
| Version | Summary | Created by | Modification | Content Size | Created at | Operation |
|---|---|---|---|---|---|---|
| 1 | Kaiser Iqbal Wani | + 3177 word(s) | 3177 | 2021-09-20 18:17:22 | | | |
| 2 | Lindsay Dong | + 1003 word(s) | 4180 | 2021-10-26 11:36:02 | | |
Video Upload Options
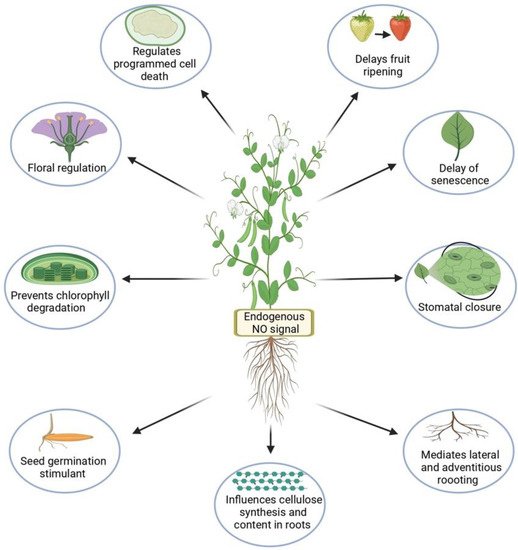
With the rapidly growing human population and changing global climate conditions, it is critical to prevent global crop losses to meet the increasing demand for food and other crop products. The reactive gaseous signaling molecule nitric oxide (NO) is involved in numerous plant developmental processes as well as plant responses to various abiotic stresses through its interactions with various molecules. Together, these interactions lead to the homeostasis of reactive oxygen species (ROS), proline and glutathione biosynthesis, post-translational modifications such as S-nitrosylation, and modulation of gene and protein expression. Exogenous application of various NO donors positively mitigates the negative effects of various abiotic stressors.
1. Introduction
| Plant Species | Drought Imposition | Concentration and Source of NO | Plant Response to NO | Reference |
|---|---|---|---|---|
| Citrullus lanatus (watermelon) | 15% PEG 600 | 100 µM SNP | Reduction in MDA content Increased activity of APX Reduced oxidative damage Increased proline content |
[37] |
| Glycine max | Withholding water | 100 µM SNP | Reduced water loss and improved biomass due to alteration of stomatal characteristics and hydraulic conductivity | [38] |
| Origanum majorana | Withholding water | 30 and 60 µM SNP | Improved water use efficiency Increased anthocyanin, soluble phenol, and flavonoid content Enhanced antioxidant capacity |
[39] |
| Brassica juncea | 10% PEG 6000 | 100 µM SNP | Antioxidant accumulation Reduction in MDA content Decreased ROS content |
[40] |
| Triticum aestivum | 15 and 30% PEG | 0.5 mM SNP | Improved antioxidant defence Enhanced glyoxalase system resulting in restoration of leaf relative water content and proline content Enhanced endogenous NO production |
[41] |
| Zea mays | Withholding water | 50, 100, 150, and 200 µM SNP | 100 µM SNP had a positive impact on chlorophyll content and water status Increased activity of CAT, SOD, and APX Improved activities of GR, GST, GOPX, nitrite and nitrate reductase activity |
[42] |
| Table | Source and Concentration of Metal | Source and Concentration of Exogenous NO | Plant Species | Impact of NO Treatment | Reference |
|---|---|---|---|---|---|
| A: Copper | 200, 400 µM CuSO4 | 200, 300 µM SNP | Lactuca sativa | Decreased DNA methylation Decreased genomic template instability Increased POX and SOD activity |
[43] |
| 5, 25, 50 μM CuSO4 | 10 μM SNP | Arabidopsis thaliana | Increased cell viability | [44] | |
| 200 µM CuCl2 | 100 μM SNP | Lolium perenne | Increased activity of SOD, CAT, APX and POX Increased chlorophyll content and photosynthesis Maintenance of Ion homeostasis |
[45] | |
| 0.2 mM Cu | 0.05 mM SNP | Nicotiana tabacum | Increased chlorophyll content, RUBISCO activity and fresh weight | [46] | |
| 450 µM CuSO4 | 200 µM SNP | Hordeum vulgare | Enhanced antioxidant enzyme activity and reduced lipid peroxidation Activation of AsA-GSH cycle |
[47] | |
| B: Cadmium | 150 μM Cd | 150 μM SNP | Hordeum vulgare | Decreased H2O2 and O2− contents Increased AsA, and GSH content Increased expression of HvAOX1a gene |
[48] |
| 200 μM CdSO4 | 200 μM SNP | Catharanthus roseus | Increased melatonin and endogenous NO concentration Increased activities of CAT, SOD, POX Decreased H2O2 and lipid peroxidation in roots |
[49] | |
| 100 μM CdSO4 | 50 μM SNP | Oryza sativa | Decreased Cd uptake by roots Restores RNS/ROS balance |
[50] | |
| 5, 7, or 9 μM CdCl2 | 300 μM SNP | Vigna radiata | Improvement adventitious formation in hypocotyl cuttings Prevents lipid peroxidation Enhanced antioxidant enzyme activity |
[51] | |
| 150 μM | 100 μM SNP | Solanum lycopersicum | Reduced Cd uptake Enhanced AsA-GSH cycle Increased activities of SOD, CAT, GR, MDHAR and APX |
[52] | |
| C: Arsenic | 75 mg/kg (NaAsO2) | 100 μM SNP | Brassica juncea | Increased activities of antioxidant enzymes Increased thiol and proline biosynthesis Decreased As uptake |
[53] |
| 50 μM (Sodium arsenate) |
100 μM SNP | Brassica seedlings | Recovery of photosynthetic pigments Increased CAT and SOD activity resulting in decreased H2O2 and Recovery of AsA and GSH content |
[54] | |
| 150 μM (Sodium meta arsenite) | 100 μM SNP | Oryza sativa | Enhanced nitrogen and thiol content Improved nitrate reductase and GOGAT activity Improved amino acid content |
[55] | |
| 1.5 mg L−1 As | 0.1 mg L−1 SNP | Pistia stratiotes Leaves |
Reduced ROS content Improved photochemical efficiency of PSII Maintained the integrity of cell organelles |
[56] | |
| D: Zinc | 500 µM ZnSO4.7H2O | 100 μM SNP | Carthamus tinctorius | Reduced Zn translocation from root to shoot Enhanced activity of AsA-GSH cycle and glyoxalase system enzymes |
[57] |
| 100, 200 µM ZnO nanoparticles | 100 μM SNP | Triticum aestivum | Decreased Zn accumulation in xylem and phloem saps Improved activity of AsA-GSH cycle |
[58] | |
| 0.05, 0.5 mM Zn (zinc sulfate) in nutrient solution | 0.1 mM SNP | Zea mays | Increased chlorophyll content Decreased leaf and root Zn content Increased nitrogen and iron content |
[59] |
2. Nitric Oxide (NO) Signaling under Abiotic Stresses
2.1. NO and Drought Stress
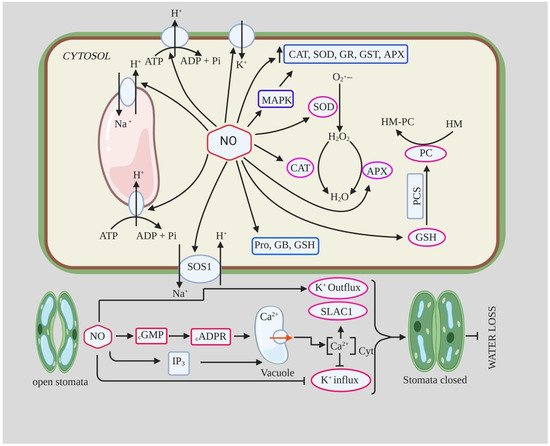
There is credible evidence that NO functions as a vital signaling molecule during both normal growth and development, and drought stress; exogenous application of NO mitigates the negative effects of drought, as seen in soybean, cucumber, and many other plants [38][60][61]. NO has been shown to mediate drought tolerance by activating the ROS scavenging enzyme system [62] and increasing osmolyte and proline metabolism [63]. NO also modulates water loss through abscisic acid-mediated stomatal response by acting as a secondary messenger in various signaling pathways, such as cyclic guanosine monophosphate (cGMP), mitogen-activated protein kinase (MAPK), and Ca2+ pathways [64].
