
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Vitaly Vodyanoy | + 2290 word(s) | 2290 | 2021-10-25 04:50:49 | | | |
| 2 | Vivi Li | Meta information modification | 2290 | 2021-11-05 04:58:51 | | |
Video Upload Options
The blood and tissues of vertebrate animals and mammals contain small endogenous metal nanoparticles. These nanoparticles were observed to be composed of individual atoms of iron, copper, zinc, silver, gold, platinum, and other metals. Metal nanoparticles can bind proteins and produce proteinaceous particles called proteons. A small fraction of the entire pool of nanoparticles is usually linked with proteins to form proteons. These endogenous metal nanoparticles, along with engineered zinc and copper nanoparticles at subnanomolar levels, were shown to be lethal to cultured cancer cells. These nanoparticles appear to be elemental crystalline metal nanoparticles. It was discovered that zinc nanoparticles produce no odor response but increase the odor reaction if mixed with an odorant. Some other metal nanoparticles, including copper, silver, gold, and platinum nanoparticles, do not affect the responses to odorants. The sources of metal nanoparticles in animal blood and tissues may include dietary plants and gut microorganisms. The solid physiological and biochemical properties of metal nanoparticles reflect their importance in cell homeostasis and disease.
1. Introduction
2. Metal Nanoparticles in Blood and Tissues of Vertebrates and Mammals
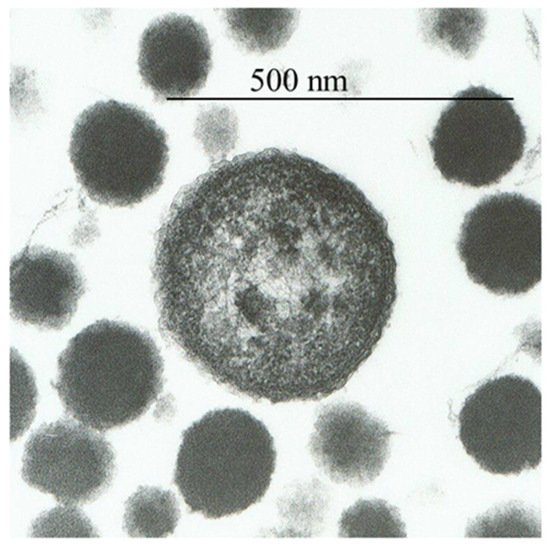
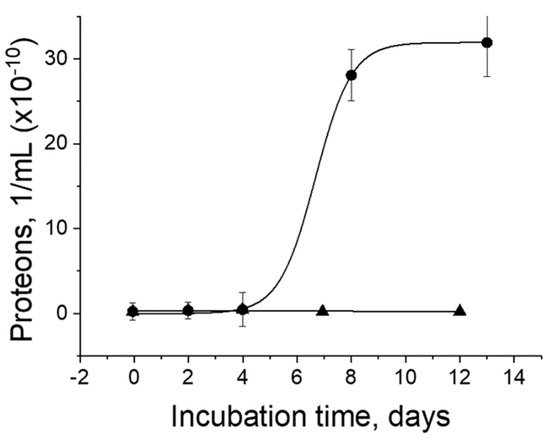
3. Metal Nanoparticles and Prions
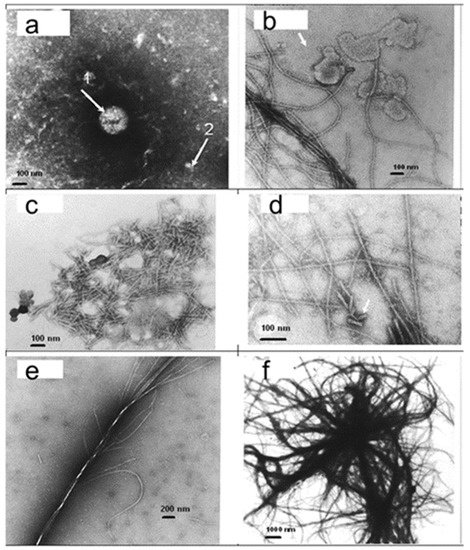
4. Metal Nanoparticles Are Lethal to Cancer Cells
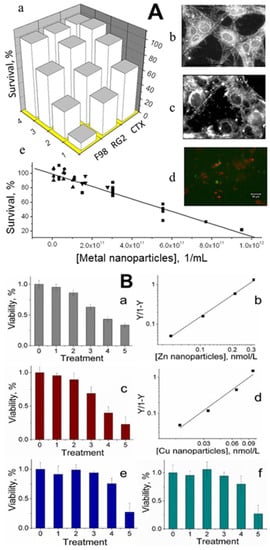
References
- Bastian, H.C. Evolution and the Origin of Life; Macmillan: London, UK, 1874; p. 215.
