Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Alexis Joavany Rodríguez-Solís | + 2073 word(s) | 2073 | 2021-11-18 04:04:47 | | | |
| 2 | Alexis Joavany Rodríguez-Solís | -24 word(s) | 2049 | 2021-11-23 15:16:12 | | | | |
| 3 | Camila Xu | Meta information modification | 2049 | 2021-11-24 02:04:05 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Rodríguez-Solís, A. Glyphosate Pollution Treatment. Encyclopedia. Available online: https://encyclopedia.pub/entry/16310 (accessed on 07 February 2026).
Rodríguez-Solís A. Glyphosate Pollution Treatment. Encyclopedia. Available at: https://encyclopedia.pub/entry/16310. Accessed February 07, 2026.
Rodríguez-Solís, Alexis. "Glyphosate Pollution Treatment" Encyclopedia, https://encyclopedia.pub/entry/16310 (accessed February 07, 2026).
Rodríguez-Solís, A. (2021, November 23). Glyphosate Pollution Treatment. In Encyclopedia. https://encyclopedia.pub/entry/16310
Rodríguez-Solís, Alexis. "Glyphosate Pollution Treatment." Encyclopedia. Web. 23 November, 2021.
Copy Citation
Glyphosate (N-Phosphonomethyl-glycine) is a broad-spectrum herbicide extensively used worldwide to eliminate weeds in agricultural areas, control vegetation in urban areas, and accelerate the harvest of several crops. Recently was classified as a potentially carcinogenic compound, due to this several countries banned or are in the process of banning its use.
glyphosate
environmental pollution
microorganisms
pesticide
remediation
1. Introduction
Glyphosate (N-Phosphonomethyl-glycine) is a broad-spectrum herbicide extensively used worldwide to eliminate weeds in agricultural areas, control vegetation in urban areas, and accelerate the harvest of several crops [1]. This phytotoxic activity of glyphosate was discovered at the beginning of the 70’s and it has been employed in different herbicide formulations since [2][3][4]. Glyphosate was patented as a pesticide with disrupting activity and lethal effects on a broad spectrum of plants with active photosynthesis [5][6]. Glyphosate herbicide activity has been attributed to its blocking effects on the shikimic acid pathway, through the inactivation of the key enzyme 5-enolpyruvylshikimate-3-phosphate synthase (EPSPS), due to its profile as a phosphoenolpyruvate analog (Figure 1). The blocking of the EPSPS enzyme prevents aromatic amino acids (phenylalanine, tyrosine, and tryptophan) biosynthesis and subsequent protein production [7], killing plants in a time of 1–3 weeks [8][9]. Glyphosate was proposed as a plant selective pesticide due to the absence of the shikimic acid pathway in animals and humans.
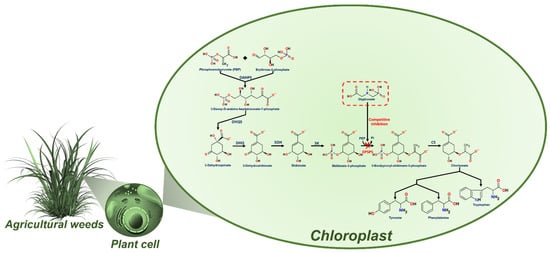
Figure 1. Glyphosate action mechanism through the inhibition of the shikimate pathway. Enzymes implicated in the pathway, DAHPS: 3-Deoxy-D-arabino-heptulosonate-7-phosphate synthase; DHQS: 3-Dehydroquinate synthase; DHQ: 3-Dehydroshikimate dehydratase; SDH: shikimate-5-dehydrogenase; SK: shikimate kinase; EPSPS: 5-Enolpyruvyl shikimate 3-phosphate synthase; and CS: chorismate synthase.
Since the glyphosate introduction to the market, it has been perceived as a less toxic weed control alternative, safe for agricultural workers and non-target organisms. In addition, glyphosate shows high effectiveness for eradicating weeds from crop fields before planting begins and for vegetation control on non-cultured areas, as the edge of roads and the sides of railways [10]. This perception of safety led to the growing popularity in the use of glyphosate-based herbicides among farmers worldwide [5], to the existence in the market of more than 750 products [11], and to be considered the most effective herbicide in all of history [12].
