
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Polly Roy | + 1890 word(s) | 1890 | 2020-07-02 11:09:48 | | | |
| 2 | Catherine Yang | Meta information modification | 1890 | 2020-08-10 04:09:24 | | |
Video Upload Options
Bluetongue virus (BTV) is an arthropod-borne virus infecting livestock. Its frequent emergence in Europe and North America had caused significant agricultural and economic loss. BTV is also of scientific interest as a model to understand the mechanisms underlying non-enveloped virus release from mammalian and insect cells. The BTV particle, which is formed of a complex double-layered capsid, was first considered as a lytic virus that needs to lyse the infected cells for cell to cell transmission. In the last decade however, a more in-depth focus on the role of the non-structural proteins has led to several examples where BTV particles are also released through different budding mechanisms at the plasma membrane. It is now clear that the non-structural protein NS3 is the main driver of BTV release, via different interactions with both viral and cellular proteins of the cell sorting and exocytosis pathway. Here are presented the main mechanisms that lead to lytic or non-lytic BTV release.
1. Introduction
Bluetongue virus (BTV) is an arthropod-borne virus vectored by biting midges from the Culicoides genus. BTV infects both domestic and wild ruminants, and the infection induces different variations of pathogenicity, from asymptomatic infection to severe symptoms such as ecchymosis, cardiac lesions, and hemorrhages, especially in sheep. Since the 1950s, BTV has spread globally from Africa to Europe, Asia, North and South America. Further, periodic outbreaks of new serotypes cause high morbidity in livestock, often with significant mortality and consequently associate with substantial economic losses in the agricultural industry.
BTV belongs to the Reoviridae family, which are characterised by a non-enveloped icosahedral capsid. The ten segments of the BTV double-stranded RNA genome encode seven structural proteins (VP1, VP2, VP3, VP4, VP5, VP6 and VP7) and six non-structural proteins (NS1, NS2, NS3, NS3A, NS4 and NS5) [1][2]. During infection, VP2 and VP5, which form the outer capsid, are responsible for BTV attachment and entry into cells via the endocytic pathway. Within early and late endosome, pH-induced conformational changes of VP2 and VP5 facilitate membrane penetration of the core [3], which is then released into the cytoplasm. The core is composed of an inner capsid (comprised of VP3 and VP7), containing the transcription complexes that are constituted by VP1 (RNA-dependent RNA polymerase), VP4 (capping enzyme), VP6 (helicase/RNA packaging) and the dsRNA segments [4][5]. The non-structural proteins fulfil essential functions within the virus life cycle, but do not form part of the virion. NS1 is necessary for BTV replication and selectively enhances viral protein synthesis [6]. It also undergoes polymerisation producing large tubules in infected cells, whose functional consequence is unknown [7]. NS2 is responsible, and sufficient, for the generation of viral inclusion bodies (VIBS), large globular structures in cytoplasm of infected cells. NS2 acts as a scaffold in the cytoplasm, recruiting positive sense ssRNA transcripts, the transcription complex components, and inner capsid proteins into VIBs. VIBs are, therefore, the sites of virus replication and are believed to be the site of viral core assembly, as inner capsid protein VP3 and VP7 have been found inside VIBs [8]. The outer capsid protein VP5 is also observed in the VIBs, suggesting that the first layer of the outer capsid could be assembled in the VIBs [9][10]. In contrast, the assembly of the VP2 protein does not happen in the VIBs [11], and the localisation of core maturation and VP2 assembly remains to be determined. However, it is likely that BTV uses the cell cytoskeleton for virus particles maturation, as it was previously demonstrated that VP2 is able to interact with the vimentin intermediate filaments [12]. The NS3 protein is the only glycoprotein containing membrane domains synthesised by BTV. NS3 is believed to be synthesised at the endoplasmic reticulum (ER), and transits through the Golgi apparatus, where it becomes glycosylated, before reaching the plasma membrane [13][14]. NS3 is the major regulatory protein of virus maturation, trafficking and egress [15][16][17][18]. Here, we review the recent advances made on the routes of BTV egress and discuss the different mechanisms, and the role played by NS3.
2. BTV Egress: Lytic Versus Non-Lytic Release
During BTV infection both lytic and non-lytic virion release has been documented. Further, electron microscopy analysis revealed different forms of virion particles, naked virion particles released by cell lysis and non-lytic viruses budding through the plasma membrane [19]. In addition to individual particle budding, a recent report demonstrated virion particles are released in aggregates within extracellular vesicles (EVs), suggesting a secretory pathway [17]. These suggest that BTV is capable of different modes of virion egress which result in the different forms of virus particle (Figure 1).