2.1.1. NO and ROS-Mediated Oxidative Stress
During drought stress, plants produce excessive amounts of ROS (oxidative burst) due to a decrease in photosynthesis, leading to an excessive reduction in the electron transport chain and subsequent photooxidative stress [65]. NO mitigates the deleterious effects of ROS by limiting lipid peroxidation, increasing the rate of photosynthesis, and promoting antioxidants through various signaling pathways, e.g., the MAP kinase pathway [66][67]. Drought tolerance of plants is significantly enhanced by the activation of antioxidant enzyme systems such as CAT, SOD, GOPX, APX, DHAR, and GR [68]. The activity of CAT and SOD is down-regulated under drought, while application of NO up-regulates antioxidant activity as seen in hull-less barley [69]. The metalloenzyme SOD catalyzes the dismutation of superoxide to form H2O2, which is then converted to H2O and O2 by CAT and POX [70]. Activation of different isoforms of SOD under drought is considered as a strategy to counteract superoxide anion (O2−) accumulation in different cellular compartments [71][72]. SOD is quite important in preventing the reaction of O2− with proteins, with unsaturated fatty acids for peroxidation, or with NO to form ONOO−; thus, transgenic plants overexpressing Cu or Zn isoforms of the SOD gene from Puccinelia tenuiflora have increased drought tolerance [73]. Fan et al. [74] showed that treatment with the NO donor, i.e., sodium nitroprusside (SNP), under drought, up-regulated the activities of SOD, CAT, and POX, resulting in lower ROS accumulation in such plants (Table 1). Increased malondialdehyde (MDA) content and electrolyte loss are important indicators of oxidative damage to cell membranes [75], and application of NO can counteract the negative effects of drought by reducing electrolyte loss and decreasing leaf H2O2 and MDA content [69]. NO generation has been reported to be up-regulated in Cucumis sativus seedlings upon polyamine (spermine and spermidine) treatment, and its exogenous application in the form of SNP counteracts lipid peroxidation and membrane damage induced by drought stress [8]. Moreover, they also found that exogenous NO application had no effect on endogenous polyamine levels in plants under drought stress but were positively correlated with mitigation of drought induced damage, indicating that polyamines act up-stream of NO in drought stress response.
2.1.2. NO and Stomatal Closure during Drought
The exact mechanistic role of NO in ABA-mediated stomatal closure is not yet clear, but it has been proposed that NO is an important component of the ABA signaling pathway for stomatal closure [76]. NO acts downstream of the ABA signaling pathway and is an important component of the drought signaling network involved in the control of stomatal transpiration [38][60]. In a study conducted by Van Meeteren [77] on leaves of Vicia faba using SNP, NO gas, and ABA, they concluded that NO modifies stomatal opening through several pathways but is probably not critical for rapid ABA-induced stomatal closure. They found that NO/SNP application did not induce stomatal closure in epidermal cells, contradicting previous studies, whereas ABA application did induce stomatal closure [77].
2.1.3. NO and Drought-Responsive Genes
Transcription factors are important molecular players that bind to gene promoters to activate or repress transcription. Interestingly, a number of transcription factors are NO-responsive and drought-dependent [78][79]. Overexpression of SlWRKY8, which belongs to the WRKY transcription factor superfamily, increases drought tolerance in tomato [80]. In contrast, SlWRKY81 negatively regulates tomato drought tolerance by repressing NR activity, leading to reduced NO accumulation and eventually to reduced stomatal closure, which in turn increases water loss [81]. Silencing of SlWRKY81 resulted in increased NO accumulation in guard cells due to increased NR expression, leading to more efficient stomatal closure and reduced water loss [81]. Thus, silencing of SlWRKY81 can be used to increase tolerance in many drought-sensitive plants. Research on NO-mediated gene regulation in plants under drought stress is limited, and further research is needed to fully elucidate its role and paint a more comprehensive mechanistic picture.
2.2. NO and Metal/Metalloid Stress
Naturally occurring metallic elements with relatively higher atomic weight and density than water are called heavy metals (HMs) [82]. Contamination of the environment with HMs mostly occurs through anthropogenic activities, such as the use of metals and metal-containing compounds in agriculture and households, mining and smelting, and industrial production [20][83].
One of the main consequences of HM stress in plants is the excessive ROS formation, due to Fenton and Haber–Weiss reactions and changes in the antioxidant system [84][85]. Certain metals such as lead and cadmium (Cd) are not directly involved in ROS formation, but they inhibit the antioxidant system and divert electrons from the electron transport chain, indirectly promoting ROS formation [86][87]. Both endogenous and exogenous NO may play a role in plant perception, signaling, and stress acclimation under HM stress [88]. NO is readily diffusible across membranes and is involved in the regulation of numerous physiological processes, including responses to HM stress [89].
2.2.1. Cadmium Stress
Cadmium (Cd), a non-essential element and one of the most hazardous pollutants, can be toxic to animals even at non-phytotoxic concentrations [90]. It is rapidly taken up by plants due to its high mobility through Fe2+, Ca2+, Zn2+, and Mn2+ transporters, such as the ZIP IRT1 transporter [91]. As reviewed by Terrón-Camero et al. [89], NO donor application correlates negatively with HM uptake, except for Cd, which showed a positive correlation in about 40% of the studies. Cd accumulation in response to NO could most likely be due to stimulation of IRT1, which has been shown to be NO-dependent and inhibited in the presence of NO synthase inhibitor [92][93][94]. Sharing of IRT1 transporters under Cd stress leads to iron deficiency, which in turn results in NO-mediated up-regulation of FRO2 (Ferric reduction oxidase 2), IRT1 (Iron-regulated transporter 1), and FIT (FER-Like Fe deficiency induced transcription factor), leading to additional Cd accumulation [92]. Cd stress has been reported to induce endogenous NO generation, which reduces root growth due to shortening of the root elongation zone, an effect that is reversed by the NOS inhibitor L- NAME (N omega-Nitro-L-arginine methyl ester hydrochloride) [77]. However, exogenous NO may prevent the reduction of root growth in response to HM stress [77]. NO accumulation under Cd stress leads to the inhibition of root meristem in Arabidopsis due to the reduced AUX level in roots, and this inhibition was alleviated by the application of NO scavengers such as L- NAME and cPTIO (2-4-carboxyphenyl-4,4,5,5-tetramethylimidazoline-1-oxyl-3-oxide) [95]. These results suggest that NO has a negative effect on root growth and development under Cd stress. Moreover, the application of NO mitigates the negative effects of Cd stress on plant growth and development [52][96].
