
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Catherine Kier | + 877 word(s) | 877 | 2021-11-16 07:42:12 | | | |
| 2 | Conner Chen | Meta information modification | 877 | 2021-11-29 07:42:35 | | |
Video Upload Options
Obstructive sleep apnea (OSA) is a form of sleep-disordered breathing consisting of upper airway collapse during sleep that results in repetitive arousals and desaturations. Estimates of disease prevalence range between 1% and 3% of the general pediatric population, with significant associated clinical consequences on individual morbidity. Consequences of untreated pediatric OSA include metabolic, endocrine, cardiovascular, and neurobehavioral implications with potential for long-term consequences. However, many pathogenic features of this syndrome in children remain unclear. Understanding principal mechanisms of OSA is critical for ultimately allowing for the development of therapeutic strategies. Much of the pathogenic mechanisms of OSA currently center around upper airway collapsibility.
1. Critical Pressure
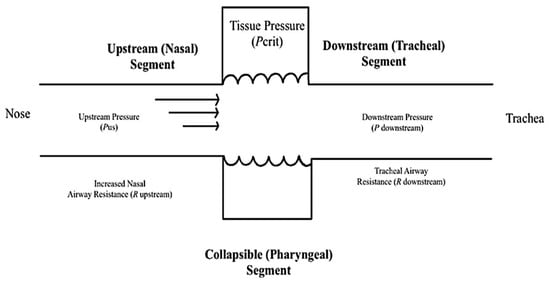
2. Oscillatory Patterns
References
- Sforza, E.; Petiau, C.; Weiss, T.; Thibault, A.; Krieger, J. Pharyngeal critical pressure in patients with obstructive sleep apnea syndrome. Clinical implications. Am. J. Respir. Crit. Care Med. 1999, 159, 149–157.
- Gold, A.R.; Schwartz, A.R. The pharyngeal critical pressure. The whys and hows of using nasal continuous positive airway pressure diagnostically. Chest 1996, 110, 1077–1088.
- Schwartz, A.R.; Smith, P.L. CrossTalk proposal: The human upper airway does behave like a Starling resistor during sleep. J. Physiol. 2013, 591, 2229–2232.
- Pham, L.V.; Schwartz, A.R. The pathogenesis of obstructive sleep apnea. J. Thorac. Dis. 2015, 7, 1358–1372.
- Wellman, A.; Genta, P.R.; Owens, R.L.; Edwards, B.A.; Sands, S.A.; Loring, S.H.; White, D.P.; Jackson, A.C.; Pedersen, O.F.; Butler, J.P. Test of the Starling resistor model in the human upper airway during sleep. J. Appl. Physiol. 2014, 117, 1478–1485.
- Schwartz, A.R.; Smith, P.L.; Wise, R.A.; Gold, A.R.; Permutt, S. Induction of upper airway occlusion in sleeping individuals with subatmospheric nasal pressure. J. Appl. Physiol. 1988, 64, 535–542.
- Katz, E.S.; D’Ambrosio, C.M. Pathophysiology of pediatric obstructive sleep apnea. Proc. Am. Thorac. Soc. 2008, 5, 253–262.
- Brockbank, J.C. Update on pathophysiology and treatment of childhood obstructive sleep apnea syndrome. Paediatr. Respir. Rev. 2017, 24, 21–23.
- Marcus, C.L.; McColley, S.A.; Carroll, J.L.; Loughlin, G.M.; Smith, P.L.; Schwartz, A.R. Upper airway collapsibility in children with obstructive sleep apnea syndrome. J. Appl. Physiol. 1994, 77, 918–924.
- Gold, A.R.; Marcus, C.L.; Dipalo, F.; Gold, M.S. Upper airway collapsibility during sleep in upper airway resistance syndrome. Chest 2002, 121, 1531–1540.
- Marcus, C.L.; Katz, E.S.; Lutz, J.; Black, C.A.; Galster, P.; Carson, K.A. Upper airway dynamic responses in children with the obstructive sleep apnea syndrome. Pediatr. Res. 2005, 57, 99–107.
- Carberry, J.C.; Jordan, A.S.; White, D.P.; Wellman, A.; Eckert, D.J. Upper Airway Collapsibility (Pcrit) and Pharyngeal Dilator Muscle Activity are Sleep Stage Dependent. Sleep 2016, 39, 511–521.
- Zhi, Y.X.; Vena, D.; Popovic, M.R.; Bradley, T.D.; Yadollahi, A. Detecting inspiratory flow limitation with temporal features of nasal airflow. Sleep Med. 2018, 48, 70–78.
- Guevarra, J.T.; Castillo, B.; Joshi, H.; Parekh, A.; Ayappa, I.; Rapoport, D.M. Immediate Physiological Responses to Inspiratory Flow Limited Events in Mild Obstructive Sleep Apnea. Ann. Am. Thorac. Soc. 2021, 1–33.
- Spina, G.; Spruit, M.A.; Alison, J.; Benzo, R.P.; Calverley, P.M.A.; Clarenbach, C.F.; Costello, R.W.; Donaire-Gonzalez, D.; Dürr, S.; Garcia-Aymerich, J.; et al. Analysis of nocturnal actigraphic sleep measures in patients with COPD and their association with daytime physical activity. Thorax 2017, 72, 694–701.
- Lin, C.; Lo, M.T.; Guilleminault, C. Exploring the Abnormal Modulation of the Autonomic Systems during Nasal Flow Limitation in Upper Airway Resistance Syndrome by Hilbert-Huang Transform. Front. Med. 2017, 4, 161.
- Khoo, M.C.K. Physiological Control Systems: Analysis, Simulation, and Estimation, 2nd ed.; IEEE Press: Piscataway, NJ, USA; Wiley: Hoboken, NJ, USA, 2018; p. xvii. 429p.
- Remmers, J.E.; deGroot, W.J.; Sauerland, E.K.; Anch, A.M. Pathogenesis of upper airway occlusion during sleep. J. Appl. Physiol. Respir. Environ. Exerc. Physiol. 1978, 44, 931–938.
- Javaheri, S.; Barbe, F.; Campos-Rodriguez, F.; Dempsey, J.A.; Khayat, R.; Javaheri, S.; Malhotra, A.; Martinez-Garcia, M.A.; Mehra, R.; Pack, A.I.; et al. Sleep Apnea: Types, Mechanisms, and Clinical Cardiovascular Consequences. J. Am. Coll. Cardiol. 2017, 69, 841–858.
- Kirkness, J.P.; Peterson, L.A.; Squier, S.B.; McGinley, B.M.; Schneider, H.; Meyer, A.; Schwartz, A.R.; Smith, P.L.; Patil, S.P. Performance characteristics of upper airway critical collapsing pressure measurements during sleep. Sleep 2011, 34, 459–467.
- Clark, C.; Ulualp, S.O. Multimodality assessment of upper airway obstruction in children with persistent obstructive sleep apnea after adenotonsillectomy. Laryngoscope 2017, 127, 1224–1230.





