
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Dimitra Kiritsi | + 1194 word(s) | 1194 | 2021-10-18 10:48:41 | | | |
| 2 | Beatrix Zheng | + 133 word(s) | 1327 | 2021-10-28 04:56:45 | | |
Video Upload Options
Plectin is a multi-faceted, 500 kDa-large protein, which due to its expression in different isoforms and distinct organs acts diversely as a cytoskeletal crosslinker and signaling scaffold. It functions as a mediator of keratinocyte mechanical stability in the skin, primarily through linking intermediate filaments to hemidesmosomes. Skin fragility may occur through the presence of mutations in the gene encoding for plectin, PLEC, or through the presence of autoantibodies against the molecule.
1. Introduction
Plectin, a member of the plakin family, is a 500 kDa-large protein with various roles as a cytoskeletal crosslinker and signaling scaffold. Twelve isoforms of the molecule have been described, arising through alternative splicing, primarily within the N-terminal domain [1]. Plectin is a multi-faceted, highly interesting molecule since different isotypes are expressed in various cells (including keratinocytes, fibroblasts, myoblasts, and Schwann cells), fulfilling distinct functions [1]. Only four are expressed in the epidermis, namely P1, P1a, P1c, and P1f [1]. Structurally, plectin has a central α-helical coiled-coil rod domain with N- and C-terminal globular domains [2]; its role in the skin and other organs is revealed by the effects of mutations in the distinct plectin domains and isoforms [3]. In the skin, plectin is a mediator of keratinocyte mechanical stability, primarily through the connection of intermediate filaments (IFs) to hemidesmosomes (HD) [4]. Specifically, the N-terminus contains an actin-binding domain and a plakin domain with binding sites for integrin α6β4 [5][6] and collagen XVII/ BP180 [7], while the C-terminus comprises six plakin domains with binding sites for IFs, vinculin, and integrins [8][9][10]. Plectin is also a component of desmosomes and focal adhesions [11]. It has been suggested that the binding of plectin to integrin β4 prevails over that with the actin cytoskeleton, resulting in the preferential distribution of plectin in the hemidesmosomes [12], which accounts for the important role of plectin in regulating keratinocyte adhesion. It also plays a role in organizing mitochondria and nuclei within the intermediate filament network [11][13], thus being involved in the cellular metabolome [14]. Skin fragility may occur through the presence of mutations in the gene encoding for plectin, PLEC , or through the presence of autoantibodies directed against the molecule. To date, more than 100 mutations have been reported to result in different inherited disease subtypes (Human Gene Mutation Database- HGMD 2021.1). The manifestations of these specific disorders are summarized below.
2. Plectin in Hereditary Skin Fragility Disorders
The PLEC gene is a huge gene on chromosome 8q24, consisting of 32 exons. Mutations have mostly been disclosed within exons 31 and 32 and cause the skin fragility disorder epidermolysis bullosa simplex (EBS) ( Figure 1 A–C). On the tissue level, plectin deficiency results in a split within the keratinocytes of the basal layer of the epidermis, and although considered exceedingly rare, around 8% of patients with EBS carry mutations in the PLEC gene [15]. Since blisters are within the basal keratinocytes directly above the HD, only fragments of the destroyed HD exist on the blister floor, and thus old blisters frequently appear to be junctional (also called “pseudojunctional” EBS) ( Figure 1 D,E) [16][17]. Electron microscopy reveals hypoplastic HD, with normal extracellular portions but a poor inner plaque assembly [16][18]. A variety of antibodies have been described to specifically detect different plectin domains, which has proven helpful for the prognostication of patients’ disease severity, as explained below.
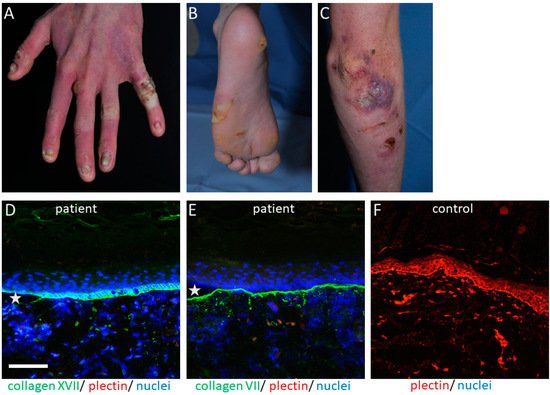
The clinical hallmark of the disease is the life-long, trauma-induced blistering of skin and mucosa. The EBS-PA subtype is characterized by congenital pyloric atresia and severe skin fragility, with areas of aplasia cutis, and was originally thought to be associated with an early demise [19]. However, a few cases have been reported where the patients survived the neonatal period, most of them developing muscular dystrophy and, interestingly, urological abnormalities [20][21][22].
