
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Aneta Słabuszewska-Jóźwiak | + 1975 word(s) | 1975 | 2020-11-09 08:37:44 | | | |
| 2 | Vicky Zhou | Meta information modification | 1975 | 2020-11-16 09:34:13 | | |
Video Upload Options
Cesarean section is a surgical procedure, which is the most frequently performed in gynecology and obstetrics. It is commonly believed that an operative delivery is a less painful and safer mode of delivery, which translates into an increasing number of the procedures performed without medical indications.
The maternal sequelae of cesarean sections are well elucidated and widely discussed in the literature, while long-term neonatal consequences still remain the issue of research and scientific dispute.
1. Introduction
Cesarean section is the most common surgical procedure performed in women worldwide. Notably, a high percentage of surgical deliveries did not translate into reduced maternal or neonatal mortality [1][2][3][4][5]. According to the recommendations of the World Health Organization (WHO) the rates of cesarean sections should range between 10 and 15% [6]. However, the fear of labor pains and simultaneous concern about the baby to be born, are more and more commonly leading to women choosing to deliver by cesarean section [7][8]. The probability of complications secondary to the implemented procedure increases with the increasing percentage of cesarean sections. Globally, perinatal mortality rates reach 19 out of 1000 children [9]. As regards African countries—1 in 23 neonates dies as a consequence of cesarean section [10], while in developed countries it is believed that the procedure may prevent severe perinatal complications.
According to an increasing number of epidemiologic studies, children delivered by cesarean section more commonly developed respiratory and neurological disorders (e.g., autism spectrum disorders [11], schizophrenia [12]) and immune-related diseases, such as asthma [13][14], skin atopy [15], juvenile arthritis, coeliac disease [16], type 1 diabetes (T1D) [17] or obesity [1][18][19][20][21][22][23]. It is worth noting differences between the occurrence of the above-mentioned conditions in cases when the surgical procedure was performed after delivery had started.
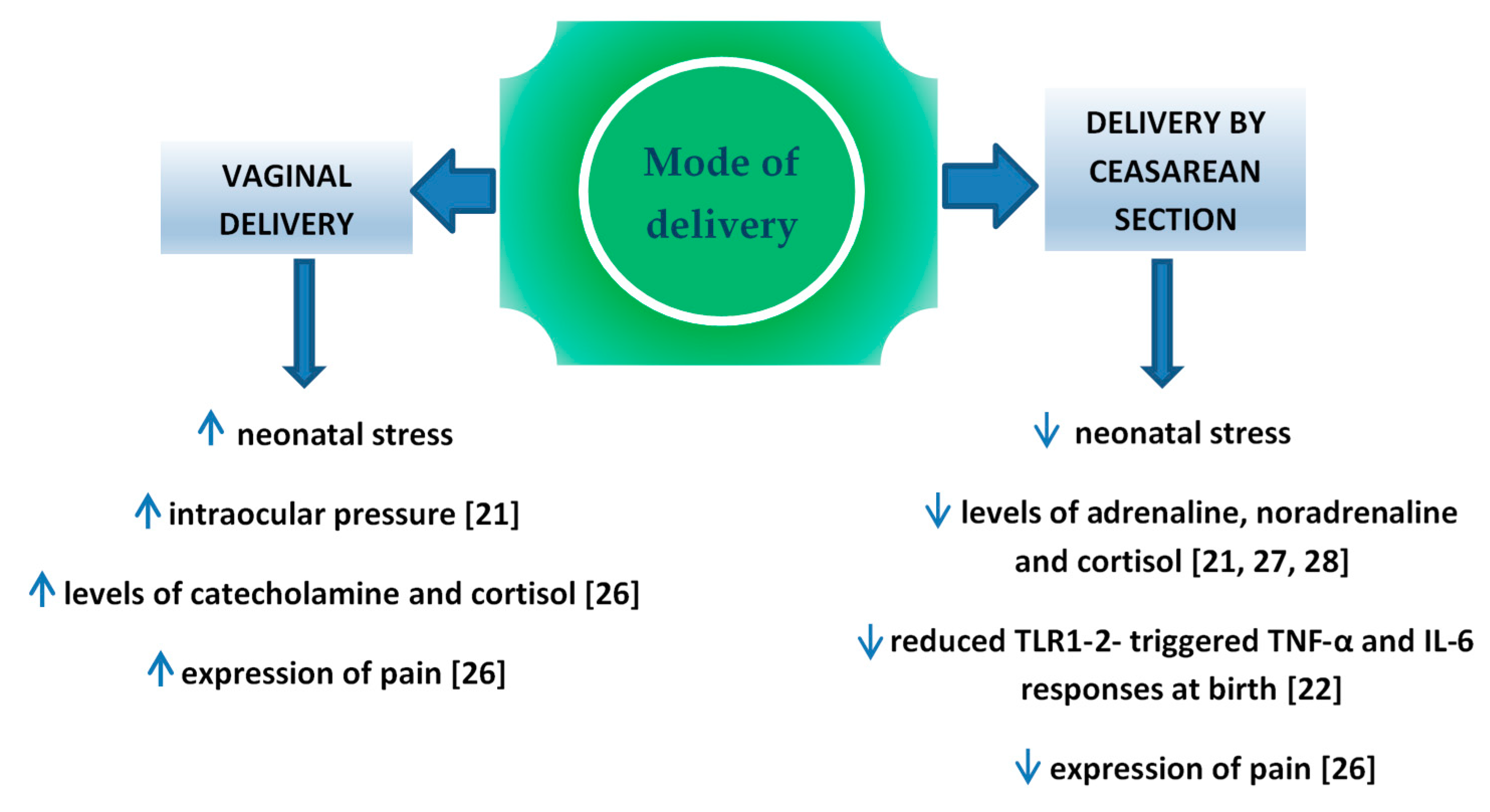
Perinatal stress leads to the increased production of catecholamine and cortisol in the infant’s blood [21]. It is important for the development of pulmonary maturity [24] and the adaptation of the circulatory system to extrauterine life [25]. The completion of pregnancy without associated neonatal stress (i.e., in case of an elective cesarean section) is a potential factor which may interfere in those processes. Schuller et al. claimed that neonates delivered vaginally were characterized by higher cortisol levels and presented higher expression of pain compared to children delivered by cesarean section [26]. In the case of infants delivered by cesarean section, the level of cortisol measured in the umbilical cord blood was significantly lower compared to vaginally delivered neonates [27][28][29] or to vacuum-assisted delivery [30], which might result in the increased percentage of adaptation complications, such as respiratory distress syndrome (RDS), persistent tachypnea or pulmonary hypertension which require hospitalization in the neonatal intensive care unit (NICU). In turn, prolonged NICU stay may be associated with a higher risk of the implementation of iatrogenic procedures [31][32] and longer total postnatal hospitalization. Delivery-related stress causes the activity of various cytokines, therefore changes in this process may have impact on the developing immune system. In the Taiwanese study, Liao et al. [22] reported also that TNF-α and IL-6 response toward TLR1–2 stimulation was significantly reduced in CS delivered neonates than in those delivered vaginally (Figure 1).
