
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Jose Ignacio Marín-Guirao | + 5662 word(s) | 5662 | 2021-11-04 10:00:08 | | | |
| 2 | Beatrix Zheng | -2731 word(s) | 2931 | 2021-11-10 03:35:50 | | |
Video Upload Options
Greenhouse agriculture typically generates large amounts of waste with plant residue (agricultural biomass) being the most abundant. This residue is generated on a seasonal basis, which complicates the external management of the material. Recently, the European Union (EU) has been implementing a policy based on sustainability through the circular economy that seeks to minimize waste generation. The effect of reusing 3.5 kg·m-2 tomato plants from the previous season as the only fertilizer versus no fertilization and inorganic fertilization in 215-day tomato cycles after transplanting was studied in this trial. The study was carried out during three seasons in greenhouse agriculture in Almeria (Spain) with the repeated use of the solarization technique. The plant debris had similar production results during two of the three seasons and fruit quality parameters were similar to inorganic fertilization. In addition, some physicochemical variables improved and the biological depressive effect of solarization was mitigated. The results suggest that the reuse of the tomato plant debris as the only fertilizer could be an alternative to conventional fertilization under the conditions tested.
1. Introduction
2. Current Insights
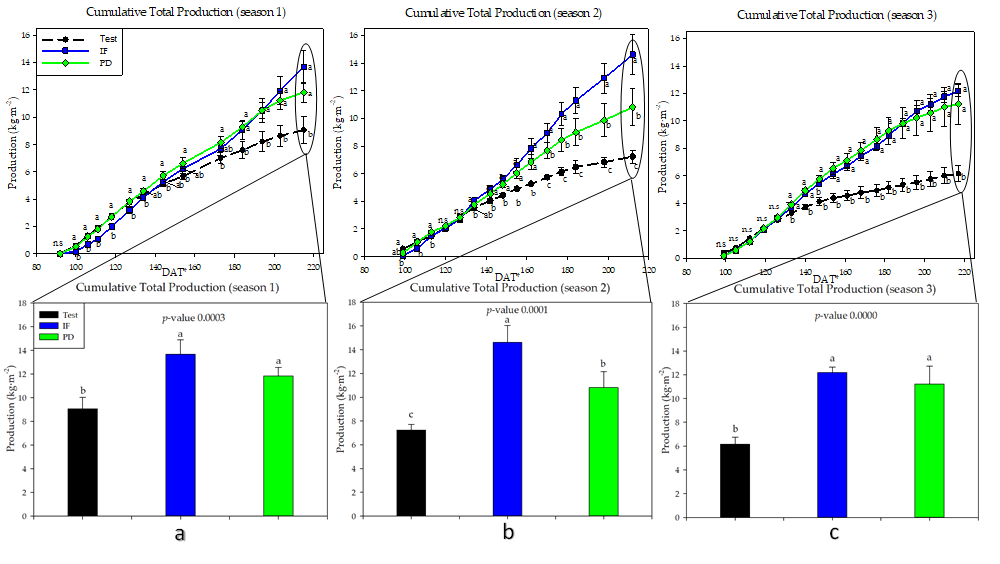
Figure 1. Cumulative tomato production in the three years of study (September–April cycles) as a function of crop nutrition: (a) Crop 1; (b): Crop 2; (c): Crop 3. Inorganic fertilization (IF); tomato plant debris (PD); no fertilization (test). Values (mean ± standard deviation). Different letters indicate significant differences (p ≤ 0.05, Tukey’s HDS test). DAT: days after transplanting.