The worldwide use of glyphosate has become an environmental problem. The volume of application of this herbicide has been increasingly high in response to the appearance of resistant weeds, the introduction of transgenic glyphosate-resistant crops, and the adoption of new patterns in its agricultural uses, as its application as a desiccant to accelerate the harvest of grains and other crops [10][13]. Due to the intensive glyphosate use, the environmental presence and adverse effects of this herbicide are each time more evident. In addition, the toxic effects of the glyphosate-based commercial formulas are more significant due to the addition of adjuvants (surfactants), which have their own toxicity, but which also enhance the toxicity of glyphosate [8][14], highlighting the use of mixtures of polyethoxylated amines (POEA) [15]. Some POEA show toxicities 10–60 times higher than glyphosate in aquatic organisms and 4–50 times higher in mammals [16][17].
Glyphosate is known as a chelating agent that binds macro- and micronutrients, essential for several plant processes and the resistance to pathogens, sequestering of such nutrient can compromise plant resistance development, but also affect animals and human health [18]. The global increase in the intensive use of glyphosate-based herbicides causes the release and presence of residues of this chemical in the soil, water, and air, compromising the integrity of the surrounding environments and constituting a threat for the organisms that inhabit these polluted areas [19]. In this sense, different studies have evidenced the presence of glyphosate residues in surface soil, and their transport to the deep soil layers or to bodies of water near agricultural fields [20][21][22].
It has been documented that glyphosate is a highly water-soluble molecule. Hence, glyphosate can be dispersed through runoff processes to different superficial water bodies as rivers and lagoons or be absorbed by the soil particles [23] causing pollution and favoring its availability for the organisms that feed through sediment filtration [24]. For example, glyphosate is highly toxic for aquatic organisms such as algae, ferns, and fishes, while moderately toxic to amphibians, crustaceans, and earthworms [25]. Furthermore, in recent studies, glyphosate residues have been identified in several agricultural products consumed by human populations [2][26]. Moreover, residues of this agrochemical were detected in food, human blood and urine, and water supplies. In a contrasting way, the initial reports considered glyphosate as a safe molecule with low toxicity on non-target organisms [2]. However, recently, the International Agency for Research on Cancer (IARC) classified glyphosate as a probable carcinogen [11], which can negatively affect mammalian biology through multiple ways, like those related to genotoxic effects and its ability to trigger oxidative stress [2].
2. Environmental Impacts of Glyphosate
For 40 years, glyphosate has been widely used as an active chemical component of more than 750 commercial herbicides under the assumption that its side effects were minimal [11][27]. However, this compound’s intensive and large-scale use in industrialized and developing countries motivated the scientific community to evaluate the risks associated with the possible accumulation of its residues in various environmental systems and its effects on environmental and human health [28]. In this sense, recent evidence shows that herbicides containing glyphosate can contaminate the soils around the treated areas, glyphosate is adsorbed to clays and organic matter, slowing down its degradation by the action of microorganisms, leading to an accumulation in soils over time [29][30][31], the persistence of glyphosate in high clay content soil reaches more than a year [31][32].
Glyphosate has a great capacity for adsorption in the clay and organic matter present in soils, being considered as a low mobility compound, so that it is assumed that it does not represent a significant risk for the contamination of the water sources [33][34][35]. However, the scientific literature has reported the presence of this compound dissolved in groundwater [36][37], the transport of soil particles with glyphosate in surface water [21][37][38], as well as its adsorption in the sediments of water bodies [35]. Moreover, the glyphosate deposited in the first centimeters of the superficial soil layer is susceptible to wind erosion and atmospheric transport [39][40], consequently, glyphosate has been detected in the air, rain, and water from melting snow [21][41]. Finally, glyphosate has also been reported in seawater, where it is very persistent [42] and in drinking water [43].
Resulting from its accumulation and persistence in the soil, glyphosate can affect exposed organisms in this environmental compartment [44]. For example, it has been reported that glyphosate can affect the activity of soil microorganisms that are involved in biogeochemical cycles, the mineralization of organic remains, the immobilization and solubilization of minerals, and the degradation of other xenobiotics [45][46][47]. Likewise, a reduction in the reproduction rate, biomass, and DNA damage in earthworms has been reported [48][49][50], as well as adverse effects in other small size organisms, such as nematodes, distributed in soils [51]. Moreover, in plant species, the direct effects of their exposure to glyphosate are related to the inhibition of the activity of antioxidant enzymes and the induction of reactive oxygen species (ROS), which promote cell damage and physiological alterations in processes such as photosynthesis and the production of secondary metabolites [52]. Furthermore, traces of this compound can be detected in plant tissues of temperate zone species up to more than 12 years after the treatment [4]. Glyphosate indirectly changes the rhizosphere microbiome, which affects plant health [28][53].