2.2.2. Copper Stress
Cu application has been reported to induce the formation of NO, which is mainly attributed to NOS, one of the most important enzymes in the production of NO [97]. However, Hu et al. [47] reported that early NO production in Hordeum vulgare under Cu stress was instead due to the activity of NR (not NOS), as the use of NR inhibitors resulted in decreased NO production in such plants. NO-mediated attenuation of Cu stress could be due to the up-regulation of defense-related genes or antioxidant enzyme activity [98]. In addition, NO also maintains the balance of cellular free metal concentrations by controlling their accumulation or excluding HMs in roots [98]. Application of SNP to B. juncea seeds increased their germination rate and alleviated Cu-induced oxidative stress due to enhancement of the antioxidant system, including SOD, GR, and APX, thereby lowering lipid peroxidation and H2O2 levels [99].
2.2.3. Arsenic Stress
Arsenic (As) is a highly toxic metalloid whose toxicity causes various symptoms such as necrosis, decreased photosynthesis, and growth inhibition [87]. First, it causes an increase in ROS, leading to increased lipid peroxidation and protein carboxylation, which negatively affect metabolism and disrupt cellular ultrastructure [100]. Arsenic also inhibits the activity of enzymes by binding to their sulfhydryl groups (- SH), thereby hindering several important cellular functions [101]. Souri et al. [102] suggested that As tolerance of Isatis capadoccica (an As hyperaccumulator) may be related to NO as SNP treatment enhances plant growth under As stress, while the application of an NO scavenger and the NOS inhibitor L- NAME reduces plant growth. They concluded that SNP treatment correlated with increased proline, GSH, thiol, and antioxidant concentrations, such as CAT, APX, SOD, and GR, which prevent lipid peroxidation and H2O2 accumulation. GSH, an important thiol compound, is also involved in As tolerance by participating in the biosynthesis of phytochelatin (metal-binding peptide), which binds As III, preventing its toxicity [102]. This phytochelatin complex formation is regulated by NO and is considered to be one of the major mechanistic reasons for As hypertolerance in I. capadoccica [103]. The application of SNP to rice plants under As stress can increase primary root length and number of lateral roots compared to As alone, indicating the role of NO in mitigating the effects of As stress on root development [104].
2.2.4. Zinc Stress
Accumulation of zinc (Zn) in the environment occurs through both natural causes (volcanic eruptions, fires, and weathering) and anthropogenic activities (electroplating, mining, ore processing, ink and battery industries, and agrochemical application) [105][106]. Zn plays an important role in various redox reactions and is an essential cofactor of several enzymes, such as SOD, when present in the homeostatic range [107]. It is required by plants in trace amounts and is involved in several enzyme-catalyzed reactions; therefore, its toxicity impairs these reactions, which in turn can lead to oxidative stress, senescence, and retarded growth [108]. As reported by Kolbert et al. [109], Zn stress in Arabidopsis leads to reduced activities of CAT and APX and decreased GSH content, resulting in an overall excess of H2O2.
2.2.5. Other Heavy Metal Stresses (Lead, Chromium, Mercury)
Lead (Pb) is one of the most important environmental pollutants, especially in regions with high anthropogenic activities [110], and in toxic concentrations it negatively affects crop biomass as well as yield [111]. At high concentrations, it leads to reduced growth, ROS accumulation, irregular phytomorphology, and cell death [112]. As reported by Okant and Kaya [113], Pb stress leads to increased NO content in maize leaves, and this has also been reported for other plants in previous studies involving different HMs [114].
Due to extensive industrial use, chromium (Cr) contamination has become a cause of environmental and scientific concern, with hexavalent Cr(VI) considered the most toxic among its various oxidation states [115]. Since it is not an essential element, there is no specific mechanism for its uptake and it competes with sulphur, phosphorus, and iron in carrier binding [116]. Once it enters the plants, it causes adverse effects in the plants from molecular level to whole plant level. Huang et al. [117] found that NO has the potential to attenuate Cr(VI) toxicity in tall fescue plants, improve the performance of the pigment system II, and improve overall physiological properties in these plants. NO has also been found to be helpful in mitigating Cr(VI)disadvantages in maize seedlings by suppressing lipoxygenase activity and enhancing antioxidant enzyme activities [118]. NO has been found to play a crucial role in germination and seedling development under Cr stress. Under Cr(VI)stress, the application of SNP improves seed germination and seedling development of tomato and increases the activity of protease and α-amylase hydrolyzing enzymes [119]. Furthermore, they reported an increase in nitrogen, proline, thiol content and antioxidants. These results suggest that exogenous NO application could be useful in Cr phytoremediation.
2.3. NO and Salinity Stress
The role of NO in salt tolerance has been studied in several plant species, and there is ample evidence that application of NO donor protects plants from salt stress by protecting against oxidative stress, maintaining ion homeostasis, regulating osmolyte accumulation, and improving physiological and biochemical parameters [124][125][126]. Treatment of pepper seedlings with 150 mM NaCl resulted in an increase in MDA and H2O2 content by ~100% and 87%, respectively, compared to the control [127]. However, they found that foliar application of 150 µM SNP to such seedlings decreased MDA and H2O2 content to 54% and 34%, respectively; it also improved leaf relative water content and antioxidant enzyme activity (SOD, POX, CAT) [127]. Ren et al. [126] reported that NO (10 µM SNP) pretreatment attenuated the inhibition of seed germination and early seedling growth of Brassica chinensis under salt stress. They found that SNP pretreatment increased antioxidant enzyme activity such as CAT, APX, and SOD and reduced H2O2 and MDA content, which reduced NaCl-induced oxidative damage. They also reported an increase in soluble sugar and proline content and increased K+/Na+ ratio in Radicula and Plumula. The maintenance of high K+/Na+ ratio and reduced Na+ accumulation is important for salt tolerance in plants as they reduce ion toxicity and contribute to the restoration of various metabolic processes [128]. The increased K+/Na+ ratio and decreased Na+ accumulation in NO-treated seedlings under salt stress is likely due to the inhibition of vacuolar Na+ compartmentation or Na+ influx through the plasma membrane of radicle [129]. Moreover, the increased K+ content and K+/Na+ ratio in NO-treated plants under salt stress could be due to decreased K+ efflux, an increase in competitive absorption sites, increased SOS1 transporter activity, and reduced H2O2 content [128][130]. In addition, NO was also able to induce the expression of H+-PPase and H+-ATPase, which detoxify the cell through Na+/H+ exchange, as well as the expression of AKT1-type K+ channels, ultimately leading to increased salinity tolerance [131]. NO assists sunflower seedlings to adapt to salinity stress (120 Mm NaCl) by regulating polyamine homeostasis by increasing the accumulation of polyamine biosynthetic enzymes, decreasing polyamine catabolism, and regulating their distribution [132]. Consistent with this, foliar application as well as pretreatment of NO also alleviates salinity-induced stress in broccoli plants by increasing antioxidant enzyme activity, decreasing MDA and H2O2 content, and improving glycine betaine, phenolics, and chlorophyll-a content [133]. NO acts as a cellular preservative that induces the expression of various genes controlling metabolic processes and also alters ROS content [12][134] (Table 3).