The disturbed colonization with bacterial microflora within the skin and digestive tract was the reason for a more frequent occurrence of immune diseases in children delivered by cesarean section [33]. The possibility of the transfer of those bacteria to the neonatal digestive tract is higher in case of long-term contact with the vaginal flora during vaginal delivery [34]. An increasing number of authors emphasized the contribution of maternal rectal microbiome to the optimal colonization of neonates [35][36]. The colonization is also promoted by pH in the stomach of neonates, which thanks to the swallowing of the amniotic fluid in utero, becomes neutral and provides conditions for the survival of aspirated bacteria. Lactobacillus, Bifidobacterium and Bacteroides are the dominant species of intestinal bacteria occurring at the early postnatal period in vaginally delivered neonates [34]. The bacteria play an important part in the regulation of the immune system [37] influencing the level of NK (natural killer) cells [38], regulating the population of T lymphocytes [39][40] the secretion of IgA antibodies [41], and the synthesis of proinflammatory cytokines [42][43][44]. Lactobacillus bacteria may prevent airway hyperresponsiveness by limiting the presence of inflammatory cells in the peribronchial tissue [45], while Bifidobacterium species prevents intestinal necrosis and plays a role in the regulation of the body weight of an infant [46][47]. Numerous authors emphasized both quantitative and qualitative differences in the intestinal microflora depending on the mode of delivery completion. It was reported that the meconium of neonates delivered by cesarean section included reduced amounts of Lactobacillus, Bifidobacterium, Bacteroides and Prevotella bacteria, while the dominant ones included iatrogenic bacterial species or ones which colonized the skin, i.e., Staphylococcus [34], Streptococcus [48], Klebsiella, Enterococcus and Clostridium [49]. Bifidobacterium species, responsible for the synthesis of short-chain fatty acids (SCFA), undergo a relatively rapid elimination from the digestive tract of neonates delivered by cesarean section [34]. SCFAs are a type of communicators between the microbiome and the immune system contributing to the maintenance of balance between pro- and anti-inflammatory reactions, e.g., by transferring the signal with a group of G protein-coupled receptors (GPR), which are present not only on the cells of the gastrointestinal system, but also of the immune and nervous system. It is believed that SCFAs may modulate the weight and reduce the amount of consumed food by the stimulation of enteroendocrine L cells responsible for releasing peptide YY (PYY) and glucagon-like peptide 1 (GLP-1) [50]. Therefore, they may influence the body weight of neonates. However, the role of bacterial microflora is still ambiguous in the development of type 1 diabetes [51].
Numerous authors of epidemiological studies emphasized a correlation between environmental factors influencing the fetus prenatally and over the early postnatal period and the development of circulatory system diseases, diabetes, obesity, tumors [52][53][54][55] and schizophrenia [56] in adulthood. The Epigenetic Impact of Childbirth (EPIIC) study demonstrated that the use of oxytocin, antibiotics and cesarean section may lead to long-term health implications [57]. The authors suspected that epigenetic mechanisms which influenced gene expression modification might be responsible for the phenomenon [57]. DNA methylation is a well elucidated epigenetic mechanism. It consists in adding a methyl group to cytosine-5-carbon in a reaction catalyzed by deoxyribonucleic acid methyltransferase (DNMT). CpG island hypermethylation within a gene promoter most commonly results in the reduction or inactivation of its expression [58][59]. Research showed that tobacco smoking, malnutrition, and long-lasting maternal stress during pregnancy might lead to silencing the expression of some genes in the fetus resulting in a variety of clinical consequences [60][61][62]. According to some researchers, cesarean section changed the global DNA methylation and the methylation of individual genes. Schlinzing et al. demonstrated a higher global methylation in the leukocytes of the umbilical cord blood in a group of elective cesarean sections [63] while Słabuszewska et al. showed a significantly lower global methylation of DNA in the placenta of women following a cesarean section [64]. Franz et al. found no differences in the global methylation of DNA between vaginal deliveries and cesarean sections, although the methylation of individual genes was significantly higher in neonates delivered by cesarean section [65]. Notably, the above mentioned studies vary in terms of methodology which may contribute to differences in the results. Therefore, the issue of the influence of cesarean section on changes in DNA methylation and its clinical implications still remains the subject of research.