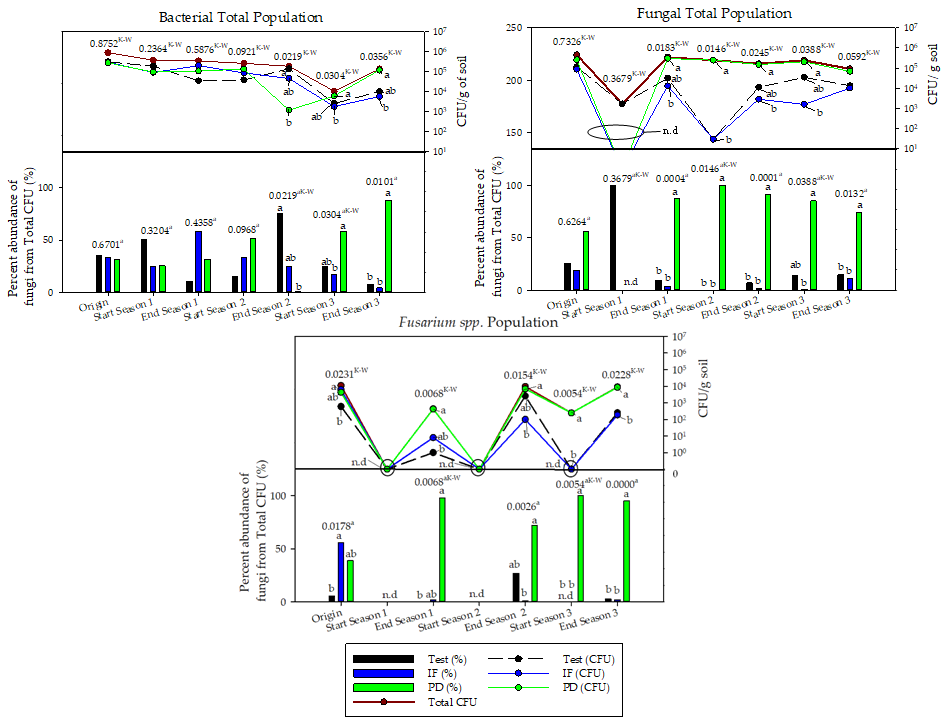
3. Conclusions
References
- Baudoin, W.; Nersisyan, A.; Shamilov, A.; Hodder, A.; Gutierrez, D.; de Pascale, S.; Nicola, S.; Chairperson, V.; Gruda, N.; Urban, L. Good Agricultural Practices for Greenhouse Vegetable Production in the South East European Countries; Food and Agriculture Organization of the United Nations: Rome, Italy, 2017; pp. 1–449. ISBN 978-92-5-109622-2.
- European Comission. EIP-AGRI Focus Group Circular Horticulture: Final Report; EIP-AGRI Agriculture & Innovation: Brussels, Belgium, 2019; pp. 1–20.
- de Andalucía, J. Cartografía de Invernaderos en Almería, Granada Y Málaga. Año 2020; Consejería de Agricultura, Ganadería, Pesca y Desarrollo Sostenible: Sevilla, España, 2020; pp. 1–24.
- Duque-Acevedo, M.; Belmonte-Ureña, L.J.; Plaza-Úbeda, J.A.; Camacho-Ferre, F. The management of agricultural waste biomass in the framework of circular economy and bioeconomy: An opportunity for greenhouse agriculture in Southeast Spain. Agronomy 2020, 10, 489.
- Camacho-Ferre, F. Técnicas de Producción de Cultivos Protegidos (Tomo II); Caja Rural Intermediterránea, Cajamar: Almería, España, 2004; pp. 389–776. ISBN 84-95531-16-X.
- Cajamar. Análisis de la Campaña Hortofrutícola 2019/2020; Cajamar Caja Rural: Almerría, España, 2020; pp. 1–9.
- Junta de Andalucía. Caracterización De Los Invernaderos De Andalucía; Consejería de Agricultura, Ganadería, Pesca y Desarrollo Sostenible: Sevilla, España, 2015; pp. 1–113.
- Valera-Martínez, D.L.; Belmonte-Ureña, L.J.; Molina Aiz, F.D.; Camacho-Ferre, F. The greenhouses of Almería, Spain: Technological analysis and profitability. Acta Hortic. 2017, 1170, 219–226.
- Vanthoor, B.H.E.; Stigter, J.D.; van Henten, E.J.; Stanghellini, C.; de Visser, P.H.B.; Hemming, S. A methodology for model-based greenhouse design: Part 5, greenhouse design optimisation for southern-Spanish and Dutch conditions. Biosyst. Eng. 2012, 111, 350–368.
- Castro, A.J.; López-Rodríguez, M.D.; Giagnocavo, C.; Giménez, M.; Céspedes, L.; La Calle, A.; Gallardo, M.; Pumares, P.; Cabello, J.; Rodríguez, E.; et al. Six Collective Challenges for Sustainability of Almería Greenhouse Horticulture. Int. J. Environ. Res. Public Health 2019, 16, 4097.
