Your browser does not fully support modern features. Please upgrade for a smoother experience.

Submitted Successfully!
Thank you for your contribution! You can also upload a video entry or images related to this topic.
For video creation, please contact our Academic Video Service.
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Govind Kannan | + 928 word(s) | 928 | 2021-10-20 03:36:59 | | | |
| 2 | Bruce Ren | Meta information modification | 928 | 2021-10-27 02:52:59 | | |
Video Upload Options
We provide professional Academic Video Service to translate complex research into visually appealing presentations. Would you like to try it?
Cite
If you have any further questions, please contact Encyclopedia Editorial Office.
Kannan, G. Prevalence of E. coli in Goats. Encyclopedia. Available online: https://encyclopedia.pub/entry/15432 (accessed on 07 February 2026).
Kannan G. Prevalence of E. coli in Goats. Encyclopedia. Available at: https://encyclopedia.pub/entry/15432. Accessed February 07, 2026.
Kannan, Govind. "Prevalence of E. coli in Goats" Encyclopedia, https://encyclopedia.pub/entry/15432 (accessed February 07, 2026).
Kannan, G. (2021, October 26). Prevalence of E. coli in Goats. In Encyclopedia. https://encyclopedia.pub/entry/15432
Kannan, Govind. "Prevalence of E. coli in Goats." Encyclopedia. Web. 26 October, 2021.
Copy Citation
Goat meat is the main source of animal protein in developing countries, particularly in Asia and Africa. Goat meat consumption has also increased in the US in the recent years due to the growing ethnic population. The digestive tract of goat is a natural habitat for Escherichia coli organisms. While researchers have long focused on postharvest intervention strategies to control E. coli outbreaks, recent works have also included preharvest methodologies. In goats, these include minimizing animal stress, manipulating diet a few weeks prior to processing, feeding diets high in tannins, controlling feed deprivation times while preparing for processing, and spray washing goats prior to slaughter.
goat meat
food safety
prevalence of E. coli
preharvest management
postharvest intervention
1. Introduction
Enterohemorrhagic Escherichia coli (E. coli) is considered one of the most economically important food-borne pathogens. Much of the research has focused on post-slaughter sanitation to improve the safety of meat products, and as a result, various strategies are practiced in meat plants to reduce carcass contamination. In recent years, researchers have also been working on developing intervention strategies in the live animal prior to slaughter to reduce foodborne pathogens. Because fecal shedding is correlated with carcass contamination, reducing bacterial loads in the gastrointestinal tracts of live animals is important in the production of safe and wholesome food products.
It is widely recognized that hygienic risks at slaughterhouses should be assessed in reference to the number of organisms indicative of fecal contamination [1]. The Agricultural Marketing Service of the US Department of Agriculture has established 500 CFU g−1 as a critical limit for generic E. coli in red meat such as beef [2]. Although E. coli organisms are normal inhabitants of the gastrointestinal tracts of ruminants, some pathogenic strains can cause hemorrhagic colitis in humans [3]. Escherichia coli O157:H7 is the most common enterohemorrhagic E. coli serotype implicated in many outbreaks of bloody diarrhea and the hemolytic–uremic syndrome resulting in kidney failure.
Goat meat is one of the most widely consumed meats in the world, especially in Asia and Africa, and importation of goat meat into the US has steadily increased mainly due to increased demand by ethnic consumers. However, research on intervention strategies to reduce pathogens in goat carcasses, cuts, and products is very limited. Data available on preslaughter intervention strategies to reduce foodborne pathogens in goats are scanty.
Dietary manipulation, feed deprivation duration prior to slaughter, supplementation with high tannin-containing diets, minimizing animal stress, and live animal washing have been studied as possible pre-harvest intervention strategies to reduce E. coli in goats. Research on postharvest methods in goats have included treating goat meat with ozonated water, electrolyzed oxidizing water, ultraviolet light, sonication, organic acids, and organic oils, to name a few. In addition, nonthermal hurdle technologies with different treatment-time combinations have also been found to be promising.
2. Prevalence of E. coli in Goats
Several countries from different continents have reported E. coli O157:H7 in humans. Although this organism has been isolated from several animal species, ruminant livestock species are regarded as natural carriers of E. coli. These food animals typically do not show any clinical signs while shedding E. coli through feces. Isolation of E. coli O157:H7 from goats was first reported in 1994 from a human outbreak of E. coli O157:H7 in United Kingdom [4].