2. Influence of Cesarean Section on Fetuses' Health
The stimulation of the hypothalamic-pituitary-adrenal axis (HPA) in the fetus leads to the increase in stress hormones contributing to lung maturation [66], thereby reducing the postnatal manifestations of respiratory insufficiency [67]. Another analysis of a multicenter World Health Organization Multicounty Survey on Maternal and Newborn Health (WHOMCS) revealed that cesarean section increased morbidity in neonates [68]. The most common complications listed by researchers include respiratory disorders, transient tachypnea or postpartum hypoglycemia [69][70]. The frequency of those complications was influenced by the co-existence of childbirth which significantly decreased the occurrence rates of such disorders [71][72].
In the case of an elective cesarean section, the risk of respiratory morbidity including transient tachypnea of the newborn (TTN), respiratory distress syndrome (RDS), and persistent pulmonary hypertension (PPH) at 37 weeks of gestation reached 10%, while with vaginal delivery the risk was 2.8% [73]. Previously, infants delivered by caesarean section were demonstrated to have significantly lower compliance of the respiratory system at the age of 1 year than those after vaginal delivery [22]. The authors however, did not observe differences in the resistance of the respiratory system and maximal expiratory flow at functional residual capacity between the groups depending on the type of delivery. Hansen et al. [71] reported that the percentage of complications was affected by cesarean section procedures and by the duration of the pregnancy. The risk of developing RDS after cesarean section at 37 gestational weeks increased 4-fold (odds ratio (OR) 3.9, 95% confidence interval 2.4 to 6.5), while at 39 weeks it was half lower (OR 1.9, 95% confidence interval (CI) 1.2 to 3.0) [71]. Similar conclusions were reached following an Israel study including 132,054 cases. Hospitalizations of offspring involving respiratory morbidity were significantly common in offspring delivered caesarean section (5.2% vs. 4.3% in vaginal deliveries [73]. Table 1 presents characteristics of studies regarding the impact of cesarean section on offspring respiratory morbidity. Currently, a prophylactic dose of corticosteroids is used in everyday clinical practice in order to avoid respiratory complications associated with elective cesarean section and premature delivery [74][75][76]. Numerous epidemiological studies demonstrated a correlation between cesarean section and an increased risk of developing immune diseases, including bronchial asthma [77][78][79][80], allergic rhinitis [81], ulcerative colitis, type 1 diabetes mellitus [17][82], celiac disease [83] and obesity [84][85].
Cesarean section is supposed to protect the neonate and its neurological consequences may differ depending on obstetric indications. In case of an elective cesarean section performed because of breech presentation or a fetal-pelvic disproportion both the mother and the fetus are subjected to limited stress unlike with intrapartum acute cesarean section. Animal research attracted attention to behavioral disorders of the offspring of females who had undergone cesarean section [86]. However, studies concerning children delivered by cesarean section demonstrated contradictory results, which assessed the prevalence of autism spectrum disorders (ASD), ADHD or behavioral disorders [87][88], especially if the analyses comprised the effect of environmental factors. A meta-analysis of the neurological consequences of surgical deliveries published in 2019 confirmed a higher risk of developing autism spectrum disorders (OR 1.33; 95% CI, 1.25–1.41) and ADHD (OR 1.17; 95% CI, 1.07–1.26) in children delivered by cesarean section. However, findings concerning intellectual deficits, obsessive-compulsive disorders, tics and eating disorders were not so explicit. The study based on 300 children at pre-school age reported that mode of delivery has no impact on IQ score [89]. In turn, the study performed in sizeable group of 5000 pre-school children showed that children delivered by caesarean section had significantly higher IQ test scores.
However, the authors observed no significant difference in IQ scores between caesarean delivery and natural vaginal delivery groups after adjusting of among others maternal and paternal education, maternal age and parity [90]. Zhang et al. [91] reported no correlation between the procedure of cesarean section and the risk of developing depression, affective and non-affective psychosis. On the other hand, Baumfeld et al. conducted a prospective cohort study and demonstrated that cesarean section was an independent factor affecting neurological disorders in children along with birth weight, maternal age, Apgar score, gestational age and the sex of the neonate [92]. Recent data by Sadowska et al. [93] demonstrated that delivery by cesarean section increased the risk of epilepsy over two-fold (OR 2.17) in the patients with cerebral palsy. Deoni et al. [94] demonstrated for the first time that caesarean section may be related with changes in brain development, at least during early infancy. The authors observed delivery mode-related differences in white matter development during infancy, which involved the frontal, temporal, and parietal lobes as well as corpus callosum. Children delivered by cesarean section showed significantly lower white matter development in widespread brain regions and simultaneously lower functional connectivity in the brain [95].
3. Summary
According to literature children delivered by cesarean section more commonly develop respiratory disorders and manifest the asthma, while the risk of developing obesity or neurological disorders is still under discussion.
References
- Betrán, A.P.; Ye, J.; Moller, A.-B.; Zhang, J.; Gülmezoglu, A.M.; Torloni, M.R. The Increasing Trend in Caesarean Section Rates: Global, Regional and National Estimates: 1990–2014. PLoS ONE 2016, 11, e0148343.