- Caparrós-Martínez, J.; Rueda-López, N.; Milán-García, J.; de Valenciano, J. Public policies for sustainability and water security: The case of Almeria (Spain). Glob. Ecol. Conserv. 2020, 23, e01037.
- European Union. Directive (EU) 2018/851 of the European Parliament and of the Council of 30 May 2018 amending Directive 2008/98/EC on Waste. Off. J. Eur. Union L Ser. 2018, 150, 109–140.
- European Comission. A new Circular Economy Action Plan. In For a Cleaner and More Competitive Europe; COM/2020/98 final. 11.3.2020; Office of the European Union: Brussels, Belgium, 2020; pp. 1–19.
- European Comission. The European Green Deal; COM(2019) 640 final. 11.12.2019; Office of the European Union: Brussels, Belgium, 2019; pp. 1–28.
- European Comission. A Sustainable Bioeconomy for Europe: Strengthening the Connection between Economy, Society and the Environment; COM(2018) 673 final. 11.10.2018; Office of the European Union: Brussels, Belgium, 2018; pp. 1–14.
- European Comission. A Farm to Fork Strategy for a Fair, Healthy and Environmentally-Friendly Food System; COM(2020) 381 final. 20.5.2020; Office of the European Union: Brussels, Belgium, 2020; pp. 1–23.
- Aznar-Sánchez, J.A.; Velasco-Muñoz, J.F.; García-Arca, D.; López-Felices, B. Identification of opportunities for applying the circular economy to intensive agriculture in Almería (South-East Spain). Agronomy 2020, 10, 1499.
- de Andalucía, J. Líneas De Actuación En Materia De Gestión De Restos Vegetales En La Horticultura De ANDALUCÍA; Consejería de Agricultura, Ganadería, Pesca y Desarrollo Sostenible: Sevilla, España, 2016; pp. 1–45.
- Camacho-Ferre, F. Diferentes alternativas para la gestión del residuo biomasa procedente de cultivos de invernadero. In Innovaciones Tecnológicas en Cultivos de Invernadero; Fernández-Rodríguez, E.J., Ed.; Ediciones Agrotécnicas: Madrid, España, 2004; pp. 211–238. ISBN 94-87480-52-7.
- Castillo-Díaz, F.J.; Ruiz-Olmos, C.A.; Gómez-Tenorio, M.Á.; Tello-Marquina, J.C. Efecto de la biosolarización sobre la producción de tomate cultivado bajo invernadero en Almería. Parte I: Evaluación de diferentes restos vegetales. Agrícola Vergel 2021, 432, 103–112.
- García-Raya, P.; Ruiz-Olmos, C.; Marín-Guirao, J.I.; Asensio-Grima, C.; Tello-Marquina, J.C.; de Cara-García, M. Greenhouse Soil Biosolarization with Tomato Plant Debris as a Unique Fertilizer for Tomato Crops Greenhouse Soil Biosolarization with Tomato Plant Debris as a Unique Fertilizer for Tomato Crops. Int. J. Environ. Res. Public Health 2019, 16, 279.
- Salinas, J.; Meca, D.; del Moral, F. Short-term effects of changing soil management practices on soil quality indicators and crop yields in greenhouses. Agronomy 2020, 10, 582.
- Contreras, J.I.; Baeza, R.; Segura, M.L. Cuantificación de los nutrientes aportados al suelo por la incorporación de los restos de los cultivos hortícolas de invernadero. XI Congr. SEAE Agric. Ecológica Fam 2014, 1, 18–19.
- Marín-Guirao, J.I.; Tello-Marquina, J.C. Microbiota edáfica y fatiga de suelo en invernaderos de la provincia de Gran. In I Jornadas de Transferencia Hortofrutícola de CIAMBITAL; Camacho-Ferre, F., Valera-Martínez, D.L., Belmonte-Ureña, L., Herrero-Sánchez, C., Reca-Cardeña, J., Marín-Membrive, P., del Pino-Gracia, A., Casa-Fernández, M., Eds.; Universidad de Almería y CIAMBITAL: Almería, España, 2017; pp. 17–36. ISBN 978-84-16389-98-8.