In water bodies, the negative impacts of glyphosate have been observed in organisms such as protozoa, mussels, crustaceans, frogs, and fish [28]. Similar to that reported in terrestrial ecosystems, the presence of glyphosate in fish produces metabolism alterations, leading to the overproduction of reactive oxygen species and oxidative stress resulting in kidney damage [54]. Likewise, other studies have linked glyphosate exposure to DNA damage and chromosomal alterations in fish [55][56]. The presence of glyphosate not only has effects at the level of individual organisms, but alterations in the interactions between species have also been documented. As an example, an increase in the levels of susceptibility of fish to its parasites has been documented [57][58]. Similarly, unwanted effects of glyphosate exposure have been reported in bee species that provide valuable ecosystem services such as pollination [59]. Finally, glyphosate has been detected in animal feed, animal meat, and urine, as well as in the food intended for human consumption, which is why the presence of this herbicide has been detected in samples of breast milk and urine [5]. Another additional environmental risk associated with the presence of glyphosate, which has not been adequately considered, is that it is a potent mineral chelator [18] whose application can lead to the reduction of macro and micronutrients that are essential cofactors in many biological processes of glyphosate-treated plants and potentially also for the organisms that feed on them. Consequently, a reduced supply of nutrients in the treated plants can compromise their resistance to diseases. In the case of humans and other animals that consume food obtained from plants treated with glyphosate, the residues of this herbicide and the reduced levels of nutrients can also have an impact on their health [18][60]. Therefore, to minimize its environmental and human health impacts, monitoring and detection of its presence in different environments, as well as the evaluation of exposure to this herbicide in humans, is of utmost importance.
3. Glyphosate Human Health Threats
Humans have been exposed to glyphosate directly through occupational exposure or indirectly through various sources [61]. Occupational exposure includes agricultural workers, farmers, gardeners, and people who work in plants that process glyphosate [62]. These people can be exposed to glyphosate through inhalation, dermal and ocular contact. In contrast, the indirect exposure includes the consumption of water or food contaminated with glyphosate residues [63] or the environmental exposure to residues or products of its transformation, such as aminomethyl phosphonic acid (AMPA) in environmental matrixes such as air, water, or soil [61][62].
The mode of action of glyphosate consists of the inhibition of the enzyme EPSPS involved in the biosynthesis of the aromatic amino acids tyrosine, tryptophan, and phenylalanine through the shikimate pathway in plants [64]. Therefore, glyphosate was proposed as a low toxicity compound for non-target organisms and was considered relatively safe for humans, according to the results of different exposure studies carried out in rodents, chickens, and amphibians [12][13][65][66].
More recently, it was determined that the toxicity of commercial herbicides based on glyphosate is exacerbated by the presence of surfactant compounds in the formulation, being polyoxyethyleneamine (POEA) the most common [5][61]. This compound uncouples elements of phosphorylation oxidative stress, causing oxidative stress and cardiotoxicity [67]. Thus, in 2015, the World Health Organization reclassified glyphosate as a possible human carcinogen [11][68][69]. Glyphosate reclassification into the group 2A of the International Agency for Research on Cancer (IARC) was based on the review of the accumulated evidence provided by experts in cancer and toxicology, which has contributed to a better understanding of the toxicity of the compound for all kinds of exposed organisms in natural areas, and in experimental animals, and its mechanisms of action [70].