- Molina, G.; Weiser, T.G.; Lipsitz, S.R.; Esquivel, M.M.; Uribe-Leitz, T.; Azad, T.; Shah, N.; Semrau, K.; Berry, W.R.; Gawande, A.A.; et al. Relationship Between Cesarean Delivery Rate and Maternal and Neonatal Mortality. JAMA 2015, 314, 2263–2270.
- Betran, A.P.; Torloni, M.R.; Zhang, J.J.; Gulmezoglu, A.M. WHO Working Group on Caesarean Section WHO Statement on Caesarean Section Rates. BJOG Int. J. Obstet. Gynaecol. 2015, 123, 667–670.
- Ye, J.; Betran, A.P.; Vela, M.G.; Souza, J.P.; Zhang, J. Searching for the Optimal Rate of Medically Necessary Cesarean Delivery. Birth 2014, 41, 237–244.
- Betran, A.P.; Torloni, M.R.; Zhang, J.; Ye, J.; Mikolajczyk, R.; Deneux-Tharaux, C.; Oladapo, O.T.; Souza, J.P.; Tunçalp, Ö.; Vogel, J.P.; et al. What is the optimal rate of caesarean section at population level? A systematic review of ecologic studies. Reprod. Health 2015, 12, 1–10.
- WHO. World Health Organization. Available online: www.who.int/reproductivehealth/ (accessed on 6 September 2020).
- Wax, J.R.; Cartin, A.; Pinette, M.G.; Blackstone, J. Patient Choice Cesarean: An Evidence-Based Review. Obstet. Gynecol. Surv. 2004, 59, 601–616.
- Ecker, J. Elective Cesarean Delivery on Maternal Request. JAMA 2013, 309, 1930–1936.
- UN-IGME. Levels and Trends in Child Mortality: Report 2018. Estimates Developed by the UN Inter-Agency Group for Child Mortality Estimation. New York, NY: UN Children’s Fund. 2018. Available online: https://www.unicef.org/publications/files/Child_Mortality_Report_2018.pdf (accessed on 6 September 2020).
- Bishop, D.; Dyer, R.A.; Maswime, S.; Rodseth, R.N.; Van Dyk, D.; Kluyts, H.-L.; Tumukunde, J.T.; Madzimbamuto, F.D.; Elkhogia, A.M.; Ndonga, A.K.N.; et al. Maternal and neonatal outcomes after caesarean delivery in the African Surgical Outcomes Study: A 7-day prospective observational cohort study. Lancet Glob. Health 2019, 7, e513–e522.
- Curran, E.A.; Dalman, C.; Kearney, P.M.; Kenny, L.C.; Cryan, J.F.; Dinan, T.G.; Khashan, A.S. Association Between Obstetric Mode of Delivery and Autism Spectrum Disorder. JAMA Psychiatry 2015, 72, 935–942.
- O’Neill, S.M.; Curran, E.A.; Dalman, C.; Kenny, L.C.; Kearney, P.M.; Clarke, G.; Cryan, J.F.; Dinan, T.G.; Khashan, A.S. Birth by Caesarean Section and the Risk of Adult Psychosis: A Population-Based Cohort Study. Schizophr. Bull. 2015, 42, 633–641.
- Hyde, M.J.; Modi, N. The long-term effects of birth by caesarean section: The case for a randomised controlled trial. Early Hum. Dev. 2012, 88, 943–949.
- Thavagnanam, S.; Fleming, J.; Bromley, A.; Shields, M.; Cardwell, C. A meta-analysis of the association between Caesarean section and childhood asthma. Clin. Exp. Allergy 2008, 38, 629–633.
- Dahlen, H.G.; Downe, S.; Wright, M.L.; Kennedy, H.P.; Taylor, J.Y. Childbirth and consequent atopic disease: Emerging evidence on epigenetic effects based on the hygiene and EPIIC hypotheses. BMC Pregnancy Childbirth 2016, 16, 4.
- Decker, E.; Engelmann, G.; Findeisen, A.; Gerner, P.; Laass, M.; Ney, D.; Posovszky, C.; Hoy, L.; Hornef, M.W. Cesarean Delivery Is Associated With Celiac Disease but Not Inflammatory Bowel Disease in Children. Pediatrics 2010, 125, e1433–e1440.
- Cardwell, C.; Stene, L.C.; Joner, G.; Cinek, O.; Svensson, J.; Goldacre, M.J.; Parslow, R.C.; Pozzilli, P.; Brigis, G.; Stoyanov, D.; et al. Caesarean section is associated with an increased risk of childhood-onset type 1 diabetes mellitus: A meta-analysis of observational studies. Diabetologia 2008, 51, 726–735.
- Sevelsted, A.; Stokholm, J.; Bønnelykke, K.; Bisgaard, H. Cesarean Section and Chronic Immune Disorders. Obstet. Gynecol. Surv. 2015, 70, 303–305.
- Sinha, A.; Bewley, S. The harmful consequences of prelabour caesarean section on the baby. Obstet. Gynaecol. Reprod. Med. 2012, 22, 54–56.
- Kristensen, K.; Henriksen, L. Cesarean section and disease associated with immune function. J. Allergy Clin. Immunol. 2016, 137, 587–590.
- Elbay, A.; Celik, U.H.; Celik, B.; Ozer, O.F.; Kilic, G.; Akkan, J.C.U.; Bayraktar, B.T.; Kaymak, N.Z. Intraocular pressure in infants and its association with hormonal changes with vaginal birth versus cesarean section. Int. Ophthalmol. 2016, 36, 855–860.