- Marín-Guirao, J.I.; de Cara-García, M.; Crisol-Martínez, E.; Gómez-Tenorio, M.A.; García-Raya, P.; Tello-Marquina, J.C. Association of plant development to organic matter and fungal presence in Association of plant development to organic matter and fungal presence in soils of horticultural crops. Ann. Appl. Biol. 2019, 1, 1–10.
- Tiessen, H.; Cuevas, E.; Chacon, P. The role of soil organic matter in sustaining soil fertility. Nature 1994, 371, 783–785.
- Hijbeek, R.; van Ittersum, M.K.; ten Berge, H.F.M.; Gort, G.; Spiegel, H.; Whitmore, A.P. Do organic inputs matter—A meta-analysis of additional yield effects for arable crops in Europe. Plant Soil 2017, 411, 293–303.
- Fernández, P.; Lacasa, A.; Guirao, P.; Larregla, S. Effects of Biosolarization with fresh sheep manure on soil physical properties of pepper greenhouses in Campo de Cartagena. In Proceedings of the 6th Workshop on Agri-Food Research, Murcia, Spain, 8–9 May 2017; Artés-Hernández, F., Cos, J.E., Fernández-Hernández, J.A., Calatrava, J.A., Aguayo, E., Alarcón, J.J., Guitiérrez-Cortines, M.E., Eds.; Universidad Politécnica de Cartagena: Cartagena, España, 2018; pp. 97–100. ISBN 9788416325641.
- Núñez-Zofio, M.; Larregla, S.; Garbisu, C. Repeated biodisinfection controls the incidence of Phytophthora root and crown rot of pepper while improving soil quality. Span. J. Agric. Res. 2012, 10, 794–805.
- Mauromicale, G.; Lo Monaco, A.; Longo, A.M.G. Improved efficiency of soil solarization for growth and yield of greenhouse tomatoes. Agron. Sustain. Dev. 2010, 30, 753–761.
- Mauromicale, G.; Longo, A.M.G.; Lo Monaco, A. The effect of organic supplementation of solarized soil on the quality of tomato fruit. Sci. Hortic. (Amsterdam) 2011, 129, 189–196.
- Marín-Guirao, J.I.; Tello-Marquina, J.C.; Díaz, M.; Boix, A.; Ruiz-Olmos, C.A.; Camacho-Ferre, F. Effect of greenhouse soil bio-disinfection on soil nitrate content and tomato fruit yield and quality. Soil Res. 2016, 54, 200–206.
- Katan, J.; Greenberger, A.; Alon, H.; Grinstein, A. Solar Heating by Polyethylene Mulching for the Control of Diseases caused by Soil-Borne Pathogens. Phytopathology 1976, 66, 683–688.
- Kirkegaard, J.A.; Gardner, J.; Desmarcherlier, J.M.; Angus, J.F. Biofumigation Using Brassica species to Control Pest and Diseases in Horticulture and Agriculture. In Preceedings of 9th Australian Research Assembly on Brassicas; Wrather, N., Mailes, R.J., Eds.; Agricultural Research Institute: Waga Wagga, Australia, 1993; pp. 77–82.
- Palmero, D.; de Cara-García, M.; Santos, M.; Tello-Marquina, J.C. Control of diseases from forma especiales of Fusarium oxysporum causing wilt in intensive horticultural crops. Res. Signpost 2011, 661, 209–228.
- Guerrero, M.M.; Lacasa, C.M.; Martínez, V.; Martínez-Lluch, M.C.; Larregla, S.; Lacasa, A. Soil biosolarization for Verticillium dahliae and Rhizoctonia solani control in artichoke crops in southeastern Spain. Span. J. Agric. Res. 2019, 17, 1–11.
- Ros, C.; Martínez, V.; Sánchez-Solana, F.; López-Marín, J.; Lacasa, C.M.; Guerrero, M.; Del Mar GUERRERO Díaz, M.; Lacasa, A. Combination of biosolarization and grafting to control Meloidogyne incognita in greenhouse pepper crops. Crop. Prot. 2018, 113, 33–39.
