4. Chemical Characterization of Humiria balsamifera (Aubl) Leaf and Stem Bark Extracts
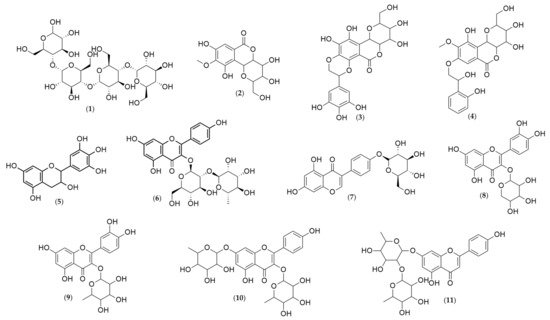
Analyses of the leaf and stem bark extracts of Humiria balsamifera (Aubl) by HPLC-ESI-IT/MS in negative-ion mode identified 11 molecular ions (Table 2, Table 3 and Table 4). Their structures were proposed (Figure 3) based on the fragments originated from the molecular ion by multi-stage mass spectrometry (MSn). The mass spectrometry ionization source was the electrospray (ESI). The ESI source may not have ionized the compounds like steroids and triterpenes. It was possible to identify only phenolic compounds in the extracts. From the 11 identified substances, seven were flavonoids (gallocatechin, kaempferol 3-neohesperidoside, sophoricoside, quercetin 3-arabinoside, quercetin-O-rhamnoside, kaempferol-dirhamnoside, and vitexin-dirhamnoside); three were coumarins (bergenin and two derivatives: galloylbergenin and hydroxybenzoyl bergenin); and one was an oligosaccharide (maltotetraose).
Figure 3. Chemical structures of the substances identified in the extracts of Humiria balsamifera (Aubl) by HPLC-ESI-IT/MS using the MassBank Spectral DataBase. Maltotetraose (1); Bergenin (2); Galloylbergenin (3); Hydroxybenzoyl bergenin (4); Gallocatechin (5); Kaempferol 3-O-neohesperidoside (6); Sophoricoside (7); Quercetin 3-arabinoside (8); Quercetin-O-rhamnoside (9); Kaempferol-dirhamnoside (10); and Vitexin-dirhamnoside (11).
Table 2. Identification of the substances present in the ethyl acetate stem bar extract of Humiria balsamifera (Aubl).
| RT (min) |
[M-H] |
MSn |
Proposed Substance |
Reference |
| 1.97 |
665 |
664, 305 |
Maltotetraose |
[28] |
| 2.85 |
327 |
249, 234, 207 |
Bergenin |
[29] |
| 3.58 |
479 |
327, 249, 234, 207 |
Galloylbergenin |
[29] |
| 4.87 |
463 |
327, 249, 234, 207 |
Hydroxybenzoyl bergenin |
[29] |
Table 3. Identification of the substances present in the Humiria balsamifera (Aubl) methanol stem bark extract.
| RT (min) |
[M-H] |
MSn |
Proposed Substance |
Reference |
| 3.01 |
305 |
179, 165 |
Gallocatechin |
[30] |
| 3.81 |
327 |
234, 207, 192 |
Bergenin |
[29] |
| 6.35 |
593 |
431, 411 |
Kaempferol 3-neohesperidoside |
[31] |
Table 4. Identification of the substances present in the Humiria balsamifera (Aubl) ethyl acetate leaf extract.
| RT (min) |
[M-H] |
MSn |
Proposed Substance |
Reference |
| 2.83 |
327 |
249, 234, 207, 192 |
Bergenin |
[29] |
| 5.27 |
431 |
311, 283, 341 |
Sophoricoside |
[32] |
| 6.09 |
433 |
300, 271, 243, 227 |
Quercetin 3-arabinoside |
[33] |
| 6.46 |
447 |
300, 271, 243 |
Quercetin-O-rhamnoside |
[33] |
| 7.17 |
577 |
431, 413, 341, 300 |
Kaempferol-dirhamnoside |
[33] |
| 8.38 |
561 |
431, 293 |
Vitexin-dirhamnoside |
[32] |
Maltotetraose (1) presented a molecular ion of m/z = 665, with a fragment of m/z = 664 after the loss of a proton. In the third stage of the fragmentation process, the loss of 341 Da as C12H21O11 and 18 Da as a water molecule produced a fragment of m/z = 305.