- Liao, S.-L.; Tsai, M.-H.; Yao, T.-C.; Hua, M.-C.; Yeh, K.-W.; Chiu, C.-Y.; Su, K.-W.; Huang, S.-Y.; Kao, C.-C.; Lai, S.-H.; et al. Caesarean Section is associated with reduced perinatal cytokine response, increased risk of bacterial colonization in the airway, and infantile wheezing. Sci. Rep. 2017, 7, 1–8.
- Keag, O.E.; Norman, J.; Stock, S.J. Long-term risks and benefits associated with cesarean delivery for mother, baby, and subsequent pregnancies: Systematic review and meta-analysis. PLoS Med. 2018, 15, e1002494.
- Nemati, B.; Atmodjo, W.; Gagnon, S.; Humes, D.; McKerlie, C.; Kaplan, F.; Sweezey, N.B. Glucocorticoid receptor disruption delays structural maturation in the lungs of newborn mice. Pediatr. Pulmonol. 2007, 43, 125–133.
- Rog-Zielinska, E.A.; Thomson, A.; Kenyon, C.J.; Brownstein, D.G.; Moran, C.M.; Szumska, D.; Michailidou, Z.; Richardson, J.; Owen, E.; Watt, A.; et al. Glucocorticoid receptor is required for fetal heart maturation. Hum. Mol. Genet. 2013, 22, 3269–3282.
- Schuller, C.; Känel, N.; Müller, O.; Kind, A.B.; Tinner, E.M.; Hösli, I.; Zimmermann, R.; Surbek, D. Stress and pain response of neonates after spontaneous birth and vacuum-assisted and cesarean delivery. Am. J. Obstet. Gynecol. 2012, 207, 416.e1–416.e6.
- Mears, K.; McAuliffe, F.; Grimes, H.; Morrison, J.J. Fetal cortisol in relation to labour, intrapartum events and mode of delivery. J. Obstet. Gynaecol. 2004, 24, 129–132.
- Vogl, S.E.; Worda, C.; Egarter, C.; Bieglmayer, C.; Szekeres, T.; Huber, J.; Husslein, P. Mode of delivery is associated with maternal and fetal endocrine stress response. BJOG Int. J. Obstet. Gynaecol. 2006, 113, 441–445.
- Słabuszewska-Jóźwiak, A.; Włodarczyk, M.; Kilian, K.; Rogulski, Z.; Ciebiera, M.; Szymańska-Majchrzak, J.; Zaręba, K.; Szymański, J.K.; Raczkiewicz, D.; Włodarczyk, M.; et al. Does the Caesarean Section Impact on 11β HSD2 and Fetal Cortisol? Int. J. Environ. Res. Public Health 2020, 17, 5566.
- Sano, Y.; Doi, T.; Kikuchi, S.; Kawai, K.; Tanaka, M. Correlations between stress hormone levels in umbilical cord blood and duration of delivery. J Pak. Med. Assoc. 2015, 65, 782–784.
- Celebi, M.; Alan, S.; Kahvecioglu, D.; Cakir, U.; Yildiz, D.; Erdeve, O.; Arsan, S.; Atasay, B. Impact of Prophylactic Continuous Positive Airway Pressure on Transient Tachypnea of the Newborn and Neonatal Intensive Care Admission in Newborns Delivered by Elective Cesarean Section. Am. J. Perinatol. 2015, 33, 99–106.
- Gizzi, C.; Klifa, R.; Pattumelli, M.; Massenzi, L.; Taveira, M.; Shankar-Aguilera, S.; De Luca, D. Continuous Positive Airway Pressure and the Burden of Care for Transient Tachypnea of the Neonate: Retrospective Cohort Study. Am. J. Perinatol. 2015, 32, 939–943.
- Azad, M.B.; Konya, T.; Maughan, H.; Guttman, D.S.; Field, C.J.; Chari, R.S.; Sears, M.R.; Becker, A.B.; Scott, J.A.; Kozyrskyj, A.L. Child Study Investigators. Gut microbiota of healthy Canadian infants: Profiles by mode of delivery and infant diet at 4 months. CMAJ 2013, 185, 385–394.
- Shao, Y.; Forster, S.C.; Tsaliki, E.; Vervier, K.; Strang, A.; Simpson, N.; Kumar, N.; Stares, M.D.; Rodger, A.; Brocklehurst, P.; et al. Stunted microbiota and opportunistic pathogen colonization in caesarean-section birth. Nature 2019, 574, 117–121.
- Makino, H.; Kushiro, A.; Ishikawa, E.; Kubota, H.; Gawad, A.; Sakai, T.; Oishi, K.; Martin, R.; Ben-Amor, K.; Knol, J.; et al. Mother-to-Infant Transmission of Intestinal Bifidobacterial Strains Has an Impact on the Early Development of Vaginally Delivered Infant’s Microbiota. PLoS ONE 2013, 8, e78331.
- Sakwinska, O.; Foata, F.; Berger, B.; Brüssow, H.; Combremont, S.; Mercenier, A.; Dogra, S.; Soh, S.E.; Yen, J.; Heong, G.; et al. Does the maternal vaginal microbiota play a role in seeding the microbiota of neonatal gut and nose? Benef. Microbes 2017, 8, 763–778.
- Fujimura, K.E.; Slusher, N.A.; Cabana, M.D.; Lynch, S.V. Role of the gut microbiota in defining human health. Expert Rev. Anti- Infect. Ther. 2010, 8, 435–454.
- Mazmanian, S.K.; Liu, C.H.; Tzianabos, A.O.; Kasper, D.L. An Immunomodulatory Molecule of Symbiotic Bacteria Directs Maturation of the Host Immune System. Cell 2005, 122, 107–118.