| Experimental Plant | NaCl Concentration | Concentration and Source of NO | Impact of NO on Plants | Reference |
|---|---|---|---|---|
| Jatropa curcas | 100 mM | 75 μM SNP | Reduced oxidative damage Decreased toxic ion and ROS accumulation Increased accumulation of AsA and GSH Increased activity of CAT, SOD and GR |
[134] |
| Brassica oleracea (Broccoli) |
120 mM | 0.02 mM SNP | Improved CAT, SOD, and POX activity Increased glycine betaine and total phenolic content Reduction in H2O2 and MDA content |
[133] |
| Crocus sativus (Saffron) |
50 and 100 mM NaCl | 10 µM SNP | Improved growth Accumulation of compatible solutes Increased antioxidant enzyme activity and secondary metabolite biosynthesis |
[135] |
| Hylotelephium erythrostictum | 200 mM NaCl | 50 μM SNP | Increased Na+ efflux and decreased K+ efflux Increased Ca2+ influx |
[136] |
| Brassica napus (Rapeseed) |
200 mM NaCl | 10 μM SNP | Redox and ion homeostasis Modulation of antioxidant defence genes SOS2 and NHX1 |
[137] |
| Cicer arietinum L. (chickpea) | 50 and 100 mM NaCl | 50 μM SNAP (S-nitroso-N-acetylpenicillamine) | Increased osmolyte accumulation Upregulation of CAT, SOD and APX genes Decreased electrolyte leakage, MDA and H2O2 content |
[125] |
| Gossypium (Cotton) seedlings | 100 mM NaCl | 0.1 and 1.00 mM SNP | Increased K+ Decreased K+/Na+ ratio Increased antioxidant enzyme activity Decreased MDA content |
[138] |
References
- He, M.; He, C.Q.; Ding, N.Z. Abiotic stresses: General defenses of land plants and chances for engineering multi stress tolerance. Front. Plant Sci. 2018, 9, 1771.
- Wani, S.H.; Kumar, V.; Shriram, V.; Sah, S.K. Phytohormones and their metabolic engineering for abiotic stress tolerance in crop plants. Crop. J. 2016, 4, 162–176.
- Fancy, N.N.; Bahlmann, A.; Loake, G.J. Nitric oxide function in plant abiotic stress. Plant Cell Environ. 2016, 40, 462–472.
- Nakashima, K.; Ito, Y.; Yamaguchi-Shinozaki, K. Transcriptional regulatory networks in response to abiotic stresses in Arabidopsis and grasses. Plant Physiol. 2009, 149, 88–95.
- Mittler, R. Oxidative stress, antioxidants and stress tolerance. Trends Plant Sci. 2002, 7, 405–410.
- Lindermayr, C.; Durner, J. Interplay of reactive oxygen species and nitric oxide: Nitric oxide coordinates reactive oxygen species homeostasis. Plant Physiol. 2015, 167, 1209–1210.
- Vranova, E.; Inze, D.; Breusegem, F.V. Signal transduction during oxidative stress. J. Exp. Bot. 2002, 53, 1227–1236.
- Furchgott, R.F.; Zawadzki, J.V. The obligatory role of endothelial cells in the relaxation of arterial smooth muscle by acetylcholine. Nature 1980, 288, 373–376.
- Gillespie, J.S.; Liu, X.; Martin, W. The effects of L-arginine and NG-monomethyl L-arginine on the response of the rat ano-coccygeus muscle to NANC nerve stimulation. Br. J. Pharmacol. 1989, 98, 1080–1082.
- Grün, S.; Lindermayr, C.; Sell, S.; Durner, J. Nitric oxide and gene regulation in plants. J. Exp. Bot. 2006, 57, 507–516.
- Schmidt, H.H.; Walter, U. NO at work. Cell 1994, 78, 919–925.
- Siddiqui, M.H.; Al-Whaibi, M.H.; Basalah, M.O. Role of nitric oxide in tolerance of plants to abiotic stress. Protoplasma 2011, 248, 447–455.
- Wink, D.A.; Mitchell, J.B. Chemical biology of nitric oxide: Insights into regulatory, cytotoxic, and cytoprotective mechanisms of nitric oxide. Free Radic. Biol. Med. 1998, 25, 434–456.
- Klepper, L. Nitric oxide (NO) and nitrogen dioxide (NO2) emissions from herbicide-treated soybean plants. Atmos. Environ. 1979, 13, 537–542.
- Beligni, M.V.; LaMattina, L. Nitric oxide stimulates seed germination and de-etiolation, and inhibits hypocotyl elongation, three light-inducible responses in plants. Planta 2000, 210, 215–221.
- García-Mata, C.; Lamattina, L. Abscisic acid (ABA) inhibits light-induced stomatal opening through calcium- and nitric oxide-mediated signaling pathways. Nitric Oxide 2007, 17, 143–151.
- Takahashi, S.; Yamasaki, H. Reversible inhibition of photophosphorylation in chloroplasts by nitric oxide. FEBS Lett. 2002, 512, 145–148.
- Sadhu, A.; Moriyasu, Y.; Acharya, K.; Bandyopadhyay, M. Nitric oxide and ROS mediate autophagy and regulate Alternaria alternata toxin-induced cell death in tobacco BY-2 cells. Sci. Rep. 2019, 9, 8973.
- Crawford, N.M.; Guo, F.-Q. New insights into nitric oxide metabolism and regulatory functions. Trends Plant Sci. 2005, 10, 195–200.
- He, Z.L.; Yang, X.E.; Stoffella, P.J. Trace elements in agroecosystems and impacts on the environment. J. Trace Elem. Med. Biol. 2005, 19, 125–140.
- Rather, B.A.; Mir, I.R.; Masood, A.; Anjum, N.A.; Khan, N.A. Nitric oxide pre-treatment advances seed germination and alle-viates copper-induced photosynthetic inhibition in indian mustard. Plants 2020, 9, 776.
- Creus, C.M.; Graziano, M.; Casanovas, E.M.; Pereyra, M.A.; Simontacchi, M.; Puntarulo, S.; Barassi, C.A.; LaMattina, L. Nitric oxide is involved in the Azospirillum brasilense-induced lateral root formation in tomato. Planta 2005, 221, 297–303.
- Pagnussat, G.C.; Lanteri, M.L.; Lombardo, M.C.; Lamattina, L. Nitric oxide mediates the indole acetic acid induction activation of a mitogen-activated protein kinase cascade involved in adventitious root development. Plant Physiol. 2004, 135, 279–286.
- Correa-Aragunde, N.; Lombardo, C.; LaMattina, L. Nitric oxide: An active nitrogen molecule that modulates cellulose synthesis in tomato roots. New Phytol. 2008, 179, 386–396.
- Guo, F.-Q.; Crawford, N.M. Arabidopsis Nitric Oxide Synthase1 is targeted to mitochondria and protects against oxidative damage and dark-induced senescence. Plant Cell 2005, 17, 3436–3450.
- Wani, K.I.; Zehra, A.; Choudhary, S.; Naeem, M.; Khan, M.M.A.; Castroverde, C.D.M.; Aftab, T. Mechanistic insights into strigolactone biosynthesis, signaling, and regulation during plant growth and development. J. Plant Growth Regul. 2020, 1–17, 1–17.
- Lamattina, L.; García-Mata, C.; Graziano, M.; Pagnussat, G. Nitric oxide: The versatility of an extensive signal molecule. Annu. Rev. Plant Biol. 2003, 54, 109–136.
- Koshland, D.E., Jr. The molecule of the year. Science 1992, 58, 1861–1862.
