, Bad↑, inhibition of the phosphorylation of Bad at Ser136 and Ser112 | [44][45] |
| 22 | Artonin E | Artocarpus elasticus | SKOV-3 | Release of cytochrome c, Activation of caspases-3, -8, and -9, Bax↑, Bcl-2↓, HSP70↓, survivin↓ | [46] |
| 23 | Myricetin | Fruits and vegetables | SNU-80 | Bax/Bcl-2↑, release of AIF | [47] |
| 24 | Xanthones | Garcinia xanthochymus | HepG2 | Bax↑, Bcl-2↓, Bcl-xL↓, Mcl-1↓, and survivin↓ | [48] |
| 25 | Cycloartobiloxanthone | Artocarpus gomezianus | H460 | Bax↑, Bcl-2↓, Mcl-1↓ | [49] |
| 26 | Paratocarpin E | Euphorbia humifusa | MCF-7 | Bax↑, Bcl-2↓, release of cytochrome c | [50] |
| 27 | Puerarin 6′’-O-xyloside | Pueraria lobata | SW480 | Bax↑, Bad↑, Bcl-2↓, caspase-3 and -9 activation | [51] |
| 28 | α-mangostin | Cratoxylum arborescens | HeLa | ROS generation, MMP loss, release of cytochrome c | [52] |
| 29 | Chrysin | Honey and propolis | Mitochondria isolated from hepatocytes of HCC rats |
ROS generation, MMP loss, release of cytochrome c, swelling in mitochondria | [53][54] |
| 30 | Fisetin | Strawberries, apples, grapes, onions, and cucumbers | SCC-4 | ROS generation, Ca2+ production, MMP loss, Bcl-2↓, Bax↑, Bid↑, release of cytochrome c, AIF, and Endo G | [55][56] |
| 31 | Baicalein | Scutellaria baicalensis, Scutellaria radix | A2780 | Combination therapy with baicalein and taxol had much higher antitumor effects compared with the monotherapy. Release of cytochrome c, and caspase-3 and -9 activation |
[57][58] |
| 32 | Alpinetin | Zingiberaceous plants |
A549 | Bcl-2↓, Bax↑, Bcl-xL↓, XIAP↓, PI3K/Akt signaling pathway, sensitized drug-resistant lung cancer cells | [59][60] |
| 33 | Chamaejasmin B | Stellerachamaejasme | KB, KBV200 | Bcl-2↓, Bax↑, MMP loss, release of cytochrome c and AIF | [61] |
| 34 | Mensacarcin | Streptomyces bacteria | SK-Mel-28, SK-Mel-5, HCT-116 | Release of cytochrome c, energy production and mitochondrial function rapidly disturbed | [62] |
| Saponins | |||||
| 35 | Gracillin | Dioscorea gracillima | H226B, H460 | Targeting mitochondrial complex II, suppressing ATP synthesis, ROS generation | [63] |
| 36 | Polyphyllin I | Paris polyphylla | MDA-MB-231 | Mitochondrial translocation of DRP1, mitochondrial fission, release of cytochrome c, mitochondrial PTEN-induced kinase 1↑ | [64][65] |
| 37 | Frondoside A | Cucumaria frondosa | CA46 | Bcl-2↓, survivin↓, release of HtrA2/Omi and cytochrome c, ROS generation | [66] |
| 38 | 3β-O-α-l- arabinopyranoside |
Clematis ganpiniana | MCF-7, MDA-MB-231 | Release of cytochrome c and Apaf-1, upregulation of caspase-9 and caspase-3 | [67] |
| 39 | Sakuraso-saponin | Aegiceras corniculatum | LNcaP, 22RV-1, C4-2 | Bcl-xL↓ | [68][69] |
| 40 | Ginsenoside compound K | Panax ginseng | SK-N-BE(2), SH-SY5Y | Bcl-2↓, Bcl-xL↓ | [70] |
| 41 | Escin | Aesculus hippocastanum | 786-O, Caki-1 | G2/M arrest and ROS-modulated mitochondrial pathways | [71] |
| 42 | α-Hederin | Hedera helix | SW620 | NF-κB signaling pathway, Bcl-2↓, Bax↑, release of cytochrome c | [72][73] |
| Alkaloids | |||||
| 43 | Cathachunine | Catharanthus roseus | HL60 | ROS-dependent mitochondria-mediated intrinsic pathway, Bcl-2/Bax↓, ROS generation, MMP loss, release of cytochrome c | [74] |
| 44 | Berberine | Rhizoma coptidis | T98G, LN18 | ERK1/2-mediated impairment of mitochondrial aerobic respiration | [75][76] |
| 45 | Papuamine | Haliclona sp. | H1299 | Intracellular ATP depleted by causing mitochondrial dysfunction, mitochondrial superoxide production | [77] |
| 46 | Bis (2-ethyl hexyl) 1H-pyrrole-3, 4-dicarboxylate | Tinospora cordifolia | MDA-MB-231 | ROS generation, increase in intracellular calcium, phosphorylation of p53, mitochondrial membrane depolarization, MPTP, and cardiolipin peroxidation, Bcl-2↓, Bax↑, release of cytochrome c, caspase activation, DNA fragmentation | [78] |
| 47 | Unantimycin A | Found in the fraction library of microbial metabolites | Semi-intact cells with specific substrates for each complex of the mitochondrial electron transport chain |
Targeted inhibition of mitochondrial complex I | [79] |
| 48 | NPL40330 | Found in chemical library | Semi-intact cells with specific substrates for each complex of the mitochondrial electron transport chain |
Targeted inhibition of mitochondrial complex III | [80] |
| 49 | Boholamide A | Marine mollusks | U87MG | Influx of Ca2+ | [81] |
| 50 | Cernumidine | Solanum cernuum | T24 | Cytotoxicity and chemosensitizing effect of cernumidine to cisplatin. Bcl-2↓, Bax↑, MMP loss | [82] |
| 51 | Lycorine | Amaryllidaceae plant family | HepG2 | mPTP opening, MMP loss, ATP depletion, release of Ca2+ and cytochrome c, caspase activation | [83] |
| 52 | Lagunamides A | Lyngbya majuscule | A549 | MMP loss, ROS generation | [84] |
| 53 | Cordycepin | Cordyceps | OVCAR-3 | Downregulation of mitochondrial function and limitation of energy production; metastasis and migration suppressed | [85][86] |
| Coumarins | |||||