Exceedingly rarely, patients might present myasthenic symptoms [23][24][25][26], although these symptoms might be disregarded in already severely muscularly impaired patients [25]. To date, phenotype–genotype correlations are still missing for the development of myasthenia in EBS-MD-affected individuals.
3. Plectin in Autoimmune Skin Fragility Disorders
Autoantibodies against plakins have been associated with autoimmune blistering disorders, specifically with paraneoplastic pemphigus (PNP), a life-threatening and rare disease, but also cases of more common bullous pemphigoid (BP) [27][28]. Due to its cytoplasmatic localization, pathogenicity, and development of anti-plectin antibodies, it is not fully understood, and the detection of plectin by blotting or immunoprecipitation is difficult due to its large size of 500 kDa [3]. Anti-plectin reactivity in BP, an acquired subepidermal blistering disease with autoreactivity to mainly hemidesmosomal proteins BP180 and/or BP230 [29], is a rare phenomenon and only rarely reported, while it is considered to occur through epitope spreading [30][31][32]. An analysis of plectin subdomains suggested the rod domain as being the immunodominant epitope [28].
Clinical hallmarks of PNP are polymorphous erosions, crusts, blisters, and/or lichenoid lesions with chronic and treatment-refractory mucositis, cheilitis, and conjunctivitis [30]. In addition, the disease shows an association with solid tumors or hematologic neoplasms (especially stomach, lung, or colon cancer, thymomas, and B-cell lymphomas) [33]. In 2001, the systemic nature of PNP was discussed by characterizing the multiorgan involvement occurring in patients, including respiratory or muscular manifestations, and the term “paraneoplastic autoimmune multiorgan syndrome” was introduced [31]. Direct immunofluorescence shows epithelial cell surface depositions of IgG and C3, but in around 50% of cases linear C3 also exists along the basement membrane zone of patients’ skin [32]. Indirect immunofluorescence on a monkey esophagus reveals IgG cell surface deposition, as occurring in pemphigus vulgaris or pemphigus foliaceus [30]. Several antigens have been associated with PNP. In the majority of patients, these include, besides plectin, the plakin family member envoplakin, desmoplakin I and II, desmogleins, desmocollins, BP230, and anti-alpha-2-macroglobulin -like 1 [34][35][36][37].
4. The Role of Plectin in Fibroblast Organization
The role of plectin in skin fibroblasts and its relevance for cutaneous disorders has remained largely unexplored. Plectin is connected to vimentin and plays a role in its network distribution within fibroblasts during the early stages of filament assembly [4]. It affects the mitosis, cell shape, polarization, and size of the cells. It also participates in forming connections between fibroblasts and their underlying extracellular matrix, while plectin-deficient fibroblasts have fewer protrusions and are rounder [38]. Their mechanical properties also seem to be impaired [39], since they are stiffer, seem to migrate less [40], and are more susceptible to stress-induced disruption [41].
5. Treatment Option for Patients with EBS-MD
References
- Castañón, M.J.; Walko, G.; Winter, L.; Wiche, G. Plectin-Intermediate Filament Partnership in Skin, Skeletal Muscle, and Peripheral Nerve. Histochem. Cell Biol. 2013, 140, 33–53.
- Wiche, G.; Becker, B.; Luber, K.; Weitzer, G.; Castañon, M.J.; Hauptmann, R.; Stratowa, C.; Stewart, M. Cloning and Sequencing of Rat Plectin Indicates a 466-KD Polypeptide Chain with a Three-Domain Structure Based on a Central Alpha-Helical Coiled Coil. J. Cell Biol. 1991, 114, 83–99.
- Natsuga, K. Plectin-Related Skin Diseases. J. Dermatol Sci 2015, 77, 139–145.
- Wiche, G.; Winter, L. Plectin Isoforms as Organizers of Intermediate Filament Cytoarchitecture. Bioarchitecture 2011, 1, 14–20.
- Geerts, D.; Fontao, L.; Nievers, M.G.; Schaapveld, R.Q.; Purkis, P.E.; Wheeler, G.N.; Lane, E.B.; Leigh, I.M.; Sonnenberg, A. Binding of Integrin Alpha6beta4 to Plectin Prevents Plectin Association with F-Actin but Does Not Interfere with Intermediate Filament Binding. J. Cell Biol. 1999, 147, 417–434.