- Becana, M.; Dalton, D.A.; Moran, J.F.; Iturbe-Ormaetxe, I.; Matamoros, M.A.; Rubio, M.C. Reactive oxygen species and antiox-idants in legume nodules. Physiol. Plant 2000, 109, 372–381.
- Foyer, C.H.; Lelandais, M.; Kunert, K.J. Photooxidative stress in plants. Physiol. Plant 1994, 92, 696–717.
- Huang, H.; Ullah, F.; Zhou, D.-X.; Yi, M.; Zhao, Y. Mechanisms of ROS regulation of plant development and stress responses. Front. Plant Sci. 2019, 10, 800.
- Sandalio, L.M.; Romero-Puertas, M.C. Peroxisomes sense and respond to environmental cues by regulating ROS and RNS signalling networks. Ann. Bot. 2015, 116, 475–485.
- Schippers, J.H.; Foyer, C.; van Dongen, J. Redox regulation in shoot growth, SAM maintenance and flowering. Curr. Opin. Plant Biol. 2016, 29, 121–128.
- Decros, G.; Baldet, P.; Beauvoit, B.; Stevens, R.; Flandin, A.; Colombié, S.; Pétriacq, P. Get the balance right: ROS homeostasis and redox signalling in fruit. Front. Plant Sci. 2019, 10, 1091.
- Muñoz, P.; Munné-Bosch, S. Photo-oxidative stress during leaf, flower and fruit development. Plant Physiol. 2018, 176, 1004–1014.
- Xie, H.-T.; Wan, Z.-Y.; Li, S.; Zhang, Y. Spatiotemporal production of reactive oxygen species by NADPH oxidase is critical for tapetal programmed cell death and pollen development in Arabidopsis. Plant Cell 2014, 26, 2007–2023.
- Hamurcu, M.; Khan, M.K.; Pandey, A.; Ozdemir, C.; Avsaroglu, Z.Z.; Elbasan, F.; Omay, A.H.; Gezgin, S. Nitric oxide regulates watermelon (Citrullus lanatus) responses to drought stress. 3 Biotech 2020, 10, 494.
- De Sousa, L.F.; de Menezes-Silva, P.E.; Lourenço, L.L.; Galmés, J.; Guimarães, A.C.; da Silva, A.F.; Farnese, F.D.S. Improving water use efficiency by changing hydraulic and stomatal characteristics in soybean exposed to drought: The involvement of nitric oxide. Physiol. Plant 2020, 168, 576–589.
- Farouk, S.; Al-Huqail, A.A. Sodium nitroprusside application regulates antioxidant capacity, improves phytopharma-ceutical production and essential oil yield of marjoram herb under drought. Ind. Crop. Prod. 2020, 158, 113034.
- Sahay, S.; Khan, E.; Gupta, M. Nitric oxide and abscisic acid protects against PEG-induced drought stress differentially in Brassica genotypes by combining the role of stress modulators, markers and antioxidants. Nitric Oxide 2019, 89, 81–92.
- Hasanuzzaman, M.; Nahar, K.; Rahman, A.; Inafuku, M.; Oku, H.; Fujita, M. Exogenous nitric oxide donor and arginine provide protection against short-term drought stress in wheat seedlings. Physiol. Mol. Biol. Plants 2018, 24, 993–1004.
- Majeed, S.; Nawaz, F.; Naeem, M.; Ashraf, M.Y. Effect of exogenous nitric oxide on sulfur and nitrate assimilation pathway enzymes in maize (Zea mays L.) under drought stress. Acta Physiol. Plant 2018, 40, 206.
- Yagci, S.; Yildirim, E.; Yildirim, N.; Shams, M.; Agar, G. Nitric oxide alleviates the effects of copper-induced DNA methylation, genomic instability, LTR retrotransposon polymorphism and enzyme activity in lettuce. Plant Physiol. Rep. 2019, 24, 289–295.
- Pető, A.; Lehotai, N.; Feigl, G.; Tugyi, N.; Ördög, A.; Gémes, K.; Kolbert, Z. Nitric oxide contributes to copper tolerance by in-fluencing ROS metabolism in Arabidopsis. Plant Cell Rep. 2013, 32, 1913–1923.
- Dong, Y.; Xu, L.; Wang, Q.; Fan, Z.; Kong, J.; Bai, X. Effects of exogenous nitric oxide on photosynthesis, antioxidative ability, and mineral element contents of perennial ryegrass under copper stress. J. Plant Interact. 2013, 9, 402–411.
- Khairy, A.I.H.; Oh, M.J.; Lee, S.M.; Kim, D.S.; Roh, K.S. Nitric oxide overcomes Cd and Cu toxicity in in vitro-grown tobacco plants through increasing contents and activities of rubisco and rubisco activase. Biochim. Open 2016, 2, 41–51.
- Hu, Y.; You, J.; Liang, X. Nitrate reductase-mediated nitric oxide production is involved in copper tolerance in shoots of hulless barley. Plant Cell Rep. 2014, 34, 367–379.
- He, L.; Wang, X.; Feng, R.; He, Q.; Wang, S.; Liang, C.; Yan, L.; Bi, Y. Alternative pathway is involved in nitric oxide-enhanced tolerance to cadmium stress in barley roots. Plants 2019, 8, 557.
- Nabaei, M.; Amooaghaie, R. Nitric oxide is involved in the regulation of melatonin-induced antioxidant responses in Catharanthus roseus roots under cadmium stress. Botany 2019, 97, 681–690.
- Piacentini, D.; Ronzan, M.; Fattorini, L.; Della Rovere, F.; Massimi, L.; Altamura, M.M.; Falasca, G. Nitric oxide alleviates cadmium-but not arsenic-induced damages in rice roots. Plant Physiol. Biochem. 2020, 151, 729–742.
- Li, S.-W.; Li, Y.; Leng, Y.; Zeng, X.-Y.; Ma, Y.-H. Nitric oxide donor improves adventitious rooting in mung bean hypocotyl cuttings exposed to cadmium and osmotic stresses. Environ. Exp. Bot. 2019, 164, 114–123.
- Ahmad, P.; Ahanger, M.A.; Alyemeni, M.N.; Wijaya, L.; Alam, P. Exogenous application of nitric oxide modulates osmolyte metabolism, antioxidants, enzymes of ascorbate-glutathione cycle and promotes growth under cadmium stress in tomato. Protoplasma 2018, 255, 79–93.
- Ahmad, A.; Khan, W.U.; Shah, A.A.; Yasin, N.A.; Naz, S.; Ali, A.; Tahir, A.; Batool, A.I. Synergistic effects of nitric oxide and silicon on promoting plant growth, oxidative stress tolerance and reduction of arsenic uptake in Brassica juncea. Chemosphere 2021, 262, 128384.
- Singh, R.; Parihar, P.; Prasad, S.M. Interplay of Calcium and nitric oxide in improvement of growth and arsenic-induced toxicity in mustard seedlings. Sci. Rep. 2020, 10, 6900.
- Praveen, A.; Pandey, A.; Gupta, M. Protective role of nitric oxide on nitrogen-thiol metabolism and amino acids profiling during arsenic exposure in Oryza sativa L. Ecotoxicology 2020, 29, 825–836.
- Farnese, F.S.; Oliveira, J.A.; Paiva, E.A.; Menezes-Silva, P.E.; da Silva, A.A.; Campos, F.V.; Ribeiro, C. The involvement of nitric oxide in integration of plant physiological and ultrastructural adjustments in response to arsenic. Front. Plant Sci. 2017, 8, 516.