| 54 | 2,3-Dihydro-7- hydroxy-2R*,3R*- dimethyl-2-[4,8-dimethyl-3(E),7- nonadienyl]-furo[3,2-c]coumarin |
Ferula ferulaeoides | C6 | MMP loss, Bcl-xL↓, Bcl-2↓, Bax↑, cleavage of Bid, FAS↑, FADD↑ | [87] |
| 55 | Dentatin | Clausena excavate | HepG2 | Bcl-xL↓, Bcl-2↓, Bax↑, release of cytochrome c | [88][89] |
| 56 | Aesculetin | Cortex Fraxini | THP-1 | Bcl-2↓, Bax↑ | [90] |
| Quinones | |||||
| 57 | Quambalarine B | Quambalaria cyanescens | Jurkat E6.1 | Inhibition of mitochondrial complex I and II, inhibition of mitochondrial respiration, metabolism reprogramming | [91][92] |
| 58 | Plumbagin | Plumbago zeylanica | MG63 | ROS generation, Bcl-2↓, Bax↑, Bcl-xL↓, and Bak↓, endoplasmic reticulum stress | [93] |
| 59 | Shikonin | Lithospermum erythrorhizon | HGC-27 | Bcl-2↓, Bax↑, survivin↓ | [94] |
| 60 | 2,7-dihydroxy-3-methylanthraquinone | Hedyotis diffusa | SGC-7901 | Bcl-xl↓, Bcl-2↓, Bax↑, Bad↑, release of cytochrome c | [95] |
| 61 | 3-hydroxy-1,5,6-trimethoxy-2-methyl-9,10-anthraquinone | Prismatomeris connate | A549, H1299 | Bcl-2↓, Mcl-1↓, Bax↑ | [96] |
| 62 | Thymoquinone | Nigella sativa | T24, 253J | Bcl-2↓, Bax↑, release of cytochrome c and AIF | [97] |
| Miscellanea | |||||
| 63 | Methylsulfonylmethane | Fruits and vegetables | YD-38 | Bcl-xL↓, Bcl-2↓, Bax↑, release of cytochrome c, MMP loss | [98][99] |
| 64 | Parameritannin A-2 | Urceola huaitingii | HGC27 | Enhanced doxorubicin-induced mitochondria-dependent apoptosis, inhibition of the PI3K/Akt, ERK1/2 and p38 pathways, Bcl-2↓, Bcl-xl↓, Bax↑, Bid↑, release of cytochrome c, caspase activation | [100] |
| 65 | Resveratrol | Polygonum cuspidatum, Veratrum nigrum, Cassia obtusifolia |
H838, H520; K562 |
Enhanced antitumor activities of cisplatin; Induced apoptosis |
[101][102] |
| 66 | Oleuropein | Olea europaea | H1299 | Bcl-2/Bax↓, release of cytochrome c, activation of caspase-3 | [103][104] |
| 67 | Homoisoflavanone-1 | Polygonatum odoratum | A549 | Mitochondria-caspase-dependent and ER stress pathways, Bcl-2/ Bak↓ | [105] |
| 68 | Gallic acid | Green tea, grapes, red wine |
H446 | ROS-dependent mitochondrial apoptotic pathway | [106] |
| 69 | Hierridin b | Cyanobium sp. | HT-29 | Proteomics identified 21 differentially expressed proteins belonging to the categories protein folding/synthesis and cell structure and reduced mitochondrial activity and as confirmed by morphological analysis of mitochondrial parameters |
[107][108] |
| 70 | Deoxyarbutin | Ecklonia cava | B16F10 | MMP loss, ATP depletion and ROS overload generation | [109] |
| 71 | Magnolol | Magnolia officinalis | OS-RC-2, 786-O | P53, Bcl-2/Bax↓, release of cytochrome c, caspase activation, ROS generation | [110] |
| 72 | Oblongifolin C | Garcinia yunnanensis | QBC939 | Mitochondrial dysfunction | [111] |
| 73 | Amorfrutin C | Glycyrrhiza foetida | HT-29 | mPTP opening, mitochondrial oxygen consumption and extracellular acidification increased | [112] |
| 74 | Allyl isothiocyanate | Cruciferous vegetables | MCF-7, MDA-MB-231 | ROS and Ca2+ production, MMP loss, release of cytochrome c, AIF, and Endo G, Bcl-2↓, Bax↑ | [113][114] |
| 75 | α-conidendrin | Taxus yunnanensis | MCF-7 and MDA-MB-231 | ROS generation, p53↑, Bax↑, Bcl-2↓, MMP loss, release of cytochrome c, activation of caspases-3 and -9 | [115] |
| 76 | Dehydrobruceine B | Brucea javanica | A549, NCI-H292 | MMP loss, release of cytochrome c, cleavage of caspase-9, caspase-3, and poly (ADP-ribose) polymerase (PARP) | [116] |
| 77 | Frugoside | Calotropis procera | M14, A375 | ROS generation | [117][118] |
| 78 | Methyl caffeate | Solanum torvum | MCF-7 | Bcl-2↓, Bax↑, Bid↑, p53↑, cleavage of caspase-3 and PARP, release of cytochrome c | [119] |
| 79 | Tetrahydrocurcumin | Curcuma longa | MCF-7 | ROS generation, Bcl-2↓, PARP↓, Bax↑, release of cytochrome c, MMP loss | [120] |
| 80 | Phloretin | Apple tree leaves and Manchurian apricot | EC-109 | Bcl-2↓, Bax↑ | [121] |
| 81 | Sesamol | Sesame seeds | HepG2 | Bcl-2↓, Bax↑, MMP loss, H2O2 production, PI3K Class III/Belin-1 pathway | [122] |
References
- Grunewald, A.; Kumar, K.R.; Sue, C.M. New insights into the complex role of mitochondria in Parkinson’s disease. Prog. Neurobiol. 2019, 177, 73–93. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.; Han, Y.; Liu, J.; Song, P.; Xu, X.; Zhao, L.; Hu, C.; Xiao, L.; Liu, F.; Zhang, H.; et al. Mitochondria: A Novel Therapeutic Target in Diabetic Nephropathy. Curr. Med. Chem. 2017, 24, 3185–3202. [Google Scholar] [CrossRef] [PubMed]
- Ishimoto, Y.; Inagi, R. Mitochondria: A therapeutic target in acute kidney injury. Nephrol. Dial. Transplant. 2016, 31, 1062–1069. [Google Scholar] [CrossRef]
- Valenti, D.; Braidy, N.; De Rasmo, D.; Signorile, A.; Rossi, L.; Atanasov, A.G.; Volpicella, M.; Henrion-Caude, A.; Nabavi, S.M.; Vacca, R.A. Mitochondria as pharmacological targets in Down syndrome. Free Radic. Biol. Med. 2018, 114, 69–83. [Google Scholar] [CrossRef]