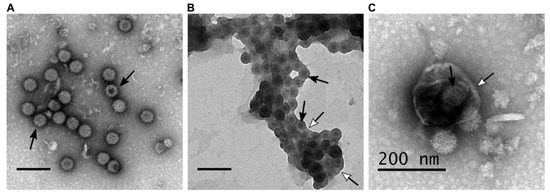
Figure 1. BTV particles purified from the supernatant of infected ovine PT cells (derived from domestic sheep) present different phenotypes. (A–C) Transmission electron microscopy of BTV virus particles that are (A) naked, (B) transiently associated with lipid membranes after budding at the plasma membrane, (C) or cloaked in EVs. Scale bars represent 200 nm, white head arrows indicate the lipid membranes, and black arrows indicate BTV particles.
Classically, non-enveloped viruses were thought to rely on lytic strategies for release, killing the infected cell in the process. In that regard, it has been shown that infection by several viruses from the Reoviridae family trigger cellular apoptosis [20][21][22][23][24]. In particular, BTV infection induces nuclear factor kappa B (NF-κB), caspase-3, DNA fragmentation and membrane disruption [22]. In mammalian cells, the expression of outer capsid proteins alone is sufficient to trigger apoptosis [25]. The mitogen activated protein kinase/extracellular signal-regulated kinase (MAPK/ERK) pathway, a key mediator of apoptosis, can be modulated by BTV in a strain-dependent manner [26][27]. Along with VP2, NS3 is a major determinant of BTV virulence [28]. Recently, it has been shown that NS3 is responsible for the activation of the MAPK/ERK pathway by interacting with the serine/threonine-protein kinase B-Raf. Although beneficial for the virus replication, this phenomenon is likely to contribute to increased expression of pro-inflammatory factors that would cause tissue damage [26].
In addition to its role in inducing apoptosis, it has also been suggested that an oligomer of NS3 possess a viroporin-like activity [29], which could induce plasma membrane permeation, similar to observations on the rotavirus NSP4 protein [30]. Indeed, rotavirus viroporin induces an increase of cytosolic calcium concentration, that is required for the formation of vesicular puncta surrounding the sites of replication. To form this viroporin, NS3 is likely to form oligomers, possibly through a predicted coiled-coil motif located in its N-terminus. However, the presence of NS3 dimers have only been identified in purified NS3 expressed in bacteria [31] and remains to be confirmed in infected eukaryotic cells.
Overall, BTV-induced apoptosis and activation of the pro-inflammatory pathways are the main events leading to BTV lytic release from infected mammalian cells. When infected with BTV in vitro, almost all mammalian cells lines, such as PT cell line (derived from sheep) or BSR cell line (derived from BHK-21 cells from hamster), are lysed after 1 to 4 days post infection, depending on the initial multiplicity of infection. However, several studies on NS3 mutant viruses revealed that when non-lytic release of BTV is inhibited, BTV is still able to spread from cell to cell but is highly attenuated, suggesting that lytic release is not the main mode of BTV egress [17][18][32].
3. BTV Non-Lytic Release in Insects
Much of the work discussed above has focused on the egress of BTV from mammalian cells. BTV is transmitted to ruminants by biting midges from the Culicoides species, and establishes a persistent, non-lethal infection in the vector species. A similar scenario is visible in KC cells, a Culicoides derived cell line; BTV replicates to a high titre in these cells but does not induce cytopathic effect. This suggests virion particles are released from insect cells by a non-lytic mechanism [33]. In contrast to mammalian cells, Culicoides vector cells respond to BTV infection using different mechanisms involving apoptotic pathway inhibition and RNA interference [34][35]. Here, virus budding is the norm for egress, a situation which is mirrored by the requirement of NS3 for non-lytic release of recombinant VLPs produced in Spodoptera frugiperda (caterpillar) insect cells using recombinant baculoviruses. VLPs release could only be achieved by co-expression of NS3/3A [15]. Additionally, NS3/3A is essential for the propagation of BTV in vector species, as knockout of this gene was found to inhibit infection in Culicoides sonorensis [36]. As in mammalian cells, budding of BTV from insect cells is dependent on the interaction of NS3 with Annexin II. Of note is that both NS3 and NS3A isoforms are required for efficient virus release from insect cells [16][37]. In contrast to the infection of mammalian cells, the outer capsid of BTV seems less important during the infection of insect cells. It has been reported that infectious subviral particles (ISVP, mature particles treated with trypsin) are more infectious than mature particles in insect cells, both in cell culture and in Culicoides vectors [38]. In insects, it was shown that the VP2 protein of BTV particles ingested by Culicoides vectors is cleaved by a trypsin-like protease present in the insect saliva [39]. BTV particles treated with insect saliva were more infectious in Culicoides derived KC cells and less infectious in mammalian cells than non-treated virus particles. It was also shown that the core protein VP7 can bind KC cells surface [40], suggesting BTV entry into insect cells is independent of the outer capsid. In support of these findings, analysis of Culicoides captured in nature revealed that segments 3 and 7, encoding the core proteins VP3 and VP7, were present in 30% of field-collected insects, whereas segment 2, encoding the VP2 protein, was detected in only 16% of the collected insects [41]. Altogether, these results suggest that outer capsid proteins may not be necessary and are downregulated during infection of the Culicoides vectors.