Researchers have reported different prevalence rates in different countries. According to Dulo et al. [5], E. coli O157:H7 was present in 2.2% of cecal content samples and 3.2% of carcass swab samples obtained from goats in Somali region of Ethiopia. The researchers suggested that poor hygiene and slaughter practices may cause contamination of meat and human health risks as consumption of raw meat is a common practice in Ethiopia. A comparison of E. coli O157:H7 contamination rate among beef, lamb, and chevon samples from slaughter plants and retail outlets in Addis Ababa, Ethiopia, showed that beef was the most frequently contaminated meat, followed by sheep and goat meat [6]. This study also revealed that contamination rates were higher at retail shops than at slaughterhouses for beef (21.9 vs. 4.7%), sheep (10.9 vs. 6.3%), and goat (9.4 vs. 6.3%) carcass and meat samples. Sixty percent of goat meat samples collected from different markets in Dhaka, Bangladesh has been reported to be positive for E. coli O157:H7 strains, although no official infections have been reported either due to improper tracking of outbreaks and causative organisms or to possible acquired immunity in the population [7]. Another report from Bangladesh also showed that prevalence of multidrug-resistant E. coli was higher in younger goats than older goats. The authors also reported that the prevalence of drug-resistant E. coli was higher in goats raised in poor hygienic conditions than those raised in good hygienic conditions, and higher in goats with recent history of transportation [8]. Shiga toxin-producing E. coli prevalence in Jordan was found to be greater in intensively reared goats with occasional grazing (65%) than in extensively reared goats with year-round grazing (50%) [9].
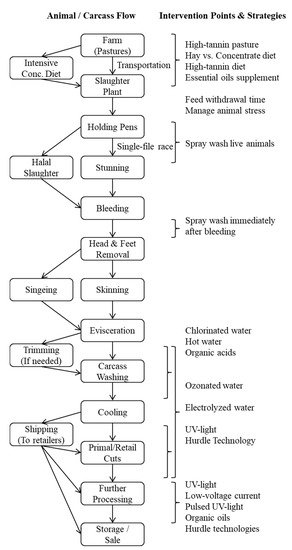
Preharvest meat goat management, slaughter and processing, and postharvest carcass and meat handling methods vary among different countries and regions. The E. coli counts on goat skin prior to slaughter generally range from 2.2 to 2.5 log10 CFU cm−2, and those on carcasses before washing range from 2.1 to 2.3 log10 CFU cm−2 [10][11]. It is not clear to what extent factors such as sex, age, and breed can influence E. coli shedding in goats. A generic flow diagram of various steps involved in pre- and post-harvest phases of chevon production is included to illustrate potential intervention points (Figure 1).
References
- Harris, L.J.; Stiles, M.E. Reliability of Escherichia coli counts for vacuum packaged ground beef. J. Food Prot. 1992, 55, 266–270.
- Agricultural Marketing Service-USDA. Microbial Testing of AMS Purchased Meat, Poultry and Egg Commodities. 2013. Available online: http://www.ams.usda.gov/resources/microbial-testing (accessed on 1 September 2021).
- Callaway, T.R.; Elder, R.O.; Keen, J.E.; Anderson, R.C.; Nisbe, D.J. Forage feeding to reduce preharvest Escherichia coli populations in cattle, a review. J. Dairy Sci. 2003, 86, 852–860.
- Shukla, R.; Slack, R.; George, A.; Cheasty, T.; Rowe, B.; Scutter, J. Escherichia coli O157 infection associated with a farm visitor center. Commun. Dis. Rep. CDR Rev. 1995, 5, R86–R90.
- Dulo, F.; Feleke, A.; Szonyi, B.; Fries, R.; Bauman, M.P.O.; Grace, D. Isolation of multidrug-resistant Escherichia coli 0157 from goats in the Somali Region of Ethiopia: A cross-sectional, abattoir-based study. PLoS ONE 2015, 10, e0142905.
- Bekele, T.; Zeede, G.; Tefera, G.; Feleke, A.; Zerom, K. Escherichia coli O157:H7 in raw meat in Addis Ababa, Ethiopia: Prevalence at an abattoir and retailers and antimicrobial susceptibility. Int. J. Food Contam. 2014, 1, 4.