- Grad, J.M.; Cepero, E.; Boise, L.H. Mitochondria as targets for established and novel anti-cancer agents. Drug Resist. Updat. 2001, 4, 85–91. [Google Scholar] [CrossRef]
- Costantini, P.; Jacotot, E.; Decaudin, D.; Kroemer, G. Mitochondrion as a novel target of anticancer chemotherapy. J. Natl. Cancer Inst. 2000, 92, 1042–1053. [Google Scholar] [CrossRef]
- Wen, S.; Zhu, D.; Huang, P. Targeting cancer cell mitochondria as a therapeutic approach. Future Med. Chem. 2013, 5, 53–67. [Google Scholar] [CrossRef]
- Wu, D.; Wang, X.; Sun, H. The role of mitochondria in cellular toxicity as a potential drug target. Cell Biol. Toxicol. 2018, 34, 87–91. [Google Scholar] [CrossRef]
- Bhat, T.A.; Kumar, S.; Chaudhary, A.K.; Yadav, N.; Chandra, D. Restoration of mitochondria function as a target for cancer therapy. Drug Discov. Today 2015, 20, 635–643. [Google Scholar] [CrossRef]
- Brenner, D.; Mak, T.W. Mitochondrial cell death effectors. Curr. Opin. Cell Biol. 2009, 21, 871–877. [Google Scholar] [CrossRef] [PubMed]
- Lindsay, J.; Esposti, M.D.; Gilmore, A.P. Bcl-2 proteins and mitochondria—Specificity in membrane targeting for death. Biochim. Biophys. Acta 2011, 1813, 532–539. [Google Scholar] [CrossRef] [PubMed]
- Chalah, A.; Khosravi-Far, R. The mitochondrial death pathway. Adv. Exp. Med. Biol. 2008, 615, 25–45. [Google Scholar] [PubMed]
- Peixoto, P.M.; Ryu, S.Y.; Kinnally, K.W. Mitochondrial ion channels as therapeutic targets. FEBS Lett. 2010, 584, 2142–2152. [Google Scholar] [CrossRef] [PubMed]
- Karbowski, M.; Youle, R.J. Dynamics of mitochondrial morphology in healthy cells and during apoptosis. Cell Death Differ. 2003, 10, 870–880. [Google Scholar] [CrossRef]
- Mancini, M.; Anderson, B.O.; Caldwell, E.; Sedghinasab, M.; Paty, P.B.; Hockenbery, D.M. Mitochondrial proliferation and paradoxical membrane depolarization during terminal differentiation and apoptosis in a human colon carcinoma cell line. J. Cell Biol. 1997, 138, 449–469. [Google Scholar] [CrossRef]
- Vyas, S.; Zaganjor, E.; Haigis, M.C. Mitochondria and Cancer. Cell 2016, 166, 555–566. [Google Scholar] [CrossRef]
- Ernster, L.; Schatz, G. Mitochondria: A historical review. J. Cell Biol. 1981, 91, 227s–255s. [Google Scholar] [CrossRef]
- Ren, M.; Phoon, C.K.; Schlame, M. Metabolism and function of mitochondrial cardiolipin. Prog. Lipid Res. 2014, 55, 1–16. [Google Scholar] [CrossRef]
- Ikon, N.; Ryan, R.O. Cardiolipin and mitochondrial cristae organization. Biochim. Biophys. Acta Biomembr. 2017, 1859, 1156–1163. [Google Scholar] [CrossRef]
- Dudek, J. Role of Cardiolipin in Mitochondrial Signaling Pathways. Front. Cell Dev. Biol. 2017, 5, 90. [Google Scholar] [CrossRef] [PubMed]
- Kameoka, S.; Adachi, Y.; Okamoto, K.; Iijima, M.; Sesaki, H. Phosphatidic Acid and Cardiolipin Coordinate Mitochondrial Dynamics. Trends Cell Biol. 2018, 28, 67–76. [Google Scholar] [CrossRef] [PubMed]
- Schlattner, U.; Tokarska-Schlattner, M.; Epand, R.M.; Boissan, M.; Lacombe, M.L.; Kagan, V.E. NME4/nucleoside diphosphate kinase D in cardiolipin signaling and mitophagy. Lab. Investig. 2018, 98, 228–232. [Google Scholar] [CrossRef] [PubMed]
- Ott, M.; Zhivotovsky, B.; Orrenius, S. Role of cardiolipin in cytochrome c release from mitochondria. Cell Death Differ. 2007, 14, 1243–1247. [Google Scholar] [CrossRef] [PubMed]
- Lucken-Ardjomande, S.; Montessuit, S.; Martinou, J.C. Contributions to Bax insertion and oligomerization of lipids of the mitochondrial outer membrane. Cell Death Differ. 2008, 15, 929–937. [Google Scholar] [CrossRef] [PubMed]
- Paradies, G.; Paradies, V.; Ruggiero, F.M.; Petrosillo, G. Role of Cardiolipin in Mitochondrial Function and Dynamics in Health and Disease: Molecular and Pharmacological Aspects. Cells 2019, 8, 728. [Google Scholar] [CrossRef]
- Kong, L.Y.; Tan, R.X. Artemisinin, a miracle of traditional Chinese medicine. Nat. Prod. Rep. 2015, 32, 1617–1621. [Google Scholar] [CrossRef]
- Ding, G.S. Important Chinese herbal remedies. Clin. Ther. 1987, 9, 345–357. [Google Scholar]
- Benelli, G.; Pavela, R.; Cianfaglione, K.; Sender, J.; Danuta, U.; Maslanko, W.; Canale, A.; Barboni, L.; Petrelli, R.; Zeppa, L.; et al. Ascaridole-rich essential oil from marsh rosemary (Ledum palustre) growing in Poland exerts insecticidal activity on mosquitoes, moths and flies without serious effects on non-target organisms and human cells. Food Chem. Toxicol. 2020, 138, 111184. [Google Scholar] [CrossRef]
- Liu, G.; Wang, K.; Kuang, S.; Cao, R.; Bao, L.; Liu, R.; Liu, H.; Sun, C. The natural compound GL22, isolated from Ganoderma mushrooms, suppresses tumor growth by altering lipid metabolism and triggering cell death. Cell Death Dis. 2018, 9, 689. [Google Scholar] [CrossRef]
- Sinha, K.; Chowdhury, S.; Banerjee, S.; Mandal, B.; Mandal, M.; Majhi, S.; Brahmachari, G.; Ghosh, J.; Sil, P.C. Lupeol alters viability of SK-RC-45 (Renal cell carcinoma cell line) by modulating its mitochondrial dynamics. Heliyon 2019, 5, e02107. [Google Scholar] [CrossRef] [PubMed]