4. Conclusions
In recent decades, extensive study of the non-structural proteins of BTV, and of the infection of mammalian and insect cells has significantly enhanced our understanding on virus replication processes. Among the key discoveries, the predominantly non-lytic release of BTV, the importance of the NS3 in mediating virus egress, and the relative absence of a viral envelope represent a shift of the egress paradigm, from lytic to non-lytic. However, several challenges remain to further understand how BTV and other arthropod-borne Orbiviruses can infect both animals and their insect vectors, with striking variations in the severity of symptoms. Further characterization of the NS3 molecular structure in a lipid environment represents a clear future goal, as this protein is central in host-pathogen interaction. Such structural information would help to understand how NS3 functions as a viroporin while mediating virus maturation and egress. Additionally, although the NS1 protein is known to upregulate viral protein synthesis, NS1 polymer tubules has been hypothesized to play a role in particle trafficking and egress [42]. Thus, the spatio-temporal co-ordination of NS1’s function with that of NS3 needs to be explored further. It is also not clear how NS2 and NS3 communicate to promote virus particle maturation. No doubt the availability of various high-resolution microscopic techniques will be able to address these issues in the near future. Similarly, the role of lipids in BTV infection and replication process offers an interesting subject to be investigated, as it has been observed that in addition to NS3, VP5 possess an independent lipid raft targeting signal, similar to the VP4 and NSP4 proteins of rotavirus [43][44]. Future studies addressing these questions will help to better understand the BTV life cycle in cells, as well as the pathogenicity and ecological cycles among alternating hosts.
References
- Ratinier, M.; Caporale, M.; Golder, M.; Franzoni, G.; Allan, K.; Nunes, S.F.; Armezzani, A.; Bayoumy, A.; Rixon, F.; Shaw, A.; et al. Identification and Characterization of a Novel Non-Structural Protein of Bluetongue Virus. PLOS Pathog. 2011, 7, e1002477.
- Stewart, M.; Hardy, A.; Barry, G.; Pinto, R.M.; Caporale, M.; Melzi, E.; Hughes, J.; Taggart, A.; Janowicz, A.; Varela, M.; et al. Characterization of a second open reading frame in genome segment 10 of bluetongue virus. J. Gen. Virol. 2015, 96, 3280–3293.
- Wu, W.; Celma, C.C.; Kerviel, A.; Roy, P. Mapping the pH Sensors Critical for Host Cell Entry by a Complex Nonenveloped Virus. J. Virol. 2019, 93, e01897-18.
- Lourenco, S.; Roy, P. In vitro reconstitution of Bluetongue virus infectious cores. Proc. Natl. Acad. Sci. USA 2011, 108, 13746–13751.
- Roy, P. Bluetongue virus structure and assembly. Curr. Opin. Virol. 2017, 24, 115–123.
- Boyce, M.; Celma, C.C.P.; Roy, P. Bluetongue virus non-structural protein 1 is a positive regulator of viral protein synthesis. Virol. J. 2012, 9, 178.
- Kerviel, A.; Ge, P.; Lai, M.; Jih, J.; Boyce, M.; Zhang, X.; Zhou, Z.H.; Roy, P. Atomic structure of the translation regulatory protein NS1 of bluetongue virus. Nat. Microbiol. 2019, 4, 837–845.
- Hyatt, A.D.; Eaton, B.T. Ultrastructural Distribution of the Major Capsid Proteins within Bluetongue Virus and Infected Cells. J. Gen. Virol. 1988, 69, 805–815.
- Mohl, B.-P.; Kerviel, A.; Labadie, T.; Matsuo, E.; Roy, P. Differential Localization of Structural and Non-Structural Proteins during the Bluetongue Virus Replication Cycle. Viruses 2020, 12, 343.
- Brookes, S.M.; Hyatt, A.D.; Eaton, B.T. Characterization of virus inclusion bodies in bluetongue virus-infected cells. J. Gen. Virol. 1993, 74, 525–530.