- Namdjoyan, S.; Kermanian, H.; Soorki, A.A.; Tabatabaei, S.M.; Elyasi, N. Interactive effects of salicylic acid and nitric oxide in alleviating zinc toxicity of Safflower (Carthamus tinctorius L.). Ecotoxicology 2017, 26, 752–761.
- Tripathi, D.K.; Mishra, R.K.; Singh, S.; Singh, S.; Singh, V.P.; Singh, P.K.; Pandey, A.C. Nitric oxide ameliorates zinc oxide nano-particles phytotoxicity in wheat seedlings: Implication of the ascorbate-glutathione cycle. Front. Plant Sci. 2017, 8, 1.
- Kaya, C. Nitric oxide improves high zinc tolerance in maize plants. J. Plant Nutr. 2015, 39, 2072–2078.
- Arasimowicz-Jelonek, M.; Floryszak-Wieczorek, J.; Kubiś, J. Interaction between polyamine and nitric oxide signaling in adaptive responses to drought in cucumber. J. Plant Growth Regul. 2009, 28, 177–186.
- Santisree, P.; Bhatnagar-Mathur, P.; Sharma, K.K. NO to drought-multifunctional role of nitric oxide in plant drought: Do we have all the answers? Plant Sci. 2015, 239, 44–55.
- Kolbert, Z.; Bartha, B.; Erdei, L. Generation of nitric oxide in roots of Pisum sativum, Triticum aestivum and Petroselinum crispum plants under osmotic and drought stress. Acta Biol. Szeged. 2005, 49, 13–16.
- Filippou, P.; Bouchagier, P.; Skotti, E.; Fotopoulos, V. Proline and reactive oxygen/nitrogen species metabolism is involved in the tolerant response of the invasive plant species Ailanthus altissima to drought and salinity. Environ. Exp. Bot. 2014, 97, 1–10.
- Gayatri, G.; Agurla, S.; Raghavendra, A.S. Nitric oxide in guard cells as an important secondary messenger during stomatal closure. Front. Plant Sci. 2013, 4, 425.
- Miller, G.; Suzuki, N.; Ciftci-Yilmaz, S.; Mittler, R. Reactive oxygen species homeostasis and signalling during drought and salinity stresses. Plant Cell Environ. 2010, 33, 453–467.
- Asai, S.; Ohta, K.; Yoshioka, H. MAPK Signaling regulates nitric oxide and NADPH oxidase-dependent oxidative bursts in Nicotiana benthamiana. Plant Cell 2008, 20, 1390–1406.
- Shi, H.; Ye, T.; Zhu, J.-K.; Chan, Z. Constitutive production of nitric oxide leads to enhanced drought stress resistance and extensive transcriptional reprogramming in Arabidopsis. J. Exp. Bot. 2014, 65, 4119–4131.
- Liu, J.; Hou, Z.-H.; Liu, G.-H.; Hou, L.-X.; Liu, X. Hydrogen sulfide may function downstream of nitric oxide in ethylene-induced stomatal closure in Vicia faba L. J. Integr. Agric. 2012, 11, 1644–1653.
- Gan, L.; Wu, X.; Zhong, Y. Exogenously applied nitric oxide enhances the drought tolerance in hulless barley. Plant Prod. Sci. 2015, 18, 52–56.
- Zhang, A.; Jiang, M.; Zhang, J.; Ding, H.; Xu, S.; Hu, X.; Tan, M. Nitric oxide induced by hydrogen peroxide mediates abscisic acid-induced activation of the mitogen-activated protein kinase cascade involved in antioxidant defense in maize leaves. New Phytol. 2007, 175, 36–50.
- Shan, C.; Zhang, S.; Ou, X. The roles of H2S and H2O2 in regulating AsA-GSH cycle in the leaves of wheat seedlings under drought stress. Protoplasma 2018, 255, 1257–1262.
- Zandalinas, S.I.; Balfagón, D.; Arbona, V.; Gómez-Cadenas, A. Modulation of antioxidant defense system is associated with combined drought and heat stress tolerance in Citrus. Front. Plant Sci. 2017, 8, 953.
- Wu, J.; Zhang, J.; Li, X.; Xu, J.J.; Wang, L. Identification and characterization of a PutCu/Zn-SOD gene from Puccinellia tenuiflora (Turcz.) Scribn. et Merr. Plant Growth Regul. 2016, 79, 55–64.
- Fan, Q.J.; Liu, J.H. Nitric oxide is involved in dehydration/drought tolerance in Poncirus trifoliata seedlings through regulation of antioxidant systems and stomatal response. Plant Cell Rep. 2012, 31, 145–154.
- Farooq, M.; Basra, S.M.A.; Wahid, A.; Cheema, Z.A.; Cheema, M.A.; Khaliq, A. Physiological role of exogenously applied gly-cinebetaine to improve drought tolerance in fine grain aromatic rice (Oryza sativa L.). J. Agron. Crop Sci. 2008, 194, 325–333.
- Murata, Y.; Mori, I.C.; Munemasa, S. Diverse stomatal signaling and the signal integration mechanism. Annu. Rev. Plant Biol. 2015, 66, 369–392.
- Van Meeteren, U.; Kaiser, E.; Malcolm Matamoros, P.; Verdonk, J.C.; Aliniaeifard, S. Is nitric oxide a critical key factor in ABA-induced stomatal closure? J. Exp. Bot. 2020, 71, 399–410.
- Nabi, R.B.S.; Tayade, R.; Hussain, A.; Kulkarni, K.P.; Imran, Q.M.; Mun, B.-G.; Yun, B.-W. Nitric oxide regulates plant responses to drought, salinity, and heavy metal stress. Environ. Exp. Bot. 2019, 161, 120–133.
- Chen, F.; Hu, Y.; Vannozzi, A.; Wu, K.; Cai, H.; Qin, Y.; Mullis, A.; Lin, Z.; Zhang, L. The WRKY Transcription Factor Family in Model Plants and Crops. Crit. Rev. Plant Sci. 2017, 36, 311–335.
- Gao, Y.; Liu, J.; Yang, F.; Zhang, G.; Wang, D.; Zhang, L.; Ou, Y.; Yao, Y. The WRKY transcription factor WRKY8 promotes resistance to pathogen infection and mediates drought and salt stress tolerance in Solanum lycopersicum. Physiol. Plant. 2019, 168, 98–117.
- Ahammed, G.J.; Li, X.; Mao, Q.; Wan, H.; Zhou, G.; Cheng, Y. The SlWRKY81 transcription factor inhibits stomatal closure by attenuating nitric oxide accumulation in the guard cells of tomato under drought. Physiol. Plant. 2021, 172, 885–895.
- Tchounwou, P.B.; Yedjou, C.G.; Patlolla, A.K.; Sutton, D.J. Heavy metal toxicity and the environment. NIH 2012, 101, 133–164.
- Shallari, S.; Schwartz, C.; Hasko, A.; Morel, J. Heavy metals in soils and plants of serpentine and industrial sites of Albania. Sci. Total Environ. 1998, 209, 133–142.
- Cui, B.; Pan, Q.; Clarke, D.; Villarreal, M.O.; Umbreen, S.; Yuan, B.; Shan, W.; Jiang, J.; Loake, G.J. S-nitrosylation of the zinc finger protein SRG1 regulates plant immunity. Nat. Commun. 2018, 9, 4226.