- Potze, L.; Di Franco, S.; Grandela, C.; Pras-Raves, M.L.; Picavet, D.I.; van Veen, H.A.; van Lenthe, H.; Mullauer, F.B.; van der Wel, N.N.; Luyf, A.; et al. Betulinic acid induces a novel cell death pathway that depends on cardiolipin modification. Oncogene 2016, 35, 427–437. [Google Scholar] [CrossRef] [PubMed]
- Xu, T.; Pang, Q.; Wang, Y.; Yan, X. Betulinic acid induces apoptosis by regulating PI3K/Akt signaling and mitochondrial pathways in human cervical cancer cells. Int. J. Mol. Med. 2017, 40, 1669–1678. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Li, H.; Wang, X.; Shen, T.; Wang, S.; Ren, D. Alisol B-23-acetate, a tetracyclic triterpenoid isolated from Alisma orientale, induces apoptosis in human lung cancer cells via the mitochondrial pathway. Biochem. Biophys. Res. Commun. 2018, 505, 1015–1021. [Google Scholar] [CrossRef]
- Wang, X.; Lu, X.; Zhu, R.; Zhang, K.; Li, S.; Chen, Z.; Li, L. Betulinic Acid Induces Apoptosis in Differentiated PC12 Cells Via ROS-Mediated Mitochondrial Pathway. Neurochem. Res. 2017, 42, 1130–1140. [Google Scholar] [CrossRef]
- Yang, C.; Li, Y.; Fu, L.; Jiang, T.; Meng, F. Betulinic acid induces apoptosis and inhibits metastasis of human renal carcinoma cells in vitro and in vivo. J. Cell. Biochem. 2018, 119, 8611–8622. [Google Scholar] [CrossRef]
- Kreiter, J.; Rupprecht, A.; Zimmermann, L.; Moschinger, M.; Rokitskaya, T.I.; Antonenko, Y.N.; Gille, L.; Fedorova, M.; Pohl, E.E. Molecular Mechanisms Responsible for Pharmacological Effects of Genipin on Mitochondrial Proteins. Biophys. J. 2019, 117, 1845–1857. [Google Scholar] [CrossRef]
- Liu, X.; Wang, J.; Sun, B.; Zhang, Y.; Zhu, J.; Li, C. Cell growth inhibition, G2M cell cycle arrest, and apoptosis induced by the novel compound Alternol in human gastric carcinoma cell line MGC803. Investig. New Drugs 2007, 25, 505–517. [Google Scholar] [CrossRef]
- Li, C.; He, C.; Xu, Y.; Xu, H.; Tang, Y.; Chavan, H.; Duan, S.; Artigues, A.; Forrest, M.L.; Krishnamurthy, P.; et al. Alternol eliminates excessive ATP production by disturbing Krebs cycle in prostate cancer. Prostate 2019, 79, 628–639. [Google Scholar] [CrossRef]
- He, L.; Han, J.; Li, B.; Huang, L.; Ma, K.; Chen, Q.; Liu, X.; Bao, L.; Liu, H. Identification of a new cyathane diterpene that induces mitochondrial and autophagy-dependent apoptosis and shows a potent in vivo anti-colorectal cancer activity. Eur. J. Med. Chem. 2016, 111, 183–192. [Google Scholar] [CrossRef]
- Zhang, J.; Liu, C.; Huang, R.Z.; Chen, H.F.; Liao, Z.X.; Sun, J.Y.; Xia, X.K.; Wang, F.X. Three new C-27-carboxylated-lupane-triterpenoid derivatives from Potentilla discolor Bunge and their in vitro antitumor activities. PLoS ONE 2017, 12, e0175502. [Google Scholar] [CrossRef] [PubMed]
- Bispo de Jesus, M.; Zambuzzi, W.F.; Ruela de Sousa, R.R.; Areche, C.; Santos de Souza, A.C.; Aoyama, H.; Schmeda-Hirschmann, G.; Rodriguez, J.A.; Monteiro de Souza Brito, A.R.; Peppelenbosch, M.P.; et al. Ferruginol suppresses survival signaling pathways in androgen-independent human prostate cancer cells. Biochimie 2008, 90, 843–854. [Google Scholar] [CrossRef] [PubMed]
- Lin, M.X.; Lin, S.H.; Li, Y.R.; Chao, Y.H.; Lin, C.H.; Su, J.H.; Lin, C.C. Lobocrassin B Induces Apoptosis of Human Lung Cancer and Inhibits Tumor Xenograft Growth. Mar. Drugs 2017, 15, 378. [Google Scholar] [CrossRef] [PubMed]
- Nabatchian, F.; Moradi, A.; Aghaei, M.; Ghanadian, M.; Jafari, S.M.; Tabesh, S. New 6(17)-epoxylathyrane diterpene: Aellinane from Euphorbia aellenii induces apoptosis via mitochondrial pathway in ovarian cancer cell line. Toxicol. Mech. Methods 2017, 27, 622–630. [Google Scholar] [CrossRef] [PubMed]
- Cevatemre, B.; Botta, B.; Mori, M.; Berardozzi, S.; Ingallina, C.; Ulukaya, E. The plant-derived triterpenoid tingenin B is a potent anticancer agent due to its cytotoxic activity on cancer stem cells of breast cancer in vitro. Chem. Biol. Interact. 2016, 260, 248–255. [Google Scholar] [CrossRef] [PubMed]
- Shin, M.; Lee, B.M.; Kim, O.; Tran, H.N.K.; Lee, S.; Hwangbo, C.; Min, B.S.; Lee, J.H. Triterpenoids from Ziziphus jujuba induce apoptotic cell death in human cancer cells through mitochondrial reactive oxygen species production. Food Funct. 2018, 9, 3895–3905. [Google Scholar] [CrossRef] [PubMed]
- Chan, M.L.; Liang, J.W.; Hsu, L.C.; Chang, W.L.; Lee, S.S.; Guh, J.H. Zerumbone, a ginger sesquiterpene, induces apoptosis and autophagy in human hormone-refractory prostate cancers through tubulin binding and crosstalk between endoplasmic reticulum stress and mitochondrial insult. Naunyn-Schmiedeberg’s Arch. Pharmacol. 2015, 388, 1223–1236. [Google Scholar] [CrossRef]
- Prasannan, R.; Kalesh, K.A.; Shanmugam, M.K.; Nachiyappan, A.; Ramachandran, L.; Nguyen, A.H.; Kumar, A.P.; Lakshmanan, M.; Ahn, K.S.; Sethi, G. Key cell signaling pathways modulated by zerumbone: Role in the prevention and treatment of cancer. Biochem. Pharmacol. 2012, 84, 1268–1276. [Google Scholar] [CrossRef]