- Olszak, T.; An, D.; Zeissig, S.; Vera, M.P.; Richter, J.; Franke, A.; Glickman, J.N.; Siebert, R.; Baron, R.M.; Kasper, D.L.; et al. Microbial Exposure During Early Life Has Persistent Effects on Natural Killer T Cell Function. Science 2012, 336, 489–493.
- Arpaia, N.; Campbell, C.; Fan, X.; Dikiy, S.; Van Der Veeken, J.; DeRoos, P.; Liu, H.; Cross, J.R.; Pfeffer, K.; Coffer, P.J.; et al. Metabolites produced by commensal bacteria promote peripheral regulatory T-cell generation. Nat. Cell Biol. 2013, 504, 451–455.
- Hapfelmeier, S.; Lawson, M.A.E.; Slack, E.; Kirundi, J.K.; Stoel, M.; Heikenwalder, M.; Cahenzli, J.; Velykoredko, Y.; Balmer, M.L.; Endt, K.; et al. Reversible Microbial Colonization of Germ-Free Mice Reveals the Dynamics of IgA Immune Responses. Science 2010, 328, 1705–1709.
- Schirmer, M.; Smeekens, S.P.; Vlamakis, H.; Jaeger, M.; Oosting, M.; Franzosa, E.A.; Ter Horst, R.; Jansen, T.; Jacobs, L.; Bonder, M.J.; et al. Linking the Human Gut Microbiome to Inflammatory Cytokine Production Capacity. Cell 2016, 167, 1897.
- Young, V.B. The intestinal microbiota in health and disease. Curr. Opin. Gastroenterol. 2012, 28, 63–69.
- Maynard, C.L.; Elson, C.O.; Hatton, R.D.; Weaver, C.T. Reciprocal interactions of the intestinal microbiota and immune system. Nat. Cell Biol. 2012, 489, 231–241.
- Yu, J.; Jang, S.-O.; Kim, B.-J.; Song, Y.-H.; Kwon, J.-W.; Kang, M.-J.; Choi, W.-A.; Jung, H.-D.; Hong, S.-J. The Effects of Lactobacillus rhamnosus on the Prevention of Asthma in a Murine Model. Allergy Asthma Immunol. Res. 2010, 2, 199–205.
- Lu, C.-Y.; Ni, Y.-H. Gut microbiota and the development of pediatric diseases. J. Gastroenterol. 2015, 50, 720–726.
- Luoto, R.; Kalliomäki, M.; Laitinen, K.; Delzenne, N.M.; Cani, P.D.; Salminen, S.; Isolauri, E. Initial Dietary and Microbiological Environments Deviate in Normal-weight Compared to Overweight Children at 10 Years of Age. J. Pediatr. Gastroenterol. Nutr. 2011, 52, 90–95.
- Akagawa, S.; Tsuji, S.; Onuma, C.; Akagawa, Y.; Yamaguchi, T.; Yamagishi, M.; Yamanouchi, S.; Kimata, T.; Sekiya, S.-I.; Ohashi, A.; et al. Effect of Delivery Mode and Nutrition on Gut Microbiota in Neonates. Ann. Nutr. Metab. 2019, 74, 132–139.
- Montoya-Williams, D.; Lemas, D.J.; Spiryda, L.; Patel, K.; Carney, O.O.; Neu, J.; Carson, T.L. The Neonatal Microbiome and Its Partial Role in Mediating the Association between Birth by Cesarean Section and Adverse Pediatric Outcomes. Neonatology 2018, 114, 103–111.
- Tan, J.; McKenzie, C.; Potamitis, M.; Thorburn, A.N.; Mackay, C.R.; Macia, L. The Role of Short-Chain Fatty Acids in Health and Disease. Adv. Immunol. 2014, 121, 91–119.
- Begum, M.; Pilkington, R.; Chittleborough, C.; Lynch, J.; Penno, M.; Smithers, L.G. Caesarean section and risk of type 1 diabetes: Whole-of-population study. Diabet. Med. 2019, 36, 1686–1693.
- Yajnik, C.S. Early Life Origins of Insulin Resistance and Type 2 Diabetes in India and Other Asian Countries. J. Nutr. 2004, 134, 205–210.
- Barker, D.J.P. The developmental origins of chronic adult disease. Acta Paediatr. 2004, 93, 26–33.
- Painter, R.C.; Roseboom, T.J.; Bleker, O.P. Prenatal exposure to the Dutch famine and disease in later life: An overview. Reprod. Toxicol. 2005, 20, 345–352.
- Gluckman, P.D.; Hanson, M.A.; Beedle, A.S. Early life events and their consequences for later disease: A life history and evolutionary perspective. Am. J. Hum. Biol. 2007, 19, 1–19.
- St Clair, D.; Xu, M.; Wang, P.; Yu, Y.; Fang, Y.; Zhang, F.; Zheng, X.; Gu, N.; Feng, G.; Sham, P.; et al. Rates of adult schizophrenia. Following prenatal exposure to the Chinese famine of 1959–1961. JAMA 2005, 294, 557–562.
- Dahlen, H.; Kennedy, H.; Anderson, C.; Bell, A.; Clark, A.; Foureur, M.; Ohm, J.; Shearman, A.; Taylor, J.; Wright, M.; et al. The EPIIC hypothesis: Intrapartum effects on the neonatal epigenome and consequent health outcomes. Med. Hypotheses 2013, 80, 656–662.
- Moarii, M.; Boeva, V.; Vert, J.-P.; Reyal, F. Changes in correlation between promoter methylation and gene expression in cancer. BMC Genom. 2015, 16, 1–14.