Among the most relevant information on the effects caused by glyphosate exposure in humans are the studies by Samsel and Seneff (2013b) [71], which indicated that exposure to the herbicide represented the main factor causing gluten intolerance and gastrointestinal disorders, as well as interference in the assimilation of micronutrients such as iron, cobalt, molybdenum, copper, and amino acids such as tryptophan, tyrosine, methionine, and selenomethionine. Subsequently, Samsel and Seneff (2013a) [60] and Schinasi and Leon (2014) [72] evidenced an association between non-Hodgkin’s lymphoma and agrochemical exposure. In particular, the latest work showed that occupational exposure to glyphosate increases the relative risk of developing this disease and the development of B-cell lymphoma. Other reports of chronic exposure to the herbicide in human populations show the association with conditions such as allergies, and asthma [73], cardiovascular diseases [74], autism, and chronic degenerative diseases such as multiple myeloma [75]. Cytotoxic damage has also been reported in chorioplacental cells of humans, which triggers inhibition in the synthesis of progesterone as a secondary effect [76]. Recent reviews suggest that glyphosate and glyphosate-based herbicides promote cytotoxic and genotoxic effects, a significant increase in oxidative stress, disruption of the estrogen pathway, adverse effects on various cognitive processes, and an association with the development of certain cancers [77][78]. The studies mentioned above compile evidence of the high glyphosate toxicity and establish this compound as a menace to the health of the agricultural population that has a history of direct exposure and for whom the exposure has been indirect through the consumption of food or water with residues of glyphosate. Human individuals exposed to this compound have presented multiple organ toxicity, nephrotoxicity, hepatotoxicity, gastrointestinal, cardiovascular, and respiratory effects [62][79].
References
- Tarazona, J.V.; Tiramani, M.; Reich, H.; Pfeil, R.; Istace, F.; Crivellente, F. Glyphosate toxicity and carcinogenicity: A review of the scientific basis of the European Union assessment and its differences with IARC. Arch. Toxicol. 2017, 91, 2723–2743.
- Myers, J.P.; Antoniou, M.N.; Blumberg, B.; Carroll, L.; Colborn, T.; Everett, L.G.; Hansen, M.; Landrigan, P.J.; Lanphear, B.P.; Mesnage, R.; et al. Concerns over use of glyphosate-based herbicides and risks associated with exposures: A consensus statement. Environ. Health. 2016, 15, 19.
- Duke, S.O. The history and current status of glyphosate. Pest. Manag. Sci. 2018, 74, 5.
- Botten, N.; Wood, L.J.; Werner, J.R. Glyphosate remains in forest plant tissues for a decade or more. For. Ecol. Manag. 2021, 493, 119259.
- Cuhra, M.; Bøhn, T.; Cuhra, P. Glyphosate: Too much of a good thing? Front. Environ. Sci. 2016, 4, 1–28.
- Zabalza, A.; Orcaray, L.; Fernández-Escalada, M.; Zulet-González, M.; Royuela, M. The pattern of shikimate pathway and phenylpropanoids after inhibition by glyphosate or quinate feeding in pea roots. Pestic. Biochem. Physiol. 2017, 141, 96–102.
- Boocock, M.R.; Coggins, J.R. Kinetics of 5-enolpyruvylshikimate-3-phosphate synthase inhibition by glyphosate. FEBS Lett. 1983, 154, 127–133.
- Gill, J.P.K.; Sethi, N.; Mohan, A.; Datta, S.; Girdhar, M. Glyphosate toxicity for animals. Environ. Chem. Lett. 2018, 16, 401–426.
- Kanissery, R.; Gairhe, B.; Kadyampakeni, D.; Batuman, O.; Alferez, F. Glyphosate: Its environmental persistence and impact on crop health and nutrition. Plants 2019, 8, 499.
- Benbrook, C.M. Trends in glyphosate herbicide use in the United States and globally. Environ. Sci. Eur. 2016, 28, 3.
- Fritschi, L.; McLaughlin, J.; Sergi, C.M.; Calaf, G.M.; Le Curieux, F.; Zeise, L. Carcinogenicity of tetrachlorvinphos, parathion, malathion, diazinon, and glyphosate. Lancet. Oncol. 2015, 16, 490–491.
- Duke, S.O.; Powles, S.B. Glyphosate: A once-in-a-century herbicide. Pest. Manag. Sci. 2008, 64, 319–325.
- Dill, G.M.; Sammons, R.D.; Feng, P.C.C.; Kohn, F.; Kretzmer, K.; Mehrsheikh, A.; Bleeke, M.; Honegger, J.L.; Farmer, D.; Wright, D.; et al. Glyphosate: Discovery, development, applications and properties. In Glyphosate Resistance in Crops and Weeds: History, Development, and Management; Wiley: Hoboken, NJ, USA, 2010; pp. 1–33.
- Mesnage, R.; Defarge, N.; Spiroux de Vendômois, J.; Seralini, G.E. Potential toxic effects of glyphosate and its commercial formulations below regulatory limits. Food Chem. Toxicol. 2015, 84, 133–153.
- Annett, R.; Habibi, H.R.; Hontela, A. Impacts of glyphosate and glyphosate-based herbicides on the freshwater environment. J. Appl. Toxicol. 2014, 34, 458–479.