- Koster, J.; van Wilpe, S.; Kuikman, I.; Litjens, S.H.M.; Sonnenberg, A. Role of Binding of Plectin to the Integrin Beta4 Subunit in the Assembly of Hemidesmosomes. Mol. Biol. Cell 2004, 15, 1211–1223.
- Koster, J.; Geerts, D.; Favre, B.; Borradori, L.; Sonnenberg, A. Analysis of the Interactions between BP180, BP230, Plectin and the Integrin Alpha6beta4 Important for Hemidesmosome Assembly. J. Cell Sci. 2003, 116, 387–399.
- Bouameur, J.-E.; Favre, B.; Fontao, L.; Lingasamy, P.; Begré, N.; Borradori, L. Interaction of Plectin with Keratins 5 and 14: Dependence on Several Plectin Domains and Keratin Quaternary Structure. J. Investig. Dermatol. 2014, 134, 2776–2783.
- Hijikata, T.; Murakami, T.; Ishikawa, H.; Yorifuji, H. Plectin Tethers Desmin Intermediate Filaments onto Subsarcolemmal Dense Plaques Containing Dystrophin and Vinculin. Histochem. Cell Biol. 2003, 119, 109–123.
- Te Molder, L.; Hoekman, L.; Kreft, M.; Bleijerveld, O.; Sonnenberg, A. Comparative Interactomics Analysis Reveals Potential Regulators of A6β4 Distribution in Keratinocytes. Biol. Open 2020, 9.
- Winter, L.; Wiche, G. The Many Faces of Plectin and Plectinopathies: Pathology and Mechanisms. Acta Neuropathol. 2013, 125, 77–93.
- Borradori, L.; Sonnenberg, A. Structure and Function of Hemidesmosomes: More than Simple Adhesion Complexes. J. Investig. Dermatol. 1999, 112, 411–418.
- Winter, L.; Abrahamsberg, C.; Wiche, G. Plectin Isoform 1b Mediates Mitochondrion-Intermediate Filament Network Linkage and Controls Organelle Shape. J. Cell Biol. 2008, 181, 903–911.
- Schröder, R.; Kunz, W.S.; Rouan, F.; Pfendner, E.; Tolksdorf, K.; Kappes-Horn, K.; Altenschmidt-Mehring, M.; Knoblich, R.; van der Ven, P.F.M.; Reimann, J.; et al. Disorganization of the Desmin Cytoskeleton and Mitochondrial Dysfunction in Plectin-Related Epidermolysis Bullosa Simplex with Muscular Dystrophy. J. Neuropathol. Exp. Neurol. 2002, 61, 520–530.
- Bolling, M.C.; Jongbloed, J.D.H.; Boven, L.G.; Diercks, G.F.H.; Smith, F.J.D.; Irwin McLean, W.H.; Jonkman, M.F. Plectin Mutations Underlie Epidermolysis Bullosa Simplex in 8% of Patients. J. Investig. Dermatol. 2014, 134, 273–276.
- Koss-Harnes, D.; Hoyheim, B.; Anton-Lamprecht, I.; Gjesti, A.; Jorgensen, R.S.; Jahnsen, F.L.; Olaisen, B.; Wiche, G.; Gedde-Dahl, T., Jr. A Site-Specific Plectin Mutation Causes Dominant Epidermolysis Bullosa Simplex Ogna: Two Identical de Novo Mutations. J. Investig. Dermatol. 2002, 118, 87–93.
- Gostyńska, K.B.; Nijenhuis, M.; Lemmink, H.; Pas, H.H.; Pasmooij, A.M.G.; Lang, K.K.; Castañón, M.J.; Wiche, G.; Jonkman, M.F. Mutation in Exon 1a of PLEC, Leading to Disruption of Plectin Isoform 1a, Causes Autosomal-Recessive Skin-Only Epidermolysis Bullosa Simplex. Hum. Mol. Genet. 2015, 24, 3155–3162.
- McMillan, J.R.; Akiyama, M.; Rouan, F.; Mellerio, J.E.; Lane, E.B.; Leigh, I.M.; Owaribe, K.; Wiche, G.; Fujii, N.; Uitto, J.; et al. Plectin Defects in Epidermolysis Bullosa Simplex with Muscular Dystrophy. Muscle Nerve 2007, 35, 24–35.
- Pfendner, E.; Rouan, F.; Uitto, J. Progress in Epidermolysis Bullosa: The Phenotypic Spectrum of Plectin Mutations. Exp. Dermatol. 2005, 14, 241–249.