- Romero-Puertas, M.C.; Terrón-Camero, L.C.; Peláez-Vico, M.Á.; Olmedilla, A.; Sandalio, L.M. Reactive oxygen and nitrogen species as key indicators of plant responses to Cd stress. Environ. Exp. Bot. 2019, 161, 107–119.
- Loix, C.; Huybrechts, M.; Vangronsveld, J.; Gielen, M.; Keunen, E.; Cuypers, A. Reciprocal interactions between cadmium-induced cell wall responses and oxidative stress in plants. Front. Plant Sci. 2017, 8, 1867.
- Souri, Z.; Karimi, N.; de Oliveira, L.M. Antioxidant enzymes responses in shoots of arsenic hyperaccumulator, Isatis cappadocica Desv., under interaction of arsenate and phosphate. Environ. Technol. 2018, 39, 1316–1327.
- Terrón-Camero, L.C.; Peláez-Vico, M.Á.; Del-Val, C.; Sandalio, L.M.; Romero-Puertas, M.C. Role of nitric oxide in plant responses to heavy metal stress: Exogenous application versus endogenous production. J. Exp. Bot. 2019, 70, 4477–4488.
- Sahay, S.; Gupta, M. An update on nitric oxide and its benign role in plant responses under metal stress. Nitric Oxide 2017, 67, 39–52.
- Ismael, M.A.; Elyamine, A.M.; Moussa, M.G.; Cai, M.; Zhao, X.; Hu, C. Cadmium in plants: Uptake, toxicity, and its interactions with selenium fertilizers. Metallomics 2019, 11, 255–277.
- Verbruggen, N.; Hermans, C.; Schat, H. Mechanisms to cope with arsenic or cadmium excess in plants. Curr. Opin. Plant Biol. 2009, 12, 364–372.
- Besson-Bard, A.; Gravot, A.; Richaud, P.; Auroy, P.; Duc, C.; Gaymard, F.; Taconnat, L.; Renou, J.-P.; Pugin, A.; Wendehenne, D. Nitric oxide contributes to cadmium toxicity in Arabidopsis by promoting cadmium accumulation in roots and by up-regulating genes related to iron uptake. Plant Physiol. 2009, 149, 1302–1315.
- Connolly, E.L.; Fett, J.; Guerinot, M.L. Expression of the IRT1 metal transporter is controlled by metals at the levels of transcript and protein accumulation. Plant Cell 2002, 14, 1347–1357.
- Graziano, M.; Lamattina, L. Nitric oxide accumulation is required for molecular and physiological responses to iron deficiency in tomato roots. Plant J. 2007, 52, 949–960.
- Zafari, S.; Sharifi, M.; Chashmi, N.A.; Mur, L. Modulation of Pb-induced stress in Prosopis shoots through an interconnected network of signaling molecules, phenolic compounds and amino acids. Plant Physiol. Biochem. 2016, 99, 11–20.
- Per, T.S.; Masood, A.; Khan, N.A. Nitric oxide improves S-assimilation and GSH production to prevent inhibitory effects of cadmium stress on photosynthesis in mustard (Brassica juncea L.). Nitric Oxide 2017, 68, 111–124.
- Chamizo-Ampudia, A.; Sanz-Luque, E.; Llamas, A.; Galvan, A.; Fernandez, E.P.-A. Nitrate reductase regulates plant nitric oxide homeostasis. Trends Plant Sci. 2017, 22, 163–174.
- Rizwan, M.; Mostofa, M.G.; Ahmad, M.Z.; Imtiaz, M.; Mehmood, S.; Adeel, M.; Dai, Z.; Li, Z.; Aziz, O.; Zhang, Y.; et al. Nitric oxide induces rice tolerance to excessive nickel by regulating nickel uptake, reactive oxygen species detoxification and defense-related gene expression. Chemosphere 2018, 191, 23–35.
- Oz, M.T.; Eyidogan, F.; Yucel, M.; Öktem, H.A. Functional Role of Nitric Oxide Under Abiotic Stress Conditions. In Nitric Oxide Action in Abiotic Stress Responses in Plants; Springer Science and Business Media LLC: Berlin/Heidelberg, Germany, 2015; pp. 21–41.
- Da-Silva, C.J.; Canatto, R.A.; Cardoso, A.A.; Ribeiro, C.; Oliveira, J.A. Arsenic-hyperaccumulation and antioxidant system in the aquatic macrophyte Spirodela intermedia W. Koch (Lemnaceae). Theor. Exp. Plant Physiol. 2017, 29, 203–213.
- Nath, S.; Panda, P.; Mishra, S.; Dey, M.; Choudhury, S.; Sahoo, L.; Panda, S.K. Arsenic stress in rice: Redox consequences and regulation by iron. Plant Physiol. Biochem. 2014, 80, 203–210.
- Souri, Z.; Karimi, N.; Farooq, M.A.; Sandalio, L.M. Nitric oxide improves tolerance to arsenic stress in Isatis cappadocica desv. Shoots by enhancing antioxidant defenses. Chemosphere 2020, 239, 124523.
- Souri, Z.; Karimi, N. Enhanced phytoextraction by As hyperaccumulator Isatis cappadocica spiked with sodium nitro-prusside. Soil Sedim. Contam. Int. J. 2017, 26, 457–468.
- Praveen, A.; Gupta, M. Nitric oxide confronts arsenic stimulated oxidative stress and root architecture through distinct gene expression of auxin transporters, nutrient related genes and modulates biochemical responses in Oryza sativa L. Environ. Pollut. 2018, 240, 950–962.
- Sharma, A.; Soares, C.; Sousa, B.; Martins, M.; Kumar, V.; Shahzad, B.; Thukral, A.K. Nitric oxide-mediated regulation of oxi-dative stress in plants under metal stress: A review on molecular and biochemical aspects. Physiol. Plant 2020, 168, 318–344.
- Tsonev, T.; Lidon, F.J.C. Zinc in plants—An overview. Emir J Food Agric. 2012, 24, 322–333.
- Soares, C.; Carvalho, M.; Azevedo, R.A.; Fidalgo, F. Plants facing oxidative challenges—A little help from the antioxidant networks. Environ. Exp. Bot. 2019, 161, 4–25.
- Lin, Y.-F.; Aarts, M.G.M. The molecular mechanism of zinc and cadmium stress response in plants. Cell. Mol. Life Sci. 2012, 69, 3187–3206.
- Kolbert, Z.; Molnár, Á.; Oláh, D.; Feigl, G.; Horváth, E.; Erdei, L.; Lindermayr, C. S-Nitrosothiol signaling is involved in regulating hydrogen peroxide metabolism of zinc-stressed Arabidopsis. Plant Cell. Physiol. 2019, 60, 2449–2463.
- Sürücü, A.; Mohammad, D.M.; Günal, E.; Budak, M. Concentration of heavy metals in soils along three major roads of Sulaimani, Northeast Iraq. Carpathian J. Earth Environ. Sci. 2018, 13, 523–538.
- Kobylińska, A.; Reiter, R.J.; Posmyk, M.M. Melatonin Protects Cultured Tobacco Cells against Lead-Induced Cell Death via Inhibition of Cytochrome c Translocation. Front. Plant Sci. 2017, 8, 1560.
- Zhou, J.; Zhang, Z.; Zhang, Y.; Wei, Y.; Jiang, Z. Effects of lead stress on the growth, physiology, and cellular structure of privet seedlings. PLoS ONE 2018, 13, e0191139.