- Martens, M.A.; Bleeke, M.S.; Leopold, V.A.; Farmer, D.R. Toxicology and human health risk assessment of polyethoxylated tallow amine surfactant used in glyphosate formulations. Regul. Toxicol. Pharmacol. 2019, 107, 104347.
- Mesnage, R.; Benbrook, C.; Antoniou, M.N. Insight into the confusion over surfactant co-formulants in glyphosate-based herbicides. Food Chem. Toxicol. 2019, 128, 137–145.
- Mertens, M.; Höss, S.; Neumann, G.; Afzal, J.; Reichenbecher, W. Glyphosate, a chelating agent relevant for ecological risk assessment? Environ. Sci. Pollut. Res. 2018, 25, 5298–5317.
- Golt, A.R.; Wood, L.J. Glyphosate-Based Herbicides Alter the Reproductive Morphology of Rosa acicularis (Prickly Rose). Front. Plant. Sci. 2021, 12, 1184.
- Borggaard, O.K.; Gimsing, A.L. Fate of Glyphosate in Soil and the Possibility of Leaching to Ground and Surface Waters: A Review. Pest. Manag. Sci. 2008, 64, 441–456.
- Battaglin, W.A.; Meyer, M.T.; Kuivila, K.M.; Dietze, J.E. Glyphosate and Its Degradation Product AMPA Occur Frequently and Widely in U.S. Soils, Surface Water, Groundwater, and Precipitation. J. Am. Water Resour. Assoc. 2014, 50, 275–290.
- Ruiz-Toledo, J.; Castro, R.; Rivero-Pérez, N.; Bello-Mendoza, R.; Sánchez, D. Occurrence of Glyphosate in Water Bodies Derived from Intensive Agriculture in a Tropical Region of Southern Mexico. Bull. Environ. Contam. Toxicol. 2014, 93, 289–293.
- Lupi, L.; Bedmar, F.; Puricelli, M.; Marino, D.; Aparicio, V.C.; Wunderlin, D.; Miglioranza, K.S. Glyphosate runoff and its occurrence in rainwater and subsurface soil in the nearby area of agricultural fields in Argentina. Chemosphere 2019, 225, 906–914.
- Bonansea, R.; Filippi, I.; Wunderlin, D.; Marino, D.; Ame, M. The fate of glyphosate and AMPA in a freshwater endorheic basin: An ecotoxicological risk assessment. Toxics 2018, 6, 3.
- Instituto Regional de Estudios en Sustancias Tóxicas (IRET). (2015). Base de Datos de Ingredientes Activos Importados en Centroamérica. Universidad Nacional. Costa Rica. Available online: http://www.plaguicidasdecentroamerica.info/index.php/base-de-datos/ingredientes-activos/306-glifosato (accessed on 1 October 2021).
- Cuhra, M. Review of GMO safety assessment studies: Glyphosate residues in Roundup Ready crops is an ignored issue. Environ. Sci. Eur. 2015, 27, 20.
- Sviridov, A.V.; Shushkova, T.V.; Ermakova, I.T.; Ivanova, E.V.; Leontievsky, A.A. Glyphosate: Safety risks, biodegradation, and bioremediation. In Current Environmental Issues and Challenges; Springer: Berlin/Heidelberg, Germany, 2014; ordrecht; pp. 183–195.
- Van Bruggen, A.H.C.; He, M.M.; Shin, K.; Mai, V.; Jeong, K.C.; Finckh, M.R.; Morris, J.G., Jr. Environmental and health effects of glyphosate. Sci. Total Environ. 2018, 616, 255–268.
- Travaglia, C.; Masciarelli, O.; Fortuna, J.; Marchetti, G.; Cardozo, P.; Lucero, M.; Zorza, E.; Luna, V.; Reinoso, H. Towards sustainable maize production: Glyphosate detoxification by Azospirillum sp. and Pseudomonas sp. Crop. Prot. 2015, 77, 102–109.
- Cassigneul, A.; Benoit, P.; Bergheaud, V.; Dumeny, V.; Etiévant, V.; Goubard, Y.; Maylin, A.; Justes, E.; Alletto, L. Fate of glyphosate and degradates in cover crop residues and underlying soil: A laboratory study. Sci. Total Environ. 2016, 545, 582–590.