- Charlesworth, A.; Chiaverini, C.; Chevrant-Breton, J.; DelRio, M.; Diociaiuti, A.; Dupuis, R.P.; El Hachem, M.; Le Fiblec, B.; Sankari-Ho, A.M.; Valhquist, A.; et al. Epidermolysis Bullosa Simplex with PLEC Mutations: New Phenotypes and New Mutations. Br. J. Dermatol. 2013, 168, 808–814.
- Kyrova, J.; Kopeckova, L.; Buckova, H.; Mrazova, L.; Vesely, K.; Hermanova, M.; Oslejskova, H.; Fajkusova, L. Epidermolysis Bullosa Simplex with Muscular Dystrophy. Review of the Literature and a Case Report. J. Dermatol. Case Rep. 2016, 10, 39–48.
- Argyropoulou, Z.; Liu, L.; Ozoemena, L.; Branco, C.C.; Senra, R.; Reis-Rego, Â.; Mota-Vieira, L. A Novel PLEC Nonsense Homozygous Mutation (c.7159G > T; p.Glu2387*) Causes Epidermolysis Bullosa Simplex with Muscular Dystrophy and Diffuse Alopecia: A Case Report. BMC Dermatol. 2018, 18, 1.
- Banwell, B.L.; Russel, J.; Fukudome, T.; Shen, X.M.; Stilling, G.; Engel, A.G. Myopathy, Myasthenic Syndrome, and Epidermolysis Bullosa Simplex Due to Plectin Deficiency. J. Neuropathol. Exp. Neurol. 1999, 58, 832–846.
- Gonzalez Garcia, A.; Tutmaher, M.S.; Upadhyayula, S.R.; Sanchez Russo, R.; Verma, S. Novel PLEC Gene Variants Causing Congenital Myasthenic Syndrome. Muscle Nerve 2019, 60, E40–E43.
- Selcen, D.; Juel, V.C.; Hobson-Webb, L.D.; Smith, E.C.; Stickler, D.E.; Bite, A.V.; Ohno, K.; Engel, A.G. Myasthenic Syndrome Caused by Plectinopathy. Neurology 2011, 76, 327–336.
- Forrest, K.; Mellerio, J.E.; Robb, S.; Dopping-Hepenstal, P.J.; McGrath, J.A.; Liu, L.; Buk, S.J.; Al-Sarraj, S.; Wraige, E.; Jungbluth, H. Congenital Muscular Dystrophy, Myasthenic Symptoms and Epidermolysis Bullosa Simplex (EBS) Associated with Mutations in the PLEC1 Gene Encoding Plectin. Neuromuscul. Disord. 2010, 20, 709–711.
- Goletz, S.; Zillikens, D.; Schmidt, E. Structural Proteins of the Dermal-Epidermal Junction Targeted by Autoantibodies in Pemphigoid Diseases. Exp. Dermatol. 2017, 26, 1154–1162.
- Buijsrogge, J.J.A.; de Jong, M.C.J.M.; Kloosterhuis, G.J.; Vermeer, M.H.; Koster, J.; Sonnenberg, A.; Jonkman, M.F.; Pas, H.H. Antiplectin Autoantibodies in Subepidermal Blistering Diseases. Br. J. Dermatol. 2009, 161, 762–771.
- Schmidt, E.; Zillikens, D. Pemphigoid Diseases. Lancet 2013, 381, 320–332.
- Anhalt, G.J.; Kim, S.C.; Stanley, J.R.; Korman, N.J.; Jabs, D.A.; Kory, M.; Izumi, H.; Ratrie, H.; Mutasim, D.; Ariss-Abdo, L. Paraneoplastic Pemphigus. An Autoimmune Mucocutaneous Disease Associated with Neoplasia. N. Engl. J. Med. 1990, 323, 1729–1735.
- Nguyen, V.T.; Ndoye, A.; Bassler, K.D.; Shultz, L.D.; Shields, M.C.; Ruben, B.S.; Webber, R.J.; Pittelkow, M.R.; Lynch, P.J.; Grando, S.A. Classification, Clinical Manifestations, and Immunopathological Mechanisms of the Epithelial Variant of Paraneoplastic Autoimmune Multiorgan Syndrome: A Reappraisal of Paraneoplastic Pemphigus. Arch. Dermatol. 2001, 137, 193–206.
- Paolino, G.; Didona, D.; Magliulo, G.; Iannella, G.; Didona, B.; Mercuri, S.R.; Moliterni, E.; Donati, M.; Ciofalo, A.; Granata, G.; et al. Paraneoplastic Pemphigus: Insight into the Autoimmune Pathogenesis, Clinical Features and Therapy. Int. J. Mol. Sci. 2017, 18, 2532.