- Tabashsum, Z.; Nazneen, M.; Ahsan, C.R.; Bari, M.L.; Yasmin, M. Isolation and characterization of Escherichia coli O157:H7 in raw goat meat in Dhaka city using conventional and molecular based technique. In Proceedings of the 2nd AFSSA Conference on Food Safety and Food Security, Dong Nai Province, Vietnam, 15–18 August 2014; pp. 89–95.
- Islam, K.; Ahad, A.; Barua, M.; Islam, A.; Chakma, S.; Dorji, C.; Uddin, M.A.; Islam, S.; Lutful Ahasan, A.S.M. Isolation and epidemiology of multidrug resistant Escherichia coli from goats in Coxs Bazar, Bangladesh. J. Adv. Vet. Anim. Res. 2016, 3, 166–172.
- Novotna, R.; Alexa, P.; Hamrik, J.; Madanat, A.; Smola, J.; Cizek, A. Isolation and characterization Shiga toxin-producing Escherichia coli from sheep and goats in Jordan with evidence of multiresistant serotype O157:H7. Vet. Med.—Czech. 2005, 50, 111–118.
- Kannan, G.; Jenkins, A.K.; Eega, K.R.; Kouakou, B.; McCommon, G.W. Preslaughter spray-washing effects on physiological stress responses and skin and carcass microbial counts in goats. Small Rumin. Res. 2007, 67, 14–19.
- Kannan, G.; Gutta, V.R.; Lee, J.H.; Kouakou, B.; Getz, W.R.; McCommon, G.W. Preslaughter diet management in sheep and goats: Effects on physiological responses and microbial loads on skin and carcass. J. Anim. Sci. Biotechnol. 2014, 5, 42–51.
- Mechineni, A.; Kommuru, D.S.; Terrill, T.H.; Kouakou, B.; Lee, J.H.; Gujja, S.; Burke, J.M.; Kannan, G. Forage type and transportation stress effects on gut microbial counts and meat quality in goats. Can. J. Anim. Sci. 2021, 101, 126–133.
- Gutta, V.R.; Kannan, G.; Lee, J.H.; Kouakou, B.; Getz, W.R. Influences of short-term pre-slaughter dietary manipulation in sheep and goats on pH and microbial loads of gastrointestinal tract. Small Rumin. Res. 2009, 81, 21–28.
- Gregory, N.G.; Jacobson, L.H.; Nagle, T.A.; Muirhead, R.W.; Leroux, G.J. Effect of preslaughter feeding system on weight loss, gut bacteria, and the physico-chemical properties of digesta in cattle. N. Z. J. Agric. Res. 2000, 43, 351–361.
- Behrends, L.L.; Blanton, J.R.; Miller, M.F.; Pond, K.R.; Allen, V.G. Tasco supplementation in feedlot cattle: Effects on pathogen load. J. Anim. Sci. 2000, 78, 106.
- Kannan, G.; Lee, J.H.; Kouakou, B.; Terrill, T.H. Reduction of microbial contamination in meat goats using dietary brown seaweed (Ascophyllum nodosum) supplementation and chlorinated wash. Can. J. Anim. Sci. 2019, 99, 570–577.
- Al-Kassie, G.A.M. Influence of two plant extracts derived from thyme and cinnamon on broiler performance. Pak. Vet. J. 2009, 29, 169–173.
- Smeti, S.; Hajji, H.; Mekki, I.; Mahouachi, M.; Atti, N. Effects of dose and administration form of rosemary essential oils on meat quality and fatty acid profile of lamb. Small Rumin. Res. 2018, 158, 62–68.
- Sheridan, J.J. Sources of contamination during slaughter and measures for control. J. Food Saf. 1998, 18, 321–339.
- Stevens, M.P.; van Diemen, P.M.; Dziva, F.; Jones, P.W.; Wallis, T. Options for the control of enterohaemorrhagic Escherichia coli in ruminants. Microbiology 2002, 148, 3767–3778.
- Gregory, N.G. Livestock Presentation and Welfare before Slaughter. In Animal Welfare and Meat Science; Gregory, N.G., Grandin, T., Eds.; CABI Publishing: New York, NY, USA, 1998; pp. 15–41.