- Sidoli, P.; Baran, N.; Angulo-Jaramillo, R. Glyphosate and AMPA adsorption in soils: Laboratory experiments and pedotransfer rules. Environ. Sci. Pollut. Res. 2016, 23, 5733–5742.
- Okada, E.; Costa, J.L.; Bedmar, F. Adsorption and mobility of glyphosate in different soils under no-till and conventional tillage. Geoderma 2016, 263, 78–85.
- Sihtmäe, M.; Blinova, I.; Kunnis-Beres, K.; Kanarbik, L.; Heinlaan, M.; Kahru, A. Ecotoxicological effects of different glyphosate formulations. Appl. Soil Ecol. 2013, 72, 215–224.
- Gill, J.P.K.; Sethi, N.; Mohan, A. Analysis of the glyphosate herbicide in water, soil and food using derivatising agents. Environ. Chem. Lett. 2017, 15, 85–100.
- Maqueda, C.; Undabeytia, T.; Villaverde, J.; Morillo, E. Behaviour of glyphosate in a reservoir and the surrounding agricultural soils. Sci. Total Environ. 2017, 593, 787–795.
- Van Stempvoort, D.R.; Spoelstra, J.; Senger, N.D.; Brown, S.J.; Post, R.; Struger, J. Glyphosate residues in rural groundwater, Nottawasaga River watershed, Ontario, Canada. Pest Manag. Sci. 2016, 72, 1862–1872.
- Rendón-von Osten, J.; Dzul-Caamal, R. Glyphosate residues in groundwater, drinking water and urine of subsistence farmers from intensive agriculture localities: A survey in Hopelchén, Campeche, Mexico. Int. J. Environ. Res. Public Health 2017, 14, 595.
- Yang, X.; Wang, F.; Bento, C.P.M.; Xue, S.; Gai, L.; van Dam, R.; Mol, H.; Ritsema, C.J.; Geissen, V. Short-term transport of glyphosate with erosion in Chinese loess soil-a flume experiment. Sci. Total Environ. 2015, 512, 406–414.
- Alonso, L.L.; Demetrio, P.M.; Etchegoyen, M.A.; Marino, D. Glyphosate and atrazine in rainfall and soils in agroproductive areas of the pampas region in Argentina. Sci. Total Environ. 2018, 645, 89–96.
- Silva, V.; Montanarella, L.; Jones, A.; Fernandez-Ugalde, O.; Mol, H.G.J.; Ritsema, C.J.; Geissen, V. Distribution of glyphosate and aminomethylphosphonic acid (AMPA) in agricultural topsoils of the European Union. Sci. Total Environ. 2018, 621, 1352–1359.
- Chang, F.C.; Simcik, M.F.; Capel, P. Occurrence and fate of the herbicide glyphosate and its degradate aminomethylphosphonic acid in the atmosphere. Environ. Toxicol. Chem. 2011, 30, 548–555.
- Mercurio, P.; Flores, F.; Mueller, J.F.; Carter, S.; Negri, A.P. Glyphosate persistence in seawater. Mar. Pollut. Bull. 2014, 85, 385–390.
- WHO (World Health Organization) Glyphosate and AMPA in Drinking-Water. 2005. Available online: http://www.who.int/water_sanitation_health/dwq/chemicals/glyphosateampa290605.pdf (accessed on 7 September 2021).
- Meena, R.S.; Kumar, S.; Datta, R.; Lal, R.; Vijayakumar, V.; Brtnicky, M.; Sharma, M.P.; Yadav, G.S.; Jhariya, M.K.; Jangir, C.K.; et al. Impact of agrochemicals on soil microbiota and management: A review. Land 2020, 9, 34.
- Sterren, M.; Uhrich, W.; Benintende, S. Residualidad de glifosato en suelos de Entre Ríos y su efecto sobre los microorganismos del suelo. Ecología Austral. 2016, 26, 46–255.
- Helander, M.; Saloniemi, I.; Omacini, M.; Druille, M.; Salminen, J.P.; Saikkonen, K. Glyphosate decreases mycorrhizal colonization and affects plant-soil feedback. Sci. Total Environ. 2018, 642, 285–291.
- Vázquez, M.B.; Moreno, M.V.; Amodeo, M.R.; Bianchinotti, M.V. Effects of glyphosate on soil fungal communities: A field study. Rev. Argent. Microbiol. 2021.