- Didona, D.; DI Zenzo, G.; Joly, P. Paraneoplastic Autoimmune Multiorgan Syndrome. Ital. J. Dermatol. Venerol. 2021, 156, 174–183.
- Kiyokawa, C.; Ruhrberg, C.; Nie, Z.; Karashima, T.; Mori, O.; Nishikawa, T.; Green, K.J.; Anhalt, G.J.; DiColandrea, T.; Watt, F.M.; et al. Envoplakin and Periplakin Are Components of the Paraneoplastic Pemphigus Antigen Complex. J. Invest. Dermatol. 1998, 111, 1236–1238.
- Joly, P.; Richard, C.; Gilbert, D.; Courville, P.; Chosidow, O.; Roujeau, J.C.; Beylot-Barry, M.; D’incan, M.; Martel, P.; Lauret, P.; et al. Sensitivity and Specificity of Clinical, Histologic, and Immunologic Features in the Diagnosis of Paraneoplastic Pemphigus. J. Am. Acad. Dermatol. 2000, 43, 619–626.
- Numata, S.; Teye, K.; Tsuruta, D.; Sogame, R.; Ishii, N.; Koga, H.; Natsuaki, Y.; Tsuchisaka, A.; Hamada, T.; Karashima, T.; et al. Anti-α-2-Macroglobulin-like-1 Autoantibodies Are Detected Frequently and May Be Pathogenic in Paraneoplastic Pemphigus. J. Investig. Dermatol. 2013, 133, 1785–1793.
- Ohzono, A.; Sogame, R.; Li, X.; Teye, K.; Tsuchisaka, A.; Numata, S.; Koga, H.; Kawakami, T.; Tsuruta, D.; Ishii, N.; et al. Clinical and Immunological Findings in 104 Cases of Paraneoplastic Pemphigus. Br. J. Dermatol. 2015, 173, 1447–1452.
- Burgstaller, G.; Gregor, M.; Winter, L.; Wiche, G. Keeping the Vimentin Network under Control: Cell-Matrix Adhesion-Associated Plectin 1f Affects Cell Shape and Polarity of Fibroblasts. Mol. Biol. Cell 2010, 21, 3362–3375.
- Na, S.; Chowdhury, F.; Tay, B.; Ouyang, M.; Gregor, M.; Wang, Y.; Wiche, G.; Wang, N. Plectin Contributes to Mechanical Properties of Living Cells. Am. J. Physiol. Cell Physiol. 2009, 296, C868–C877.
- Abrahamsberg, C.; Fuchs, P.; Osmanagic-Myers, S.; Fischer, I.; Propst, F.; Elbe-Bürger, A.; Wiche, G. Targeted Ablation of Plectin Isoform 1 Uncovers Role of Cytolinker Proteins in Leukocyte Recruitment. Proc. Natl. Acad. Sci. USA 2005, 102, 18449–18454.
- Spurny, R.; Abdoulrahman, K.; Janda, L.; Rünzler, D.; Köhler, G.; Castañón, M.J.; Wiche, G. Oxidation and Nitrosylation of Cysteines Proximal to the Intermediate Filament (IF)-Binding Site of Plectin: Effects on Structure and Vimentin Binding and Involvement in IF Collapse. J. Biol. Chem. 2007, 282, 8175–8187.
- Winter, L.; Staszewska, I.; Mihailovska, E.; Fischer, I.; Goldmann, W.H.; Schroder, R.; Wiche, G. Chemical Chaperone Ameliorates Pathological Protein Aggregation in Plectin-Deficient Muscle. J. Clin. Investig. 2014, 124, 1144–1157.
- Spörrer, M.; Prochnicki, A.; Tölle, R.C.; Nyström, A.; Esser, P.R.; Homberg, M.; Athanasiou, I.; Zingkou, E.; Schilling, A.; Gerum, R.; et al. Treatment of Keratinocytes with 4-Phenylbutyrate in Epidermolysis Bullosa: Lessons for Therapies in Keratin Disorders. EBioMedicine 2019, 44, 502–515.
- Wally, V.; Klausegger, A.; Koller, U.; Lochmüller, H.; Krause, S.; Wiche, G.; Mitchell, L.G.; Hintner, H.; Bauer, J.W. 5′ Trans-Splicing Repair of the PLEC1 Gene. J. Investig. Dermatol. 2008, 128, 568–574.