- Assini, J.M.; Mulvihill, E.E.; Huff, M.W. Citrus flavonoids and lipid metabolism. Curr. Opin. Lipidol. 2013, 24, 34–40. [Google Scholar] [CrossRef]
- Manach, C.; Scalbert, A.; Morand, C.; Remesy, C.; Jimenez, L. Polyphenols: Food sources and bioavailability. Am. J. Clin. Nutr. 2004, 79, 727–747. [Google Scholar] [CrossRef]
- Pei, R.; Liu, X.; Bolling, B. Flavonoids and gut health. Curr. Opin. Biotechnol. 2020, 61, 153–159. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Wang, Y.; Mai, Y.; Li, H.; Wang, Z.; Xu, J.; He, X. Health Benefits of the Flavonoids from Onion: Constituents and Their Pronounced Antioxidant and Anti-Neuroinflammatory Capacities. J. Agric. Food Chem. 2020, 68, 799–807. [Google Scholar] [CrossRef] [PubMed]
- Androutsopoulos, V.P.; Papakyriakou, A.; Vourloumis, D.; Tsatsakis, A.M.; Spandidos, D.A. Dietary flavonoids in cancer therapy and prevention: Substrates and inhibitors of cytochrome P450 CYP1 enzymes. Pharmacol. Ther. 2010, 126, 9–20. [Google Scholar] [CrossRef] [PubMed]
- Duteil, D.; Metzger, E.; Willmann, D.; Karagianni, P.; Friedrichs, N.; Greschik, H.; Gunther, T.; Buettner, R.; Talianidis, I.; Metzger, D.; et al. LSD1 promotes oxidative metabolism of white adipose tissue. Nat. Commun. 2014, 5, 4093. [Google Scholar] [CrossRef]
- Hino, S.; Sakamoto, A.; Nagaoka, K.; Anan, K.; Wang, Y.; Mimasu, S.; Umehara, T.; Yokoyama, S.; Kosai, K.; Nakao, M. FAD-dependent lysine-specific demethylase-1 regulates cellular energy expenditure. Nat. Commun. 2012, 3, 758. [Google Scholar] [CrossRef]
- Sakamoto, A.; Hino, S.; Nagaoka, K.; Anan, K.; Takase, R.; Matsumori, H.; Ojima, H.; Kanai, Y.; Arita, K.; Nakao, M. Lysine Demethylase LSD1 Coordinates Glycolytic and Mitochondrial Metabolism in Hepatocellular Carcinoma Cells. Cancer Res. 2015, 75, 1445–1456. [Google Scholar] [CrossRef]
- Xu, X.; Peng, W.; Liu, C.; Li, S.; Lei, J.; Wang, Z.; Kong, L.; Han, C. Flavone-based natural product agents as new lysine-specific demethylase 1 inhibitors exhibiting cytotoxicity against breast cancer cells in vitro. Bioorg. Med. Chem. 2019, 27, 370–374. [Google Scholar] [CrossRef]
- Valentova, K.; Vrba, J.; Bancirova, M.; Ulrichova, J.; Kren, V. Isoquercitrin: Pharmacology, toxicology, and metabolism. Food Chem. Toxicol. 2014, 68, 267–282. [Google Scholar] [CrossRef]
- Li, Z.; Zhang, Y.; Chen, L.; Li, H. The dietary compound luteolin inhibits pancreatic cancer growth by targeting BCL-2. Food Funct. 2018, 9, 3018–3027. [Google Scholar] [CrossRef]
- Zhang, Z.; Zhang, H.; Chen, S.; Xu, Y.; Yao, A.; Liao, Q.; Han, L.; Zou, Z.; Zhang, X. Dihydromyricetin induces mitochondria-mediated apoptosis in HepG2 cells through down-regulation of the Akt/Bad pathway. Nutr. Res. 2017, 38, 27–33. [Google Scholar] [CrossRef]
- Hou, X.L.; Tong, Q.; Wang, W.Q.; Shi, C.Y.; Xiong, W.; Chen, J.; Liu, X.; Fang, J.G. Suppression of Inflammatory Responses by Dihydromyricetin, a Flavonoid from Ampelopsis grossedentata, via Inhibiting the Activation of NF-κB and MAPK Signaling Pathways. J. Nat. Prod. 2015, 78, 1689–1696. [Google Scholar] [CrossRef] [PubMed]
- Rahman, M.A.; Ramli, F.; Karimian, H.; Dehghan, F.; Nordin, N.; Ali, H.M.; Mohan, S.; Hashim, N.M. Artonin E Induces Apoptosis via Mitochondrial Dysregulation in SKOV-3 Ovarian Cancer Cells. PLoS ONE 2016, 11, e0151466. [Google Scholar] [CrossRef] [PubMed]
- Jo, S.; Ha, T.K.; Han, S.H.; Kim, M.E.; Jung, I.; Lee, H.W.; Bae, S.K.; Lee, J.S. Myricetin Induces Apoptosis of Human Anaplastic Thyroid Cancer Cells via Mitochondria Dysfunction. Anticancer Res. 2017, 37, 1705–1710. [Google Scholar] [PubMed]
- Gao, S.; Sun, D.; Wang, G.; Zhang, J.; Jiang, Y.; Li, G.; Zhang, K.; Wang, L.; Huang, J.; Chen, L. Growth inhibitory effect of paratocarpin E, a prenylated chalcone isolated from Euphorbia humifusa Wild. by induction of autophagy and apoptosis in human breast cancer cells. Bioorg. Chem. 2016, 69, 121–128. [Google Scholar] [CrossRef]
- Zhang, X.L.; Wang, B.B.; Mo, J.S. Puerarin 6″-O-xyloside possesses significant antitumor activities on colon cancer through inducing apoptosis. Oncol. Lett. 2018, 16, 5557–5564. [Google Scholar] [CrossRef]
- El Habbash, A.I.; Mohd Hashim, N.; Ibrahim, M.Y.; Yahayu, M.; Omer, F.A.E.; Abd Rahman, M.; Nordin, N.; Lian, G.E.C. In vitro assessment of anti-proliferative effect induced by alpha-mangostin from Cratoxylum arborescens on HeLa cells. PeerJ 2017, 5, e3460. [Google Scholar] [CrossRef]
- Seydi, E.; Rahimpour, Z.; Salimi, A.; Pourahmad, J. Selective toxicity of chrysin on mitochondria isolated from liver of a HCC rat model. Bioorg. Med. Chem. 2019, 27, 115163. [Google Scholar] [CrossRef]
- Khoo, B.Y.; Chua, S.L.; Balaram, P. Apoptotic effects of chrysin in human cancer cell lines. Int. J. Mol. Sci. 2010, 11, 2188–2199. [Google Scholar] [CrossRef]
- Su, C.H.; Kuo, C.L.; Lu, K.W.; Yu, F.S.; Ma, Y.S.; Yang, J.L.; Chu, Y.L.; Chueh, F.S.; Liu, K.C.; Chung, J.G. Fisetin-induced apoptosis of human oral cancer SCC-4 cells through reactive oxygen species production, endoplasmic reticulum stress, caspase-, and mitochondria-dependent signaling pathways. Environ. Toxicol. 2017, 32, 1725–1741. [Google Scholar] [CrossRef]