- Correia, F.V.; Moreira, J.C. Effects of glyphosate and 2, 4-D on earthworms (Eisenia foetida) in laboratory tests. Bull. Environ. Contam. Toxicol. 2010, 85, 264–268.
- Piola, L.; Fuchs, J.; Oneto, M.L.; Basack, S.; Kesten, E.; Casabe, N. Comparative toxicity of two glyphosate-based formulations to Eisenia andrei under laboratory conditions. Chemosphere 2013, 91, 545–551.
- García-Torre, T.; Giufre, L.; Romaniuk, R.; Rios, R.P.; Pagano, E.A. Exposure assessment to glyphosate of two species of annelids. Bull. Environ. Contam. Toxicol. 2014, 93, 209–214.
- Hagner, M.; Mikola, J.; Saloniemi, I.; Saikkonen, K.; Helander, M. Effects of a glyphosate-based herbicide on soil animal trophic groups and associated ecosystem functioning in a northern agricultural field. Sci. Rep. 2019, 9, 8540.
- Gomes, M.P.; Manaćh, L.; Sarah, G.; Henault-Ethier, L.; Labrecque, M.; Lucotte, M.; Juneau, P. Glyphosate-dependent inhibition of photosynthesis in willow. Front. Plant Sci. 2017, 8, 207.
- Van Bruggen, A.H.C.; Finckh, M.R. Plant diseases and management approaches in organic farming systems. Annu. Rev. Phytopathol. 2016, 54, 25–54.
- Li, M.H.; Ruan, L.Y.; Zhou, J.W.; Fu, Y.H.; Jiang, L.; Zhao, H.; Wang, J.S. Metabolic profiling of goldfish (Carassius auratis) after long-term glyphosate based herbicide exposure. Aquat. Toxicol. 2017, 188, 159–169.
- Moreno, N.C.; Sofia, S.H.; Martinez, C.B.R. Genotoxic effects of the herbicide Roundup Transorb® and its active ingredient glyphosate on the fish Prochilodus lineatus. Eviron. Toxicol. Pharmacol. 2014, 37, 448–454.
- Caramello, C.S.; Jorge, M.J.; Jorge, N.L.; Jorge, L.C. Evaluation of herbicide glyphosate effects in the fish Prochilodus lineatus using chromosome aberration test. Rev. Vet. 2017, 28, 65–68.
- Kreutz, L.C.; Gil Barcellos, L.J.; Marteninghe, E.; Davidos Santos, E.; Zanatta, R. Exposure to sublethal concentration of glyphosate or atrazine-based herbicides alters the phagocytic function and increases the susceptibility of silver catfish fingerlings (Rhamdia quelen) to Aeromonas hydrophila challenge. Fish Shellfish. Immunol. 2010, 29, 694–697.
- Kreutz, L.C.; Gil Barcellos, L.J.; Valle, S.F.; Silva, T.O.; Anziliero, D.; Davidos Santos, E.; Pivato, M.; Zanatta, R. Altered hematological and immunological parameters in silver catfish (Rhamdia quelen) following short term exposure to sublethal concentration of glyphosate. Fish Shellfish. Immunol. 2011, 30, 51–57.
- Balbuena, M.S.; Tison, L.; Hahn, M.L.; Greggers, U.; Menzel, R.; Farina, W.M. Effects of sublethal doses of glyphosate on honeybee navigation. J. Exp. Biol. 2015, 218, 2799–2805.
- Samsel, A.; Seneff, S. Glyphosate’s suppression of cytochrome P450 enzymes and amino acid biosynthesis by the gut microbiome: Pathways to modern diseases. Entroy 2013, 15, 1416–1463.
- Gandhi, K.; Khan, S.; Patrikar, M.; Markad, A.; Kumar, N.; Choudhari, A.; Sagar, P.; Indurkar, S. Exposure risk and environmental impacts of glyphosate: Highlights on the toxicity of herbicide co-formulants. Environ. Chall. 2021, 4, 100149.
- Gillezeau, C.; Van Gerwen, M.; Shaffer, R.; Rana, I.; Zhang, L.; Sheppard, L.; Taioli, E. The evidence of human exposure to glyphosate: A review. Environ. Health 2019, 18, 2.
- WHO Europe. Results of Joint FAO/WHO Meeting on Pesticide Residues (JMPR). 2016. Available online: https://www.euro.who.int/en/health-topics/disease-prevention/food-safety/news/news/2016/05/results-of-joint-faowho-meeting-on-pesticide-residues-jmpr (accessed on 1 October 2021).