- Byrne, C.M.; Bolton, D.J.; Sheridan, J.J.; McDowell, D.A.; Blair, I.S. The effects of preslaughter washing on the reduction of Escherichia coli O157:H7 transfer from cattle hides to carcasses during slaughter. Lett. Appl. Microbiol. 2000, 30, 142–145.
- Ba, H.V.; Seo, H.W.; Pil-Nam, S.; Kim, Y.S.; Park, B.Y.; Moon, S.S.; Kang, S.J.; Choi, Y.M.; Kim, J.H. The effects of pre- and post-slaughter spray application with organic acids on microbial population reductions on beef carcasses. Meat Sci. 2018, 137, 16–23.
- James, C.; Thornton, J.; Ketteringham, L.; James, S. Effect of steam condensation, hot water or chlorinated water immersion on bacterial numbers and quality of lamb carcass. J. Food Eng. 2000, 43, 219–225.
- Dorsa, W.J. New and established carcass decontamination procedures commonly used in the beef-processing industry. J. Food Prot. 1997, 60, 1146–1151.
- Smulders, F.J.M.; Greer, G.G. Integrating microbial decontamination with organic acids in HACCP programme for muscle foods: Prospects and controversies. Int. J. Food Microbiol. 1998, 44, 149–169.
- Cutter, C.N.; Siragusa, G.R. Efficacy of organic acids against Escherichia coli O157:H7 attached to beef carcass tissue using a pilot scale carcass washer. J. Food Prot. 1994, 57, 97–103.
- Berry, E.D.; Cutter, C.N. Effects of acid adaptation of Escherichia coli O157:H7 on efficacy of acetic acid spray washes to decontaminate beef carcass tissue. Appl. Environ. Microbiol. 2000, 66, 1493–1498.
- Mahapatra, A.K.; Muthukumarappan, K.; Julson, J.L. Application of ozone, bacteriocins and irradiation in food processing: A review. Crit. Rev. Food Sci. Nutr. 2005, 45, 447–461.
- Degala, H.L.; Mahapatra, A.K.; Demirci, A.; Kannan, G. Evaluation of non-thermal hurdle technology for ultraviolet-light to inactivate Escherichia coli K12 on goat meat surfaces. Food Control 2018, 90, 113–120.
- Degala, H.L.; Scott, J.R.; Nakkiran, P.; Mahapatra, A.K.; Kannan, G. Inactivation of E. coli O157:H7 on goat meat surface using ozonated water alone and in combination with electrolyzed oxidizing water. In American Society of Agricultural and Biological Engineers; Paper No. 2462209; ASABE: St. Joseph, MI, USA, 2016.
- Arya, R.; Bryant, M.; Degala, H.L.; Mahapatra, A.K.; Kannan, G. Effectiveness of a low-cost household electrolyzed water generator in reducing the populations of Escherichia coli K12 on inoculated beef, chevon, and pork surfaces. J. Food Process. Preserv. 2018, 42, e13636.
- Saif, S.M.H.; Lan, Y.; Williams, L.L.; Joshee, L.; Wang, S. Reduction of Escherichia coli O157:H7 on goat meat surface with pulsed dc square wave signal. J. Food Eng. 2006, 77, 281–288.
- Bryant, M.; Degala, H.L.; Mahapatra, A.K.; Gosukonda, R.M.; Kannan, G. Inactivation of Escherichia coli K12 by pulsed UV-light on goat meat and beef: Microbial responses and modeling. Int. J. Food Sci. Technol. 2020, 56, 563–572.
More
Information
Subjects:
Agriculture, Dairy & Animal Science
Contributor
MDPI registered users' name will be linked to their SciProfiles pages. To register with us, please refer to https://encyclopedia.pub/register
:
View Times:
1.5K
Revisions:
2 times
(View History)
Update Date:
27 Oct 2021
Notice
You are not a member of the advisory board for this topic. If you want to update advisory board member profile, please contact office@encyclopedia.pub.
OK
Confirm
Only members of the Encyclopedia advisory board for this topic are allowed to note entries. Would you like to become an advisory board member of the Encyclopedia?
Yes
No
${ textCharacter }/${ maxCharacter }
Submit
Cancel
Back
Comments
${ item }
|
More
No more~
There is no comment~
${ textCharacter }/${ maxCharacter }
Submit
Cancel
${ selectedItem.replyTextCharacter }/${ selectedItem.replyMaxCharacter }
Submit
Cancel
Confirm
Are you sure to Delete?
Yes
No