Bergenin (2) originated four ionic fragments: from an initial loss of 60 Da as C2H4O2, and 18 Da as a water molecule, resulted the fragment C12H10O6− (m/z = 249); the second one was produced from the loss of 93 Da as C2H4O2, a methyl group, and a water molecule, resulting in the ion C11H7O6− (m/z = 234); the third fragment C10H8O5− (m/z = 207) was the result of the loss of 120 Da as C4H8O4; and finally, the loss of 135 Da as C4H8O4 and a methyl group led to the fourth fragment, C9H5O5− (m/z = 192).
All these ions were also present in the spectra of the bergenin derivatives, plus one other fragment, observed in both spectra.
The fragment C14H15O9− (m/z = 327) was observed in both spectra—galloylbergenin (3) and hydroxybenzoyl bergenin (4). For galloylbergenin, this fragment was a result of the loss of 152 Da as the galloyl group. For hydroxybenzoyl bergenin, this fragment resulted from the elimination of the hydroxybenzoyl group (136 Da).
Gallocatechin (5) produced two fragment ions: C9H8O4− (m/z = 179), resulting from the loss of 126 Da as C6H6O3, and C8H8O4 (m/z = 165) from the loss of 140 Da as C7H8O3.
Kaempferol 3-O-neohesperidoside (6) also produced only two fragment ions: C21H19O10− (m/z = 431), generated by the loss of 162 Da as C6H10O5, and C21H15O9− (m/z = 411), originated by the ion C21H19O10−, after a water loss. Bergenin (2) was also identified.
Sophoricoside (7) originated three ions: C21H19O10− (m/z = 431), as a result of the loss of 90 Da as C3H6O3, C17H11O6− (m/z = 311) from the loss of 120 Da as C4H8O4−, and from C17H11O6−, the third fragment, C16H11O5− (m/z = 283), was formed as a result of the loss of 28 Da as a carbon monoxide molecule.
In the quercetin 3-arabinoside spectrum (8), four fragment ions were observed: C15H9O7− (m/z = 300) from the loss of 133 Da as C5H9O4, C14H7O6− (m/z = 271) from the loss of 162 Da as C6H10O5, and the fragments C13H7O5− (m/z = 243) and C13H7O4− (m/z = 227), resulting from the loss of a carbon monoxide and dioxide, respectively.
The fragmentation of quercetin-O-rhamnoside (9) were similar to what was observed for quercetin 3-arabinoside. The loss of a rhamnose molecule, C6H11O4 (m/z = 147 Da), led to the fragment ion C15H9O7− (m/z = 300). Then, the loss of carbon monoxide produced the fragments C14H7O6− (m/z = 271) and C13H7O5− (m/z = 243).
The fragmentation of kaempferol-dirhamnoside produced four ions. The first one, C21H19O10− (m/z = 431), was generated by the loss of a rhamnose molecule (C6H11O4, m/z = 147 Da). From the C21H19O10− fragment (m/z = 431), the loss of a water molecule produced the second fragment ion, C21H17O9− (m/z = 413), which, in turn, generated the fragment C18H13O7− (m/z = 341) by the loss of 72 Da as C3H4O2. Finally, the loss of 41 Da as C2OH produced the fragment C16H12O6− (m/z = 300).
The fragmentation of vitexin-dirhamnoside resulted in two ionic products: C21H17O9− (m/z = 413), generated by the loss of a rhamnose molecule (C6H11O4, 147 Da), and C17H9O5− (m/z = 293), produced by the loss of C4H6O3 (m/z = 102), followed by a water molecule.