- Becket, E.; Chopra, S.; Duymich, C.E.; Lin, J.J.; You, J.S.; Pandiyan, K.; Nichols, P.W.; Siegmund, K.D.; Charlet, J.; Weisenberger, D.J.; et al. Identification of DNA Methylation–Independent Epigenetic Events Underlying Clear Cell Renal Cell Carcinoma. Cancer Res. 2016, 76, 1954–1964.
- Odom, L.N.; Taylor, H.S. Environmental induction of the fetal epigenome. Expert Rev. Obstet. Gynecol. 2010, 5, 657–664.
- Oberlander, T.F.; Weinberg, J.; Papsdorf, M.; Grunau, R.; Misri, S.; Devlin, A.M. Prenatal exposure to maternal depression, neonatal methylation of human glucocorticoid receptor gene (NR3C1) and infant cortisol stress responses. Epigenetics 2008, 3, 97–106.
- Breton, C.V.; Byun, H.-M.; Wenten, M.; Pan, F.; Yang, A.; Gilliland, F.D. Prenatal Tobacco Smoke Exposure Affects Global and Gene-specific DNA Methylation. Am. J. Respir. Crit. Care Med. 2009, 180, 462–467.
- Schlinzig, T.; Johansson, S.; Gunnar, A.; Ekström, T.J.; Norman, M. Epigenetic modulation at birth–altered DNA-methylation in white blood cells after Caesarean section. Acta Paediatr. 2009, 98, 1096–1099.
- Słabuszewska-Jóźwiak, A.; Włodarczyk, M.; Ciebiera, M.; Zwolińska, J.; Wojtyła, C.; Nowicka, G.; Jakiel, G.; Raczkiewicz, D. Placental DNA methylation in caesarean sections–A pilot study. Arch. Med. Sci. 2020, 16.
- Franz, M.B.; Poterauer, M.; Elhenicky, M.; Stary, S.; Birner, P.; Vinatzer, U.; Husslein, P.; Streubel, B.; Husslein, H. Global and single gene DNA methylation in umbilical cord blood cells after elective caesarean: A pilot study. Eur. J. Obstet. Gynecol. Reprod. Biol. 2014, 179, 121–124.
- Lagercrantz, H. The good stress of being born. Acta Paediatr. 2016, 105, 1413–1416.
- Tutdibi, E.; Gries, K.; Bücheler, M.; Misselwitz, B.; Schlosser, R.L.; Gortner, L. Impact of Labor on Outcomes in Transient Tachypnea of the Newborn: Population-Based Study. Pediatrics 2010, 125, e577–e583.
- Ganchimeg, T.; Nagata, C.; Vogel, J.P.; Morisaki, N.; Pileggi-Castro, C.; Ortiz-Panozo, E.; Jayaratne, K.; Mittal, S.; Ota, E.; Souza, J.P.; et al. Optimal Timing of Delivery among Low-Risk Women with Prior Caesarean Section: A Secondary Analysis of the WHO Multicountry Survey on Maternal and Newborn Health. PLoS ONE 2016, 11, e0149091.
- Wilmink, F.A.; Hukkelhoven, C.W.; Lunshof, S.; Mol, B.W.J.; Van Der Post, J.A.; Papatsonis, D.N. Neonatal outcome following elective cesarean section beyond 37 weeks of gestation: A 7-year retrospective analysis of a national registry. Am. J. Obstet. Gynecol. 2010, 202, 250.e1–250.e8.
- Tita, A.T.; Landon, M.B.; Spong, C.Y.; Lai, Y.; Leveno, K.J.; Varner, M.W.; Moawad, A.H.; Caritis, S.N.; Meis, P.J.; Wapner, R.J.; et al. Eunice Kennedy Shriver NICHD Maternal-Fetal Medicine Units Network. Timing of elective repeat cesarean delivery at term and neonatal out- comes. N. Engl. J. Med. 2009, 360, 111–120.
- Hansen, A.K.; Wisborg, K.; Uldbjerg, N.; Henriksen, T.B. Risk of respiratory morbidity in term infants delivered by elective caesarean section: Cohort study. BMJ 2007, 336, 85–87.
- Hefny, S.M.; Hashem, A.M.T.; Abdel-Razek, A.-R.A.; Ayad, S.M. The neonatal respiratory outcome in relation to timing of elective cesarean section at 38 versus 39week gestation: A single center based study. Egypt. Pediatr. Assoc. Gaz. 2013, 61, 78–82.
- Yael, B.; Walfisch, A.; Wainstock, T.; Segal, I.; Sergienko, R.; Landau, D.; Sheiner, E. Elective cesarean delivery at term and the long-term risk for respiratory morbidity of the offspring. Eur. J. Nucl. Med. Mol. Imaging 2018, 177, 1653–1659.
- Nada, A.M.; Shafeek, M.; El Maraghy, M.; Nageeb, A.; El-Din, A.S.S.; Awad, M.; Salaheldine, A. Antenatal corticosteroid administration before elective caesarean section at term to prevent neonatal respiratory morbidity: A randomized controlled trial. Eur. J. Obstet. Gynecol. Reprod. Biol. 2016, 199, 88–91.
- Sotiriadis, A.; Makrydimas, G.; Papatheodorou, S.; Ioannidis, J.P.; McGoldrick, E. Corticosteroids for preventing neonatal respiratory morbidity after elective caesarean section at term. Cochrane Database Syst. Rev. 2018, 8, CD006614.