- Bastian, H.C. The Beginnings of Life: Being some Account of the Nature, Modes of Origin and Transformations; Macmillan: London, UK, 1872; p. 180.
- Samoylov, A.M.; Samoylova, T.I.; Pustovyy, O.M.; Samoylov, A.A.; Toivio-Kinnucan, M.A.; Morrison, N.E.; Globa, L.P.; Gale, W.F.; Vodyanoy, V. Novel Metal Clusters Isolated from Blood Are Lethal to Cancer Cells. Cells Tissues Organs 2005, 179, 115–124.
- Vodyanoy, V.; Daniels, Y.; Pustovyy, O.; MacCrehan, W.A.; Muramoto, S.; Stan, G. Engineered metal nanoparticles in the sub-nanomolar levels kill cancer cells. Int. J. Nanomed. 2016, 11, 1–10.
- Singletary, M.; Lau, J.W.; Hagerty, S.; Pustovyy, O.; Globa, L.; Vodyanoy, V. Endogenous zinc nanoparticles in the rat olfactory epithelium are functionally significant. Sci. Rep. 2020, 10, 18435.
- Miller, V.M.; Rodgers, G.; Charlesworth, J.A.; Kirkland, B.; Severson, S.R.; Rasmussen, T.E.; Yagubyan, M.; Rodgers, J.C.; Franklin, R.; Cockerill, I.; et al. Evidence of nanobacterial-like structures in calcified human arteries and cardiac valves. Am. J. Physiol. Heart Circ. Physiol. 2004, 287, H1115–H1124.
- Kumar, V.; Farell, G.; Yu, S.; Harrington, S.; Fitzpatrick, L.; Rzewuska, E.; Miller, V.M.; Lieske, J.C. Cell biology of pathologic renal calcification: Contribution of crystal transcytosis, cell-mediated calcification, and nanoparticles. J. Investig. Med. 2006, 54, 412–424.
- Kajander, E.O.; Ciftcioglu, N. Nanobacteria: An alternative mechanism for pathogenic intra- and extracellular calcification and stone formation. Proc. Natl. Acad. Sci. USA 1998, 95, 8274–8279.
- Cisar, J.O.; Xu, D.-Q.; Thompson, J.; Swaim, W.; Hu, L.; Kopecko, D.J. An alternative interpretation of nanobacteria-induced biomineralization. Proc. Natl. Acad. Sci. USA 2000, 97, 11511–11515.
- Puskás, L.G.; Tiszlavicz, L.; Rázga, Z.; Torday, L.L.; Krenács, T.; Papp, J.G. Detection of nanobacteria-like particles in human atherosclerotic plaques. Acta. Biol. Hung. 2005, 56, 233–245.
- Tanner, M.A.; Goebel, B.M.; Dojka, M.A.; Pace, N.R. Specific ribosomal DNA sequences from diverse environmental settings correlate with experimental contaminants. Appl. Environ. Microbiol. 1998, 64, 3110–3113.
- Vali, H.; McKee, M.D.; Ciftcioglu, N.; Sears, S.K.; Plows, F.L.; Chevet, E.; Ghiabi, P.; Plavsic, M.; Kajander, E.O.; Zare, R.N. Nanoforms: A new type of protein-associated mineralization. Geochim. Et Cosmochim. Acta 2001, 65, 63–74.
- Vainrub, A.; Pustovyy, O.; Vodyanoy, V. Resolution of 90 nm (λ/5) in an optical transmission microscope with an annular condenser. Opt. Lett. 2006, 31, 2855–2857.
- Vodyanoy, V.J.; Samoylov, A.M.; Pustovyy, O.M. Method of Isolation and Self-assembly of Small Protein Particles from Blood and other Biological Materials. US Patent 7,138,255, 21 November 2006.
- Vodyanoy, V.; Pustovyy, O.; Vainrub, A. High.-Resolution Light Microscopy of Nanoforms; SPIE: Bellingham, WA, USA, 2007; Volume 6694.
- Föller, M.; Huber, S.M.; Lang, F. Erythrocyte programmed cell death. Lubmb Life 2008, 60, 661–668.