5. Discussion
This research aimed to characterize and evaluate the antimicrobial potential of the extracts of
Humiria balsamifera (Aubl), also known as “mirim”. This species belongs to the Humiriaceae family, and its tea is used in many Brazilian regions for its anti-inflammatory action, especially for treating uterine inflammation
[22][23].
Analyses of the leaf extracts by HPLC-ESI-MS and FIA-ESI-IT/MS led to the identification of 11 substances, 10 of which had not yet been reported for this species: seven flavonoids (gallocatechin, kaempferol 3-neohesperidoside, sophoricoside, quercetin 3-arabinoside, quercetin-O-rhamnoside, kaempferol-dirhamnoside, and vitexin-dirhamnoside); one oligosaccharide (maltotetraose), bergenin; and two derivatives (galloylbergenin and hydroxybenzoyl bergenin).
The substances present in the chemical composition of
H. balsamifera tell a lot about the species. Flavonoids, according to the characterization presented in this study, are the most abundant class of compounds. These substances exhibit high bioactive potential and present anti-ulcer, antioxidant, anti-inflammatory, anti-allergic, antitumor, antiviral, antifungal, and antidiabetic activities
[34][35][36][37].
Antimicrobial tests with flavonoids have received increasing attention in recent years, since these compounds are synthesized by plants in response to various types of stress, including microbial infections
[38][39][40]. Researchers are also interested in how flavonoids are able to exhibit antibacterial activity through mechanisms different from conventional drugs, hindering the development of microbial resistance
[41][42].
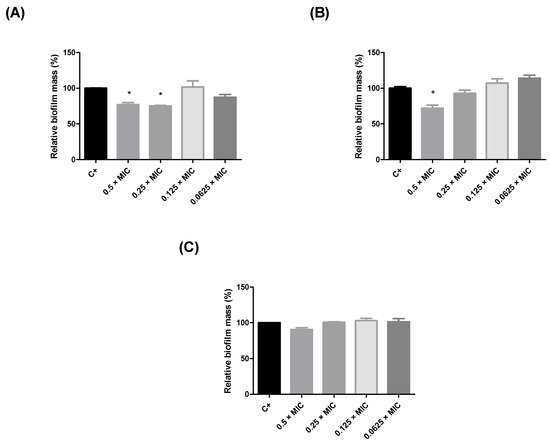
During this study, the
H. balsamifera extracts were subjected to three tests for the evaluation of their antimicrobial potential against different bacteria strains. By the end of the in vitro tests, the extracts which presented the best results were selected for the in vivo anti-infective assay using
T. molitor larvae. Our results showed that the
H. balsamifera ethyl acetate leaf extract (EALE) showed efficacy against
S. aureus, one of the most resistant pathogens in existence, in all three tests (MIC, antibiofilm potential, and the tests in vivo). The efficacy of this extract is believed to be due to the flavonoids present in its composition. Flavonoids are well-known in the literature, as other polyphenols, to be able to inhibit microbial growth through several mechanisms, such as the inhibition of ATP synthesis in the electron transport chain, inhibition of nucleic acid synthesis, inhibition of the efflux pump, inhibition of biofilm formation, inhibition of virulence factors, inhibition of quorum sensing, membrane disruption, inhibitors of bacterial toxins, and inhibition of cell envelope synthesis
[41][43][44].
Bergenin, identified in the three extracts analyzed, has already been reported in
H. balsamifera [24] and other two species from the Humiriaceae family:
Endopleura uchi and
Sacoglottis gabonensis [45][46]. This isocoumarin and its derivatives, such as the identified flavonoids, can be directly related to the antimicrobial activity of the extracts against
S. aureus. A recent study showed that six synthetic derivatives of bergenin obtained by Williamson synthesis inhibited
S. aureus growth, especially 8,10-dihexyl-bergenin and 8,10-didecyl-bergenin, which presented the most promising MIC value: 3.12 µg/mL
[47].
Besides antimicrobial activity, bergenin has also been pointed out as one of the main substances responsible for antimalarial
[24], anti-inflammatory
[48][49], antinociceptive
[50], anxiolytic
[51], and antioxidant activities
[52][53].