- Roberts, D.; Brown, J.; Medley, N.; Dalziel, S.R. Antenatal corticosteroids for accelerating fetal lung maturation for women at risk of preterm birth. Cochrane Database Syst. Rev. 2017, 3, CD004454.
- Magnus, M.C.; Håberg, S.E.; Stigum, H.; Nafstad, P.; London, S.J.; Vangen, S.; Nystad, W. Delivery by Cesarean Section and Early Childhood Respiratory Symptoms and Disorders: The Norwegian Mother and Child Cohort Study. Am. J. Epidemiol. 2011, 174, 1275–1285.
- Almqvist, C.; Cnattingius, S.; Lichtenstein, P.; Lundholm, C. The impact of birth mode of delivery on childhood asthma and allergic diseases—A sibling study. Clin. Exp. Allergy 2012, 42, 1369–1376.
- Van Berkel, A.C.; Dekker, H.T.D.; Jaddoe, V.W.V.; Reiss, I.; Gaillard, R.; Hofman, A.; De Jongste, J.C.; Duijts, L. Mode of delivery and childhood fractional exhaled nitric oxide, interrupter resistance and asthma: The Generation R study. Pediatr. Allergy Immunol. 2015, 26, 330–336.
- Cho, C.E.; Norman, M. Cesarean section and development of the immune system in the offspring. Am. J. Obstet. Gynecol. 2013, 208, 249–254.
- Cotten, C.M.; Klebanoff, M.A.; Signore, C. Delivery after Previous Cesarean: Long-Term Outcomes in the Child. Semin. Perinatol. 2010, 34, 281–292.
- Khashan, A.S.; Kenny, L.C.; Lundholm, C.; Kearney, P.M.; Gong, T.; Almqvist, C. Mode of Obstetrical Delivery and Type 1 Diabetes: A Sibling Design Study. Pediatrics 2014, 134, e806–e813.
- Mårild, K.; Stephansson, O.; Montgomery, S.M.; Murray, J.A.; Ludvigsson, J.F. Pregnancy Outcome and Risk of Celiac Disease in Offspring: A Nationwide Case-Control Study. Gastroenterology 2012, 142, 39–45.e3.
- Ajslev, T.A.; Andersen, C.S.; Gamborg, M.; Sorensen, T.I.; Jess, T. Childhood overweight after establishment of the gut microbiota: The role of delivery mode, pre-pregnancy weight and early administration of antibiotics. Int. J. Obes. 2011, 35, 522–529.
- Li, H.-T.; Zhou, Y.-B.; Liu, J.-M. The impact of cesarean section on offspring overweight and obesity: A systematic review and meta-analysis. Int. J. Obes. 2012, 37, 893–899.
- Boksa, P.; El-Khodor, B. Birth insult interacts with stress at adulthood to alter dopaminergic function in animal models: Possible implications for schizophrenia and other disorders. Neurosci. Biobehav. Rev. 2003, 27, 91–101.
- Curran, E.A.; Khashan, A.S.; Dalman, C.; Kenny, L.C.; Cryan, J.F.; Dinan, T.G.; Kearney, P.M. Obstetric mode of delivery and attention-deficit/hyperactivity disorder: A sibling-matched study. Int. J. Epidemiol. 2016, 45, 532–542.
- Curran, E.A.; O’Neill, S.M.; Cryan, J.F.; Kenny, L.C.; Dinan, T.G.; Khashan, A.S.; Kearney, P.M. Research Review: Birth by caesarean section and development of autism spectrum disorder and attention-deficit/hyperactivity disorder: A systematic review and meta-analysis. J. Child Psychol. Psychiatry 2014, 56, 500–508.
- Hasab, M.F. Effect of Mode of Delivery on Children Intelligence Quotient at Pre-School Age in El-Minia City. Assiut Sci. Nurs. J. 2013, 1, 153–164.
- Khadem, N.; Khadivzadeh, T. The intelligence quotient of school aged children delivered by cesarean section and vaginal delivery. Iran. J. Nurs. Midwifery Res. 2010, 15, 135–140.
- Zhang, T.; Sidorchuk, A.; Sevilla-Cermeño, L.; Vilaplana-Pérez, A.; Chang, Z.; Larsson, H.; Mataix-Cols, D.; De La Cruz, L.F. Association of Cesarean Delivery With Risk of Neurodevelopmental and Psychiatric Disorders in the Offspring. JAMA Netw. Open 2019, 2, e1910236.
- Sheiner, E.; Wainstock, T.; Segal, I.; Sergienko, R.; Landau, D.; Walfisch, A.; Yael, B. Elective Cesarean Delivery at Term and the Long-Term Risk for Neurological Morbidity of the Offspring. Am. J. Perinatol. 2018, 35, 1038–1043.
- Sadowska, M.; Sarecka-Hujar, B.; Kopyta, I. Evaluation of Risk Factors for Epilepsy in Pediatric Patients with Cerebral Palsy. Brain Sci. 2020, 10, 481.
- Deoni, S.C.L.; Adams, S.H.; Li, X.; Badger, T.; Pivik, R.; Glasier, C.; Ramakrishnaiah, R.; Rowell, A.; Ou, X. Cesarean Delivery Impacts Infant Brain Development. Am. J. Neuroradiol. 2018, 40, 169–177.
- Macharey, G.; Toijonen, A.; Hinnenberg, P.; Gissler, M.; Heinonen, S.; Ziller, V. Term cesarean breech delivery in the first pregnancy is associated with an increased risk for maternal and neonatal morbidity in the subsequent delivery: A national cohort study. Arch. Gynecol. Obstet. 2020, 302, 85–91.