- Duke, S.O. Overview of herbicide mechanisms of action. Environ. Health Perspect. 1990, 87, 263–271.
- Mann, R.M.; Bidwell, J.R. The toxicity of glyphosate and several glyphosate formulations to four species of southwestern Australian frogs. Arch. Environ. Contam. Toxicol. 1999, 36, 193–199.
- Williams, G.M.; Kroes, R.; Munro, I.C. Safety evaluation and risk assessment of the herbicide Roundup and its active ingredient, glyphosate, for humans. Regul. Toxicol. Pharmacol. 2000, 31, 117–165.
- Gress, S.; Lemoine, S.; Séralini, G.E.; Puddu, P.E. Glyphosate-based herbicides potently affect cardiovascular system in mammals: Review of the literature. Cardiovasc. Toxicol. 2015, 15, 117–126.
- IARC. Glyphosate. In Some Organophosphate Insecticides and Herbicides: Diazinon, Glyphosate, Malathion, Parathion, Tetrachlorvinphos. IARC Working Group, 3–10 March 2015; World Health Organization (WHO), International Agency for Research on Cancer (IARC) (IARC Monographs on the Evaluation of Carcinogen Risks to Humans): Lyon, France, 2015; Volume 112, pp. 1–92. Available online: http://monographs.iarc.fr/ENG/Monographs/vol112/index.php (accessed on 1 October 2021).
- Bai, S.H.; Ogbourne, S.M. Glyphosate: Environmental contamination, toxicity and potential risks to human health via food contamination. Environ. Sci. Pollut. Res. 2016, 23, 18988–19001.
- Arellano-Aguilar, O.; Montero-Montoya, R. Glifosato y los cultivos transgénicos en México. In Los plaguicidas altamente peligrosos en México; Bejarano-González, F., Ed.; Red de Acción sobre Plaguicidas y Alternativas en México, A.C. (RAPAM): Estado de México, México, 2015; pp. 153–166.
- Samsel, A.; Seneff, S. Glyphosate, pathways to modern diseases II: Celiac sprue and gluten intolerance. Interdiscip. Toxicol. 2013, 6, 159–184.
- Schinasi, L.; Leon, M.E. Non-Hodgkin lymphoma and occupational exposure to agricultural pesticide chemical groups and active ingredients: A systematic review and meta-analysis. Int. J. Environ. Res. Public. Health. 2014, 11, 4449–4527.
- Henneberger, P.K.; Liang, X.; London, S.J.; Umbach, D.M.; Sandler, D.P.; Hoppin, J.A. Exacerbation of symptoms in agricultural pesticide applicators with asthma. Int. Arch. Occup. Environ. Health 2014, 87, 423–432.
- Zouaoui, K.; Dulaurent, S.; Gaulier, J.M.; Moesch, C.; Lachatre, G. Determination of glyphosate and AMPA in blood and urine from humans: About cases of acute intoxication. Forensic. Sci. Int. 2013, 226, 20–25.
- Beecham, J.E.; Seneff, S. The possible link between autism and glyphosate acting as glycine mimetic—A review of evidence from the literature with analysis. J. Mol. Genet. Med. 2015, 9, 1000197.
- Young, F.; Ho, D.; Glynn, D.; Edwards, V. Endocrine disruption and cytotoxicity of glyphosate and roundup in human JAr cells in vitro. Integr. Pharm. Toxicol. Gentocicol. 2015, 1, 12–19.
- Peillex, C.; Pelletier, M. The impact and toxicity of glyphosate and glyphosate-based herbicides on health and immunity. J. Immunotoxicol. 2020, 17, 163–174.
- Weisenburger, D.D. A review and update with perspective of evidence that the herbicide glyphosate (Roundup) is a Cause of non-Hodgkin lymphoma. Clin. Lymphoma Myeloma Leuk. 2021, 21, 621–630.
- Venugopal, K.; Suresh, C.; Vishwanath, H.; Lingaraja, M.; Bharath Raj, M.Y. Glyphosate: Surfactant herbicide poisoning—Is it mild? Med. J. DY. Patil. Univ. 2015, 8, 816–818.
More
Information
Subjects:
Biochemistry & Molecular Biology; Soil Science
Contributor
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
1.5K
Entry Collection:
Environmental Sciences
Revisions:
3 times
(View History)
Update Date:
24 Nov 2021
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No