- Okant, M.; Kaya, C. The role of endogenous nitric oxide in melatonin-improved tolerance to lead toxicity in maize plants. Environ. Sci. Pollut. Res. 2019, 26, 11864–11874.
- Corpas, F.J.; Barroso, J.B. Peroxynitrite (ONOO−) is endogenously produced in Arabidopsis peroxisomes and is overproduced under cadmium stress. Ann. Bot. 2013, 113, 87–96.
- Oliveira, H. Chromium as an environmental pollutant: Insights on induced plant toxicity, Hindawi Publishing Corporation. J. Bot. 2012, 2012, 375843.
- Shanker, A.K.; Cervantes, C.; Loza-Tavera, H.; Avudainayagam, S. Chromium toxicity in plants. Environ Int. 2005, 31, 739–753.
- Huang, M.; Ai, H.; Xu, X.; Chen, K.; Niu, H.; Zhu, H.; Sun, J.; Du, D.; Chen, L. Nitric oxide alleviates toxicity of hexavalent chromium on tall fescue and improves performance of photosystem II. Ecotoxicol. Environ. Saf. 2018, 164, 32–40.
- Kharbech, O.; Sakouhi, L.; Ben Massoud, M.; Mur, L.A.J.; Corpas, F.J.; Djebali, W.; Chaoui, A. Nitric oxide and hydrogen sulfide protect plasma membrane integrity and mitigate chromium-induced methylglyoxal toxicity in maize seedlings. Plant Physiol. Biochem. 2020, 157, 244–255.
- Khan, M.N.; Alamri, S.; Al-Amri, A.A.; Alsubaie, Q.D.; Al-Munqedi, B.; Ali, H.M.; Siddiqui, M.H. Effect of nitric oxide on seed germination and seedling development of tomato under chromium toxicity. J. Plant Growth Regul. 2020, 1–13.
- Han, F.X.; Banin, A.; Su, Y.; Monts, D.L.; Plodinec, J.M.; Kingery, W.L.; Triplett, G.E. Industrial age anthropogenic inputs of heavy metals into the pedosphere. Naturwissenschaften 2002, 89, 497–504.
- Han, F.X.; Su, Y.; Monts, D.L.; Waggoner, C.A.; Plodinec, M.J. Binding, distribution, and plant uptake of mercury in a soil from Oak Ridge, Tennessee, USA. Sci. Total Environ. 2006, 368, 753–768.
- Ahmad, P.; Alyemeni, M.N.; Wijaya, L.; Ahanger, M.A.; Ashraf, M.; Alam, P.; Paray, B.A.; Rinklebe, J. Nitric oxide donor, sodium nitroprusside, mitigates mercury toxicity in different cultivars of soybean. J. Hazard. Mater. 2021, 408, 124852.
- Chen, Z.; Zhang, L.; Zhu, C. Exogenous nitric oxide mediates alleviation of mercury toxicity by promoting auxin transport in roots or preventing oxidative stress in leaves of rice seedlings. Acta Physiol. Plant. 2015, 37, 194.
- Ahmad, P.; Ahanger, M.A.; Alyemeni, M.N.; Wijaya, L.; Alam, P.; Ashraf, M. Mitigation of sodium chloride toxicity in Solanum lycopersicum L. by supplementation of jasmonic acid and nitric oxide. J. Plant Interact. 2018, 13, 64–72.
- Ahmad, P.; Abdel Latef, A.A.; Hashem, A.; Abd_Allah, E.F.; Gucel, S.; Tran, L.S.P. Nitric oxide mitigates salt stress by regulating levels of osmolytes and antioxidant enzymes in chickpea. Front. Plant Sci. 2016, 7, 347.
- Ren, Y.; Wang, W.; He, J.; Zhang, L.; Wei, Y.; Yang, M. Nitric oxide alleviates salt stress in seed germination and early seedling growth of pakchoi (Brassica chinensis L.) by enhancing physiological and biochemical parameters. Ecotoxicol. Environ. Saf. 2020, 187, 109785.
- Shams, M.; Ekinci, M.; Ors, S.; Turan, M.; Agar, G.; Kul, R.; Yildirim, E. Nitric oxide mitigates salt stress effects of pepper seedlings by altering nutrient uptake, enzyme activity and osmolyte accumulation. Physiol. Mol. Biol. Plants 2019, 25, 1149–1161.
- Assaha, D.V.M.; Ueda, A.; Saneoka, H.; Al-Yahyai, R.; Yaish, M.W. The role of Na+ and K+ transporters in salt stress adaptation in glycophytes. Front. Physiol. 2017, 8, 509.
- Zhang, Y.; Wang, L.; Liu, Y.; Zhang, Q.; Wei, Q.; Zhang, W. Nitric oxide enhances salt tolerance in maize seedlings through increasing activities of proton-pump and Na+/H+ antiport in the tonoplast. Planta 2006, 224, 545–555.
- Campos, F.V.; Oliveira, J.A.; Pereira, M.G.; Farnese, F.S. Nitric oxide and phytohormone interactions in the response of Lactuca sativa to salinity stress. Planta 2019, 250, 1475–1489.
- Hasanuzzaman, M.; Oku, H.; Nahar, K.; Bhuyan, M.B.; Al Mahmud, J.; Baluska, F.; Fujita, M. Nitric oxide-induced salt stress tolerance in plants: ROS metabolism, signaling, and molecular interactions. Plant Biotechnol. Rep. 2018, 12, 77–92.
- Tailor, A.; Tandon, R.; Bhatla, S.C. Nitric oxide modulates polyamine homeostasis in sunflower seedling cotyledons under salt stress. Plant Signal. Behav. 2019, 14, 1667730.
- Akram, N.A.; Hafeez, N.; Farid-Ul-Haq, M.; Ahmad, A.; Sadiq, M.; Ashraf, M. Foliage application and seed priming with nitric oxide causes mitigation of salinity-induced metabolic adversaries in broccoli (Brassica oleracea L.) plants. Acta Physiol. Plant. 2020, 42, 155.
- Gadelha, C.G.; Miranda, R.D.S.; Alencar, N.L.M.; Costa, J.H.; Prisco, J.T.; Gomes-Filho, E. Exogenous nitric oxide improves salt tolerance during establishment of Jatropha curcas seedlings by ameliorating oxidative damage and toxic ion accumulation. J. Plant Physiol. 2017, 212, 69–79.
- Babaei, S.; Niknam, V.; Behmanesh, M. Comparative effects of nitric oxide and salicylic acid on salinity tolerance in saffron (Crocus sativus). Plant Biosyst. Int. J. Deal. All Asp. Plant Biol. 2021, 155, 73–82.
- Chen, Z.; Zhao, X.; Hu, Z.; Leng, P. Nitric oxide modulating ion balance in Hylotelephium erythrostictum roots subjected to NaCl stress based on the analysis of transcriptome, fluorescence, and ion fluxes. Sci. Rep. 2019, 9, 18317.
- Zhao, G.; Zhao, Y.; Yu, X.; Kiprotich, F.; Han, H.; Guan, R.; Wang, R.; Shen, W. Nitric oxide is required for melatonin-enhanced tolerance against salinity stress in rapeseed (Brassica napus L.) seedlings. Int. J. Mol. Sci. 2018, 19, 1912.
- Dong, Y.; Jinc, S.; Liu, S.; Xu, L.; Kong, J. Effects of exogenous nitric oxide on growth of cotton seedlings under NaCl stress. J. Soil Sci. Plant Nutr. 2014, 14, 1–13.