- Gómez-Tenorio, M.A.; Lupión-Rodríguez, B.; Boix-Ruiz, A.; Ruiz-Olmos, C.; Marín-Guirao, J.I.; Tello-Marquina, J.C.; Camacho-Ferre, F.; De Cara-García, M. Meloidogyne-infested tomato crop residues are a suitable material for biodisinfestation to manage Meloidogyne sp. in greenhouses in Almería (south-east Spain). Acta Hortic. 2018, 1207, 217–221.
- Pankhurst, C.E.; Lynch, J.M. The role of soil microbiology in sustainable intensive agriculture. Adv. Plant Pathol. 1995, 11, 229–247.
- FAO. Keep Soil Alive, Protect Soil Biodiversity; Food and Agriculture Organization of the United Nations: Rome, Italy, 2021; pp. 1–29.
- Gómez-Tenorio, M.Á.; Magdaleno-González, J.; Tello-Marquina, J.C. Evaluación e Implementación de Técnicas Regenerativas Para la Mejora de la Fertilidad en el Cultivo del Almendro en las Provincias de Almería y Granada; Portal TecnoAgrícola: Madrid, España, 2021; pp. 1–132. ISBN 978-84-17596-98-9.
- Bilalis, D.; Krokida, M.; Roussis, I.; Papastylianou, P.; Travlos, I.; Cheimona, N.; Dede, A. Effects of organic and inorganic fertilization on yield and quality of processing tomato (Lycopersicon esculentum Mill). Folia Hortic. 2018, 30, 321–332.
- Pieper, J.R.; Barrett, D.M. Effects of organic and conventional production systems on quality and nutritional parameters of processing tomatoes. J. Sci. Food Agric. 2009, 89, 177–194.
- Guajardo-ríos, O.; Lozano-cavazos, C.J.; Valdez-Aguilar, L.A.; Benavides-mendoza, A.; Ibarra-jiménez, L.; Ascacio-Valdés, J.A.; Aguilar-gonzález, C.N. Animal-based organic nutrition can substitute inorganic fertigation in soilless-grown grape tomato. Acta Agric. Scand. Sect. B Soil Plant Sci. 2018, 68, 77–85.
- Polat, E.; Demir, H.; Erler, F. Yield and quality criteria in organically and conventionally grown tomatoes in Turkey e convencional na Turquia. Sci. Agric. 2010, 67, 424–429.
- Martín-Expósito, E.; Marín-Guirao, J.I.; García-García, M.C.; de Cara-García, M. ¿Es suficiente un único abonado de fondo con estiércol en un cultivo de tomate ecológico bajo invernadero? In Proceedings of the VIII International Congress on Agroecology, Vigo, Spain, 2–3 July 2020; p. 686.
- Gómez-Tenorio, M.A.; Ruiz-Olmos, C.A.; Marín-Guirao, J.I.; Martín, F.; Camacho-Ferre, F.; Tello-Marquina, J.C. La biodesinfección de un suelo arenado y relación con el lixiviado de nitratos. Acta Hortic. 2016, 327, 16–21.
- Guerrero, M.M.; Ros, C.; Lacasa, C.M.; Martínez, V.; Lacasa, A.; Fernández, P.; Núñez-Zofío, M.; Larreglac, S.; Martíneza, M.A.; Díez-Rojo, M.A.; et al. Effect of biosolarization using pellets of brassica carinata on soil-borne pathogens in protected pepper crops. Acta Hortic. 2010, 883, 337–344.
- Seo, M.W.; Lee, S.W.; Lee, S.H.; Jang, I.B.; Heo, H.J. Effect of Green Manure Incorporation and Solarization on Root Rot Disease of 3-year-old Ginseng in Soil of Continuous Cropping Ginseng. Korean J. Med. Crop. Sci. 2019, 27, 284–291.
- Ruiz-Olmos, C.A.; Gómez-Tenorio, M.Á.; Camacho-ferre, F.; Belmonte-Ureña, L.J.; Tello-Marquina, J.C. Control de nematodos del género Meloidogyne en un suelo de invernadero cultivado con papaya utilizando la técnica de biosolarización de suelos. Terralia 2018, 116, 53–62.
- Marín-Guirao, J.I.; de Cara-García, M.; Tello-Marquina, J.C. Effect of soil biodisinfection on soil fungal communities associated to horticultural crops. Ecosistemas 2019, 28, 63–72.