- Murtaza, I.; Adhami, V.M.; Hafeez, B.B.; Saleem, M.; Mukhtar, H. Fisetin, a natural flavonoid, targets chemoresistant human pancreatic cancer AsPC-1 cells through DR3-mediated inhibition of NF-κB. Int. J. Cancer 2009, 125, 2465–2473. [Google Scholar] [CrossRef]
- Pan, Q.; Xue, M.; Xiao, S.S.; Wan, Y.J.; Xu, D.B. A Combination Therapy with Baicalein and Taxol Promotes Mitochondria-Mediated Cell Apoptosis: Involving in Akt/beta-Catenin Signaling Pathway. DNA Cell Biol. 2016, 35, 646–656. [Google Scholar] [CrossRef] [PubMed]
- Kong, E.K.; Yu, S.; Sanderson, J.E.; Chen, K.B.; Huang, Y.; Yu, C.M. A novel anti-fibrotic agent, baicalein, for the treatment of myocardial fibrosis in spontaneously hypertensive rats. Eur. J. Pharmacol. 2011, 658, 175–181. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Yang, W.; Zhang, S.N.; Lu, J.B. Alpinetin inhibits lung cancer progression and elevates sensitization drug-resistant lung cancer cells to cis-diammined dichloridoplatium. Drug Des. Dev. Ther. 2015, 9, 6119–6127. [Google Scholar]
- Umehara, K.; Nemoto, K.; Kimijima, K.; Matsushita, A.; Terada, E.; Monthakantirat, O.; De-Eknamkul, W.; Miyase, T.; Warashina, T.; Degawa, M.; et al. Estrogenic constituents of the heartwood of Dalbergia parviflora. Phytochemistry 2008, 69, 546–552. [Google Scholar] [CrossRef] [PubMed]
- Makkar, H.P.S.; Siddhuraju, P.; Becker, K. Plant Secondary Metabolites; Walker, J.M., Ed.; Publisher: Stuttgart, Germany, 2007; Volume 393, pp. 1–122. [Google Scholar]
- Wang, Y.J.; Li, Q.; Xiao, H.B.; Li, Y.J.; Yang, Q.; Kan, X.X.; Chen, Y.; Liu, X.N.; Weng, X.G.; Chen, X.; et al. Chamaejasmin B exerts anti-MDR effect in vitro and in vivo via initiating mitochondria-dependant intrinsic apoptosis pathway. Drug Des. Dev. Ther. 2015, 9, 5301–5313. [Google Scholar] [CrossRef] [PubMed]
- Plitzko, B.; Kaweesa, E.N.; Loesgen, S. The natural product mensacarcin induces mitochondrial toxicity and apoptosis in melanoma cells. J. Biol. Chem. 2017, 292, 21102–21116. [Google Scholar] [CrossRef]
- Del Hierro, J.N.; Cueva, C.; Tamargo, A.; Nunez-Gomez, E.; Moreno-Arribas, M.V.; Reglero, G.; Martin, D. In Vitro Colonic Fermentation of Saponin-Rich Extracts from Quinoa, Lentil, and Fenugreek. Effect on Sapogenins Yield and Human Gut Microbiota. J. Agric. Food Chem. 2020, 68, 106–116. [Google Scholar] [CrossRef]
- Min, H.Y.; Jang, H.J.; Park, K.H.; Hyun, S.Y.; Park, S.J.; Kim, J.H.; Son, J.; Kang, S.S.; Lee, H.Y. The natural compound gracillin exerts potent antitumor activity by targeting mitochondrial complex II. Cell Death Dis. 2019, 10, 810. [Google Scholar] [CrossRef]
- Li, G.B.; Fu, R.Q.; Shen, H.M.; Zhou, J.; Hu, X.Y.; Liu, Y.X.; Li, Y.N.; Zhang, H.W.; Liu, X.; Zhang, Y.H.; et al. Polyphyllin I induces mitophagic and apoptotic cell death in human breast cancer cells by increasing mitochondrial PINK1 levels. Oncotarget 2017, 8, 10359–10374. [Google Scholar] [CrossRef]
- Chang, J.; Wang, H.; Wang, X.; Zhao, Y.; Zhao, D.; Wang, C.; Li, Y.; Yang, Z.; Lu, S.; Zeng, Q.; et al. Molecular mechanisms of Polyphyllin I-induced apoptosis and reversal of the epithelial-mesenchymal transition in human osteosarcoma cells. J. Ethnopharmacol. 2015, 170, 117–127. [Google Scholar] [CrossRef]
- Dyshlovoy, S.A.; Rast, S.; Hauschild, J.; Otte, K.; Alsdorf, W.H.; Madanchi, R.; Kalinin, V.I.; Silchenko, A.S.; Avilov, S.A.; Dierlamm, J.; et al. Frondoside A induces AIF-associated caspase-independent apoptosis in Burkitt lymphoma cells. Leuk. Lymphoma 2017, 58, 2905–2915. [Google Scholar] [CrossRef]
- Cheng, L.; Shi, L.; Wu, J.; Zhou, X.; Li, X.; Sun, X.; Zhu, L.; Xia, T.S.; Ding, Q. A hederagenin saponin isolated from Clematis ganpiniana induces apoptosis in breast cancer cells via the mitochondrial pathway. Oncol. Lett. 2018, 15, 1737–1743. [Google Scholar] [CrossRef] [PubMed]
- Song, I.S.; Jeong, Y.J.; Kim, J.; Seo, K.H.; Baek, N.I.; Kim, Y.; Kim, C.S.; Jang, S.W. Pharmacological inhibition of androgen receptor expression induces cell death in prostate cancer cells. Cell. Mol. Life Sci. 2020, 77, 4663–4673. [Google Scholar] [CrossRef]
- Vinh, L.B.; Nguyet, N.T.M.; Yang, S.Y.; Kim, J.H.; Thanh, N.V.; Cuong, N.X.; Nam, N.H.; Minh, C.V.; Hwang, I.; Kim, Y.H. Cytotoxic triterpene saponins from the mangrove Aegiceras corniculatum. Nat. Prod. Res. 2019, 33, 628–634. [Google Scholar] [CrossRef] [PubMed]
- Oh, J.M.; Kim, E.; Chun, S. Ginsenoside Compound K Induces Ros-Mediated Apoptosis and Autophagic Inhibition in Human Neuroblastoma Cells In Vitro and In Vivo. Int. J. Mol. Sci. 2019, 20, 4279. [Google Scholar] [CrossRef] [PubMed]
- Zou, K.; Li, Z.; Zhang, Y.; Zhang, H.Y.; Li, B.; Zhu, W.L.; Shi, J.Y.; Jia, Q.; Li, Y.M. Advances in the study of berberine and its derivatives: A focus on anti-inflammatory and anti-tumor effects in the digestive system. Acta Pharmacol. Sin. 2017, 38, 157–167. [Google Scholar] [CrossRef]