- Rother, R.P.; Bell, L.; Hillmen, P.; Gladwin, M.T. The clinical sequelae of intravascular hemolysis and extracellular plasma hemoglobin: A novel mechanism of human disease. Jama 2005, 293, 1653–1662.
- Aronson, S.; Blumenthal, R. Perioperative renal dysfunction and cardiovascular anesthesia: Concerns and controversies. J. Cardiothorac. Vasc. Anesth. 1998, 12, 567–586.
- Kristiansen, M.; Graversen, J.H.; Jacobsen, C.; Sonne, O.; Hoffman, H.J.; Law, S.K.; Moestrup, S.K. Identification of the haemoglobin scavenger receptor. Nature 2001, 409, 198–201.
- Schlüter, K.; Drenckhahn, D. Co-clustering of denatured hemoglobin with band 3: Its role in binding of autoantibodies against band 3 to abnormal and aged erythrocytes. Proc. Natl. Acad. Sci. USA 1986, 83, 6137–6141.
- Kannan, R.; Labotka, R.; Low, P.S. Isolation and characterization of the hemichrome-stabilized membrane protein aggregates from sickle erythrocytes. Major site of autologous antibody binding. J. Biol. Chem. 1988, 263, 13766–13773.
- Papalexis, V.; Siomos, M.A.; Campanale, N.; Guo, X.; Kocak, G.; Foley, M.; Tilley, L. Histidine-rich protein 2 of the malaria parasite, Plasmodium falciparum, is involved in detoxification of the by-products of haemoglobin degradation. Mol. Biochem. Parasitol. 2001, 115, 77–86.
- Brown, D.R.; Qin, K.; Herms, J.W.; Madlung, A.; Manson, J.; Strome, R.; Fraser, P.E.; Kruck, T.; von Bohlen, A.; Schulz-Schaeffer, W.; et al. The cellular prion protein binds copper in vivo. Nature 1997, 390, 684–687.
- Bradley, R.; Verwoerd, D.W. Unclassified virus-like agents, transmissible spongiform encephalopathies and prion diseases. In Infectious Diseases of Livestock; Coetzer, J.A.W., Tustin, R.C., Eds.; Oxford University Press: New York, NY, USA, 2004; pp. 1388–1390.
- Prusiner, S.B. Novel proteinaceous infectious particles cause scrapie. Science 1982, 216, 136–144.
- Leach, S.P.; Salman, M.D.; Hamar, D. Trace elements and prion diseases: A review of the interactions of copper, manganese and zinc with the prion protein. Anim. Health Res. Rev. 2006, 7, 97–105.
- Brown, D.R. Metallic prions. Biochem. Soc. Symp. 2004, 71, 193–202.
- Tsenkova, R.N.; Iordanova, I.K.; Toyoda, K.; Brown, D.R. Prion protein fate governed by metal binding. Biochem. Biophys Res. Commun. 2004, 325, 1005–1012.
- Que, E.L.; Domaille, D.W.; Chang, C.J. Metals in neurobiology: Probing their chemistry and biology with molecular imaging. Chem. Rev. 2008, 108, 1517–1549.
- Baskakov, I.V.; Legname, G.; Prusiner, S.B.; Cohen, F.E. Folding of Prion Protein to Its Native α-Helical Conformation Is under Kinetic Control*. J. Biol. Chem. 2001, 276, 19687–19690.
- Ghosh, R.; Goswami, U.; Ghosh, S.S.; Paul, A.; Chattopadhyay, A. Synergistic Anticancer Activity of Fluorescent Copper Nanoclusters and Cisplatin Delivered through a Hydrogel Nanocarrier. Acs Appl. Mater. Interfaces 2015, 7, 209–222.
- Jose, G.P.; Santra, S.; Mandal, S.K.; Sengupta, T.K. Singlet oxygen mediated DNA degradation by copper nanoparticles: Potential towards cytotoxic effect on cancer cells. J. Nanobiotechnol. 2011, 9, 9.
- Studer, A.M.; Limbach, L.K.; Van Duc, L.; Krumeich, F.; Athanassiou, E.K.; Gerber, L.C.; Moch, H.; Stark, W.J. Nanoparticle cytotoxicity depends on intracellular solubility: Comparison of stabilized copper metal and degradable copper oxide nanoparticles. Toxicol. Lett. 2010, 197, 169–174.
- Bastow, M.; Kriedt, C.L.; Baldassare, J.; Shah, M.; Klein, C. Zinc is a potential therapeutic for chemoresistant ovarian cancer. J. Exp. Oncol. 2011, 9, 175–181.