- Gould, A.R.; Hyatt, A.D.; Eaton, B.T. Morphogenesis of a bluetongue virus variant with an amino acid alteration at a neutralization site in the outer coat protein, VP2. Virology 1988, 165, 23–32.
- Bhattacharya, B.; Noad, R.J.; Roy, P. Interaction between Bluetongue virus outer capsid protein VP2 and vimentin is necessary for virus egress. Virol. J. 2007, 4, 1–12.
- Wu, X.; Chen, S.Y.; Iwata, H.; Compans, R.W.; Roy, P. Multiple glycoproteins synthesized by the smallest RNA segment (S10) of bluetongue virus. J. Virol. 1992, 66, 7104–7112.
- Bansal, O.B.; Stokes, A.; Bansal, A.; Bishop, D.; Roy, P. Membrane Organization of Bluetongue Virus Nonstructural Glycoprotein NS3. J. Virol. 1998, 72, 3362–3369.
- Hyatt, A.D.; Zhao, Y.; Roy, P. Release of Bluetongue Virus-like Particles from Insect Cells is Mediated by BTV Nonstructural Protein NS3/NS3A. Virology 1993, 193, 592–603.
- Beaton, A.R.; Rodriguez, J.; Reddy, Y.K.; Roy, P. The membrane trafficking protein calpactin forms a complex with bluetongue virus protein NS3 and mediates virus release. Proc. Natl. Acad. Sci. USA 2002, 99, 13154–13159.
- Celma, C.C.P.; Roy, P. A Viral Nonstructural Protein Regulates Bluetongue Virus Trafficking and Release. J. Virol. 2009, 83, 6806–6816.
- Labadie, T.; Jegouic, S.; Roy, P. Bluetongue Virus Nonstructural Protein 3 Orchestrates Virus Maturation and Drives Non-Lytic Egress via Two Polybasic Motifs. Viruses 2019, 11, 1107.
- Labadie, T.; Roy, P. A non-enveloped arbovirus released in secretory lysosomes induces super-infection exclusion. bioRxiv 2020.
- Clarke, P.; Meintzer, S.M.; Gibson, S.; Widmann, C.; Garrington, T.P.; Johnson, G.L.; Tyler, K.L. Reovirus-Induced Apoptosis Is Mediated by TRAIL. J. Virol. 2000, 74, 8135–8139.
- Tyler, K.L.; Squier, M.K.; Rodgers, S.E.; Schneider, B.E.; Oberhaus, S.M.; Grdina, T.A.; Cohen, J.J.; Dermody, T.S. Differences in the capacity of reovirus strains to induce apoptosis are determined by the viral attachment protein sigma 1. J. Virol. 1995, 69, 6972–6979.
- DeMaula, C.D.; Jutila, M.A.; Wilson, D.W.; MacLachlan, N.J. Infection kinetics, prostacyclin release and cytokine-mediated modulation of the mechanism of cell death during bluetongue virus infection of cultured ovine and bovine pulmonary artery and lung microvascular endothelial cells. J. Gen. Virol. 2001, 82, 787–794.
- Chaïbi, C.; Cotte-Laffitte, J.; Sandré, C.; Esclatine, A.; Servin, A.L.; Quéro, A.-M.; Géniteau-Legendre, M. Rotavirus induces apoptosis in fully differentiated human intestinal Caco-2 cells. Virology 2005, 332, 480–490.
- Mortola, E.; Larsen, A. Bluetongue virus infection: Activation of the MAP kinase-dependent pathway is required for apoptosis. Res. Vet. Sci. 2010, 89, 460–464.
- Mortola, E.; Noad, R.; Roy, P. Bluetongue Virus Outer Capsid Proteins Are Sufficient To Trigger Apoptosis in Mammalian Cells. J. Virol. 2004, 78, 2875–2883.
- Kundlacz, C.; Pourcelot, M.; Fablet, A.; Moraes, R.A.D.S.; Léger, T.; Morlet, B.; Viarouge, C.; Sailleau, C.; Turpaud, M.; Gorlier, A.; et al. Novel Function of Bluetongue Virus NS3 Protein in Regulation of the MAPK/ERK Signaling Pathway. J. Virol. 2019, 93, e0036–19.
- Mohl, B.-P.; Emmott, E.; Roy, P. Phosphoproteomic Analysis Reveals the Importance of Kinase Regulation During Orbivirus Infection. Mol. Cell. Proteom. 2017, 16, 1990–2005.