- Sun, Y.; Yu, J.; Liu, X.; Zhang, C.; Cao, J.; Li, G.; Liu, X.; Chen, Y.; Huang, H. Oncosis-like cell death is induced by berberine through ERK1/2-mediated impairment of mitochondrial aerobic respiration in gliomas. Biomed. Pharmacother. 2018, 102, 699–710. [Google Scholar] [CrossRef]
- Min, H.Y.; Jung, Y.; Park, K.H.; Lee, H.Y. Papuamine Inhibits Viability of Non-small Cell Lung Cancer Cells by Inducing Mitochondrial Dysfunction. Anticancer Res. 2020, 40, 323–333. [Google Scholar] [CrossRef]
- Wang, X.D.; Li, C.Y.; Jiang, M.M.; Li, D.; Wen, P.; Song, X.; Chen, J.D.; Guo, L.X.; Hu, X.P.; Li, G.Q.; et al. Induction of apoptosis in human leukemia cells through an intrinsic pathway by cathachunine, a unique alkaloid isolated from Catharanthus roseus. Phytomedicine 2016, 23, 641–653. [Google Scholar] [CrossRef]
- Rashmi, K.C.; Harsha Raj, M.; Paul, M.; Girish, K.S.; Salimath, B.P.; Aparna, H.S. A new pyrrole based small molecule from Tinospora cordifolia induces apoptosis in MDA-MB-231 breast cancer cells via ROS mediated mitochondrial damage and restoration of p53 activity. Chem. Biol. Interact. 2019, 299, 120–130. [Google Scholar] [CrossRef]
- Futamura, Y.; Muroi, M.; Aono, H.; Kawatani, M.; Hayashida, M.; Sekine, T.; Nogawa, T.; Osada, H. Bioenergetic and proteomic profiling to screen small molecule inhibitors that target cancer metabolisms. Biochim. Biophys. Acta Proteins Proteom. 2019, 1867, 28–37. [Google Scholar] [CrossRef] [PubMed]
- Torres, J.P.; Lin, Z.; Fenton, D.S.; Leavitt, L.U.; Niu, C.; Lam, P.Y.; Robes, J.M.; Peterson, R.T.; Concepcion, G.P.; Haygood, M.G.; et al. Boholamide A, an APD-Class, Hypoxia-Selective Cyclodepsipeptide. J. Nat. Prod. 2020, 83, 1249–1257. [Google Scholar] [CrossRef] [PubMed]
- Miranda, M.A.; Mondal, A.; Sachdeva, M.; Cabral, H.; Neto, Y.; Khan, I.; Groppo, M.; McChesney, J.D.; Bastos, J.K. Chemosensitizing Effect of Cernumidine Extracted from Solanum cernuum on Bladder Cancer Cells In Vitro. Chem. Biodivers. 2019, 16, e1900334. [Google Scholar] [CrossRef]
- Liu, W.Y.; Tang, Q.; Zhang, Q.; Hu, C.P.; Huang, J.B.; Sheng, F.F.; Liu, Y.L.; Zhou, M.; Lai, W.J.; Li, G.B.; et al. Lycorine Induces Mitochondria-Dependent Apoptosis in Hepatoblastoma HepG2 Cells Through ROCK1 Activation. Front. Pharmacol. 2019, 10, 651. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Huang, W.; Li, L.; Sun, X.; Song, S.; Xu, Q.; Zhang, L.; Wei, B.G.; Deng, X. Structure Determinants of Lagunamide A for Anticancer Activity and Its Molecular Mechanism of Mitochondrial Apoptosis. Mol. Pharm. 2016, 13, 3756–3763. [Google Scholar] [CrossRef]
- Wang, C.W.; Hsu, W.H.; Tai, C.J. Antimetastatic effects of cordycepin mediated by the inhibition of mitochondrial activity and estrogen-related receptor alpha in human ovarian carcinoma cells. Oncotarget 2017, 8, 3049–3058. [Google Scholar] [CrossRef]
- Kostova, I. Studying plant-derived coumarins for their pharmacological and therapeutic properties as potential anticancer drugs. Expert Opin. Drug Discov. 2007, 2, 1605–1618. [Google Scholar] [CrossRef]
- Venugopala, K.N.; Rashmi, V.; Odhav, B. Review on natural coumarin lead compounds for their pharmacological activity. Biomed. Res. Int. 2013, 2013, 963248. [Google Scholar] [CrossRef]
- Peng, X.M.; Damu, G.L.; Zhou, C. Current developments of coumarin compounds in medicinal chemistry. Curr. Pharm. Des. 2013, 19, 3884–3930. [Google Scholar] [CrossRef]
- Zhang, L.; Tong, X.; Zhang, J.; Huang, J.; Wang, J. DAW22, a natural sesquiterpene coumarin isolated from Ferula ferulaeoides (Steud.) Korov. that induces C6 glioma cell apoptosis and endoplasmic reticulum (ER) stress. Fitoterapia 2015, 103, 46–54. [Google Scholar] [CrossRef]
- Andas, A.R.; Abdul, A.B.; Rahman, H.S.; Sukari, M.A.; Abdelwahab, S.I.; Samad, N.A.; Anasamy, T.; Arbab, I.A. Dentatin from Clausena excavata Induces Apoptosis in HepG2 Cells via Mitochondrial Mediated Signaling. Asian Pac. J. Cancer Prev. 2015, 16, 4311–4316. [Google Scholar] [CrossRef]
- Arbab, I.A.; Abdul, A.B.; Sukari, M.A.; Abdullah, R.; Syam, S.; Kamalidehghan, B.; Ibrahim, M.Y.; Taha, M.M.; Abdelwahab, S.I.; Ali, H.M.; et al. Dentatin isolated from Clausena excavata induces apoptosis in MCF-7 cells through the intrinsic pathway with involvement of NF-κB signaling and G0/G1 cell cycle arrest: A bioassay-guided approach. J. Ethnopharmacol. 2013, 145, 343–354. [Google Scholar] [CrossRef] [PubMed]
- Gong, J.; Zhang, W.G.; Feng, X.F.; Shao, M.J.; Xing, C. Aesculetin (6,7-dihydroxycoumarin) exhibits potent and selective antitumor activity in human acute myeloid leukemia cells (THP-1) via induction of mitochondrial mediated apoptosis and cancer cell migration inhibition. J. BUON 2017, 22, 1563–1569. [Google Scholar] [PubMed]
- Grobarova, V.; Valis, K.; Talacko, P.; Pavlu, B.; Hernychova, L.; Novakova, J.; Stodulkova, E.; Flieger, M.; Novak, P.; Cerny, J. Quambalarine B, a Secondary Metabolite from Quambalaria cyanescens with Potential Anticancer Properties. J. Nat. Prod. 2016, 79, 2304–2314. [Google Scholar] [CrossRef]