- Martínez-Francés, M.A.; Lacasa-Plasencia, A.; Tello-Marquina, J.C. Ecología de la Microbiota Fúngica de los Suelos de los Invernaderos de Pimiento y su Interés Agronómico; Ministerio: Madrid, España, 2009; Volume 148, ISBN 9788449109874.
- Martínez, M.A.; Martínez, M.C.; Bielza, P.; Tello-Marquina, J.C.; Lacasa-Plasencia, A. Effect of biofumigation with manure amendments and repeated biosolarization on Fusarium densities in pepper crops. J. Ind. Microbiol. Biotechnol. 2011, 38, 3–11.
- Zhang, M.; Zhao, G.; Li, Y.; Wang, Q.; Dang, P.; Qin, X.; Zou, Y.; Chen, Y.; Siddique, K.H.M. Straw incorporation with ridge–furrow plastic film mulch alters soil fungal community and increases maize yield in a semiarid region of China. Appl. Soil Ecol. 2021, 167, 104038.
- Brizuela, A.M.; De la Lastra, E.; Marín-Guirao, J.I.; Gálvez, L.; De Cara-García, M.; Capote, N.; Palmero, D. Fusarium Consortium Populations Associated with Asparagus Crop in Spain and Their Role on Field Decline Syndrome. J. Fungi 2020, 6, 336.
- Mendes, G.D.O.; Luiz, A.; Freitas, M. De Mechanisms of phosphate solubilization by fungal isolates when exposed to different P sources. Ann. Microbiol. 2014, 64, 239–249.
- López, J.E.; Gallego, J.L.; Vargas-ruiz, A.; Peña-mosquera, A.L.; Zapata-zapata, A.D.; López-sánchez, I.J.; Botero-botero, L.R. Aspergillus tubingensis and Talaromyces islandicus Solubilize Rock Phosphate Under Saline and Fungicide Stress and Improve Zea mays Growth and Phosphorus Nutrition. J. Soil Sci. Plant Nutr. 2020, 3, 2490–2501.
- Ceci, A.; Pinzari, F.; Russo, F.; Maggi, O. Saprotrophic soil fungi to improve phosphorus solubilisation and release: In vitro abilities of several species. Ambio 2018, 47, 30–40.
- Hora, T.S.; Iyengar, M.R.S. Nitrification by soil fungi. Arch. Mikrobiol. 1960, 35, 252–257.
- Marín-guirao, J.I.; Rodríguez-Romera, B.; Lupión-Rodriguez, B.; Camacho-Ferre, F.; Tello-Marquina, J.C. Effect of Trichoderma on horticultural seedlings growth promotion depending on inoculum and substrate type Effect of Trichoderma on horticultural seedlings growth promotion depending on inoculum and substrate type. J. Appl. Microbiol. 2016, 121, 1095–1102.
- Vassileva, M.; Malus, E.; Eichler-löbermann, B.; Vassilev, N. Aspegillus terreus: From Soil to Industry and Back. Microorganisms 2020, 8, 1655.
- Sahu, A.; Manna, M.C.; Bhattacharjya, S.; Thakur, J.K.; Mandal, A.; Mahmudur, M.; Singh, U.B.; Bhargav, V.K.; Srivastava, S.; Patra, A.K.; et al. Thermophilic ligno-cellulolytic fungi: The future of e ffi cient and rapid bio-waste management. J. Environ. Manag. 2019, 244, 144–153.
- Medina, J.J.; Miranda, L.; Soria, C.; Palencia, P. Non-Chemical Alternatives to Methyl Bromide for Strawberry: Biosolarization as Case-Study in Huelva (Spain). Acta Hortic. 2009, 842, 961–964.
- Gómez-Tenorio, M.Á.; Magdaleno-González, J.; Castillo-Díaz, F.J.; Tello-Marquina, J.C. Influence of sheep manure on soil microbiota and the vigor of cucumber seedlings in soils cultivated with almond trees. Mod. Environ. Sci. Eng. 2021, accepted.
- Usero, F.M.; Ármas, C.; Morillo, J.; Gallardo, M.; Thompson, R.B.; Pugnaire, F.I. Effects of soil microbial communities associated to different soil fertilization practices on tomato growth in intensive greenhouse agriculture. Appl. Soil Ecol. 2021, 162, 103896.