- Janowicz, A.; Caporale, M.; Shaw, A.; Gulletta, S.; Di Gialleonardo, L.; Ratinier, M.; Palmarini, M. Multiple genome segments determine virulence of bluetongue virus serotype 8. J. Virol. 2015, 89, 5238–5249.
- Han, Z.; Harty, R.N. The NS3 Protein of Bluetongue Virus Exhibits Viroporin-like Properties. J. Biol. Chem. 2004, 279, 43092–43097.
- Hyser, J.M.; Collinson-Pautz, M.R.; Utama, B.; Estes, M.K. Rotavirus Disrupts Calcium Homeostasis by NSP4 Viroporin Activity. MBio 2010, 1, e00265–10.
- Chacko, N.; Mohanty, N.N.; Biswas, S.K.; Chand, K.; Yogisharadhya, R.; Pandey, A.B.; Mondal, B.; Shivachandra, S.B. A coiled-coil motif in non-structural protein 3 (NS3) of bluetongue virus forms an oligomer. Virus Genes 2015, 51, 244–251.
- Van Gennip, R.G.P.; van de Water, S.G.P.; van Rijn, P.A. Bluetongue Virus Nonstructural Protein NS3/NS3a Is Not Essential for Virus Replication. PLoS ONE 2014, 9, e85788.
- Mellor, P.S. Infection of the vectors and bluetongue epidemiology in Europe. Vet. Ital. 2004, 40, 167–174.
- Vermaak, E.; Maree, F.F.; Theron, J. The Culicoides sonorensis inhibitor of apoptosis 1 protein protects mammalian cells from apoptosis induced by infection with African horse sickness virus and bluetongue virus. Virus Res. 2017, 232, 152–161.
- Schnettler, E.; Ratinier, M.; Watson, M.; Shaw, A.E.; McFarlane, M.; Varela, M.; Elliott, R.M.; Palmarini, M.; Kohl, A. RNA Interference Targets Arbovirus Replication in Culicoides Cells. J. Virol. 2013, 87, 2441–2454.
- Feenstra, F.; Drolet, B.S.; Boonstra, J.; van Rijn, P.A. Non-structural protein NS3/NS3a is required for propagation of bluetongue virus in Culicoides sonorensis. Parasites Vectors 2015, 8, 1–9.
- Saxena, V.; Lai, C.-K.; Chao, T.-C.; Jeng, K.-S.; Lai, M.M.C. Annexin A2 Is Involved in the Formation of Hepatitis C Virus Replication Complex on the Lipid Raft. J. Virol. 2012, 86, 4139–4150.
- Mertens, P.P.C.; Burroughs, J.N.; Walton, A.; Wellby, M.P.; Fu, H.; O’hara, R.S.; Brookes, S.M.; Mellor, P.S. Enhanced Infectivity of Modified Bluetongue Virus Particles for Two Insect Cell Lines and for TwoCulicoidesVector Species. Virology 1996, 217, 582–593.
- Darpel, K.E.; Langner, K.F.A.; Nimtz, M.; Anthony, S.J.; Brownlie, J.; Takamatsu, H.-H.; Mellor, P.S.; Mertens, P.P.C. Saliva Proteins of Vector Culicoides Modify Structure and Infectivity of Bluetongue Virus Particles. PLoS ONE 2011, 6, e17545.
- Tan, B.-H.; Nason, E.; Staeuber, N.; Jiang, W.; Monastryrskaya, K.; Roy, P. RGD Tripeptide of Bluetongue Virus VP7 Protein Is Responsible for Core Attachment to Culicoides Cells. J. Virol. 2001, 75, 3937–3947.
- White, D.M.; Wilson, W.C.; Blair, C.D.; Beaty, B.J. Studies on overwintering of bluetongue viruses in insects. J. Gen. Virol. 2005, 86, 453–462.
- Owens, R.J.; Limn, C.; Roy, P. Role of an Arbovirus Nonstructural Protein in Cellular Pathogenesis and Virus Release. J. Virol. 2004, 78, 6649–6656.
- Bhattacharya, B.; Roy, P. Bluetongue Virus Outer Capsid Protein VP5 Interacts with Membrane Lipid Rafts via a SNARE Domain. J. Virol. 2008, 82, 10600–10612.
- Sapin, C.; Colard, O.; Delmas, O.; Tessier, C.; Breton, M.; Enouf, V.; Chwetzoff, S.; Ouanich, J.; Cohen, J.; Wolf, C.; et al. Rafts Promote Assembly and Atypical Targeting of a Nonenveloped Virus, Rotavirus, in Caco-2 Cells. J. Virol. 2002, 76, 4591–4602.