- Valis, K.; Grobarova, V.; Hernychova, L.; Buganova, M.; Kavan, D.; Kalous, M.; Cerny, J.; Stodulkova, E.; Kuzma, M.; Flieger, M.; et al. Reprogramming of leukemic cell metabolism through the naphthoquinonic compound Quambalarine B. Oncotarget 2017, 8, 103137–103153. [Google Scholar] [CrossRef] [PubMed]
- Chao, C.C.; Hou, S.M.; Huang, C.C.; Hou, C.H.; Chen, P.C.; Liu, J.F. Plumbagin induces apoptosis in human osteosarcoma through ROS generation, endoplasmic reticulum stress and mitochondrial apoptosis pathway. Mol. Med. Rep. 2017, 16, 5480–5488. [Google Scholar] [CrossRef] [PubMed]
- Hou, Y.; Xu, J.; Liu, X.; Xia, X.; Li, N.; Bi, X. Shikonin induces apoptosis in the human gastric cancer cells HGC-27 through mitochondria-mediated pathway. Pharmacogn. Mag. 2015, 11, 250–256. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.; Zheng, Z.; Zhang, J.; Liu, X.; Liu, Y.; Yang, W.; Liu, Y.; Zhang, T.; Zhao, Y.; Liu, Y.; et al. Anticancer effect of 2,7-dihydroxy-3-methylanthraquinone on human gastric cancer SGC-7901 cells in vitro and in vivo. Pharm. Biol. 2016, 54, 285–292. [Google Scholar] [CrossRef]
- Feng, S.; Wang, Z.; Zhang, M.; Zhu, X.; Ren, Z. HG30, a tetrahydroanthraquinone compound isolated from the roots of Prismatomeris connate, induces apoptosis in human non-small cell lung cancer cells. Biomed. Pharmacother. 2018, 100, 124–131. [Google Scholar] [CrossRef]
- Zhang, M.; Du, H.; Huang, Z.; Zhang, P.; Yue, Y.; Wang, W.; Liu, W.; Zeng, J.; Ma, J.; Chen, G.; et al. Thymoquinone induces apoptosis in bladder cancer cell via endoplasmic reticulum stress-dependent mitochondrial pathway. Chem. Biol. Interact. 2018, 292, 65–75. [Google Scholar] [CrossRef]
- Jacobsen, K.M.; Villadsen, N.L.; Torring, T.; Nielsen, C.B.; Salomon, T.; Nielsen, M.M.; Tsakos, M.; Sibbersen, C.; Scavenius, C.; Nielsen, R.; et al. APD-Containing Cyclolipodepsipeptides Target Mitochondrial Function in Hypoxic Cancer Cells. Cell Chem. Biol. 2018, 25, 1337–1349.e12. [Google Scholar] [CrossRef] [PubMed]
- Nipin, S.P.; Kang, D.Y.; Kim, B.J.; Joung, Y.H.; Darvin, P.; Byun, H.J.; Kim, J.G.; Park, J.U.; Yang, Y.M. Methylsulfonylmethane Induces G1 Arrest and Mitochondrial Apoptosis in YD-38 Gingival Cancer Cells. Anticancer Res. 2017, 37, 1637–1646. [Google Scholar]
- Wang, Z.; Li, S.; Ren, R.; Li, J.; Cui, X. Recombinant Buckwheat Trypsin Inhibitor Induces Mitophagy by Directly Targeting Mitochondria and Causes Mitochondrial Dysfunction in Hep G2 Cells. J. Agric. Food Chem. 2015, 63, 7795–7804. [Google Scholar] [CrossRef] [PubMed]
- Liang, L.; Amin, A.; Cheung, W.Y.; Xu, R.; Yu, R.; Tang, J.; Yao, X.; Liang, C. Parameritannin A-2 from Urceola huaitingii enhances doxorubicin-induced mitochondria-dependent apoptosis by inhibiting the PI3K/Akt, ERK1/2 and p38 pathways in gastric cancer cells. Chem. Biol. Interact. 2020, 316, 108924. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.; Li, W.; Wang, R.; Nan, Y.; Wang, Q.; Liu, W.; Jin, F. Resveratrol enhanced anticancer effects of cisplatin on non-small cell lung cancer cell lines by inducing mitochondrial dysfunction and cell apoptosis. Int. J. Oncol. 2015, 47, 1460–1468. [Google Scholar] [CrossRef]
- Wang, B.; Liu, J.; Gong, Z. Resveratrol induces apoptosis in K562 cells via the regulation of mitochondrial signaling pathways. Int. J. Clin. Exp. Med. 2015, 8, 16926–16933. [Google Scholar]
- Wang, W.; Wu, J.; Zhang, Q.; Li, X.; Zhu, X.; Wang, Q.; Cao, S.; Du, L. Mitochondria-mediated apoptosis was induced by oleuropein in H1299 cells involving activation of p38 MAP kinase. J. Cell. Biochem. 2019, 120, 5480–5494. [Google Scholar] [CrossRef]
- Shamshoum, H.; Vlavcheski, F.; Tsiani, E. Anticancer effects of oleuropein. Biofactors 2017, 43, 517–528. [Google Scholar] [CrossRef]
- Ning, D.; Jin, M.; Xv, T.; Sun, J.; Li, M. Homoisoflavanone-1 isolated from Polygonatum odoratum arrests the cell cycle and induces apoptosis in A549 cells. Oncol. Lett. 2018, 16, 3545–3554. [Google Scholar] [CrossRef]
- Wang, R.; Ma, L.; Weng, D.; Yao, J.; Liu, X.; Jin, F. Gallic acid induces apoptosis and enhances the anticancer effects of cisplatin in human small cell lung cancer H446 cell line via the ROS-dependent mitochondrial apoptotic pathway. Oncol. Rep. 2016, 35, 3075–3083. [Google Scholar] [CrossRef]
- Freitas, S.; Martins, R.; Costa, M.; Leao, P.N.; Vitorino, R.; Vasconcelos, V.; Urbatzka, R. Hierridin B Isolated from a Marine Cyanobacterium Alters VDAC1, Mitochondrial Activity, and Cell Cycle Genes on HT-29 Colon Adenocarcinoma Cells. Mar. Drugs 2016, 14, 158. [Google Scholar] [CrossRef] [PubMed]
- Kitai, Y.; Zhang, X.; Hayashida, Y.; Kakehi, Y.; Tamura, H. Induction of G2/M arrest and apoptosis through mitochondria pathway by a dimer sesquiterpene lactone from Smallanthus sonchifolius in HeLa cells. J. Food Drug Anal. 2017, 25, 619–627. [Google Scholar] [CrossRef] [PubMed]





